69f9484dc293fb797004d30ef52474bc.ppt
- Количество слайдов: 37

Summary of Proceedings: FDA Transmissible Spongiform Encephalopathies Advisory Committee 17 th Meeting 08 February 2005 FDA Blood Products Advisory Committee 82 nd Meeting 17 March 2005 Gaithersburg MD David M. Asher, MD <asher@cber. fda. gov> Laboratory of Bacterial, Parasitic and Unconventional Agents Division of Emerging & Transfusion-Transmitted Diseases Office of Blood Research & Review Center for Biologics Evaluation & Research US Food & Drug Administration

m m l Worldwide BSE update (Lisa Ferguson, APHIS, USDA) UK notifications of recipients plasma derivatives re possible v. CJD risk (Kate Soldan, Anna Molesworth, UK CJD Incidents Panel) [TSEs and surgical instrument decontamination (Lynn Sehulster, CDC)] Developing a risk assessment model and initial risk estimates for exposure to agent of variant Creutzfeldt-Jakob disease (v. CJD) via investigational use of UK factor XI in USA (Mark Weinstein, Dorothy Scott, Steven Anderson, FDA) l m Developing risk assessment models for exposure to v. CJD agent via other coagulation factors (Steven Anderson, FDA) v. CJD in France (Jean-Philippe Brandel, Epidemio. Surveillance. Network, Paris [presented in absentia and revised by Pedro Piccardo and Steven Anderson, FDA]) and UK (Sheila Bird, MRC Biostat Unit, Inst of Public Health, Cambridge) l l Possible deferral of blood and plasma donors with history of prior transfusion in European countries besides UK (Alan Williams, FDA --------------------------------= Decisional issues

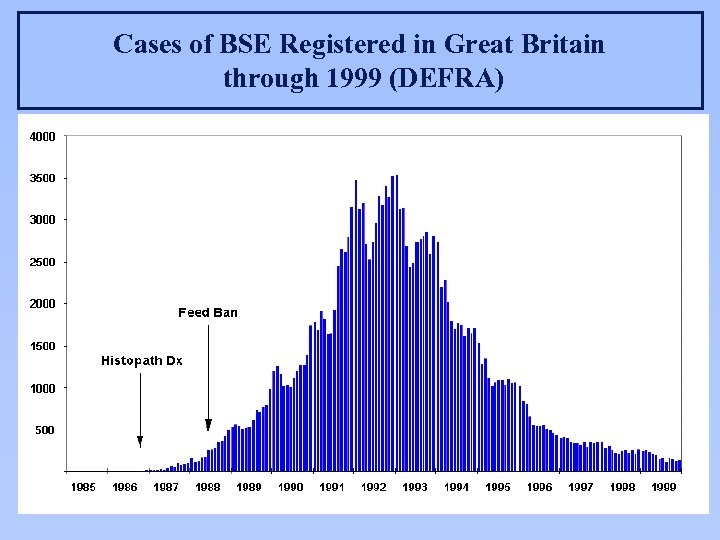
Cases of BSE Registered in Great Britain through 1999 (DEFRA)


23 Countries with BSE in Native Cattle [yr first reported & approx. total cases reported to OIE thro’ 28 Feb 2005] l UK 1986 (>183 000) [1202 ‘ 01; 1144 ’ 02; 612 ’ 03; 338 ’ 04] l l l l l Ireland 1989 (1489) Switzerland 1990 (519) France 1991 (945) Portugal 1994 (950) Belgium 1997 (129) Netherlands 1997 (77) Luxembourg 1997 (2) Liechtenstein 1998 (2) Denmark 2000 (13) Germany 2000 (357) l l l ¡ Spain 2000 (519) Italy 2000 (124) Greece 2001 (1) Czech Repub 2001 (15) Slovakia 2001 (15) Japan 2001 (15) Slovenia 2001 (5) Finland 2001 (1) Austria 2001 (1) Poland 2002 (22) Israel 2002 (1) Canada 2003 (1 ex UK+4) [USA 2003 (1 ex Canada)]
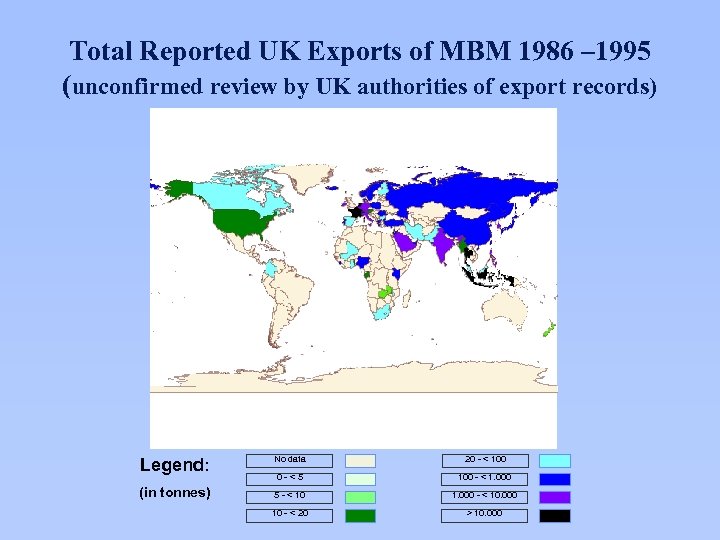
Total Reported UK Exports of MBM 1986 – 1995 (unconfirmed review by UK authorities of export records) Legend: (in tonnes) No data 20 - < 100 0 -<5 100 - < 1. 000 5 - < 10 1. 000 - < 10. 000 10 - < 20 > 10. 000
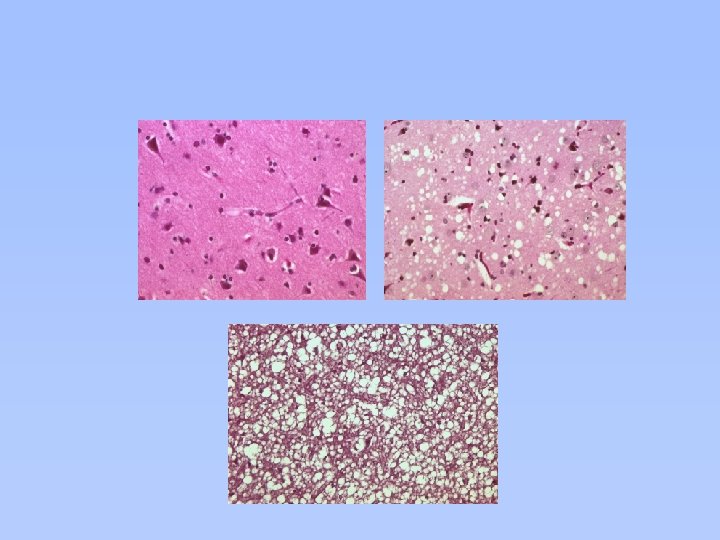
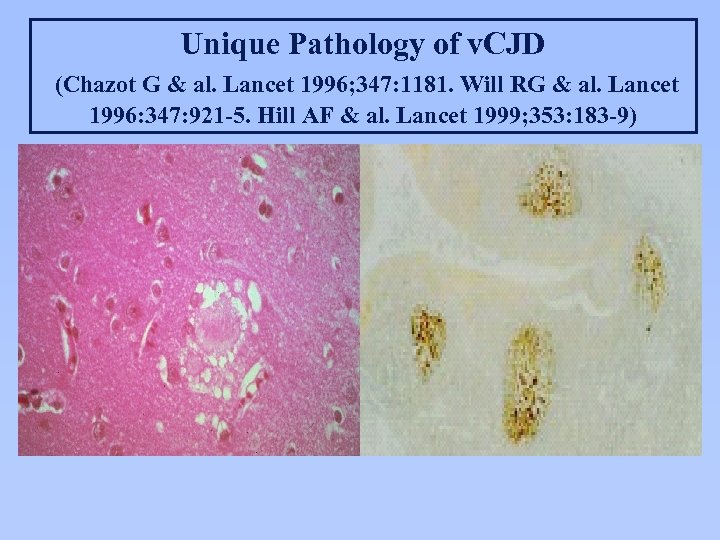
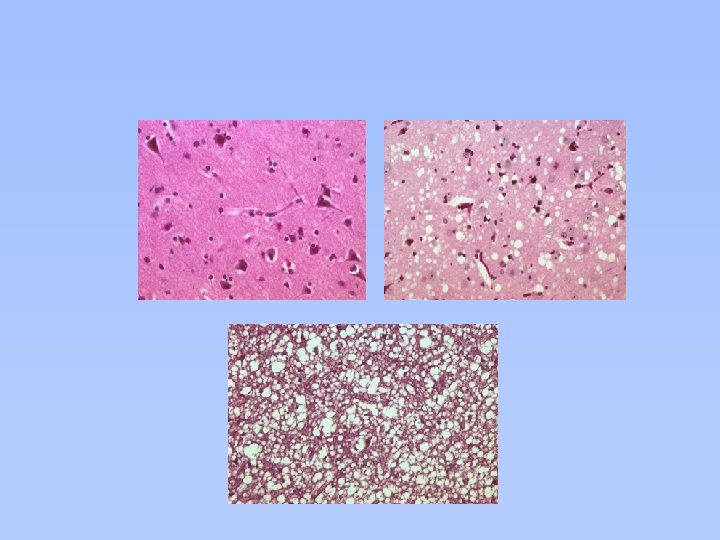
Unique Pathology of v. CJD (Chazot G & al. Lancet 1996; 347: 1181. Will RG & al. Lancet 1996: 347: 921 -5. Hill AF & al. Lancet 1999; 353: 183 -9)

Variant CJD: Reasons for Greater FDA Concern about Potential Infectivity of Blood l l Lymphoid tissues of patients with v. CJD contain much more protease-resistant prion protein than do those of patients with conventional forms of CJD. Infectivity of those tissues is not yet clear. (Note: Lymphoid tissues of some patients with conventional sporadic CJD [s. CJD] have been infectious for non-human primates [Brown P et al. Ann Neurol 1994; 35: 513]. ) Implication: Blood, containing lymphoid cells, might be more infectious in v. CJD than in classical forms of CJD. v. CJD differs markedly from s. CJD; distribution of infectivity in patients with s. CJD might not be predictive for v. CJD is a new emerging disease not found in the USA except in one long-time UK resident. Actions of UK authorities implied lack of confidence in safety of blood of UK donors. -------------------------------------- è Two cases of probable TT v. CJD

Final Guidance for Industry: Revised Precautionary Measures to Reduce the Possible Risk of Transmission of Creutzfeldt-Jakob Disease (CJD) & Variant Creutzfeldt. Jakob Disease (v. CJD) by Blood and Blood Products Jan 9, 2002 www. fda. gov/cber/guidelines. htm l Phase I (for implementation by May 31, 2002) Indefinitely defer all donors who - have any form of CJD or are at increased risk of CJD - (no change from previous FDA guidance) - spent 3 mo in UK from Jan 1, 1980 to Dec 31, 1996 v v or who ever had blood transfusion in UK from 1980 to present or who ever injected UK bovine insulin prepared in or after 1980 - spent 5 yr in France from Jan 1, 1980 to the present - spent 6 mo on US military bases from Jan 1, 1980 to end of 1990 north of Alps or end of 1996 south of Alps

Final Guidance for Industry: Revised Precautionary Measures to Reduce the Possible Risk of Transmission of Creutzfeldt-Jakob Disease (CJD) & Variant Creutzfeldt. Jakob Disease (v. CJD) by Blood and Blood Products Jan 9, 2002 www. fda. gov/cber/guidelines. htm Phase II (for implementation by October 31, 2002) - Indefinitely defer all donors of Whole Blood but not donors of Source Plasma who spent 5 yr in Europe from Jan 1, 1980 to the present (including time in spent in UK 19801996 and France 1980 -present) l Exempt from deferral are v Donors of Source Plasma who spent any period of time in Europe except UK and France v Donors of plasma/serum to manufacture CBERapproved non-injectable products (specially labeled)
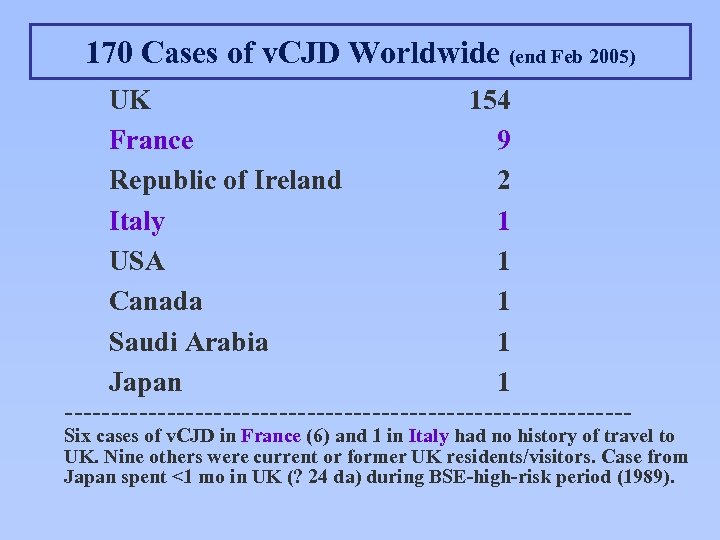
170 Cases of v. CJD Worldwide (end Feb 2005) UK 154 France 9 Republic of Ireland 2 Italy 1 USA 1 Canada 1 Saudi Arabia 1 Japan 1 ------------------------------- Six cases of v. CJD in France (6) and 1 in Italy had no history of travel to UK. Nine others were current or former UK residents/visitors. Case from Japan spent <1 mo in UK (? 24 da) during BSE-high-risk period (1989).
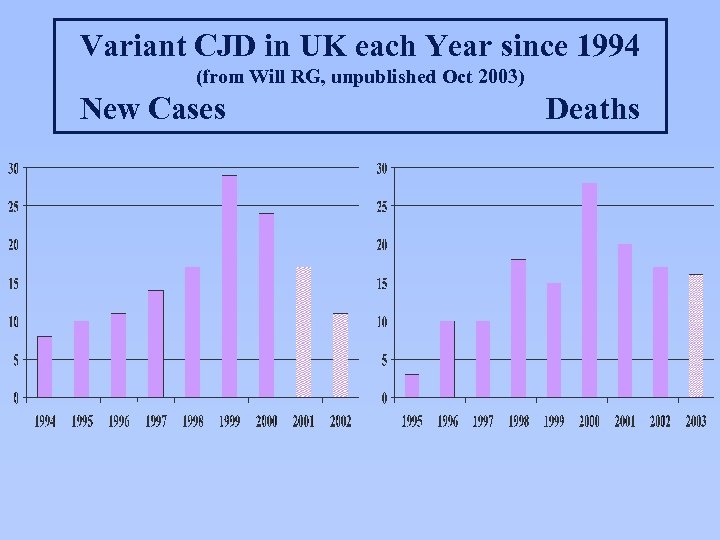
Variant CJD in UK each Year since 1994 (from Will RG, unpublished Oct 2003) New Cases Deaths
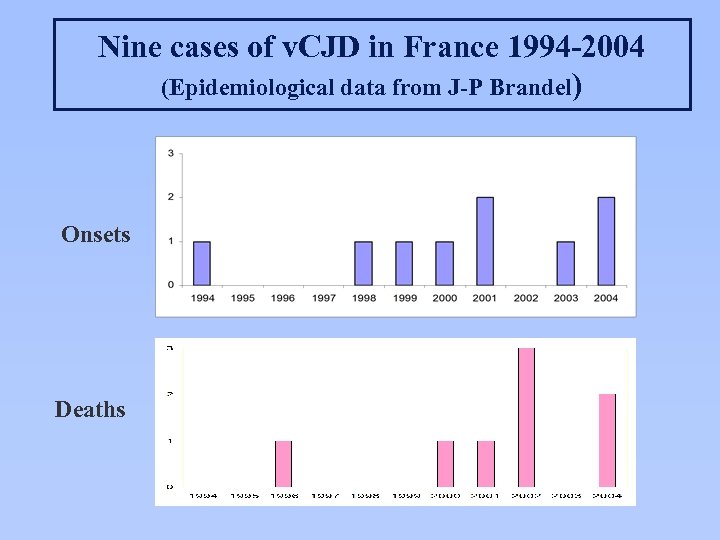
Nine cases of v. CJD in France 1994 -2004 (Epidemiological data from J-P Brandel) Onsets Deaths

1 st Transfusion-Transmitted Case of v. CJD (UK Parliament 17 Dec 2003; CA Llewelyn & al. Lancet 2004; 363: 417 -421) March 1996 l Clinically healthy young blood donor donated Whole Blood to the UK National Blood Service. l RBC—not leukoreduced—transfused into 63 yr-old surgical patient. About March 1999 l Three yr later, donor developed signs of variant CJD, died; v. CJD was confirmed by autopsy. l UK Transfusion Medicine Epidemiology Review (TMER) enrolled recipient (with 49 other recipients of v. CJD-implicated labile blood components from 16 donors later Dx with v. CJD— 13 now living >5 yr). December 2003 l The recipient died age 69; post-mortem diagnosis was typical v. CJD (PRNP-129 -met/met homozygous). l The recipient's age-adjusted food-borne risk of v. CJD estimated by UK authorities to have been ~ 1: 15, 000 to 1: 30, 000.

2 nd Transfusion-Transmitted Case of v. CJD (Peden AH & al. Lancet 2004; 264: 527 -529) 1999 l Clinically healthy young blood donor donated Whole Blood to the UK National Blood Service. l RBC—not leukoreduced—transfused into a surgical patient. 2000 l Donor developed signs of variant CJD 18 mo later, died in 2001; v. CJD confirmed by autopsy. l UK Transfusion Medicine Epidemiology Review (TMER) enrolled recipient (with 49 other recipients of v. CJD-implicated labile blood components from 16 donors later Dx with v. CJD— 13 now living >5 yr). 2003 l The recipient died of ruptured abdominal aortic aneurysm. l No history of dementia and CNS, tonsils and appendix were normal. l Pr. Psc was present in several areas of spleen, cervical lymph node. l The recipient's age-adjusted food-borne risk of v. CJD was estimated by UK authorities to have been ~ 1: 15, 000 to 1: 30, 000. l Recipient’s genotype was PRNP 129 -met/val heterozygous).

General Approaches to Assessing Risk FDA is committed to risk-based regulatory decision making. -----------------------------Recent Example: TSEAC Meeting 08 February 2005 -----------------------------TSE Risk Assessment for Plasma Derivatives Steven Anderson, Ph. D, MPP Office of Biostatistics & Epidemiology Center for Biologics Evaluation and Research U. S. Food and Drug Administration -----------------------------<http: //www. fda. gov/ohrms/dockets/ac/05/transcripts/2005 -4088 t 1. htm>

Risk of Product Contamination Source of raw materials l l Animal: low-risk origin, low-risk tissue - Certificate - Test for contaminant or Dx (better ones needed) Human: low-risk population - Donor or surrogate questionnaire - Donor screening test (needed) Manufacturing process l l Eliminate contaminating agent (validate method) - Inactivation - Removal Prevent cross contamination (“downstream” contamination) - Cleaning - Disinfection End use of product l l Route Dose (course, year, lifetime)
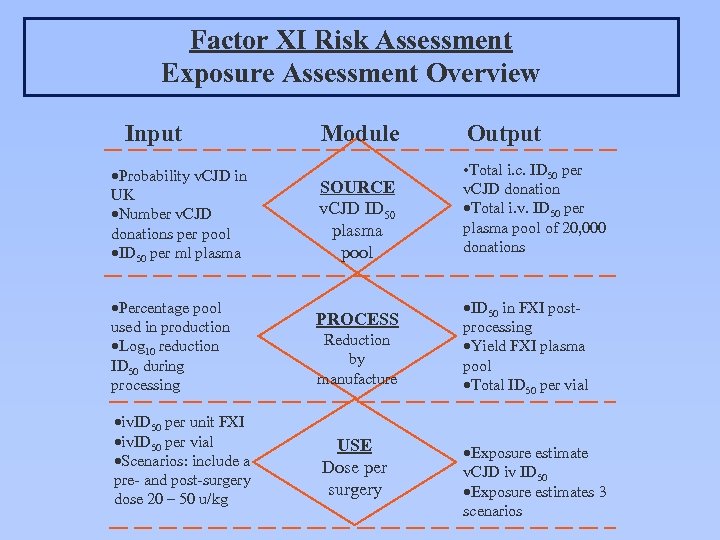
Factor XI Risk Assessment Exposure Assessment Overview Input ·Probability v. CJD in UK ·Number v. CJD donations per pool ·ID 50 per ml plasma ·Percentage pool used in production ·Log 10 reduction ID 50 during processing ·iv. ID 50 per unit FXI ·iv. ID 50 per vial ·Scenarios: include a pre- and post-surgery dose 20 – 50 u/kg Module Output SOURCE v. CJD ID 50 plasma pool • Total i. c. ID 50 per v. CJD donation ·Total i. v. ID 50 per plasma pool of 20, 000 donations PROCESS Reduction by manufacture USE Dose per surgery ·ID 50 in FXI postprocessing ·Yield FXI plasma pool ·Total ID 50 per vial ·Exposure estimate v. CJD iv ID 50 ·Exposure estimates 3 scenarios
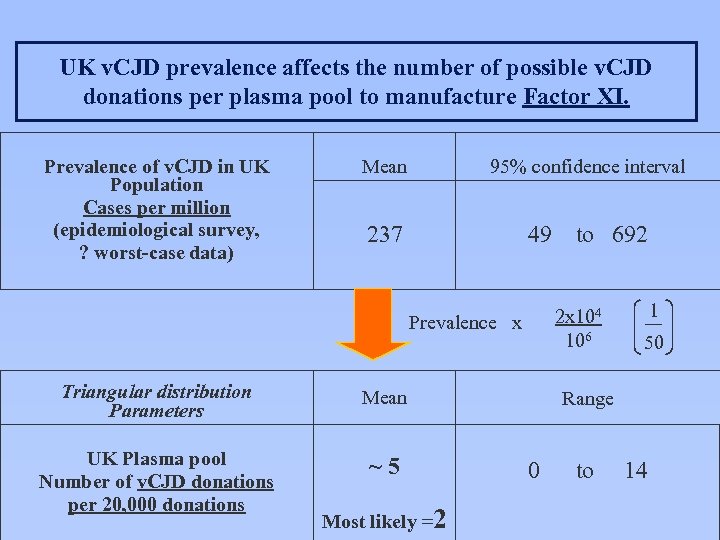
UK v. CJD prevalence affects the number of possible v. CJD donations per plasma pool to manufacture Factor XI. Prevalence of v. CJD in UK Population Cases per million (epidemiological survey, ? worst-case data) Mean 95% confidence interval 237 49 2 x 104 106 Prevalence x Triangular distribution Parameters Mean UK Plasma pool Number of v. CJD donations per 20, 000 donations ~ 5 Most likely =2 to 692 1 50 Range 0 to 14

UK FXI v. CJD Exposure Assessment: Possible v. CJD infectivity in UK plasma pool Quantity of v. CJD ID 50 present per ml plasma · Intracerebral (ic) ID 50 per ml blood (animal ID 50) – Minimum 0. 1 – Most likely 10 – Maximum 1, 000 · Assume 58% associated with plasma (Gregori, et al. 2004) · Adjust down 5 -fold to 10 -fold for reduced efficiency of intravenous (iv) vs ic route of exposure

FXI v. CJD Exposure Assessment Reduction during manufacturing PROCESS Log 10 reduction ID 50 during plasma processing Reduction based on processing steps l Variability in processing and reductions achieved l Assumption Log 10 reduction v. CJD agent infectivity Minimum Most Likely Maximum 0 2. 0 4. 0
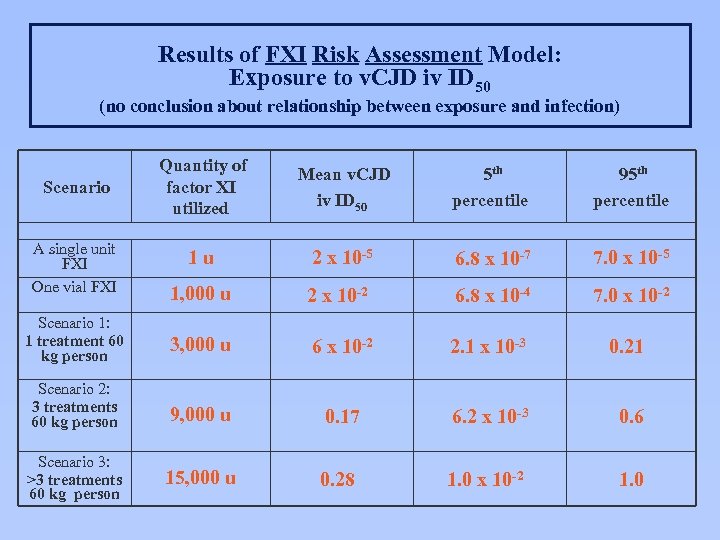
Results of FXI Risk Assessment Model: Exposure to v. CJD iv ID 50 (no conclusion about relationship between exposure and infection) Scenario A single unit FXI One vial FXI Scenario 1: 1 treatment 60 kg person Scenario 2: 3 treatments 60 kg person Scenario 3: >3 treatments 60 kg person Quantity of factor XI utilized Mean v. CJD iv ID 50 5 th percentile 95 th percentile 1 u 2 x 10 -5 6. 8 x 10 -7 7. 0 x 10 -5 1, 000 u 2 x 10 -2 6. 8 x 10 -4 7. 0 x 10 -2 3, 000 u 6 x 10 -2 2. 1 x 10 -3 0. 21 9, 000 u 0. 17 6. 2 x 10 -3 0. 6 15, 000 u 0. 28 1. 0 x 10 -2 1. 0

Exposure Risks Advantages of quantitative models 1. Transparent: assumptions articulated and available for modification 2. Data gaps: identified 3. Sensitivity Analysis (Importance Analysis): Most important elements “driving” overall risk identified, even without agreement on assumptions
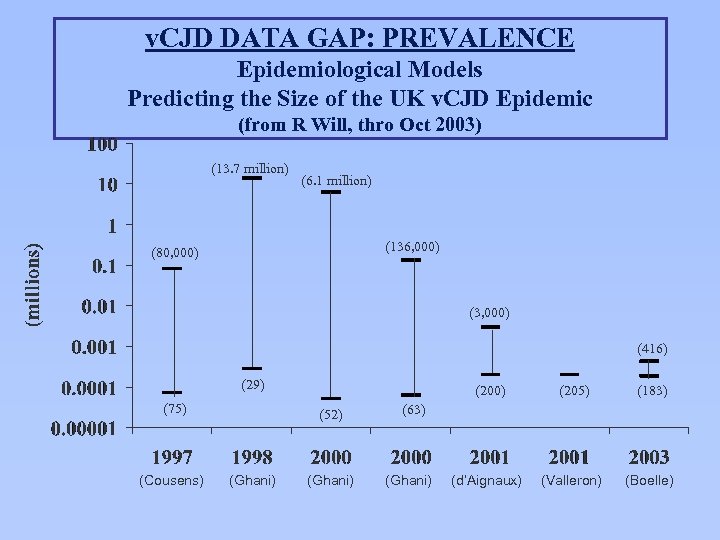
v. CJD DATA GAP: PREVALENCE Epidemiological Models Predicting the Size of the UK v. CJD Epidemic (from R Will, thro Oct 2003) (13. 7 million) (6. 1 million) (136, 000) (80, 000) (3, 000) (416) (29) (75) (Cousens) (200) (52) (Ghani) (205) (183) (d’Aignaux) (Valleron) (Boelle) (63) (Ghani)
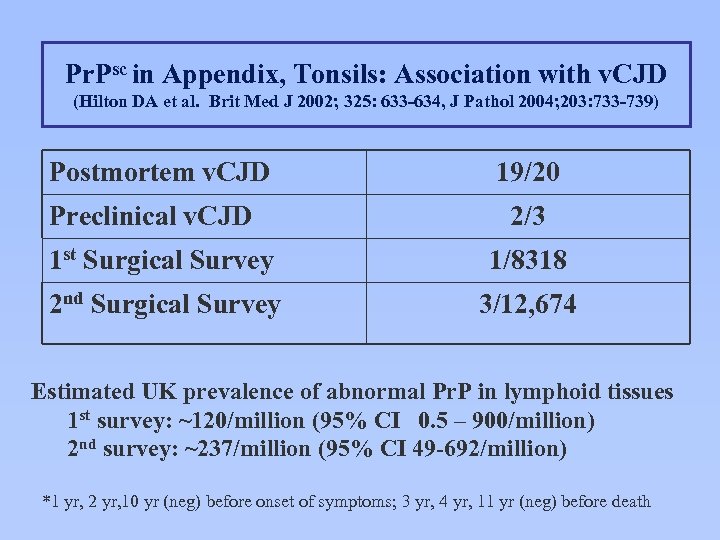
Pr. Psc in Appendix, Tonsils: Association with v. CJD (Hilton DA et al. Brit Med J 2002; 325: 633 -634, J Pathol 2004; 203: 733 -739) Postmortem v. CJD Preclinical v. CJD 19/20 2/3 1 st Surgical Survey 1/8318 2 nd Surgical Survey 3/12, 674 Estimated UK prevalence of abnormal Pr. P in lymphoid tissues 1 st survey: ~120/million (95% CI 0. 5 – 900/million) 2 nd survey: ~237/million (95% CI 49 -692/million) *1 yr, 2 yr, 10 yr (neg) before onset of symptoms; 3 yr, 4 yr, 11 yr (neg) before death

v. CJD Data Gaps: Incubation periods, duration of blood infectivity, amounts infectivity in blood, human iv. ID 50) l Incubation - Food-borne cases US Canada Japan [Ireland - Blood-borne cases First case Second case ¡ 9 -21 yr 11 -19 yr 12 yr 5 -10 yr] 6 yr > 5 yr Blood infectivity before clinical v. CJD: 3 yr

Sensitivity Analysis: Risk Drivers Estimates of FXI v. CJD Exposure Risk Two major factors influenced FXI v. CJD risk: 1. Number of v. CJD donations per plasma pool v Risk reduction measure=donor deferrals 2. Reduction of v. CJD agent during manufacture v Robustness (inactivation>removal) v Orthogonality (≥two processes preferred) v Additivity of process steps (must justify) v Validity (and relevance) of experimental data

Recipients surviving >5 yrs post transfusion of blood components from v. CJD/CJD Donors (data from UK TMER and US ARC look-back studies [R Dodd], presented by S. Anderson, FDA TSEAC 14 Oct 2004) No Infection v. CJD 2 13 CJD 0 116 Fisher's Exact Test comparing rates of infection after transfusions from v. CJD and CJD donors suggests a statistically significant difference between the two groups ( 1. 2% likelihood of difference by chance). Conclusion: Risk of TT CJD is less than TT v. CJD.

Policies to Reduce Risk of Transmitting v. CJD by Blood Products by Donor Selection: General Approaches l Reduce risk that donor was exposed to BSE agent - Dietary exposure: Lived in BSE country (or military base importing beef from UK) =“geographic” deferrals - Other exposure: Use of UK bovine insulin l Reduce risk that donor was exposed to v. CJD agent of human origin - Transfusion, UK after 1980 - [Transfusion, other BSE country—especially France] - [Surgery in BSE country after 1980: suggested by TSEAC member (not addressed by FDA or TSEAC)]
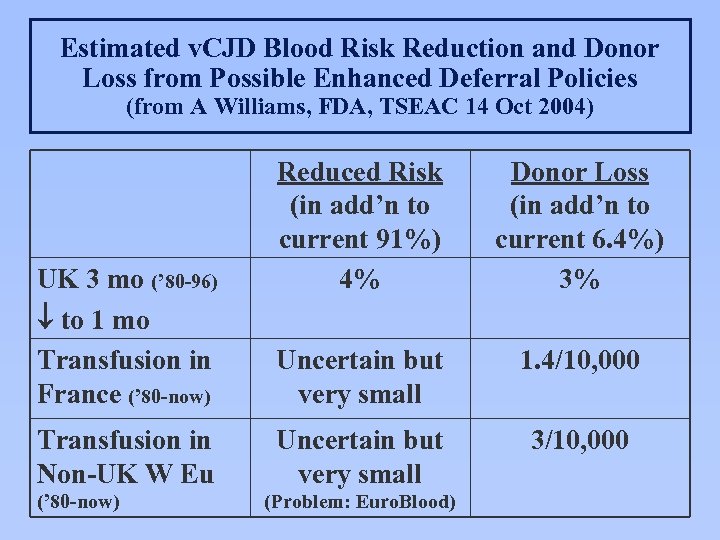
Estimated v. CJD Blood Risk Reduction and Donor Loss from Possible Enhanced Deferral Policies (from A Williams, FDA, TSEAC 14 Oct 2004) UK 3 mo (’ 80 -96) to 1 mo Transfusion in France (’ 80 -now) Reduced Risk (in add’n to current 91%) 4% Donor Loss (in add’n to current 6. 4%) 3% Uncertain but very small 1. 4/10, 000 Transfusion in Non-UK W Eu Uncertain but very small 3/10, 000 (’ 80 -now) (Problem: Euro. Blood)

TSEAC Advice to FDA 14 October 2004 1. Are measures currently recommended by FDA to reduce the risk of transmitting CJD and v. CJD by blood products still justified? Yes 14, No 0 2. Do recent scientific data on v. CJD warrant consideration by FDA of any additional potentially risk-reducing measures for blood and blood products? Yes 1, No 13 3. If so, please comment on the additional risk-reducing measures that FDA should consider at this time. [Investigate v. CJD cases outside UK for ? exposure to blood. Consider deferral of donors transfused in non-UK BSE countries. ]

TSEAC Advice to FDA 08 February 2005 1. Should FDA recommend deferral of Whole Blood donors transfused after the beginning of 1980? l In France Yes 12, No 3, Abstain 1 l Other W Europe Yes 0, No 15, Abstain 1 2. Should FDA recommend deferral of Source Plasma donors transfused in/after 1980? l In France Yes 5, No 7, Abstain 4 l Other W Europe Yes 0, No 16, Abstain 0

l l Limitation of deferral-based approaches to reduce the risk of blood-borne v. CJD Problem Many (? most) deferred donors are probably not infected with a TSE agent (needlessly deferred and alarmed). Possible solutions l Develop validated and reliable screening tests to detect infected donors and re-enter/reassure uninfected donors. Develop validated and reliable methods to remove (inactivate or separate) infectious TSE agents from products. Current status - No FDA-licensed/approved TSE test (for diagnosis or donor screening) No FDA-licensed method validated to remove TSE infectivity from red cells, platelets and plasma
69f9484dc293fb797004d30ef52474bc.ppt