2ae016b571b3dacb11e2513def5a106d.ppt
- Количество слайдов: 19
 Synthesis of Propionic acid from Syngas Date: 25 January 2011 Team Echo: Sabah Basrawi Alex Guerrero Sarah Hanif Mrunal Patel Kevin Thompson Client Mentor: Shannon Brown
Synthesis of Propionic acid from Syngas Date: 25 January 2011 Team Echo: Sabah Basrawi Alex Guerrero Sarah Hanif Mrunal Patel Kevin Thompson Client Mentor: Shannon Brown
 Project Outline 1. Design Basis 2. Block Flow Diagram 3. Process Flow Diagram 4. Material and Energy Balance 5. Calculations 6. Annotated Equipment List 7. Economic Evaluation 8. Utilities
Project Outline 1. Design Basis 2. Block Flow Diagram 3. Process Flow Diagram 4. Material and Energy Balance 5. Calculations 6. Annotated Equipment List 7. Economic Evaluation 8. Utilities
 Project Outline Pt. 2 9. Conceptual Control Scheme 10. General Arrangement 11. Distribution and End-use Issues Review 12. Constraints Review 13. Applicable Standards 14. Project Communications File 15. Information Sources and References
Project Outline Pt. 2 9. Conceptual Control Scheme 10. General Arrangement 11. Distribution and End-use Issues Review 12. Constraints Review 13. Applicable Standards 14. Project Communications File 15. Information Sources and References
 Focal Points Design Basis Competing Processes Block Flow Diagram
Focal Points Design Basis Competing Processes Block Flow Diagram
 Project Goal Synthesize 30, 000 ton/year of Propionic Acid from syngas retrieved from group “Foxtrot. ” Split syngas feed to synthesize Ethylene needed to carry out reaction. The reactant will react with a second syngas feed to create a propionic acid precursor. Precursor undergoes oxidation to yield propionic acid The product can either be sold or used to create plastics, preservatives or painkillers.
Project Goal Synthesize 30, 000 ton/year of Propionic Acid from syngas retrieved from group “Foxtrot. ” Split syngas feed to synthesize Ethylene needed to carry out reaction. The reactant will react with a second syngas feed to create a propionic acid precursor. Precursor undergoes oxidation to yield propionic acid The product can either be sold or used to create plastics, preservatives or painkillers.
 Design Basis Propionic acid is a commonly used chemical in industry that will be synthesized from syngas, ethylene and water catalyzed by metal ion in solution Propionic acid facts: Naturally occurring Made from ethylene and carbon monoxide Analgesic Intermediate product for thermoplastic Plastics used in daily processes; will increase affordability and stimulate economy
Design Basis Propionic acid is a commonly used chemical in industry that will be synthesized from syngas, ethylene and water catalyzed by metal ion in solution Propionic acid facts: Naturally occurring Made from ethylene and carbon monoxide Analgesic Intermediate product for thermoplastic Plastics used in daily processes; will increase affordability and stimulate economy
 Environmental Review Industrially, the majority of propionic acid is used as a bactericide and fungicide to protect hay and grains that are being stored as well as an ingredient for pesticides (EPA). These uses have, according to the EPA, “low toxicity to fish, invertebrates, birds and mammals, ” as well as to non-target organisms and is not expected to result in any adverse health effects.
Environmental Review Industrially, the majority of propionic acid is used as a bactericide and fungicide to protect hay and grains that are being stored as well as an ingredient for pesticides (EPA). These uses have, according to the EPA, “low toxicity to fish, invertebrates, birds and mammals, ” as well as to non-target organisms and is not expected to result in any adverse health effects.
 Competing Processes Coal based syngas Major feed Abundant Ethylene Cheap to buy, but making will allow more independence from the market. Wacker Process Similar to hydroformylation Hydroformylation promotes chain extension and uses Rh based catalyst. Wacker process uses a Tetrachloropalladate catalyst
Competing Processes Coal based syngas Major feed Abundant Ethylene Cheap to buy, but making will allow more independence from the market. Wacker Process Similar to hydroformylation Hydroformylation promotes chain extension and uses Rh based catalyst. Wacker process uses a Tetrachloropalladate catalyst
 Competing Processes Pt. 2 Syngas is chosen as the feed for the process because it is the most thermodynamically efficient Rhodium is the most commonly used catalyst in hydroformylation While Rh was considered for this process due its high poison tolerance it is very expensive Aerobic Oxidation is what we have chosen to oxidize propionate. Other methods include: Larson Process: Ethylene, CO and steam Reppe Process: Ethanol. CO, BF 3 Fischer-Tropsch Process: Byproduct of the reaction Pyrolysis: Byproduct of the reaction
Competing Processes Pt. 2 Syngas is chosen as the feed for the process because it is the most thermodynamically efficient Rhodium is the most commonly used catalyst in hydroformylation While Rh was considered for this process due its high poison tolerance it is very expensive Aerobic Oxidation is what we have chosen to oxidize propionate. Other methods include: Larson Process: Ethylene, CO and steam Reppe Process: Ethanol. CO, BF 3 Fischer-Tropsch Process: Byproduct of the reaction Pyrolysis: Byproduct of the reaction
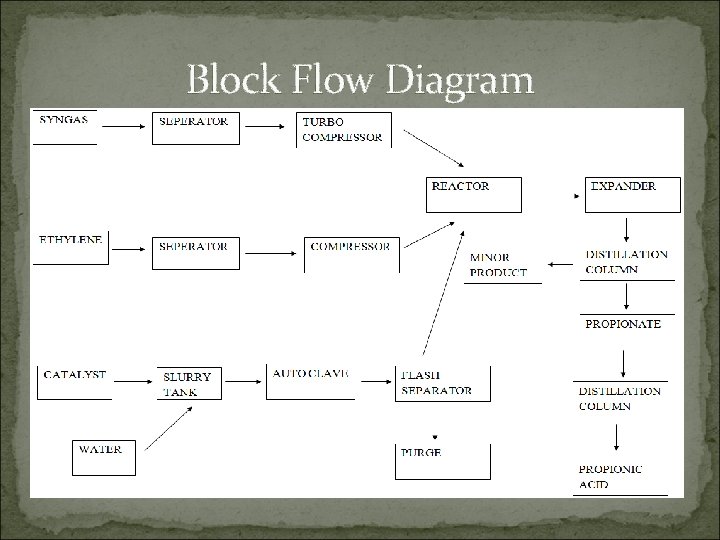 Block Flow Diagram
Block Flow Diagram
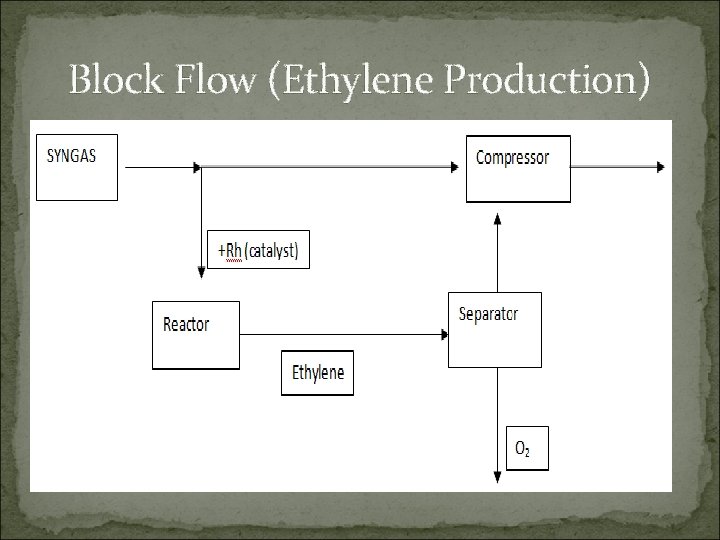 Block Flow (Ethylene Production)
Block Flow (Ethylene Production)
 Equipment List Turbo Compressors Turbo Expanders Reactors Storage Tanks Water Reservoir Heat Exchangers Distillation Column
Equipment List Turbo Compressors Turbo Expanders Reactors Storage Tanks Water Reservoir Heat Exchangers Distillation Column
 Economic Evaluation Once the amounts of feed stock have been determined, equipment costs can be calculated as well as any feeds costs that will be added outside of the syngas obtained from group Foxtrot. If Ethylene is to be purchased it will run around $650750/ton The current price of Propionic acid is $2000 -2100/ton
Economic Evaluation Once the amounts of feed stock have been determined, equipment costs can be calculated as well as any feeds costs that will be added outside of the syngas obtained from group Foxtrot. If Ethylene is to be purchased it will run around $650750/ton The current price of Propionic acid is $2000 -2100/ton
 Estimates Material Balance (1) 2 CO + 4 H 2 + RCOOH C 2 H 5 OOCR + 2 H 2 O (2) C 2 H 5 OOCR C 2 H 4 +HOOCR (3) CO + H 2 +C 2 H 4 CH 3 CH 2 CHO Calcalation: TBD Utilities: TBD
Estimates Material Balance (1) 2 CO + 4 H 2 + RCOOH C 2 H 5 OOCR + 2 H 2 O (2) C 2 H 5 OOCR C 2 H 4 +HOOCR (3) CO + H 2 +C 2 H 4 CH 3 CH 2 CHO Calcalation: TBD Utilities: TBD
 Estimates Pt. 2 Conceptual control scheme: Dual man and mechanically run process Constraints: Managing syngas composition for each feed
Estimates Pt. 2 Conceptual control scheme: Dual man and mechanically run process Constraints: Managing syngas composition for each feed
 Applicable Standards Analgesic properties Used in pharmaceutical Used in plastics Synthesis of propionates to make methyl methacrylate Commonly used as pesticides and bactericides Anticonvulsant Preservatives Used as solvents
Applicable Standards Analgesic properties Used in pharmaceutical Used in plastics Synthesis of propionates to make methyl methacrylate Commonly used as pesticides and bactericides Anticonvulsant Preservatives Used as solvents
 Project Communication Files Wiki link: http: //seniorecho. wikispaces. com/ Email: designecho@listserv. uic. edu
Project Communication Files Wiki link: http: //seniorecho. wikispaces. com/ Email: designecho@listserv. uic. edu
 References "Acetic Acid Production. " Acetic Acid Production. 2009. Web. 22 Jan. 2011.
References "Acetic Acid Production. " Acetic Acid Production. 2009. Web. 22 Jan. 2011.
 QUESTIONS
QUESTIONS


