09a - Биогенез и транспорт.ppt
- Количество слайдов: 28
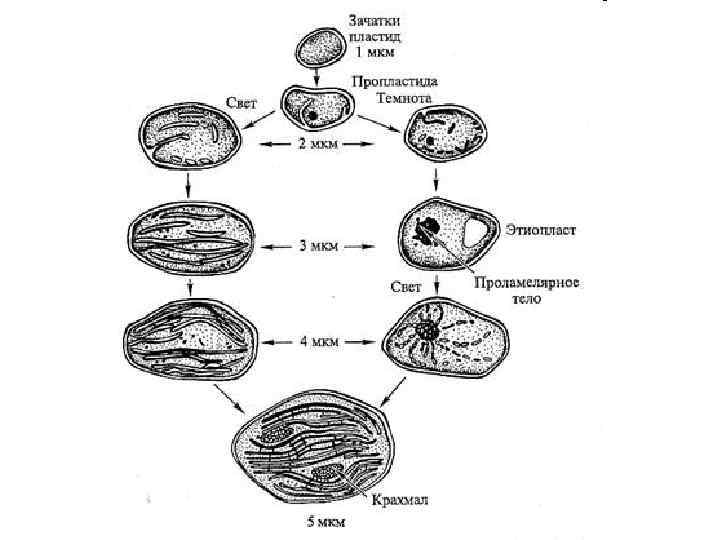
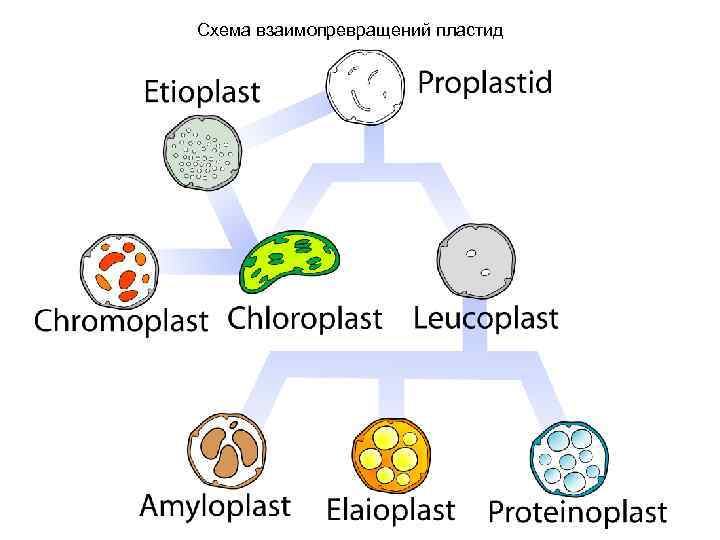
Схема взаимопревращений пластид

Prolamellar bodies
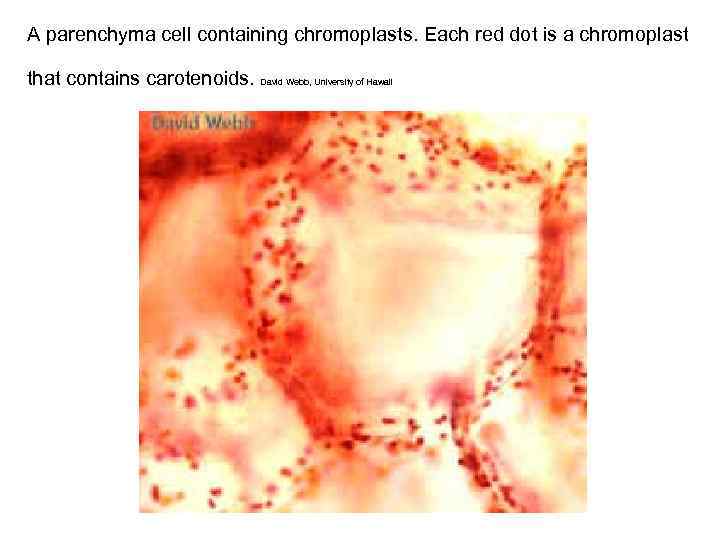
A parenchyma cell containing chromoplasts. Each red dot is a chromoplast that contains carotenoids. David Webb, University of Hawaii
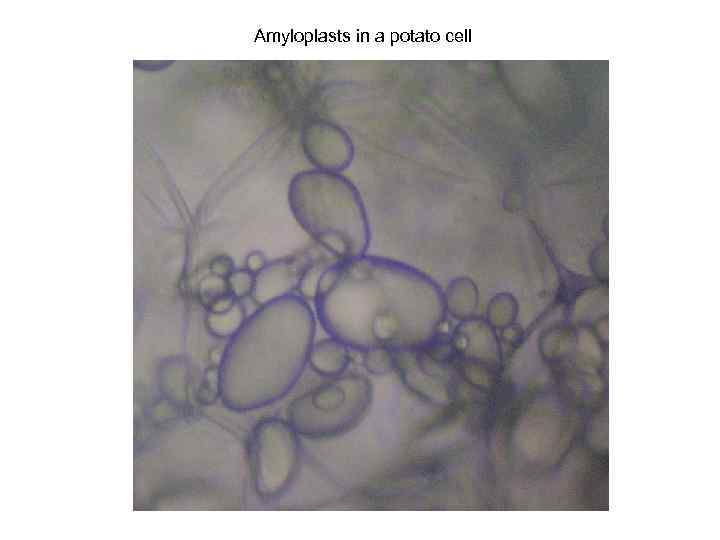
Amyloplasts in a potato cell
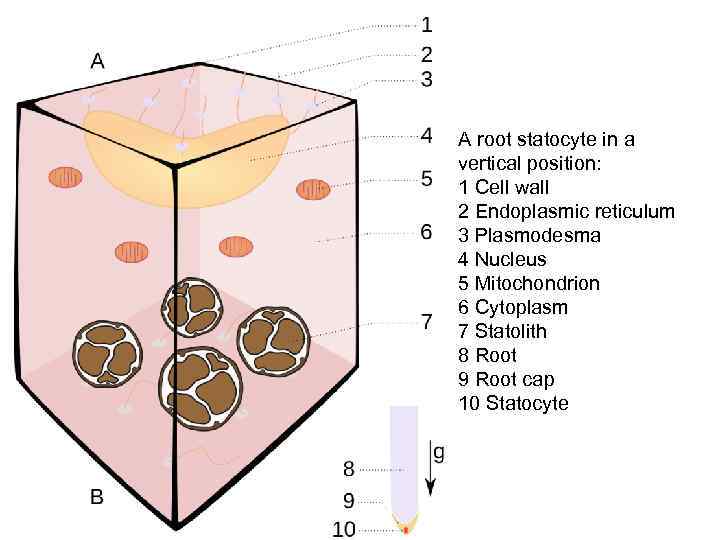
A root statocyte in a vertical position: 1 Cell wall 2 Endoplasmic reticulum 3 Plasmodesma 4 Nucleus 5 Mitochondrion 6 Cytoplasm 7 Statolith 8 Root 9 Root cap 10 Statocyte
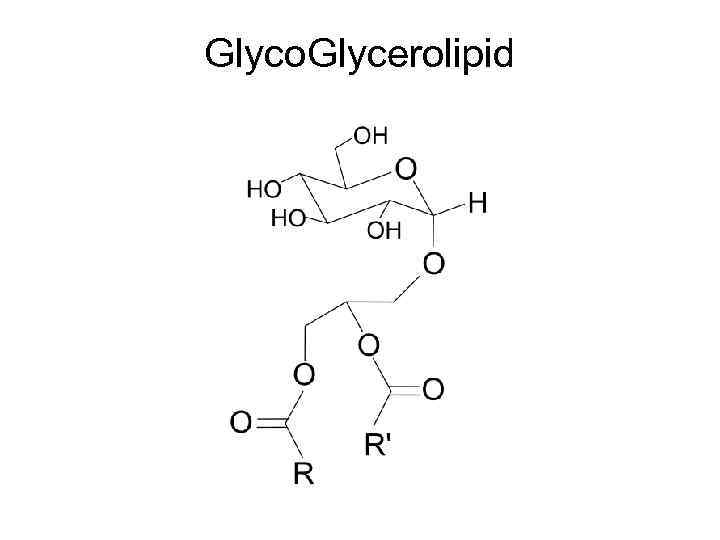
Glyco. Glycerolipid
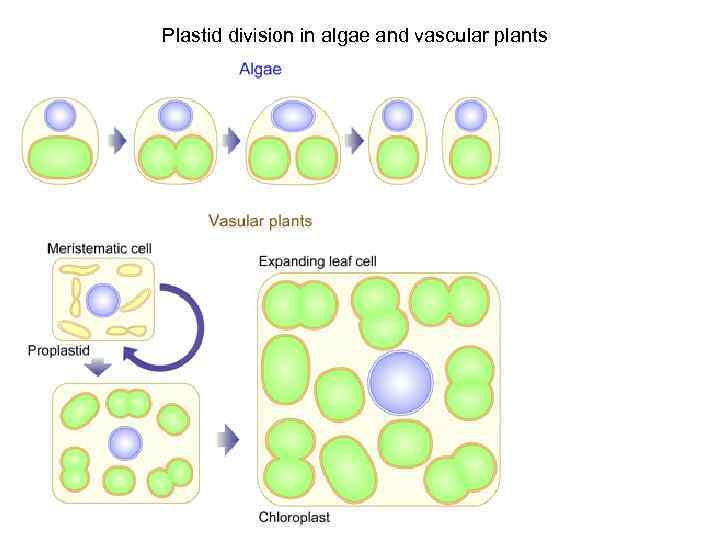
Plastid division in algae and vascular plants
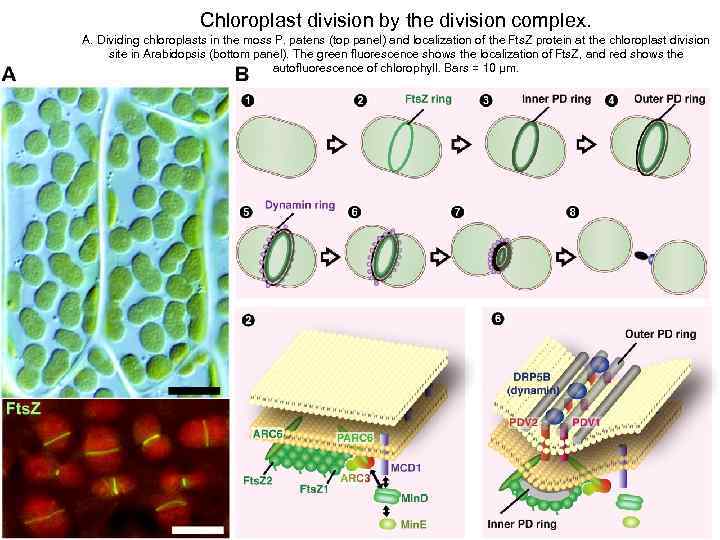
Chloroplast division by the division complex. A. Dividing chloroplasts in the moss P. patens (top panel) and localization of the Fts. Z protein at the chloroplast division site in Arabidopsis (bottom panel). The green fluorescence shows the localization of Fts. Z, and red shows the autofluorescence of chlorophyll. Bars = 10 μm.
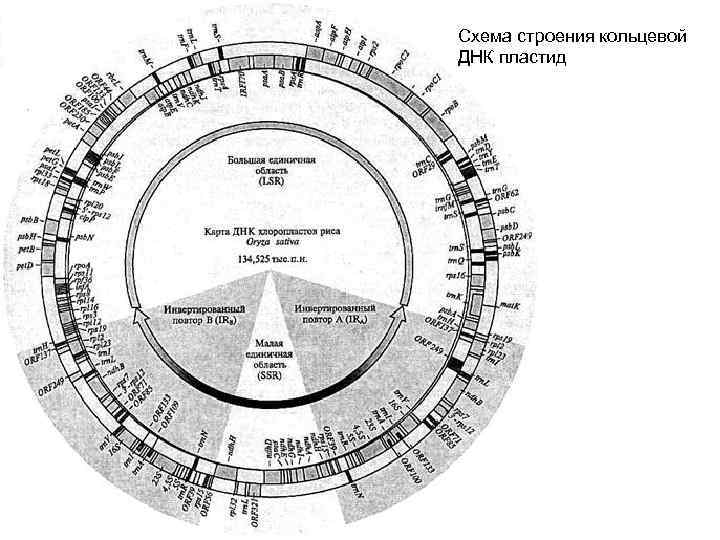
Схема строения кольцевой ДНК пластид
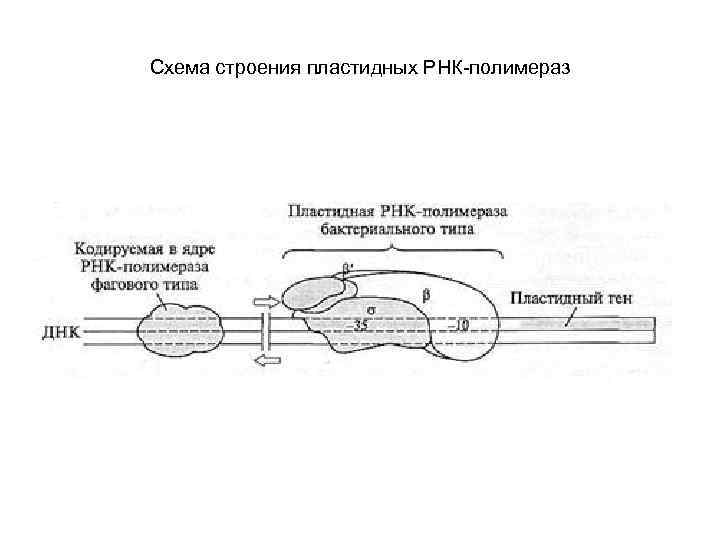
Схема строения пластидных РНК-полимераз
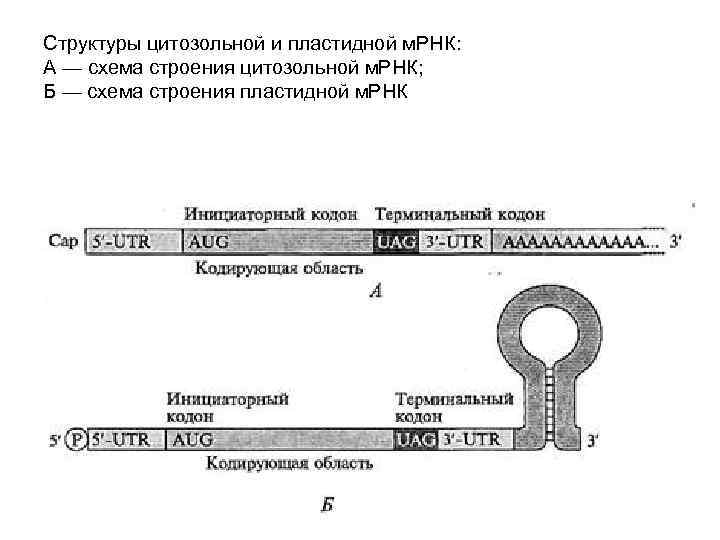
Cтруктуры цитозольной и пластидной м. РНК: А — схема строения цитозольной м. РНК; Б — cхема строения пластидной м. РНК
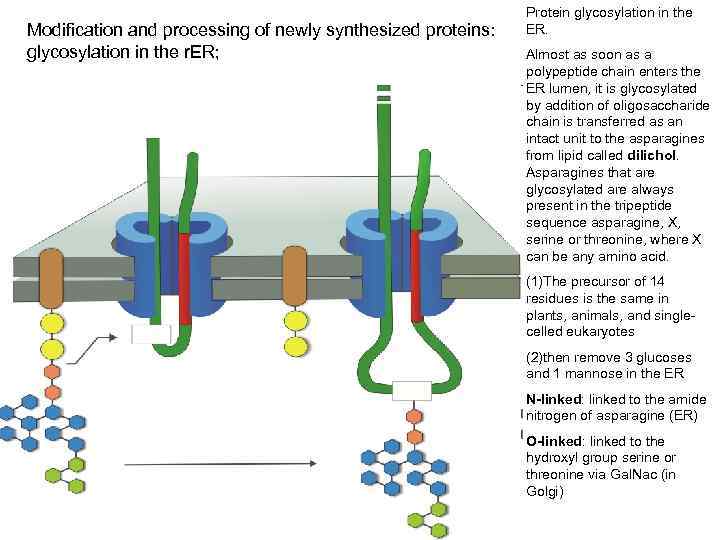
Modification and processing of newly synthesized proteins: glycosylation in the r. ER; Protein glycosylation in the ER. Almost as soon as a polypeptide chain enters the ER lumen, it is glycosylated by addition of oligosaccharide chain is transferred as an intact unit to the asparagines from lipid called dilichol. Asparagines that are glycosylated are always present in the tripeptide sequence asparagine, X, serine or threonine, where X can be any amino acid. (1)The precursor of 14 residues is the same in plants, animals, and singlecelled eukaryotes (2)then remove 3 glucoses and 1 mannose in the ER N-linked: linked to the amide nitrogen of asparagine (ER) O-linked: linked to the hydroxyl group serine or threonine via Gal. Nac (in Golgi)
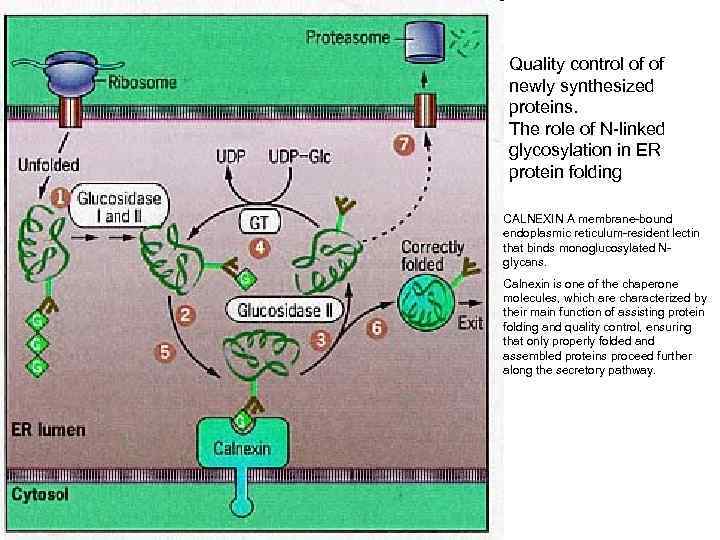
Quality control of of newly synthesized proteins. The role of N-linked glycosylation in ER protein folding CALNEXIN A membrane-bound endoplasmic reticulum-resident lectin that binds monoglucosylated Nglycans. Calnexin is one of the chaperone molecules, which are characterized by their main function of assisting protein folding and quality control, ensuring that only properly folded and assembled proteins proceed further along the secretory pathway.
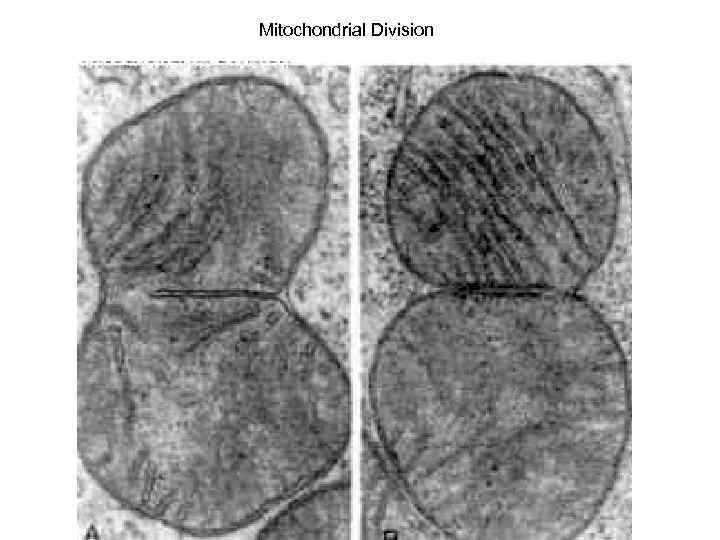
Mitochondrial Division
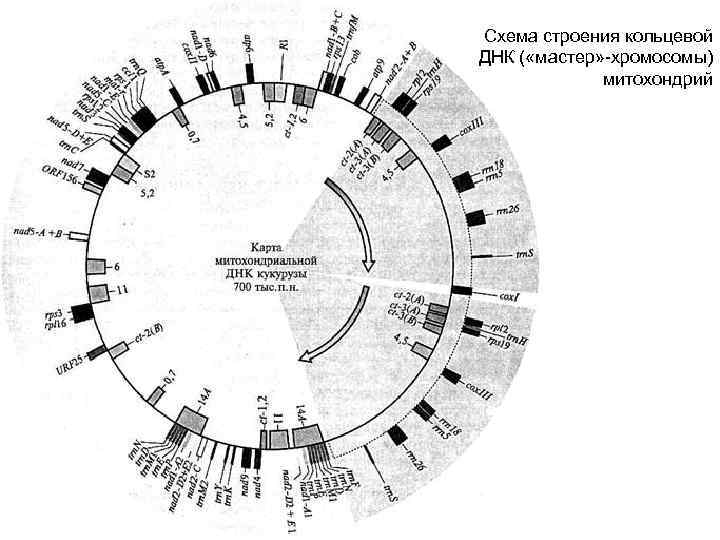
Схема строения кольцевой ДНК ( «мастер» -хромосомы) митохондрий
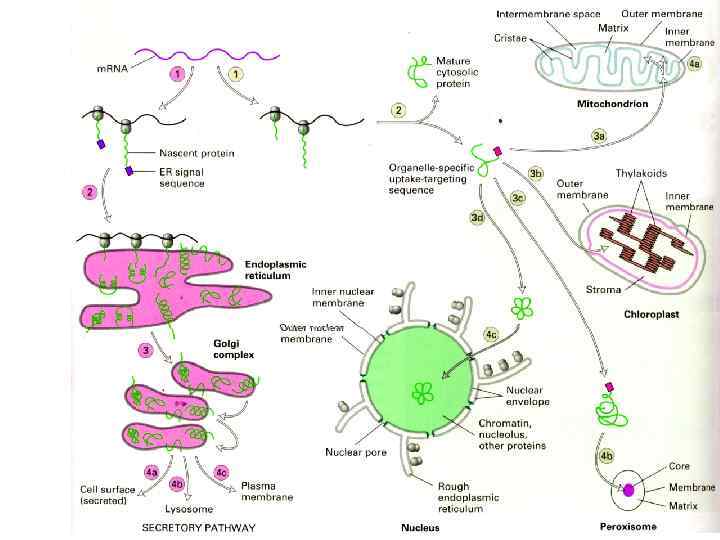
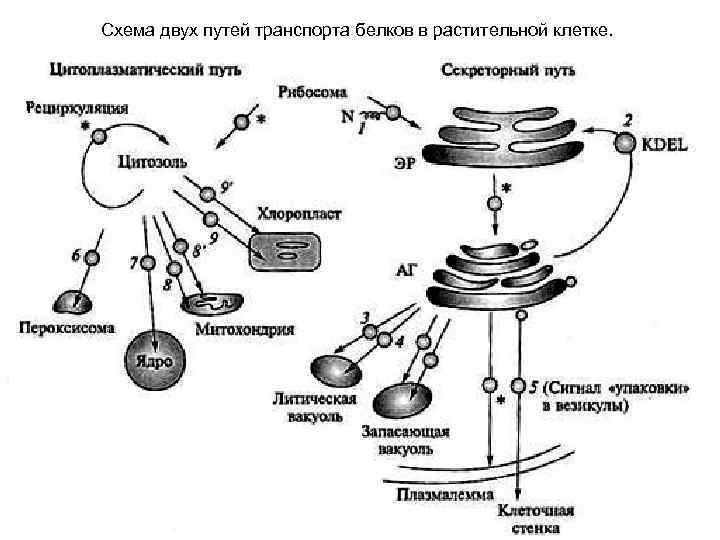
Схема двух путей транспорта белков в растительной клетке.
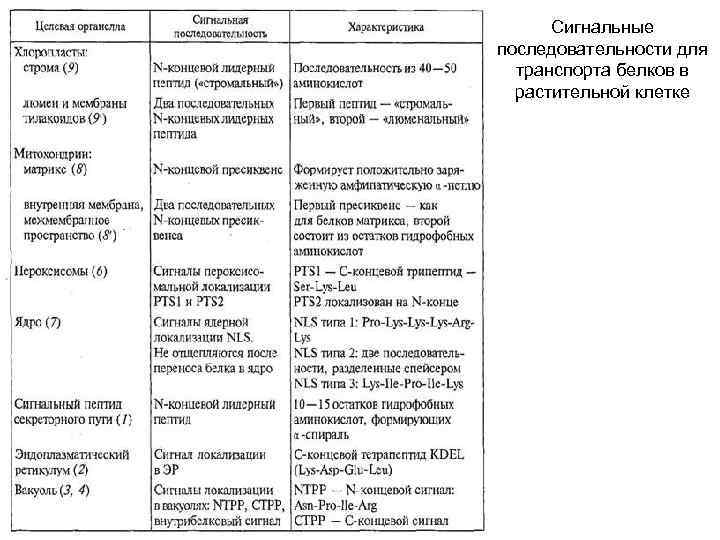
Сигнальные последовательности для транспорта белков в растительной клетке
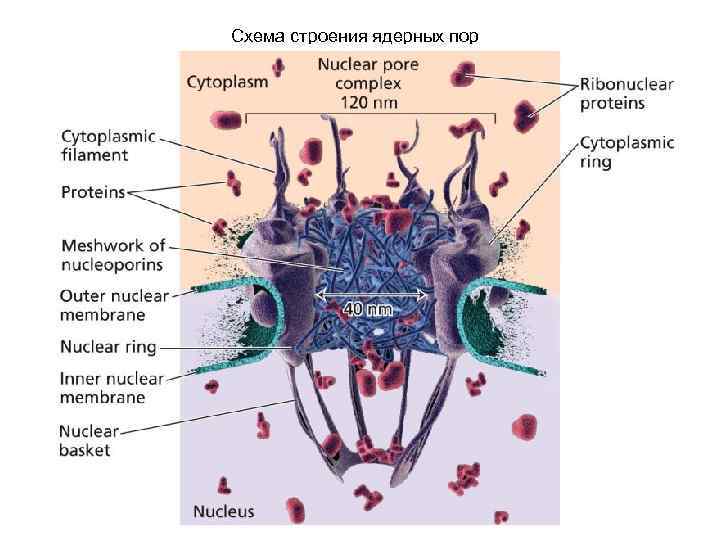
Схема строения ядерных пор
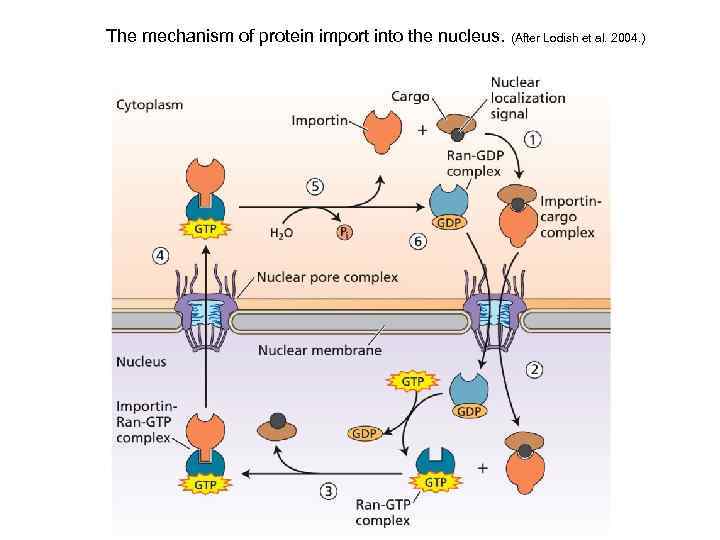
The mechanism of protein import into the nucleus. (After Lodish et al. 2004. )
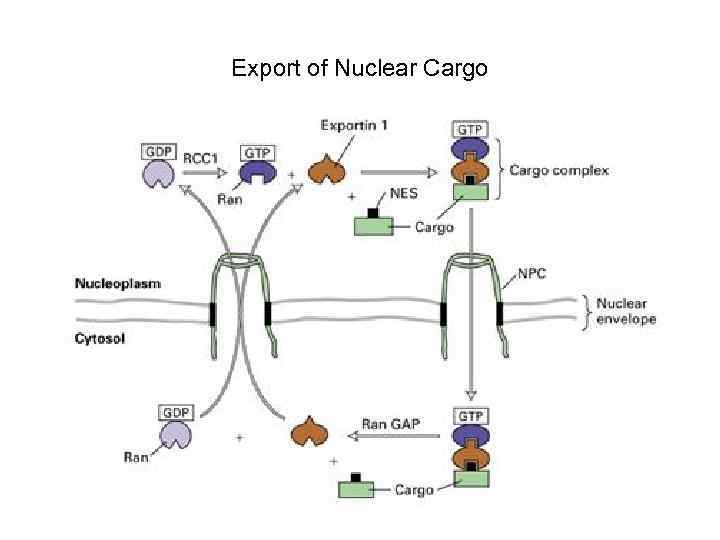
Export of Nuclear Cargo
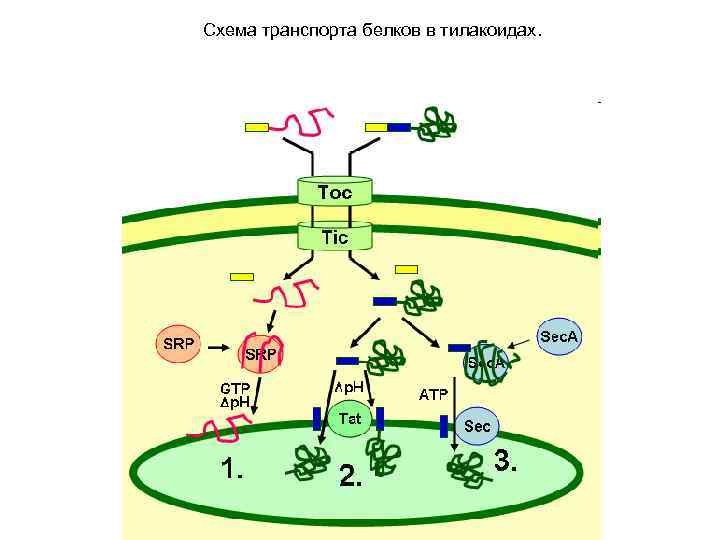
Схема транспорта белков в тилакоидах.
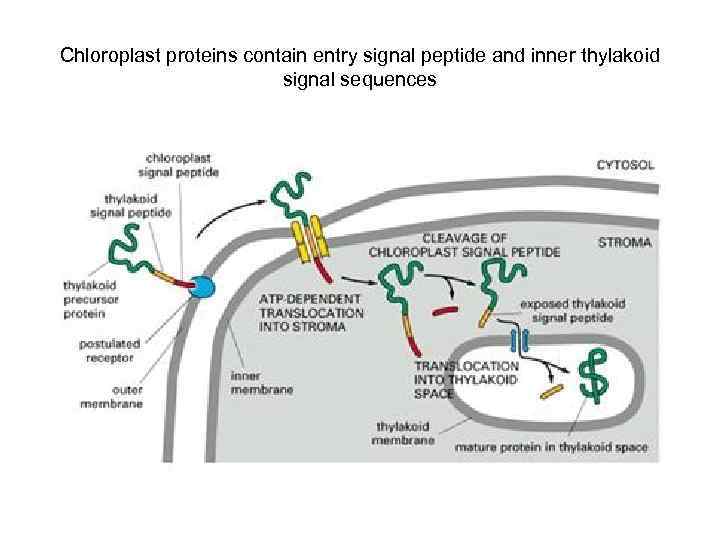
Chloroplast proteins contain entry signal peptide and inner thylakoid signal sequences
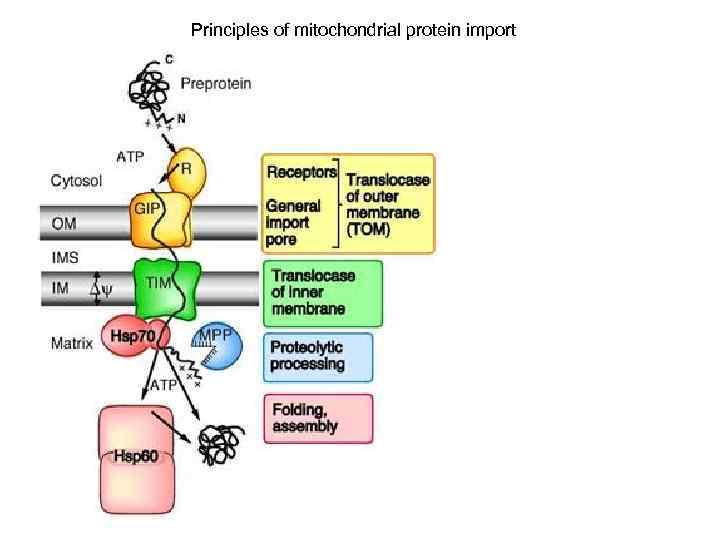
Principles of mitochondrial protein import

Protein sorting: Protein molecules move from the cytosol to their target organelles or cell surface directed by the sorting signals in the proteins. Some typical signal sequences: Import into nucleus: -Pro-Lys-Lys-Arg-Lys-Val. Export from nucleus: -Leu-Ala-Leu-Lys-Leu-Ala-Gly-Leu-Asp-Ile. Import into mitochondris: +H 3 N-Met-Leu-Ser-Leu-Arg-Gln-Ser-Ile-Arg-Phe-Lys-Pro. Ala-Thr-Arg-Thr-Leu-Cys-Ser-Arg-Tyr-Leu. Import into peroxisomes: -Ser-Lys-Leu-COOImport into ER: +H 3 N-Met-Ser-Phe-Val-Ser-Leu-Leu-Val-Gly-Ile-Leu-Phe-Try. Ala-Thr-Glu-Ala-Glu-Gln-Leu-Thr-Lys-Cys-Glu-Val-Phe-Gln. Return to ER: -Lys-Asp-Glu-Leu--COOSome characteristic features of the differences of the different classes of signal sequences are highlighted in color. Where they are known to be important for the function of the signal sequence, positively charged amino acids are shown in blue. Similarly, important hydrophobic amino acids are shown in red. +H 3 N indicates the Nterminus of a protein; COO- indicates the C-terminus.
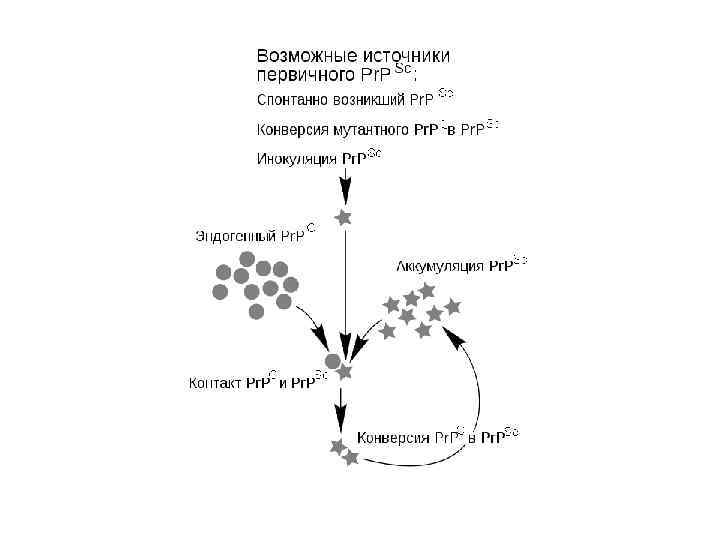
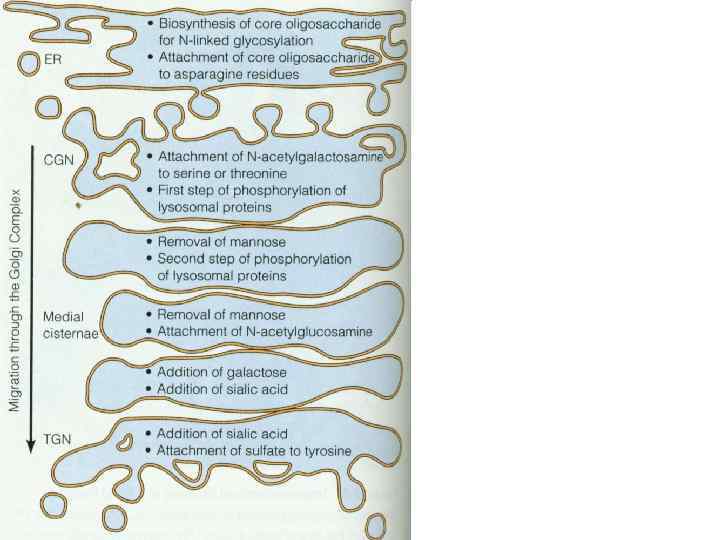
09a - Биогенез и транспорт.ppt