cf4bd96c4e71632ddcf15596a72fb025.ppt
- Количество слайдов: 1
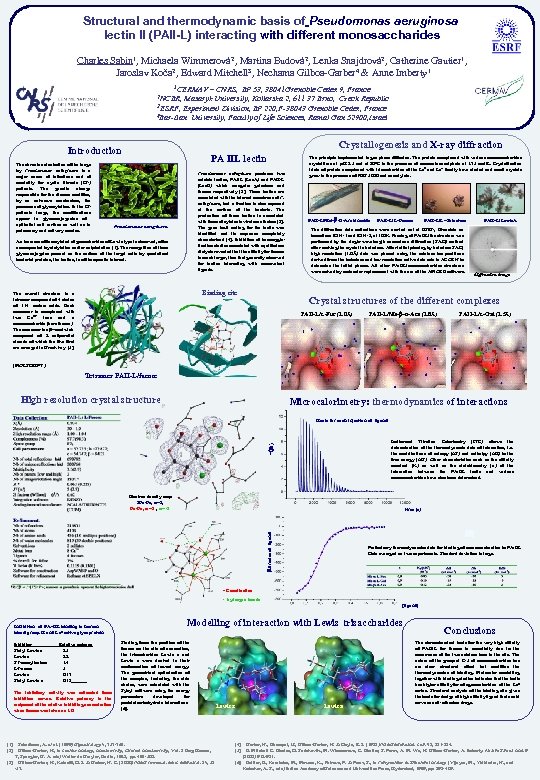
Structural and thermodynamic basis of Pseudomonas aeruginosa lectin II (PAII-L) interacting with different monosaccharides Charles Sabin 1, Michaela Wimmerová 2, Martina Budová 2, Lenka Snajdrová 2, Catherine Gautier 1, Jaroslav Koča 2, Edward Mitchell 3, Nechama Gilboa-Garber 4 & Anne Imberty 1 1 CERMAV – CNRS, BP 53, 38041 Grenoble Cedex 9, France Masaryk University, Kotlarska 2, 611 37 Brno, Czech Republic 3 ESRF, Experiment Division, BP 220, F-38043 Grenoble Cedex, France 4 Bar-Ilan University, Faculty of Life Sciences, Ramat Gan 52900, Israel 2 NCBR, Crystallogenesis and X-ray diffraction Introduction The chronic colonisation of the lungs by Pseudomonas aeruginosa is a major cause of infections and of mortality for cystic fibrosis (CF) patients. The genetic change responsible for the disease modifies, by an unknown mechanism, the processes of glycosylation. In the CF patients lungs, the modifications appear in glycoconjuguates of epithelial cell surface as well as in pulmonary and salivary mucins. PA IIL lectin Pseudomonas aeruginosa produces two soluble lectins, PAI-L (Lec. A) and PAII-L (Lec. B) which recognise galactose and fucose respectively [2]. These lectins are associated with the internal membrane of P. aeruginosa, but a fraction is also exposed at the surface of the bacteria. The production of these lectins is associated with those of cytotoxic virulence factors [3]. The gene lec. A coding for the lectin was identified and its sequence completely characterised [4]. Inhibition of haemagglutination studies associated with equilibrium dialysis revealed that the affinity for fucose is much larger, than that generally observed for lectins interacting with monovalent ligands. Pseudomonas aeruginosa An increase of fucosylated oligosaccharides of Lewis type is observed, often accompanied by sialylation and/or sulphatation [1]. The recognition of these glycoconjugates present on the surface of the target cells by specialised bacterial proteins, the lectins, is of therapeutic interest. Binding site The overall structure is a tetramer composed of 4 chains of 114 amino acids. Each monomer is complexed with two Ca 2+ ions and a monosaccharide (here fucose). The monomer is a b-sandwich composed of 8 antiparallel strands of which the five first are arranged in Greek key [5]. The principle implemented is gas phase diffusion. The protein complexed with various monosaccharides crystallises at p. H 8. 5 and at 20°C in the presence of ammonium sulphate at 1. 75 mol/L. Crystsllisation trials of protein complexed with trisaccharides of the Lea and Lex family have started and small crystals grow in the presence of PEG 5000 and cacodylate. PAII-L/Met- -D-Arabinoside PAII-L/L-Fucose PAII-L/L-Galactose The diffraction data collections were carried out at ESRF, Grenoble on beamlines ID 14 -1 and ID 14 -2, at 100 K. Phasing of PA-IIL/fuc structure was performed by the single-wavelength anomalous diffraction (SAD) method after soaking the crystal in holmium. After initial phasing by holmium SAD, high resolution (1. 0Å) data was phased using the calcium ion positions derived from the holmium and low resolution native data sets in ACORN to determine the initial phases. All other PA-IIL/monosaccharides structures were solved by molecular replacement with the use of the AMORE software. PAII-L/Lewis. A Diffraction image Crystal structures of the different complexes PAII-L/L-Fuc (1. 0Å) PAII-L/Me- -D-Ara (1. 8Å) PAII-L/L-Gal (1. 5Å) (MOLSCRIPT ) Tetramer PAII-L/fucose High resolution crystal structure Microcalorimetry: thermodynamics of interactions Heats for each injection of ligand -(m. J) Isothermal Titration Calorimetry (ITC) allows the determination of thermodynamic data of interaction, i. e. the contributions of entropy (DS) and enthalpy (DH) to the free energy (DG). Other characteristics such as the affinity constant (K) as well as the stoichiometry (n) of the interaction between the PA-IIL lectin and various monosaccharides have also been determined. Electron density map 2 Fo-Fc, s=2, Fo-Fc, s =3 , s= -3 m. J/moles of ligand time (s) Preliminary thermodynamics data for binding of monosacchardies to PA-IIL Data averaged on two experiments. Standard deviation is large. - Coordination - hydrogen bonds [ligand] Inhibition of PA-IIL binding to human blood group H and Leb active glycoprotein Inhibitor Sialyl Lewis a 3’Fucosyllactose L-Fucose Lewis x Sialyl Lewix x Relative potence 3. 5 2. 8 1. 4 1 0. 17 0. 13 The inhibitory activity was estimated from inhibition curves. Relative potency is the reciprocal of the relative inhibiting concentration when fucose was taken as 1. 0 Modelling of interaction with Lewis trisaccharides Starting from the position of the fucose on the site of connection, the trisaccharides Lewis x and Lewis a were docked in their conformation of lowest energy. The geometrical optimisation of the complex, including the side chains, were calculated with the Sybyl software using the energy parameters developed for protein/carbohydrate interactions [6]. [1] Scharfman, A. et al. (1999) Glycobiology 9, 757 -764. [2] Gilboa-Garber, N. , in Lectins Biology, Biochemistry, Clinical Biochemistry, Vol. 3 Bog-Hansen, T, Spengler, G. A. eds) Walter de Gruyter, Berlin, 1983, pp. 495 -502. [3] Gilboa-Garber, N. , Katcoff, D. J. & Garber, N. C. (2000) FEMS Immunol. Med. Microbiol. 29, 53 -57. Lewis x Lewis a Conclusions The stereochemical basis for the very high affinity of PAII-L for fucose is essentially due to the occurrence of the two calcium ions in the site. The nature of the group at C-5 of monosaccharides has no clear structural effect but modifies thermodynamics of binding. Molecular modelling together with binding studies indicates that the lectin has higher affinity for oliogosaccharides of the Lea series. Structural analysis of the binding site gives the basis for design of high affinity ligand that could serve as anti-adhesion drugs. [4] Garber, N. , Guempel, U. , Gilboa-Garber, N. & Doyle, R. J. (1987) FEMS Microbiol. Lett. 48, 331 -334. [5] E. Mitchell C. Houles, D. Sudakevitz, M. Wimmerova, C. Gautier, S. Pérez, A. M. Wu, N. Gilboa-Garber, A. Imberty. Nature Struct. Biol. 9 (2002) 918 -921. [6] Bettler, E. , Karababa, M. , Mazeau, K. , Petrova, P. & Pérez, S. , in Perspectives in Structural Biology (Vijayan, M. , Yathindra, N. , and Kolaskar, A. S. , eds) Indian Academy of Sciences and Universities Press, Hyderabad, 1999, pp. 392 -409.
cf4bd96c4e71632ddcf15596a72fb025.ppt