CCO_Targeting_CD33_AML_Slides.pptx
- Количество слайдов: 31
 Targeting CD 33 in AML: Therapeutic Strategies With New Antibody–Drug Conjugates William Donnellan, MD Investigator, Hematologic Malignancies Tennessee Oncology Sarah Cannon Research Institute Nashville, Tennessee This activity is supported by an educational grant from Seattle Genetics.
Targeting CD 33 in AML: Therapeutic Strategies With New Antibody–Drug Conjugates William Donnellan, MD Investigator, Hematologic Malignancies Tennessee Oncology Sarah Cannon Research Institute Nashville, Tennessee This activity is supported by an educational grant from Seattle Genetics.
 About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details
About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details
 Faculty William Donnellan, MD Investigator, Hematologic Malignancies Tennessee Oncology Sarah Cannon Research Institute Nashville, Tennessee William Donnellan, MD, has disclosed that he has received funds for research support from Seattle Genetics.
Faculty William Donnellan, MD Investigator, Hematologic Malignancies Tennessee Oncology Sarah Cannon Research Institute Nashville, Tennessee William Donnellan, MD, has disclosed that he has received funds for research support from Seattle Genetics.
 WHO AML Classification § AML with recurrent genetic abnormalities – t(8; 21)(q 22; q 22); RUNX 1 T 1 – inv(16)(p 13, 1 q 22) or t(16/16)(p 13. 1; q 22); CBFBMYH 11 § Other AML-related clinical presentations – AML with minimal differentiation – AML without maturation – AML with maturation – t(15; 17)(q 22; q 12); PML-RARA – Acute myelomonocytic leukemia – t(9; 11)(p 22; q 23); MLLT 3 -MLL – Acute erythroid leukemia – t(6; 9)(p 23/q 34); DEK-NUP 214 – Acute megakaryoblastic leukemia – Mutated NPM 1 (provisional) Mutated CEBPA (provisional) – Acute panmyelosis with myelofibrosis Swerdlow SH, et al. WHO Classification. IARC Press. 2008; 109 -138. Vardiman JW, et al. Blood. 2009; 114: 937 -951. Arber, et al. Blood. 2016; 127: 2391 -2405. Slide credit: clinicaloptions. com
WHO AML Classification § AML with recurrent genetic abnormalities – t(8; 21)(q 22; q 22); RUNX 1 T 1 – inv(16)(p 13, 1 q 22) or t(16/16)(p 13. 1; q 22); CBFBMYH 11 § Other AML-related clinical presentations – AML with minimal differentiation – AML without maturation – AML with maturation – t(15; 17)(q 22; q 12); PML-RARA – Acute myelomonocytic leukemia – t(9; 11)(p 22; q 23); MLLT 3 -MLL – Acute erythroid leukemia – t(6; 9)(p 23/q 34); DEK-NUP 214 – Acute megakaryoblastic leukemia – Mutated NPM 1 (provisional) Mutated CEBPA (provisional) – Acute panmyelosis with myelofibrosis Swerdlow SH, et al. WHO Classification. IARC Press. 2008; 109 -138. Vardiman JW, et al. Blood. 2009; 114: 937 -951. Arber, et al. Blood. 2016; 127: 2391 -2405. Slide credit: clinicaloptions. com
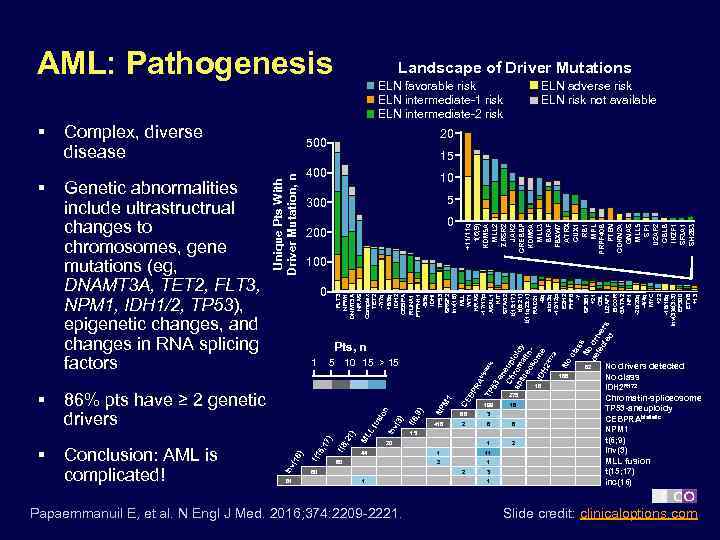 AML: Pathogenesis ELN adverse risk ELN risk not available 20 500 15 400 10 300 5 0 200 +11/11 q t(6; 9) KDM 5 A MLL 2 ZRSR 2 JAK 2 CREBBP KDM 6 A MLL 3 BRAF FBXW 7 ATRX CUX 1 RB 1 MPL PRPF 40 B PTEN CDKN 2 A GNAS MLL 5 SF 1 U 2 AF 2 CBLB IKZF 1 SF 3 A 1 SH 2 B 3 Genetic abnormalities include ultrastructrual changes to chromosomes, gene mutations (eg, DNAMT 3 A, TET 2, FLT 3, NPM 1, IDH 1/2, TP 53), epigenetic changes, and changes in RNA splicing factors ELN favorable risk ELN intermediate-1 risk ELN intermediate-2 risk 100 0 FLT 3 NPM 1 DNMT 3 A NRAS ML Complex L fu TET 2 sio -7/7 q n +8/8 q in v( IDH 2 3) CEBPA RUNX 1 t(6 , 9) PTPN 11 -5/5 q IDHI NP TP 53 M 1 SRSF 2 inv(16) MLL CE WT 1 BP RA KRAS bi all -17/17 p eli TP c ASXL 1 53 -a KIT ne STAG 2 Ch up t(15; 17) ro loid sp t(8; 21) lic mat y eo in t(11 q 23; x) so RAD 21 m ID -9 q H 2 R e abn 3 q 17 2 -12/12 p EZH 2 No PHF 6 cla ss -Y No SF 3 B 1 de dri +21 te ve CBL ct ed rs U 2 AF 1 BCOR GATA 2 NF 1 -20/20 q -4/4 q MYC +22 -18/18 q inv(3)/t(3; 3) EP 300 ETV 6 +13 § Complex, diverse disease Unique Pts With Driver Mutation, n § Landscape of Driver Mutations Pts, n 1 5 10 15 > 15 62 166 86% pts have ≥ 2 genetic drivers 199 2 8 6 2 1) 15 ; 2 ) 5; 17 81 t(1 ) 16 Conclusion: AML is complicated! 3 1 418 in v( § 16 66 t(8 § 18 275 20 44 1 11 2 60 1 Papaemmanuil E, et al. N Engl J Med. 2016; 374: 2209 -2221. 3 1 No drivers detected No class IDH 2 R 172 Chromatin-spliceosome TP 53 -aneuploidy CEBPRAbiallelic NPM 1 t(6; 9) Inv(3) MLL fusion t(15; 17) inc(16) Slide credit: clinicaloptions. com
AML: Pathogenesis ELN adverse risk ELN risk not available 20 500 15 400 10 300 5 0 200 +11/11 q t(6; 9) KDM 5 A MLL 2 ZRSR 2 JAK 2 CREBBP KDM 6 A MLL 3 BRAF FBXW 7 ATRX CUX 1 RB 1 MPL PRPF 40 B PTEN CDKN 2 A GNAS MLL 5 SF 1 U 2 AF 2 CBLB IKZF 1 SF 3 A 1 SH 2 B 3 Genetic abnormalities include ultrastructrual changes to chromosomes, gene mutations (eg, DNAMT 3 A, TET 2, FLT 3, NPM 1, IDH 1/2, TP 53), epigenetic changes, and changes in RNA splicing factors ELN favorable risk ELN intermediate-1 risk ELN intermediate-2 risk 100 0 FLT 3 NPM 1 DNMT 3 A NRAS ML Complex L fu TET 2 sio -7/7 q n +8/8 q in v( IDH 2 3) CEBPA RUNX 1 t(6 , 9) PTPN 11 -5/5 q IDHI NP TP 53 M 1 SRSF 2 inv(16) MLL CE WT 1 BP RA KRAS bi all -17/17 p eli TP c ASXL 1 53 -a KIT ne STAG 2 Ch up t(15; 17) ro loid sp t(8; 21) lic mat y eo in t(11 q 23; x) so RAD 21 m ID -9 q H 2 R e abn 3 q 17 2 -12/12 p EZH 2 No PHF 6 cla ss -Y No SF 3 B 1 de dri +21 te ve CBL ct ed rs U 2 AF 1 BCOR GATA 2 NF 1 -20/20 q -4/4 q MYC +22 -18/18 q inv(3)/t(3; 3) EP 300 ETV 6 +13 § Complex, diverse disease Unique Pts With Driver Mutation, n § Landscape of Driver Mutations Pts, n 1 5 10 15 > 15 62 166 86% pts have ≥ 2 genetic drivers 199 2 8 6 2 1) 15 ; 2 ) 5; 17 81 t(1 ) 16 Conclusion: AML is complicated! 3 1 418 in v( § 16 66 t(8 § 18 275 20 44 1 11 2 60 1 Papaemmanuil E, et al. N Engl J Med. 2016; 374: 2209 -2221. 3 1 No drivers detected No class IDH 2 R 172 Chromatin-spliceosome TP 53 -aneuploidy CEBPRAbiallelic NPM 1 t(6; 9) Inv(3) MLL fusion t(15; 17) inc(16) Slide credit: clinicaloptions. com
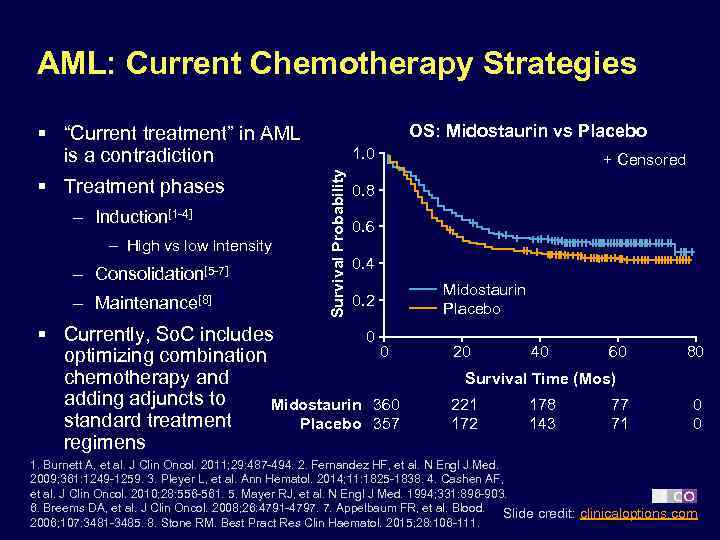 AML: Current Chemotherapy Strategies OS: Midostaurin vs Placebo § “Current treatment” in AML is a contradiction – Induction[1 -4] – High vs low intensity – Consolidation[5 -7] – Maintenance[8] Survival Probability § Treatment phases 1. 0 + Censored 0. 8 0. 6 0. 4 0. 2 § Currently, So. C includes 0 0 optimizing combination chemotherapy and adding adjuncts to Midostaurin 360 standard treatment Placebo 357 regimens Midostaurin Placebo 20 40 60 80 Survival Time (Mos) 221 172 178 143 77 71 0 0 1. Burnett A, et al. J Clin Oncol. 2011; 29: 487 -494. 2. Fernandez HF, et al. N Engl J Med. 2009; 361: 1249 -1259. 3. Pleyer L, et al. Ann Hematol. 2014; 11: 1825 -1838. 4. Cashen AF, et al. J Clin Oncol. 2010; 28: 556 -561. 5. Mayer RJ, et al. N Engl J Med. 1994; 331: 896 -903. 6. Breems DA, et al. J Clin Oncol. 2008; 26: 4791 -4797. 7. Appelbaum FR, et al. Blood. Slide credit: clinicaloptions. com 2006; 107: 3481 -3485. 8. Stone RM. Best Pract Res Clin Haematol. 2015; 28: 106 -111.
AML: Current Chemotherapy Strategies OS: Midostaurin vs Placebo § “Current treatment” in AML is a contradiction – Induction[1 -4] – High vs low intensity – Consolidation[5 -7] – Maintenance[8] Survival Probability § Treatment phases 1. 0 + Censored 0. 8 0. 6 0. 4 0. 2 § Currently, So. C includes 0 0 optimizing combination chemotherapy and adding adjuncts to Midostaurin 360 standard treatment Placebo 357 regimens Midostaurin Placebo 20 40 60 80 Survival Time (Mos) 221 172 178 143 77 71 0 0 1. Burnett A, et al. J Clin Oncol. 2011; 29: 487 -494. 2. Fernandez HF, et al. N Engl J Med. 2009; 361: 1249 -1259. 3. Pleyer L, et al. Ann Hematol. 2014; 11: 1825 -1838. 4. Cashen AF, et al. J Clin Oncol. 2010; 28: 556 -561. 5. Mayer RJ, et al. N Engl J Med. 1994; 331: 896 -903. 6. Breems DA, et al. J Clin Oncol. 2008; 26: 4791 -4797. 7. Appelbaum FR, et al. Blood. Slide credit: clinicaloptions. com 2006; 107: 3481 -3485. 8. Stone RM. Best Pract Res Clin Haematol. 2015; 28: 106 -111.
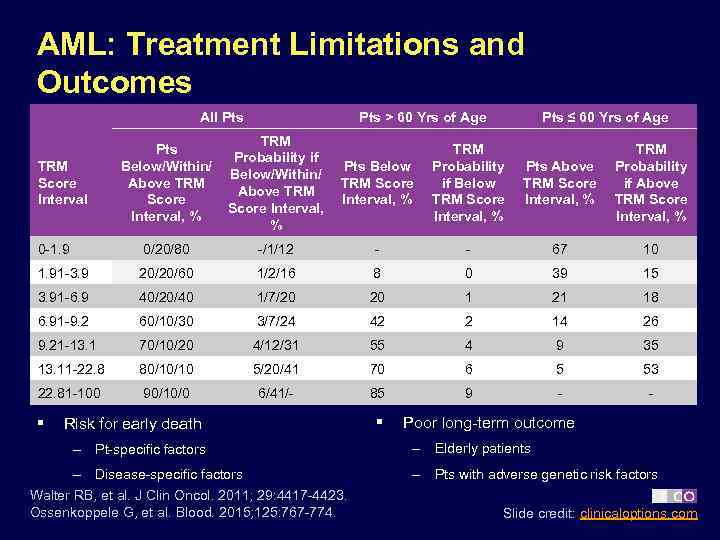 AML: Treatment Limitations and Outcomes All Pts > 60 Yrs of Age Pts Below/Within/ Above TRM Score Interval, % TRM Probability if Below/Within/ Above TRM Score Interval, % 0 -1. 9 0/20/80 1. 91 -3. 9 Pts ≤ 60 Yrs of Age Pts Below TRM Score Interval, % TRM Probability if Below TRM Score Interval, % Pts Above TRM Score Interval, % TRM Probability if Above TRM Score Interval, % -/1/12 - - 67 10 20/20/60 1/2/16 8 0 39 15 3. 91 -6. 9 40/20/40 1/7/20 20 1 21 18 6. 91 -9. 2 60/10/30 3/7/24 42 2 14 26 9. 21 -13. 1 70/10/20 4/12/31 55 4 9 35 13. 11 -22. 8 80/10/10 5/20/41 70 6 5 53 22. 81 -100 90/10/0 6/41/- 85 9 - - TRM Score Interval § Risk for early death – Pt-specific factors – Disease-specific factors Walter RB, et al. J Clin Oncol. 2011; 29: 4417 -4423. Ossenkoppele G, et al. Blood. 2015; 125: 767 -774. § Poor long-term outcome – Elderly patients – Pts with adverse genetic risk factors Slide credit: clinicaloptions. com
AML: Treatment Limitations and Outcomes All Pts > 60 Yrs of Age Pts Below/Within/ Above TRM Score Interval, % TRM Probability if Below/Within/ Above TRM Score Interval, % 0 -1. 9 0/20/80 1. 91 -3. 9 Pts ≤ 60 Yrs of Age Pts Below TRM Score Interval, % TRM Probability if Below TRM Score Interval, % Pts Above TRM Score Interval, % TRM Probability if Above TRM Score Interval, % -/1/12 - - 67 10 20/20/60 1/2/16 8 0 39 15 3. 91 -6. 9 40/20/40 1/7/20 20 1 21 18 6. 91 -9. 2 60/10/30 3/7/24 42 2 14 26 9. 21 -13. 1 70/10/20 4/12/31 55 4 9 35 13. 11 -22. 8 80/10/10 5/20/41 70 6 5 53 22. 81 -100 90/10/0 6/41/- 85 9 - - TRM Score Interval § Risk for early death – Pt-specific factors – Disease-specific factors Walter RB, et al. J Clin Oncol. 2011; 29: 4417 -4423. Ossenkoppele G, et al. Blood. 2015; 125: 767 -774. § Poor long-term outcome – Elderly patients – Pts with adverse genetic risk factors Slide credit: clinicaloptions. com
 Rationale for CD 33 as a Target in AML § CD 33 broadly expressed in AML: 87% to 98% of cases[1, 2] § Function of CD 33 poorly understood § CD 33 internalized upon binding[4] § In some myeloid leukemias, CD 33 thought to be expressed on LSCs[5] – Implicated in cell adhesion and activation – May function as an inhibitory receptor dampening immune response[3] 1. Ehninger A, et al. Blood Cancer J. 2014; 4: e 218. 2. Andrews RG, et al. J Exp Med. 1989: 169: 1721 -1731. 3. Crocker PR, et al. Ann N Y Acad Sci. 2012; 1253: 102 -111. 4. Walter RB, et al. J Leukoc Biol. 2008; 83: 200 -211. 5. Walter RB, et al. Blood. 2012; 119: 6198 -6208. 6. Crocker PR, et al. Nat Rev Immunol. 2007; 7: 255 -266. Slide credit: clinicaloptions. com
Rationale for CD 33 as a Target in AML § CD 33 broadly expressed in AML: 87% to 98% of cases[1, 2] § Function of CD 33 poorly understood § CD 33 internalized upon binding[4] § In some myeloid leukemias, CD 33 thought to be expressed on LSCs[5] – Implicated in cell adhesion and activation – May function as an inhibitory receptor dampening immune response[3] 1. Ehninger A, et al. Blood Cancer J. 2014; 4: e 218. 2. Andrews RG, et al. J Exp Med. 1989: 169: 1721 -1731. 3. Crocker PR, et al. Ann N Y Acad Sci. 2012; 1253: 102 -111. 4. Walter RB, et al. J Leukoc Biol. 2008; 83: 200 -211. 5. Walter RB, et al. Blood. 2012; 119: 6198 -6208. 6. Crocker PR, et al. Nat Rev Immunol. 2007; 7: 255 -266. Slide credit: clinicaloptions. com
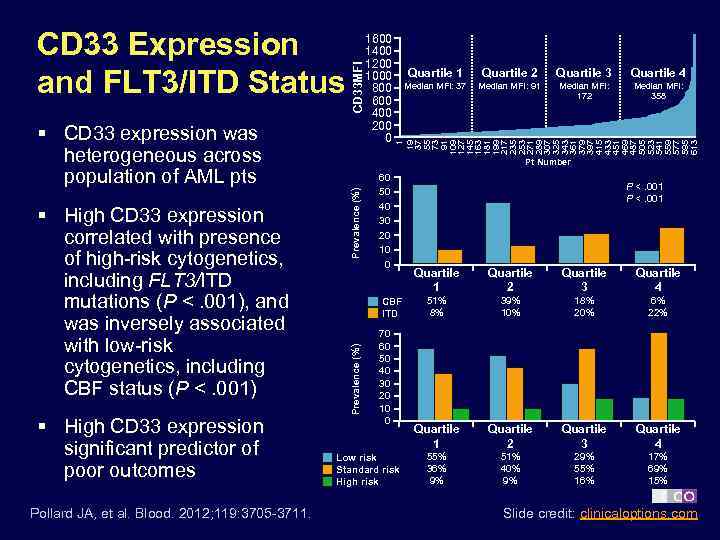 § High CD 33 expression significant predictor of poor outcomes Pollard JA, et al. Blood. 2012; 119: 3705 -3711. Prevalence (%) § High CD 33 expression correlated with presence of high-risk cytogenetics, including FLT 3/ITD mutations (P <. 001), and was inversely associated with low-risk cytogenetics, including CBF status (P <. 001) Quartile 1 Quartile 2 Quartile 3 Quartile 4 Median MFI: 37 Median MFI: 91 Median MFI: 172 Median MFI: 358 1 19 37 55 73 91 109 127 145 163 181 199 217 235 253 271 289 307 325 343 361 379 397 415 433 451 469 487 505 523 541 559 577 595 613 1600 1400 1200 1000 800 600 400 200 0 Pt Number 60 50 40 30 20 10 0 CBF ITD Prevalence (%) § CD 33 expression was heterogeneous across population of AML pts CD 33 MFI CD 33 Expression and FLT 3/ITD Status 70 60 50 40 30 20 10 0 Low risk Standard risk High risk P <. 001 Quartile 2 Quartile 3 Quartile 4 51% 8% 39% 10% 18% 20% 6% 22% Quartile 1 Quartile 2 Quartile 3 Quartile 4 55% 36% 9% 51% 40% 9% 29% 55% 16% 17% 69% 15% Slide credit: clinicaloptions. com
§ High CD 33 expression significant predictor of poor outcomes Pollard JA, et al. Blood. 2012; 119: 3705 -3711. Prevalence (%) § High CD 33 expression correlated with presence of high-risk cytogenetics, including FLT 3/ITD mutations (P <. 001), and was inversely associated with low-risk cytogenetics, including CBF status (P <. 001) Quartile 1 Quartile 2 Quartile 3 Quartile 4 Median MFI: 37 Median MFI: 91 Median MFI: 172 Median MFI: 358 1 19 37 55 73 91 109 127 145 163 181 199 217 235 253 271 289 307 325 343 361 379 397 415 433 451 469 487 505 523 541 559 577 595 613 1600 1400 1200 1000 800 600 400 200 0 Pt Number 60 50 40 30 20 10 0 CBF ITD Prevalence (%) § CD 33 expression was heterogeneous across population of AML pts CD 33 MFI CD 33 Expression and FLT 3/ITD Status 70 60 50 40 30 20 10 0 Low risk Standard risk High risk P <. 001 Quartile 2 Quartile 3 Quartile 4 51% 8% 39% 10% 18% 20% 6% 22% Quartile 1 Quartile 2 Quartile 3 Quartile 4 55% 36% 9% 51% 40% 9% 29% 55% 16% 17% 69% 15% Slide credit: clinicaloptions. com
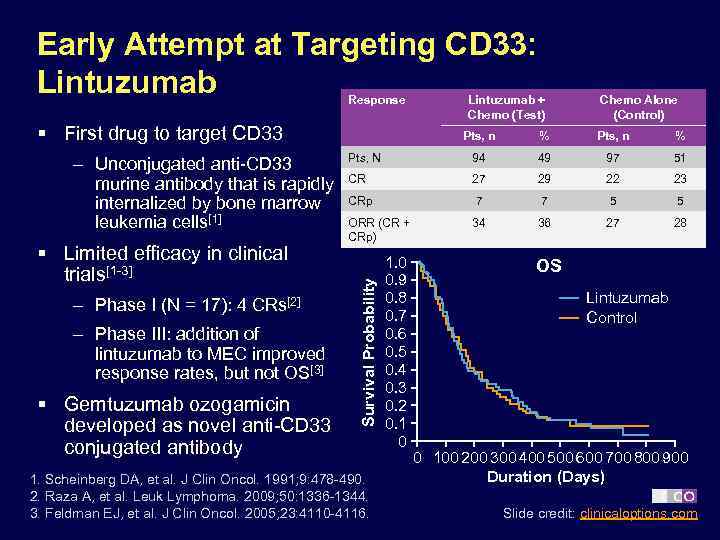 Early Attempt at Targeting CD 33: Lintuzumab Response § First drug to target CD 33 § Limited efficacy in clinical trials[1 -3] – Phase I (N = 17): 4 CRs[2] – Phase III: addition of lintuzumab to MEC improved response rates, but not OS[3] § Gemtuzumab ozogamicin developed as novel anti-CD 33 conjugated antibody Chemo Alone (Control) Pts, n % Pts, N 94 49 97 51 CR 27 29 22 23 CRp 7 7 5 5 ORR (CR + CRp) 34 36 27 28 Survival Probability – Unconjugated anti-CD 33 murine antibody that is rapidly internalized by bone marrow leukemia cells[1] Lintuzumab + Chemo (Test) 1. Scheinberg DA, et al. J Clin Oncol. 1991; 9: 478 -490. 2. Raza A, et al. Leuk Lymphoma. 2009; 50: 1336 -1344. 3. Feldman EJ, et al. J Clin Oncol. 2005; 23: 4110 -4116. 1. 0 0. 9 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0 OS Lintuzumab Control 0 100 200 300 400 500 600 700 800 900 Duration (Days) Slide credit: clinicaloptions. com
Early Attempt at Targeting CD 33: Lintuzumab Response § First drug to target CD 33 § Limited efficacy in clinical trials[1 -3] – Phase I (N = 17): 4 CRs[2] – Phase III: addition of lintuzumab to MEC improved response rates, but not OS[3] § Gemtuzumab ozogamicin developed as novel anti-CD 33 conjugated antibody Chemo Alone (Control) Pts, n % Pts, N 94 49 97 51 CR 27 29 22 23 CRp 7 7 5 5 ORR (CR + CRp) 34 36 27 28 Survival Probability – Unconjugated anti-CD 33 murine antibody that is rapidly internalized by bone marrow leukemia cells[1] Lintuzumab + Chemo (Test) 1. Scheinberg DA, et al. J Clin Oncol. 1991; 9: 478 -490. 2. Raza A, et al. Leuk Lymphoma. 2009; 50: 1336 -1344. 3. Feldman EJ, et al. J Clin Oncol. 2005; 23: 4110 -4116. 1. 0 0. 9 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0 OS Lintuzumab Control 0 100 200 300 400 500 600 700 800 900 Duration (Days) Slide credit: clinicaloptions. com
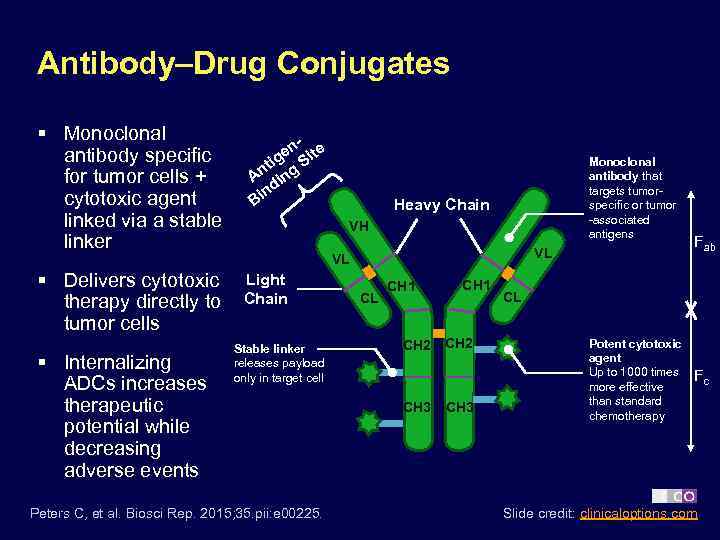 Antibody–Drug Conjugates § Monoclonal antibody specific for tumor cells + cytotoxic agent linked via a stable linker § Delivers cytotoxic therapy directly to tumor cells § Internalizing ADCs increases therapeutic potential while decreasing adverse events en ite ig S nt ng A i d in B Monoclonal antibody that targets tumorspecific or tumor -associated antigens Heavy Chain VH VL VL Light Chain Stable linker releases payload only in target cell Peters C, et al. Biosci Rep. 2015; 35. pii: e 00225. CL CH 1 CH 2 CH 3 Fab CL Potent cytotoxic agent Up to 1000 times more effective than standard chemotherapy Fc Slide credit: clinicaloptions. com
Antibody–Drug Conjugates § Monoclonal antibody specific for tumor cells + cytotoxic agent linked via a stable linker § Delivers cytotoxic therapy directly to tumor cells § Internalizing ADCs increases therapeutic potential while decreasing adverse events en ite ig S nt ng A i d in B Monoclonal antibody that targets tumorspecific or tumor -associated antigens Heavy Chain VH VL VL Light Chain Stable linker releases payload only in target cell Peters C, et al. Biosci Rep. 2015; 35. pii: e 00225. CL CH 1 CH 2 CH 3 Fab CL Potent cytotoxic agent Up to 1000 times more effective than standard chemotherapy Fc Slide credit: clinicaloptions. com
 FDA-Approved ADCs ADC Indication Drug Approval Date Gemtuzumab ozogamicin AML Humanized Ig. G 4 anti-CD 33 antibody + calicheamicin Approved in 2000, withdrawn in 2010 Brentuximab vedotin Relapsed HL and systemic anaplastic large cell lymphoma Chimeric Ig. G 1 anti. CD 30 antibody + MMAE Accelerated approval in 2011 Relapsed or chemotherapy refractory HER 2+ breast cancer Humanized Ig. G 1 anti-HER 2 antibody + DM 1 2013 T-DM 1 § Many others in clinical development – Inotuzumab, denintuzumab, vadastuximab Peters C, et al. Biosci Rep. 2015; 35. pii: e 00225. Slide credit: clinicaloptions. com
FDA-Approved ADCs ADC Indication Drug Approval Date Gemtuzumab ozogamicin AML Humanized Ig. G 4 anti-CD 33 antibody + calicheamicin Approved in 2000, withdrawn in 2010 Brentuximab vedotin Relapsed HL and systemic anaplastic large cell lymphoma Chimeric Ig. G 1 anti. CD 30 antibody + MMAE Accelerated approval in 2011 Relapsed or chemotherapy refractory HER 2+ breast cancer Humanized Ig. G 1 anti-HER 2 antibody + DM 1 2013 T-DM 1 § Many others in clinical development – Inotuzumab, denintuzumab, vadastuximab Peters C, et al. Biosci Rep. 2015; 35. pii: e 00225. Slide credit: clinicaloptions. com
![Gemtuzumab Ozogamicin: MOA § Monoclonal anti-CD 33 antibody linked to calicheamicin-y 1[1] Growth Factors, Gemtuzumab Ozogamicin: MOA § Monoclonal anti-CD 33 antibody linked to calicheamicin-y 1[1] Growth Factors,](https://present5.com/presentation/12886429_438531200/image-13.jpg) Gemtuzumab Ozogamicin: MOA § Monoclonal anti-CD 33 antibody linked to calicheamicin-y 1[1] Growth Factors, Cytokines GO CD 33 Target Levels § Internalized and cleaved in lysosomes to release free calicheamicin moiety[2] § Calicheamicin moiety enters nucleus and interacts with DNA causing double-strand breaks initiating apoptosis[1 -3] Drug Transporter Activity Survival Signaling P 13 K/AKT MEK/ERK MDR-1/MRP-1 JAK/STAT GO-Induced DNA Damage GO Internalization/ Processing Mitochondria p-CHk 2 γH 2 AX GO-Induced Apoptosis and Cell Death c-Casp 3 c-PARP Cell Death Rosen DB, et al. PLo. S One. 2013; 8: e 53518. 1. Zein N, et al. Science. 1988; 240: 1198 -1201. 2. Naito K, et al. Leukemia. 2000; Slide credit: clinicaloptions. com 14: 1436 -1443. 3. Elmroth K, et al. DNA Repair (Amst). 2003; 2: 363 -374.
Gemtuzumab Ozogamicin: MOA § Monoclonal anti-CD 33 antibody linked to calicheamicin-y 1[1] Growth Factors, Cytokines GO CD 33 Target Levels § Internalized and cleaved in lysosomes to release free calicheamicin moiety[2] § Calicheamicin moiety enters nucleus and interacts with DNA causing double-strand breaks initiating apoptosis[1 -3] Drug Transporter Activity Survival Signaling P 13 K/AKT MEK/ERK MDR-1/MRP-1 JAK/STAT GO-Induced DNA Damage GO Internalization/ Processing Mitochondria p-CHk 2 γH 2 AX GO-Induced Apoptosis and Cell Death c-Casp 3 c-PARP Cell Death Rosen DB, et al. PLo. S One. 2013; 8: e 53518. 1. Zein N, et al. Science. 1988; 240: 1198 -1201. 2. Naito K, et al. Leukemia. 2000; Slide credit: clinicaloptions. com 14: 1436 -1443. 3. Elmroth K, et al. DNA Repair (Amst). 2003; 2: 363 -374.
 Gemtuzumab Ozogamicin in AML: Development Overview § Approved for AML in 2000, withdrawn in 2010 – Voluntarily withdrawn from the market after addition of gemtuzumab to frontline chemotherapy shown to increase mortality[1] § Critics of the withdrawal noted increased mortality only slightly higher than normally seen in AML § Subsequently, studies of gemtuzumab in AML have demonstrated survival benefits, particularly in pts younger than 60 yrs of age and favorable-risk pts 1. Petersdorf S, et al. Blood. ASH 2009. Abstract 790. Slide credit: clinicaloptions. com
Gemtuzumab Ozogamicin in AML: Development Overview § Approved for AML in 2000, withdrawn in 2010 – Voluntarily withdrawn from the market after addition of gemtuzumab to frontline chemotherapy shown to increase mortality[1] § Critics of the withdrawal noted increased mortality only slightly higher than normally seen in AML § Subsequently, studies of gemtuzumab in AML have demonstrated survival benefits, particularly in pts younger than 60 yrs of age and favorable-risk pts 1. Petersdorf S, et al. Blood. ASH 2009. Abstract 790. Slide credit: clinicaloptions. com
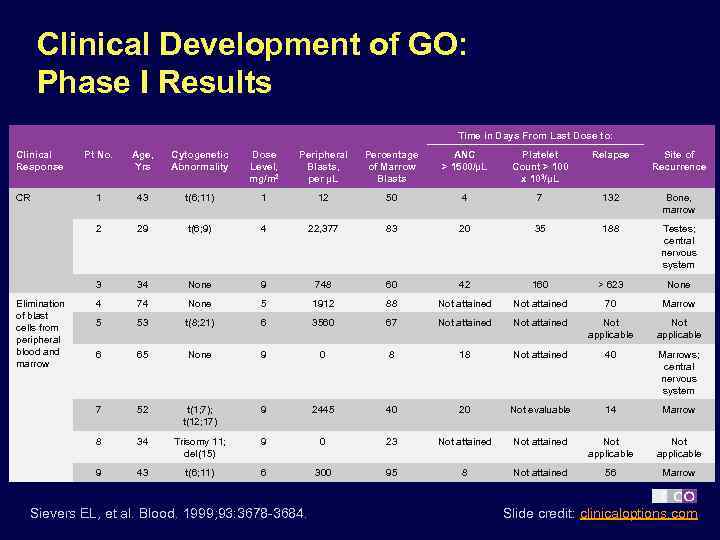 Clinical Development of GO: Phase I Results Time in Days From Last Dose to: Clinical Response Cytogenetic Abnormality Dose Level, mg/m 2 Peripheral Blasts, per µL Percentage of Marrow Blasts ANC > 1500/µL Platelet Count > 100 x 103/µL Relapse Site of Recurrence 1 43 t(6; 11) 1 12 50 4 7 132 Bone, marrow 29 t(6; 9) 4 22, 377 83 20 35 188 Testes; central nervous system 3 34 None 9 748 60 42 160 > 623 None 4 74 None 5 1912 88 Not attained 70 Marrow 5 53 t(8; 21) 6 3560 67 Not attained Not applicable 6 65 None 9 0 8 18 Not attained 40 Marrows; central nervous system 7 52 t(1; 7); t(12; 17) 9 2445 40 20 Not evaluable 14 Marrow 8 34 Trisomy 11; del(15) 9 0 23 Not attained Not applicable 9 Elimination of blast cells from peripheral blood and marrow Age, Yrs 2 CR Pt No. 43 t(6; 11) 6 300 95 8 Not attained 56 Marrow Sievers EL, et al. Blood. 1999; 93: 3678 -3684. Slide credit: clinicaloptions. com
Clinical Development of GO: Phase I Results Time in Days From Last Dose to: Clinical Response Cytogenetic Abnormality Dose Level, mg/m 2 Peripheral Blasts, per µL Percentage of Marrow Blasts ANC > 1500/µL Platelet Count > 100 x 103/µL Relapse Site of Recurrence 1 43 t(6; 11) 1 12 50 4 7 132 Bone, marrow 29 t(6; 9) 4 22, 377 83 20 35 188 Testes; central nervous system 3 34 None 9 748 60 42 160 > 623 None 4 74 None 5 1912 88 Not attained 70 Marrow 5 53 t(8; 21) 6 3560 67 Not attained Not applicable 6 65 None 9 0 8 18 Not attained 40 Marrows; central nervous system 7 52 t(1; 7); t(12; 17) 9 2445 40 20 Not evaluable 14 Marrow 8 34 Trisomy 11; del(15) 9 0 23 Not attained Not applicable 9 Elimination of blast cells from peripheral blood and marrow Age, Yrs 2 CR Pt No. 43 t(6; 11) 6 300 95 8 Not attained 56 Marrow Sievers EL, et al. Blood. 1999; 93: 3678 -3684. Slide credit: clinicaloptions. com
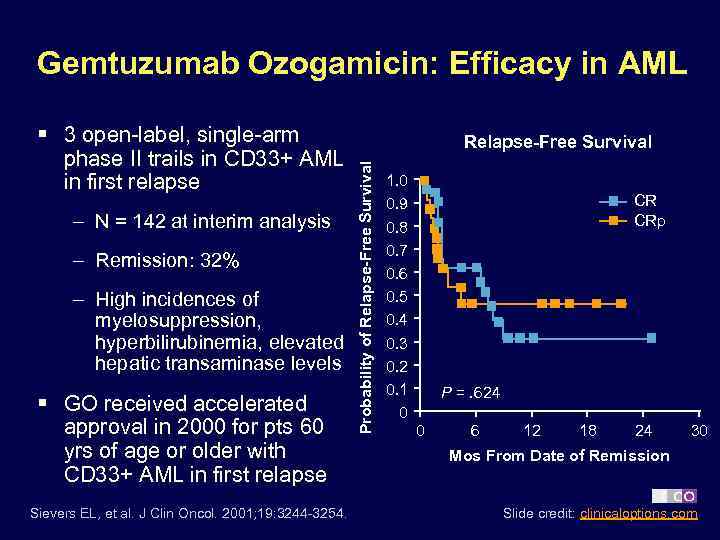 Gemtuzumab Ozogamicin: Efficacy in AML – N = 142 at interim analysis – Remission: 32% – High incidences of myelosuppression, hyperbilirubinemia, elevated hepatic transaminase levels § GO received accelerated approval in 2000 for pts 60 yrs of age or older with CD 33+ AML in first relapse Sievers EL, et al. J Clin Oncol. 2001; 19: 3244 -3254. Relapse-Free Survival Probability of Relapse-Free Survival § 3 open-label, single-arm phase II trails in CD 33+ AML in first relapse 1. 0 0. 9 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0 CR CRp P =. 624 0 6 12 18 24 30 Mos From Date of Remission Slide credit: clinicaloptions. com
Gemtuzumab Ozogamicin: Efficacy in AML – N = 142 at interim analysis – Remission: 32% – High incidences of myelosuppression, hyperbilirubinemia, elevated hepatic transaminase levels § GO received accelerated approval in 2000 for pts 60 yrs of age or older with CD 33+ AML in first relapse Sievers EL, et al. J Clin Oncol. 2001; 19: 3244 -3254. Relapse-Free Survival Probability of Relapse-Free Survival § 3 open-label, single-arm phase II trails in CD 33+ AML in first relapse 1. 0 0. 9 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0 CR CRp P =. 624 0 6 12 18 24 30 Mos From Date of Remission Slide credit: clinicaloptions. com
![Gemtuzumab Ozogamicin in AML: Phase III Results Study[1] N Treatment Results of GO vs Gemtuzumab Ozogamicin in AML: Phase III Results Study[1] N Treatment Results of GO vs](https://present5.com/presentation/12886429_438531200/image-17.jpg) Gemtuzumab Ozogamicin in AML: Phase III Results Study[1] N Treatment Results of GO vs Comparator § Improved 5 -yr OS for favorable risk group § No difference in ORR, TRM, relapse, survival MRC/NCRI AML 15[2] 1113 GO (3 mg/m 2) + either ADE, DA, or FLAG-IDA ALFA 0701[3] 280 GO (3 mg/m 2) + DA § Improved 2 -yr EFS, RFS, OS § No difference in ORR or mortality 238 GO (6 mg/m 2) + DA induction and MA consolidation § Improved EFS in pts who did not have allogeneic HCT § No difference in OS, ORR, TRM, 3 yr EFS 1115 GO (3 mg/m 2) + either DA or DCLo 595 GO (6 mg/m 2) + DA GOELAMS AML 2006 IR[4] MRC/NCRI AML SWOG S 0106[6] 16[5] § Reduced 3 -yr relapse risk, and superior DFS and OS § No difference in TRM § Increased TRM § No difference in ORR, DFS, or OS 1. Cowan AJ, et al. Front Biosci (Landmark Ed. ) 2013; 18: 1311 -1334. 2. Burnett AK, et al. J Clin Oncol. 2011; 29: 369 -377. 3. Castaigne S, et al. Lancet. 2012; 379: 1508 -1516. 4. Delaunay J, et al. ASH 2011. Abstract 79. 5. Burnett AK, et al. J Clin Oncol. Slide credit: clinicaloptions. com 2012; 30: 3924 -3931. 6. Petersdorf S, et al. Blood. 2013; 121: 4854 -4860.
Gemtuzumab Ozogamicin in AML: Phase III Results Study[1] N Treatment Results of GO vs Comparator § Improved 5 -yr OS for favorable risk group § No difference in ORR, TRM, relapse, survival MRC/NCRI AML 15[2] 1113 GO (3 mg/m 2) + either ADE, DA, or FLAG-IDA ALFA 0701[3] 280 GO (3 mg/m 2) + DA § Improved 2 -yr EFS, RFS, OS § No difference in ORR or mortality 238 GO (6 mg/m 2) + DA induction and MA consolidation § Improved EFS in pts who did not have allogeneic HCT § No difference in OS, ORR, TRM, 3 yr EFS 1115 GO (3 mg/m 2) + either DA or DCLo 595 GO (6 mg/m 2) + DA GOELAMS AML 2006 IR[4] MRC/NCRI AML SWOG S 0106[6] 16[5] § Reduced 3 -yr relapse risk, and superior DFS and OS § No difference in TRM § Increased TRM § No difference in ORR, DFS, or OS 1. Cowan AJ, et al. Front Biosci (Landmark Ed. ) 2013; 18: 1311 -1334. 2. Burnett AK, et al. J Clin Oncol. 2011; 29: 369 -377. 3. Castaigne S, et al. Lancet. 2012; 379: 1508 -1516. 4. Delaunay J, et al. ASH 2011. Abstract 79. 5. Burnett AK, et al. J Clin Oncol. Slide credit: clinicaloptions. com 2012; 30: 3924 -3931. 6. Petersdorf S, et al. Blood. 2013; 121: 4854 -4860.
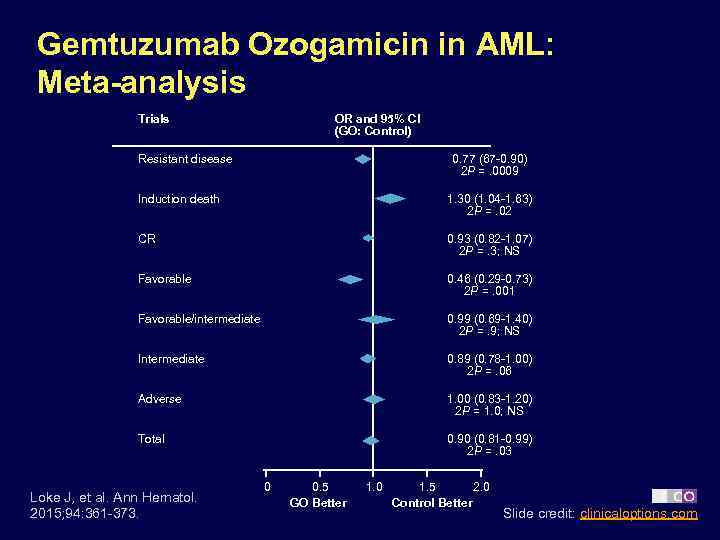 Gemtuzumab Ozogamicin in AML: Meta-analysis Trials OR and 95% CI (GO: Control) Resistant disease 0. 77 (67 -0. 90) 2 P =. 0009 Induction death 1. 30 (1. 04 -1. 63) 2 P =. 02 CR 0. 93 (0. 82 -1. 07) 2 P =. 3; NS Favorable 0. 46 (0. 29 -0. 73) 2 P =. 001 Favorable/intermediate 0. 99 (0. 69 -1. 40) 2 P =. 9; NS Intermediate 0. 89 (0. 78 -1. 00) 2 P =. 06 Adverse 1. 00 (0. 83 -1. 20) 2 P = 1. 0; NS Total 0. 90 (0. 81 -0. 99) 2 P =. 03 Loke J, et al. Ann Hematol. 2015; 94: 361 -373. 0 0. 5 GO Better 1. 0 1. 5 2. 0 Control Better Slide credit: clinicaloptions. com
Gemtuzumab Ozogamicin in AML: Meta-analysis Trials OR and 95% CI (GO: Control) Resistant disease 0. 77 (67 -0. 90) 2 P =. 0009 Induction death 1. 30 (1. 04 -1. 63) 2 P =. 02 CR 0. 93 (0. 82 -1. 07) 2 P =. 3; NS Favorable 0. 46 (0. 29 -0. 73) 2 P =. 001 Favorable/intermediate 0. 99 (0. 69 -1. 40) 2 P =. 9; NS Intermediate 0. 89 (0. 78 -1. 00) 2 P =. 06 Adverse 1. 00 (0. 83 -1. 20) 2 P = 1. 0; NS Total 0. 90 (0. 81 -0. 99) 2 P =. 03 Loke J, et al. Ann Hematol. 2015; 94: 361 -373. 0 0. 5 GO Better 1. 0 1. 5 2. 0 Control Better Slide credit: clinicaloptions. com
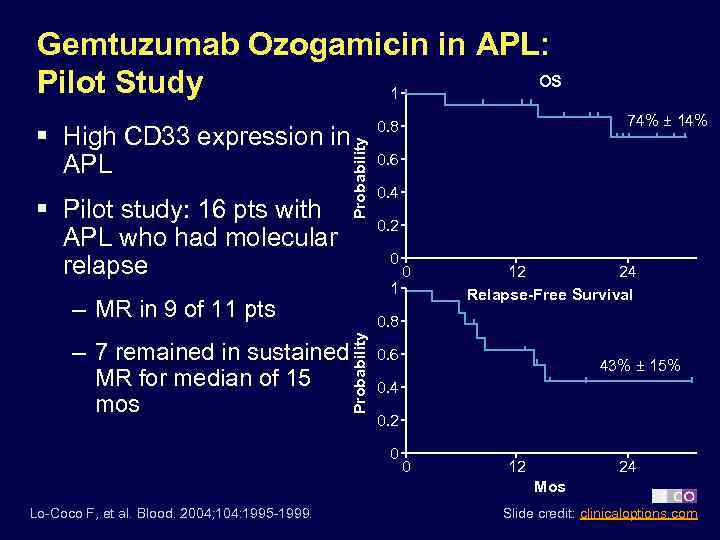 Gemtuzumab Ozogamicin in APL: OS Pilot Study 1 § Pilot study: 16 pts with APL who had molecular relapse Probability § High CD 33 expression in APL 0. 4 0. 2 1 0 12 24 Relapse-Free Survival 0. 8 Probability – 7 remained in sustained MR for median of 15 mos 0. 6 0 – MR in 9 of 11 pts 74% ± 14% 0. 8 0. 6 43% ± 15% 0. 4 0. 2 0 0 12 24 Mos Lo-Coco F, et al. Blood. 2004; 104: 1995 -1999. Slide credit: clinicaloptions. com
Gemtuzumab Ozogamicin in APL: OS Pilot Study 1 § Pilot study: 16 pts with APL who had molecular relapse Probability § High CD 33 expression in APL 0. 4 0. 2 1 0 12 24 Relapse-Free Survival 0. 8 Probability – 7 remained in sustained MR for median of 15 mos 0. 6 0 – MR in 9 of 11 pts 74% ± 14% 0. 8 0. 6 43% ± 15% 0. 4 0. 2 0 0 12 24 Mos Lo-Coco F, et al. Blood. 2004; 104: 1995 -1999. Slide credit: clinicaloptions. com
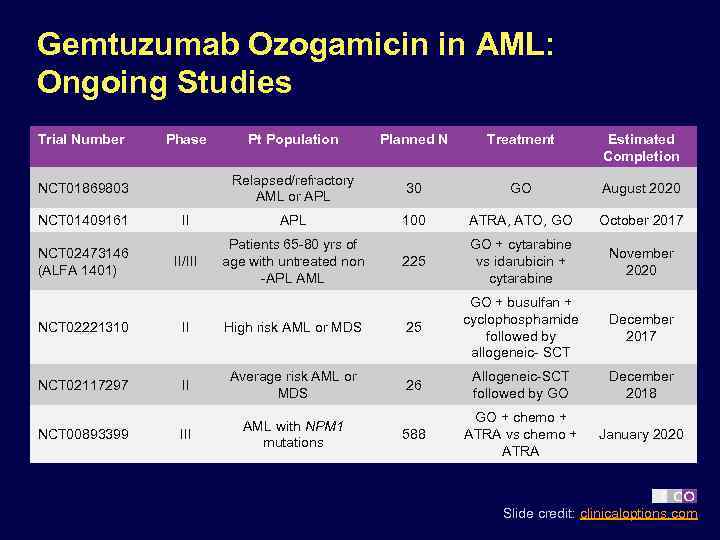 Gemtuzumab Ozogamicin in AML: Ongoing Studies Trial Number Phase Pt Population Planned N Treatment Estimated Completion Relapsed/refractory AML or APL 30 GO August 2020 II APL 100 ATRA, ATO, GO October 2017 II/III Patients 65 -80 yrs of age with untreated non -APL AML 225 GO + cytarabine vs idarubicin + cytarabine November 2020 December 2017 NCT 01869803 NCT 01409161 NCT 02473146 (ALFA 1401) NCT 02221310 II High risk AML or MDS 25 GO + busulfan + cyclophosphamide followed by allogeneic- SCT NCT 02117297 II Average risk AML or MDS 26 Allogeneic-SCT followed by GO December 2018 III AML with NPM 1 mutations 588 GO + chemo + ATRA vs chemo + ATRA January 2020 NCT 00893399 Slide credit: clinicaloptions. com
Gemtuzumab Ozogamicin in AML: Ongoing Studies Trial Number Phase Pt Population Planned N Treatment Estimated Completion Relapsed/refractory AML or APL 30 GO August 2020 II APL 100 ATRA, ATO, GO October 2017 II/III Patients 65 -80 yrs of age with untreated non -APL AML 225 GO + cytarabine vs idarubicin + cytarabine November 2020 December 2017 NCT 01869803 NCT 01409161 NCT 02473146 (ALFA 1401) NCT 02221310 II High risk AML or MDS 25 GO + busulfan + cyclophosphamide followed by allogeneic- SCT NCT 02117297 II Average risk AML or MDS 26 Allogeneic-SCT followed by GO December 2018 III AML with NPM 1 mutations 588 GO + chemo + ATRA vs chemo + ATRA January 2020 NCT 00893399 Slide credit: clinicaloptions. com
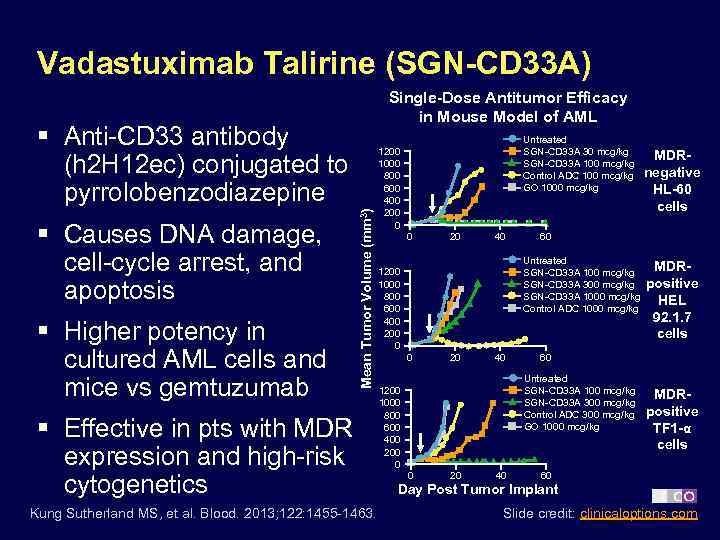 Vadastuximab Talirine (SGN-CD 33 A) Single-Dose Antitumor Efficacy in Mouse Model of AML § Causes DNA damage, cell-cycle arrest, and apoptosis § Higher potency in cultured AML cells and mice vs gemtuzumab Mean Tumor Volume (mm 3) § Anti-CD 33 antibody (h 2 H 12 ec) conjugated to pyrrolobenzodiazepine § Effective in pts with MDR expression and high-risk cytogenetics Kung Sutherland MS, et al. Blood. 2013; 122: 1455 -1463. Untreated SGN-CD 33 A 30 mcg/kg SGN-CD 33 A 100 mcg/kg Control ADC 100 mcg/kg GO 1000 mcg/kg 1200 1000 800 600 400 200 0 0 20 40 60 Untreated SGN-CD 33 A 100 mcg/kg SGN-CD 33 A 300 mcg/kg SGN-CD 33 A 1000 mcg/kg Control ADC 1000 mcg/kg 1200 1000 800 600 400 200 0 0 20 40 MDRpositive HEL 92. 1. 7 cells 60 Untreated SGN-CD 33 A 100 mcg/kg SGN-CD 33 A 300 mcg/kg Control ADC 300 mcg/kg GO 1000 mcg/kg 1200 1000 800 600 400 200 0 MDRnegative HL-60 cells MDRpositive TF 1 -α cells 60 Day Post Tumor Implant Slide credit: clinicaloptions. com
Vadastuximab Talirine (SGN-CD 33 A) Single-Dose Antitumor Efficacy in Mouse Model of AML § Causes DNA damage, cell-cycle arrest, and apoptosis § Higher potency in cultured AML cells and mice vs gemtuzumab Mean Tumor Volume (mm 3) § Anti-CD 33 antibody (h 2 H 12 ec) conjugated to pyrrolobenzodiazepine § Effective in pts with MDR expression and high-risk cytogenetics Kung Sutherland MS, et al. Blood. 2013; 122: 1455 -1463. Untreated SGN-CD 33 A 30 mcg/kg SGN-CD 33 A 100 mcg/kg Control ADC 100 mcg/kg GO 1000 mcg/kg 1200 1000 800 600 400 200 0 0 20 40 60 Untreated SGN-CD 33 A 100 mcg/kg SGN-CD 33 A 300 mcg/kg SGN-CD 33 A 1000 mcg/kg Control ADC 1000 mcg/kg 1200 1000 800 600 400 200 0 0 20 40 MDRpositive HEL 92. 1. 7 cells 60 Untreated SGN-CD 33 A 100 mcg/kg SGN-CD 33 A 300 mcg/kg Control ADC 300 mcg/kg GO 1000 mcg/kg 1200 1000 800 600 400 200 0 MDRnegative HL-60 cells MDRpositive TF 1 -α cells 60 Day Post Tumor Implant Slide credit: clinicaloptions. com
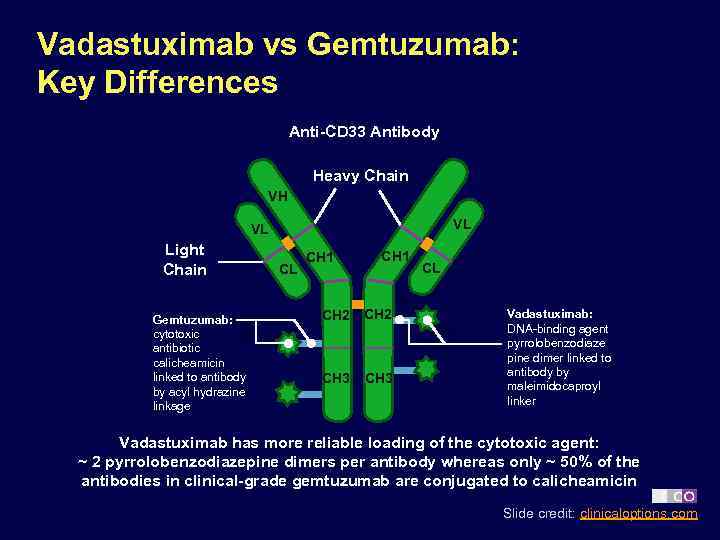 Vadastuximab vs Gemtuzumab: Key Differences Anti-CD 33 Antibody Heavy Chain VH VL VL Light Chain Gemtuzumab: cytotoxic antibiotic calicheamicin linked to antibody by acyl hydrazine linkage CL CH 1 CH 2 CH 3 CL Vadastuximab: DNA-binding agent pyrrolobenzodiaze pine dimer linked to antibody by maleimidocaproyl linker Vadastuximab has more reliable loading of the cytotoxic agent: ~ 2 pyrrolobenzodiazepine dimers per antibody whereas only ~ 50% of the antibodies in clinical-grade gemtuzumab are conjugated to calicheamicin Slide credit: clinicaloptions. com
Vadastuximab vs Gemtuzumab: Key Differences Anti-CD 33 Antibody Heavy Chain VH VL VL Light Chain Gemtuzumab: cytotoxic antibiotic calicheamicin linked to antibody by acyl hydrazine linkage CL CH 1 CH 2 CH 3 CL Vadastuximab: DNA-binding agent pyrrolobenzodiaze pine dimer linked to antibody by maleimidocaproyl linker Vadastuximab has more reliable loading of the cytotoxic agent: ~ 2 pyrrolobenzodiazepine dimers per antibody whereas only ~ 50% of the antibodies in clinical-grade gemtuzumab are conjugated to calicheamicin Slide credit: clinicaloptions. com
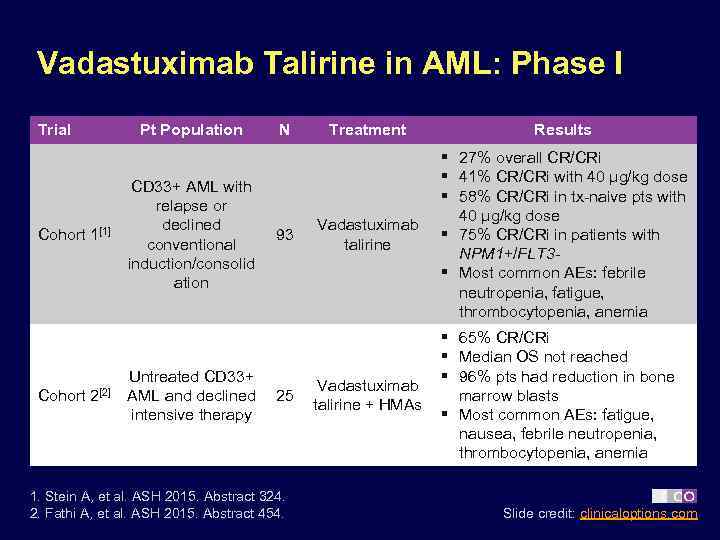 Vadastuximab Talirine in AML: Phase I Trial Pt Population § Preliminary results N Cohort 1[1] Cohort 2[2] CD 33+ AML with relapse or declined conventional induction/consolid ation Untreated CD 33+ AML and declined intensive therapy 93 25 1. Stein A, et al. ASH 2015. Abstract 324. 2. Fathi A, et al. ASH 2015. Abstract 454. Treatment Results Vadastuximab talirine § 27% overall CR/CRi § 41% CR/CRi with 40 µg/kg dose § 58% CR/CRi in tx-naive pts with 40 µg/kg dose § 75% CR/CRi in patients with NPM 1+/FLT 3§ Most common AEs: febrile neutropenia, fatigue, thrombocytopenia, anemia Vadastuximab talirine + HMAs § 65% CR/CRi § Median OS not reached § 96% pts had reduction in bone marrow blasts § Most common AEs: fatigue, nausea, febrile neutropenia, thrombocytopenia, anemia Slide credit: clinicaloptions. com
Vadastuximab Talirine in AML: Phase I Trial Pt Population § Preliminary results N Cohort 1[1] Cohort 2[2] CD 33+ AML with relapse or declined conventional induction/consolid ation Untreated CD 33+ AML and declined intensive therapy 93 25 1. Stein A, et al. ASH 2015. Abstract 324. 2. Fathi A, et al. ASH 2015. Abstract 454. Treatment Results Vadastuximab talirine § 27% overall CR/CRi § 41% CR/CRi with 40 µg/kg dose § 58% CR/CRi in tx-naive pts with 40 µg/kg dose § 75% CR/CRi in patients with NPM 1+/FLT 3§ Most common AEs: febrile neutropenia, fatigue, thrombocytopenia, anemia Vadastuximab talirine + HMAs § 65% CR/CRi § Median OS not reached § 96% pts had reduction in bone marrow blasts § Most common AEs: fatigue, nausea, febrile neutropenia, thrombocytopenia, anemia Slide credit: clinicaloptions. com
 Identifying Patients Who Are Candidates for Current Clinical Trials of CD 33 -Directed Therapy
Identifying Patients Who Are Candidates for Current Clinical Trials of CD 33 -Directed Therapy
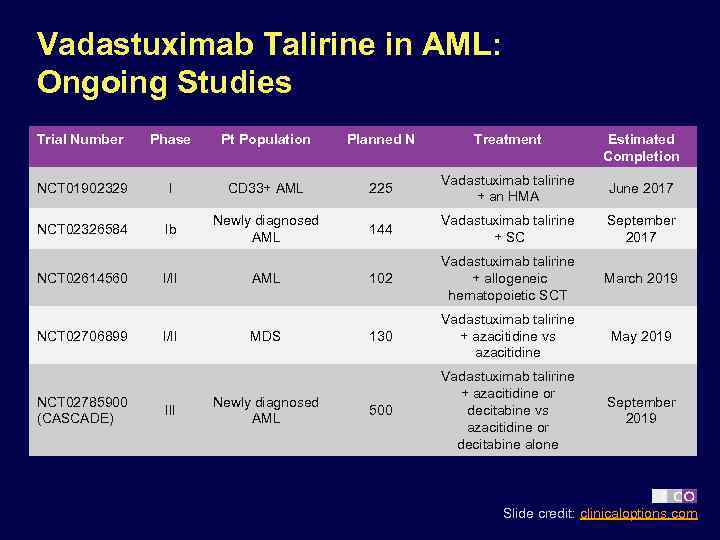 Vadastuximab Talirine in AML: Ongoing Studies Trial Number Phase Pt Population Planned N Treatment NCT 01902329 I CD 33+ AML 225 Vadastuximab talirine + an HMA June 2017 NCT 02326584 Ib Newly diagnosed AML 144 Vadastuximab talirine + SC September 2017 102 Vadastuximab talirine + allogeneic hematopoietic SCT March 2019 130 Vadastuximab talirine + azacitidine vs azacitidine May 2019 500 Vadastuximab talirine + azacitidine or decitabine vs azacitidine or decitabine alone September 2019 NCT 02614560 NCT 02706899 NCT 02785900 (CASCADE) I/II III AML MDS Newly diagnosed AML Estimated Completion Slide credit: clinicaloptions. com
Vadastuximab Talirine in AML: Ongoing Studies Trial Number Phase Pt Population Planned N Treatment NCT 01902329 I CD 33+ AML 225 Vadastuximab talirine + an HMA June 2017 NCT 02326584 Ib Newly diagnosed AML 144 Vadastuximab talirine + SC September 2017 102 Vadastuximab talirine + allogeneic hematopoietic SCT March 2019 130 Vadastuximab talirine + azacitidine vs azacitidine May 2019 500 Vadastuximab talirine + azacitidine or decitabine vs azacitidine or decitabine alone September 2019 NCT 02614560 NCT 02706899 NCT 02785900 (CASCADE) I/II III AML MDS Newly diagnosed AML Estimated Completion Slide credit: clinicaloptions. com
 Phase Ib Study: Vadastuximab Talirine + So. C in Newly Diagnosed AML § Dose-escalation study – Nonrandomized, open label – 4 treatment arms: induction, consolidation, maintenance, induction plus consolidation § Eligibility: all AML subtypes (excluding APL, aka M 3) – ECOG PS 0/1, no prior treatment for MDS or MPN (dose escalation cohorts) § Primary endpoint: safety (AEs, lab abnormalities, DLTs) § Secondary outcomes: CR after induction, LFS, OS Clinical. Trials. gov. NCT 02326584. Slide credit: clinicaloptions. com
Phase Ib Study: Vadastuximab Talirine + So. C in Newly Diagnosed AML § Dose-escalation study – Nonrandomized, open label – 4 treatment arms: induction, consolidation, maintenance, induction plus consolidation § Eligibility: all AML subtypes (excluding APL, aka M 3) – ECOG PS 0/1, no prior treatment for MDS or MPN (dose escalation cohorts) § Primary endpoint: safety (AEs, lab abnormalities, DLTs) § Secondary outcomes: CR after induction, LFS, OS Clinical. Trials. gov. NCT 02326584. Slide credit: clinicaloptions. com
 Phase I/II Study: Vadastuximab Talirine Before or After Allogeneic SCT in AML § Nonrandomized, open-label study – 2 treatment arms: pre- and posttransplant § Eligibility: relapsed/refractory AML (excluding APL) – Arm A: relapsed AML, available HLA MRD or MUD, eligible for allogeneic SCT – Arm B: transplanted with active AML and achieved CR/CRi posttransplant, 60 -100 days posttransplant, no grade ≥ 2 GVHD, no SOS § Primary outcomes: safety, 1 -yr OS Clinical. Trials. gov. NCT 02614560. Slide credit: clinicaloptions. com
Phase I/II Study: Vadastuximab Talirine Before or After Allogeneic SCT in AML § Nonrandomized, open-label study – 2 treatment arms: pre- and posttransplant § Eligibility: relapsed/refractory AML (excluding APL) – Arm A: relapsed AML, available HLA MRD or MUD, eligible for allogeneic SCT – Arm B: transplanted with active AML and achieved CR/CRi posttransplant, 60 -100 days posttransplant, no grade ≥ 2 GVHD, no SOS § Primary outcomes: safety, 1 -yr OS Clinical. Trials. gov. NCT 02614560. Slide credit: clinicaloptions. com
 Phase I/II Study: Vadastuximab Talirine + Azacitidine in Untreated, Higher-Risk MDS § Randomized, double-blind, placebo-controlled trial – Treatment arms – Azacitidine + vadastuximab talirine – Azacitidine + placebo § Eligibility: untreated IPSS int-2 or high-risk MDS § Primary outcomes – Phase I: safety, recommended phase II dose – Phase II: ORR § Secondary outcomes: safety (AEs, laboratory abnormalities) Clinical. Trials. gov. NCT 02706899. Slide credit: clinicaloptions. com
Phase I/II Study: Vadastuximab Talirine + Azacitidine in Untreated, Higher-Risk MDS § Randomized, double-blind, placebo-controlled trial – Treatment arms – Azacitidine + vadastuximab talirine – Azacitidine + placebo § Eligibility: untreated IPSS int-2 or high-risk MDS § Primary outcomes – Phase I: safety, recommended phase II dose – Phase II: ORR § Secondary outcomes: safety (AEs, laboratory abnormalities) Clinical. Trials. gov. NCT 02706899. Slide credit: clinicaloptions. com
 Phase III Trial: Vadastuximab Talirine + HMAs in Older, Newly Diagnosed AML § Randomized, double-blind, placebo-controlled CASCADE study – Treatment arms – Vadastuximab talirine + azacitidine – Vadastuximab talirine + decitabine § Eligibility: newly diagnosed, untreated, de novo, or secondary AML (excluding APL) § Primary outcome: OS § Secondary outcomes: CR, EFS, LFS, remission duration, AEs, time to response, mortality at Days 30 and 60, MRD Clinical. Trials. gov. NCT 02785900. Slide credit: clinicaloptions. com
Phase III Trial: Vadastuximab Talirine + HMAs in Older, Newly Diagnosed AML § Randomized, double-blind, placebo-controlled CASCADE study – Treatment arms – Vadastuximab talirine + azacitidine – Vadastuximab talirine + decitabine § Eligibility: newly diagnosed, untreated, de novo, or secondary AML (excluding APL) § Primary outcome: OS § Secondary outcomes: CR, EFS, LFS, remission duration, AEs, time to response, mortality at Days 30 and 60, MRD Clinical. Trials. gov. NCT 02785900. Slide credit: clinicaloptions. com
 Summary § AML is the most common acute leukemia in adults § Genetically, AML is a complex, diverse disease § Despite its diversity, the standard treatment is uniform and unchanged for many yrs § CD 33 is expressed on the majority of AML cells and a prime target for therapeutic exploitation § Early attempts at targeting CD 33 have resulted in mixed results § Newer CD 33 -targeted agents have enhanced potency and are being developed diversely in myeloid stem cell disorders Slide credit: clinicaloptions. com
Summary § AML is the most common acute leukemia in adults § Genetically, AML is a complex, diverse disease § Despite its diversity, the standard treatment is uniform and unchanged for many yrs § CD 33 is expressed on the majority of AML cells and a prime target for therapeutic exploitation § Early attempts at targeting CD 33 have resulted in mixed results § Newer CD 33 -targeted agents have enhanced potency and are being developed diversely in myeloid stem cell disorders Slide credit: clinicaloptions. com
 Go Online for More CCO Coverage of CD 33 -Targeted Agents! Expert-Authored Interactive Module clinicaloptions. com/oncology
Go Online for More CCO Coverage of CD 33 -Targeted Agents! Expert-Authored Interactive Module clinicaloptions. com/oncology


