9cb16ee225071bca19ab449d431fb2d4.ppt
- Количество слайдов: 42

State of Global Rectal Microbicide Research Ian Mc. Gowan MD Ph. D FRCP Magee-Womens Research Institute University of Pittsburgh

Microbicides are products that can be applied to the vaginal or rectal mucosa with the intent of preventing or significantly reducing the risk of acquiring STIs including HIV
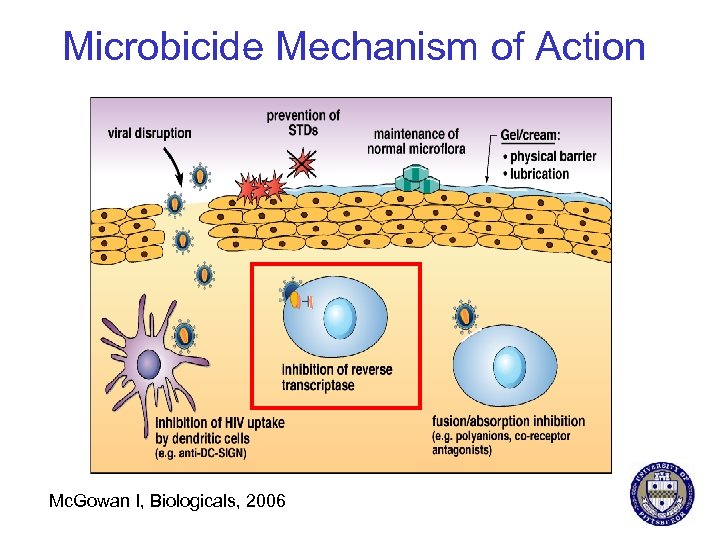
Microbicide Mechanism of Action Mc. Gowan I, Biologicals, 2006

Rationale for Rectal Microbicides § Unprotected receptive anal intercourse (RAI) is the highest risk sexual activity for HIV transmission § Men and women in the developed and developing world practice RAI § Murine and non human primate studies have shown proof of concept that rectal application of ARV microbicides can prevent SIV/HIV infection

Lubricants As a Drug Delivery System
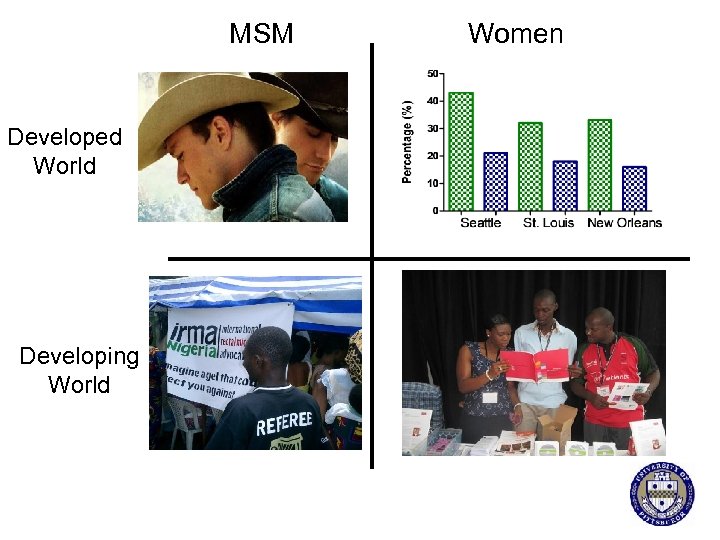
MSM Developed World Developing World Women

Clinical Development of Rectal Microbicides

Phase 1 Studies

Early Nonoxynol-9 Studies § Low-dose N-9 gel was not associated with macroscopic rectal and penile epithelial disruption or inflammation, but histologic abnormalities were commonly observed during N-9 gel as well as during placebo gel use. § Tabet S et al. Sex Trans Dis 1999 § 2% N-9 showed rapid exfoliation of the rectal epithelium. § Phillips D et al. Contraception 2004
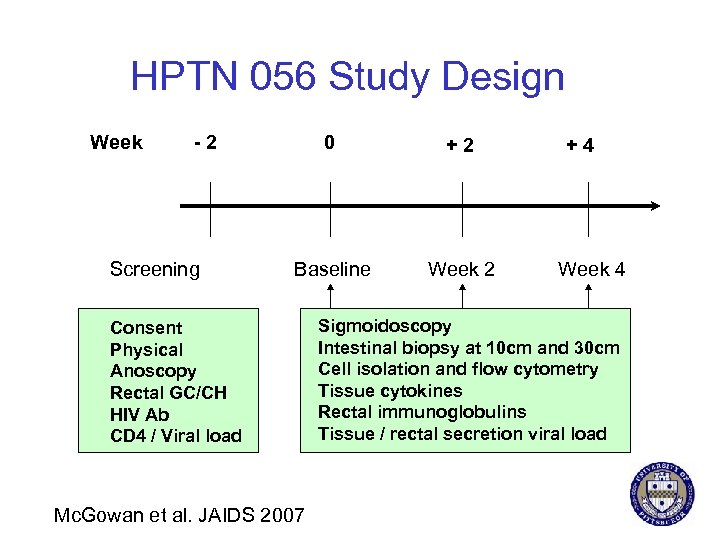
HPTN 056 Study Design Week -2 Screening 0 +2 Baseline Week 2 Consent Physical Anoscopy Rectal GC/CH HIV Ab CD 4 / Viral load Mc. Gowan et al. JAIDS 2007 +4 Week 4 Sigmoidoscopy Intestinal biopsy at 10 cm and 30 cm Cell isolation and flow cytometry Tissue cytokines Rectal immunoglobulins Tissue / rectal secretion viral load
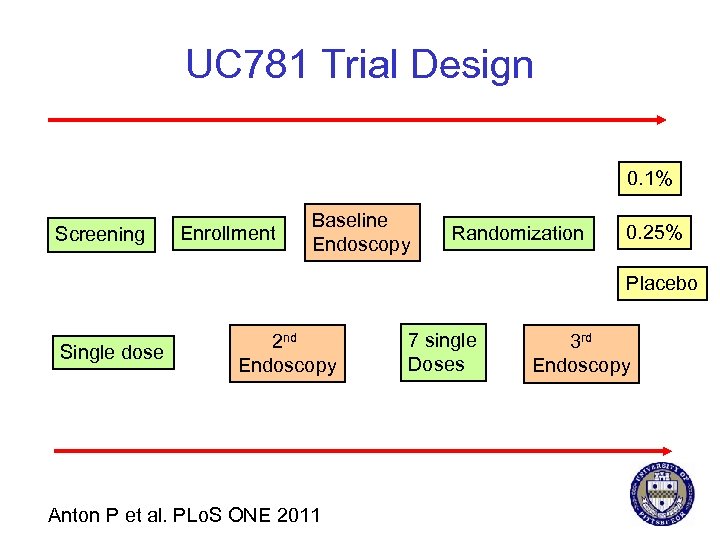
UC 781 Trial Design 0. 1% Screening Enrollment Baseline Endoscopy Randomization 0. 25% Placebo Single dose 2 nd Endoscopy Anton P et al. PLo. S ONE 2011 7 single Doses 3 rd Endoscopy
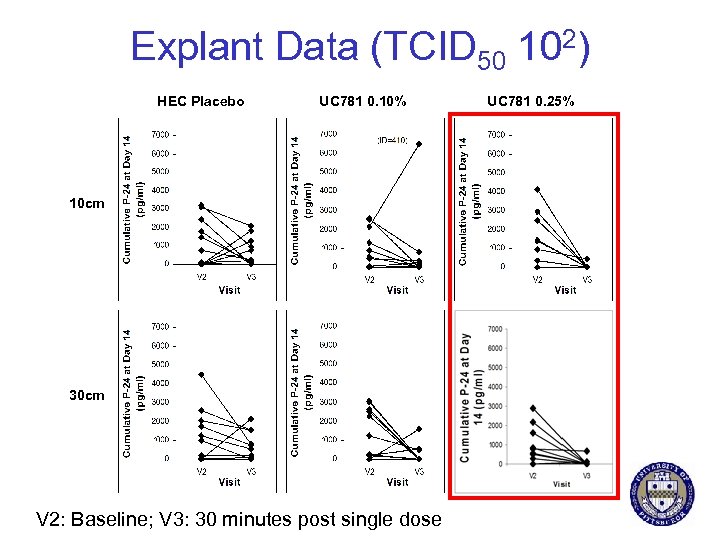
Explant Data (TCID 50 102) HEC Placebo UC 781 0. 10% 10 cm 30 cm V 2: Baseline; V 3: 30 minutes post single dose UC 781 0. 25%
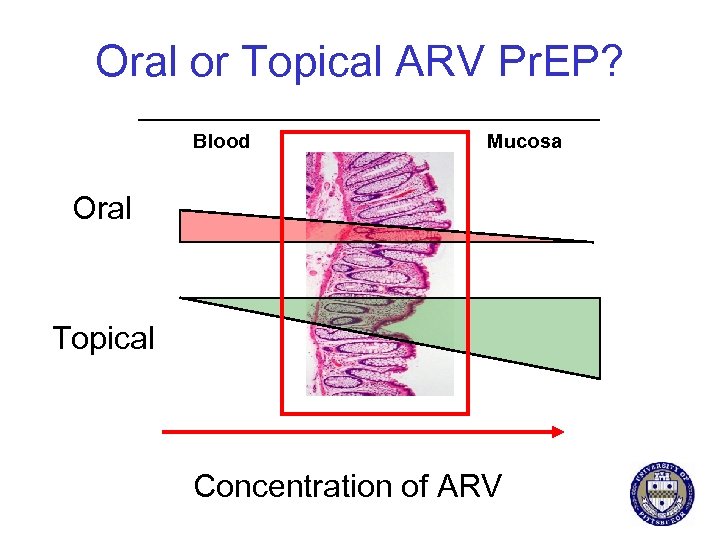
Oral or Topical ARV Pr. EP? Blood Mucosa Oral Topical Concentration of ARV
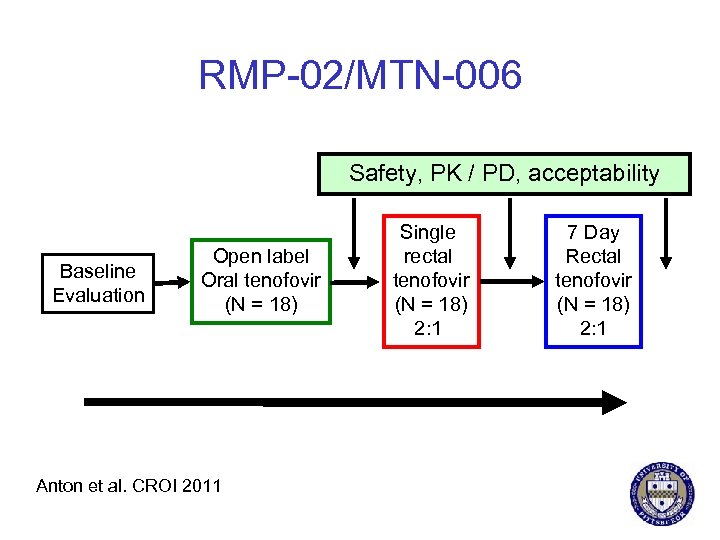
RMP-02/MTN-006 Safety, PK / PD, acceptability Baseline Evaluation Open label Oral tenofovir (N = 18) Anton et al. CROI 2011 Single rectal tenofovir (N = 18) 2: 1 7 Day Rectal tenofovir (N = 18) 2: 1
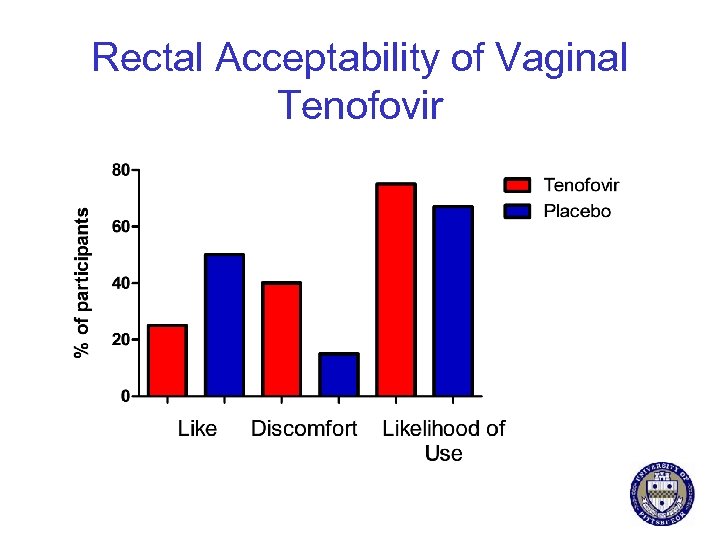
Rectal Acceptability of Vaginal Tenofovir
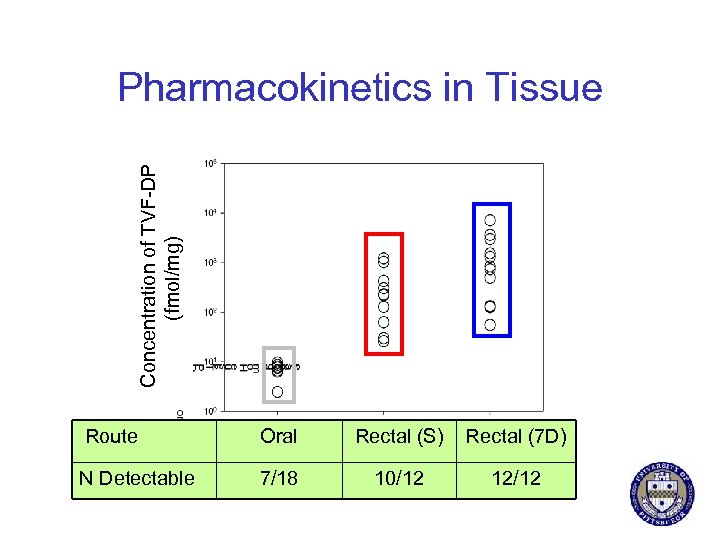
Concentration of TVF-DP (fmol/mg) Pharmacokinetics in Tissue Route Oral Rectal (S) Rectal (7 D) N Detectable 7/18 10/12 12/12
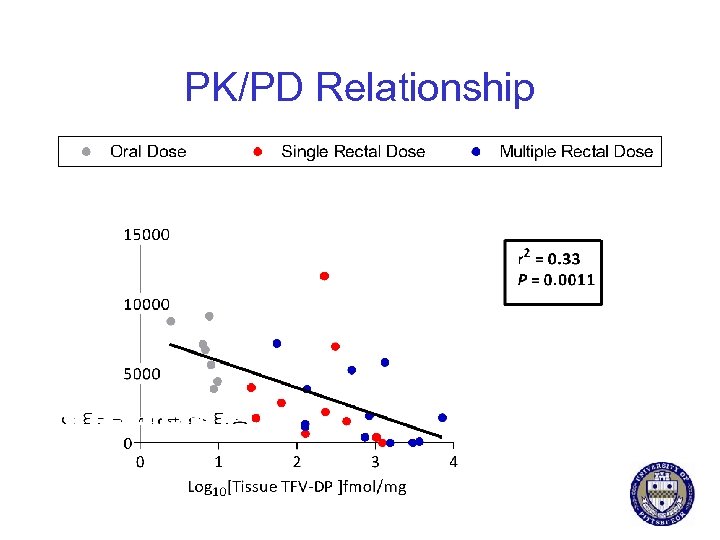
PK/PD Relationship
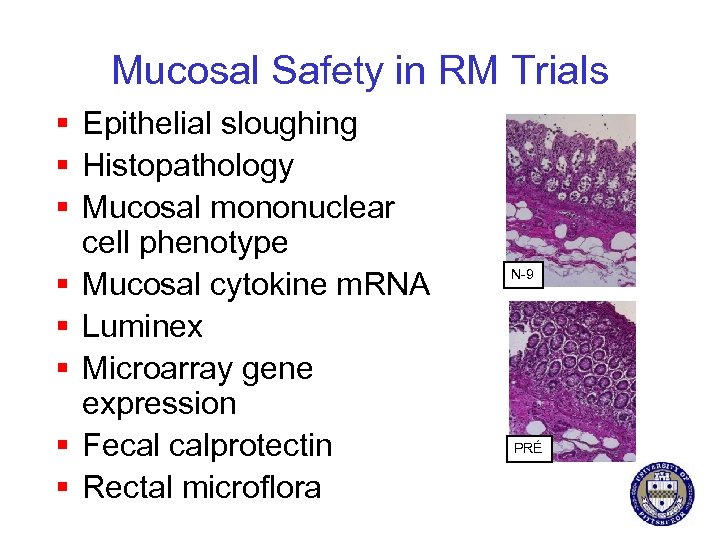
Mucosal Safety in RM Trials § Epithelial sloughing § Histopathology § Mucosal mononuclear cell phenotype § Mucosal cytokine m. RNA § Luminex § Microarray gene expression § Fecal calprotectin § Rectal microflora N-9 PRÉ
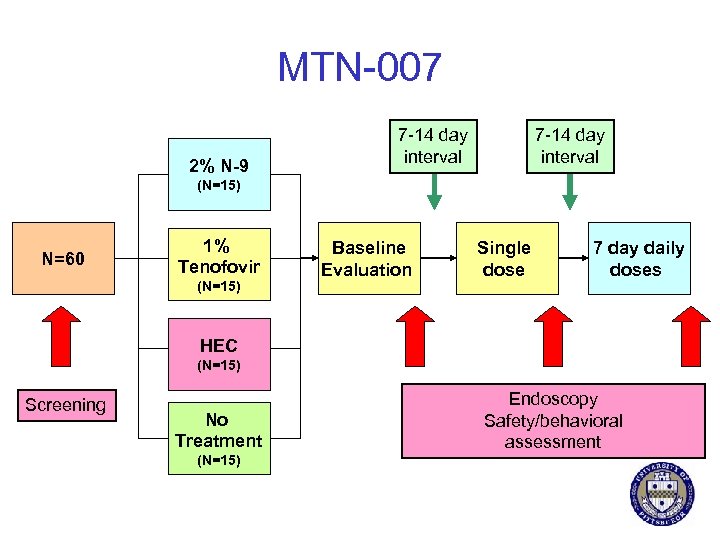
MTN-007 2% N-9 7 -14 day interval (N=15) N=60 1% Tenofovir (N=15) Baseline Evaluation Single dose 7 day daily doses HEC (N=15) Screening No Treatment (N=15) Endoscopy Safety/behavioral assessment

DAIDS Integrated Preclinical Clinical Program for HIV Topical Microbicides

Microbicide Development Program § First IPCP focusing on rectal microbicide development § Provided proof of concept in the SIV NHP model and development of explant platform § Phase 1 clinical trials of the vaginal formulation of tenofovir gel § UC 781 (RMP-01) § Tenofovir (RMP-02/MTN-006) § Behavioral correlates of RAI Anton: IPCP U 19 AI 060614 / August 2004

CHARM Program § Combination HIV Antiretroviral Rectal Microbicide Program § NIAID/DAIDS Integrated Preclinical Clinical Program § Consortium § § § University of Pittsburgh UCLA Johns Hopkins UNC CONRAD / Gilead Mc. Gowan: IPCP U 19 AI 082637 / September 2009

CHARM Program Overview § Development of rectal specific ARV microbicides § PK/PD evaluation in humanized mouse model § Phase 1 studies § Tenofovir § Maraviroc § Tenofovir & Maraviroc

CHARM-01 § Pre-Phase 1 single dose comparison of current formulations of tenofovir 1% gel: § Vaginal formulation § Reduced glycerin formulation § Rectal specific formulation § Endpoints § General and mucosal safety § PK/PD § Current status § Version 1. 0

CHARM-02 § Pre-Phase 1 single dose comparison of current formulations of tenofovir 1% gel with and without simulated RAI § Endpoints § Pharmacokinetics § Drug distribution using SPECT/CT imaging § Current status § Version 1. 0

Project Gel Mc. Gowan & Carballo-Dieguez: NICHD R 01 / September 2009

Microbicide Safety and Acceptability in Young Men § NICHD R 01 § Pittsburgh, Boston, Puerto Rico § Phase 1 safety and acceptability of tenofovir 1% gel § Ethnically diverse MSM (18 -30) § Consensual RAI in last month § Unprotected RAI in last year
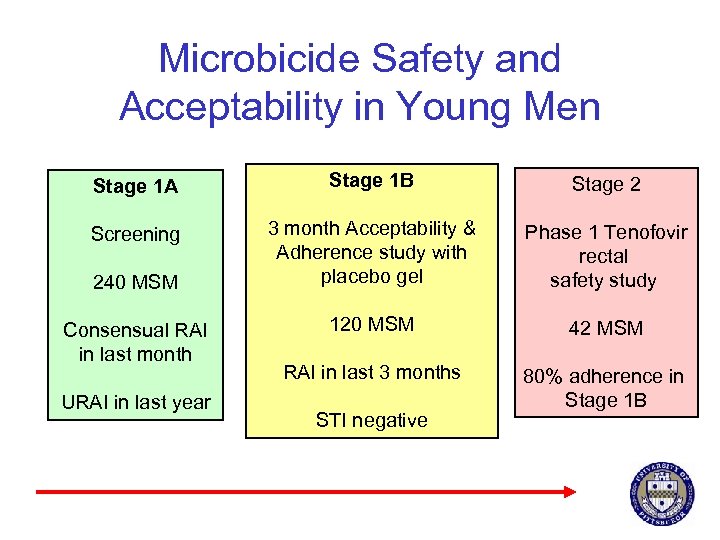
Microbicide Safety and Acceptability in Young Men Stage 1 A Stage 1 B Stage 2 Screening 3 month Acceptability & Adherence study with placebo gel Phase 1 Tenofovir rectal safety study 120 MSM 42 MSM RAI in last 3 months 80% adherence in Stage 1 B 240 MSM Consensual RAI in last month URAI in last year STI negative

Phase 2 Studies

MTN-017 § Phase 2 rectal safety study of tenofovir gel § N = 210 § International sites § § United States (3) Thailand (2) South Africa (1) Peru (1) § Endpoints § Safety § Adherence § Self report § Objective measures § Acceptability § PK/PD
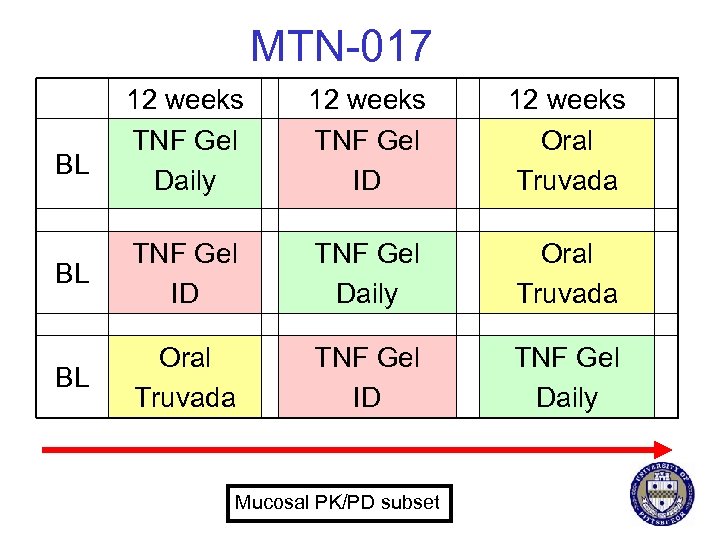
MTN-017 12 weeks TNF Gel Daily 12 weeks TNF Gel ID 12 weeks Oral Truvada BL TNF Gel ID TNF Gel Daily Oral Truvada BL Oral Truvada TNF Gel ID TNF Gel Daily BL Mucosal PK/PD subset

Phase 2 B/3 Studies
Combination Prevention Conventional HIV Prevention Package + Pr. EP SC ± Oral ± Rectal ± HIV Vaccine ± Vaginal
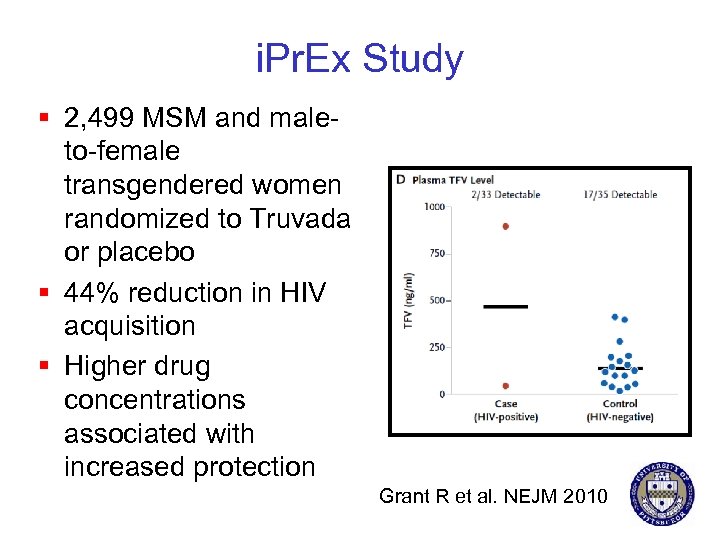
i. Pr. Ex Study § 2, 499 MSM and maleto-female transgendered women randomized to Truvada or placebo § 44% reduction in HIV acquisition § Higher drug concentrations associated with increased protection Grant R et al. NEJM 2010

TMC-278 LA § Rilpivirine NNRTI § IM Nanosuspension § Potential for 1 -3 month delivery § Phase 1 PK/PD studies ongoing § Colorectal explants § Cervicovaginal explants § MWRI-01* § University of Pittsburgh § Liverpool University *Funded by the Bill and Melinda Gates Foundation
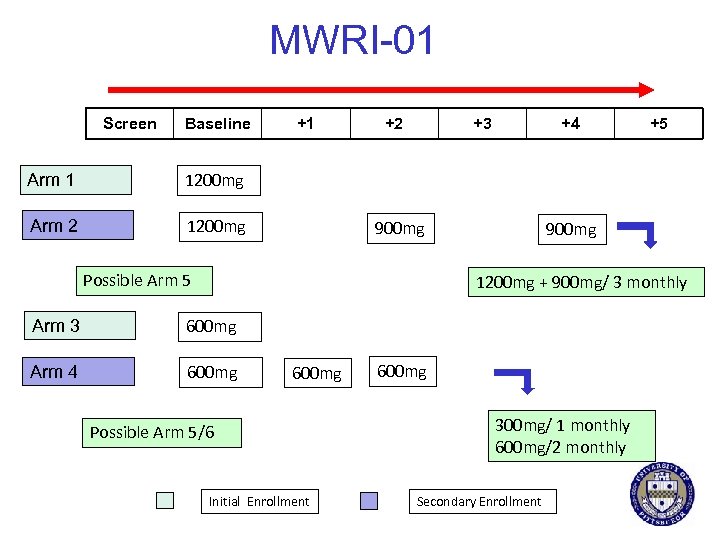
MWRI-01 Screen Baseline Arm 1 1200 mg +2 +3 +4 +5 1200 mg Arm 2 +1 900 mg Possible Arm 5 900 mg 1200 mg + 900 mg/ 3 monthly Arm 3 600 mg Arm 4 600 mg Possible Arm 5/6 Initial Enrollment 600 mg 300 mg/ 1 monthly 600 mg/2 monthly Secondary Enrollment

Effectiveness Study Designs § Option 1: Tenofovir gel versus placebo + standard prevention package § Option 2: Tenofovir gel versus placebo + standard prevention package + permission to use Pr. EP (HVTN 505) § Option 3: Tenofovir gel versus placebo + standard prevention package + Truvada § Option 4: Tenofovir gel versus Truvada versus TMC 278 LA + standard prevention package

Sample Size Parameters § Assumptions § § Incidence rate ~ 5% Effect size 60% Lower bound 25% Minimum FU 12 months § Enrollment 300/month § Pr. EP effect 46% § Endpoints: 120 § Option 1 § N = 2, 400; FU 24 months § Option 2 § N = 2, 400; FU 30 months § Option 3 § N = 2, 400; FU 37 months

The VOICE Trial § Both oral and topical tenofovir arms stopped for futility § Reasons for failure not yet known § Non adherence § Compartmental PK § Biology § Implications for rectal microbicide development?
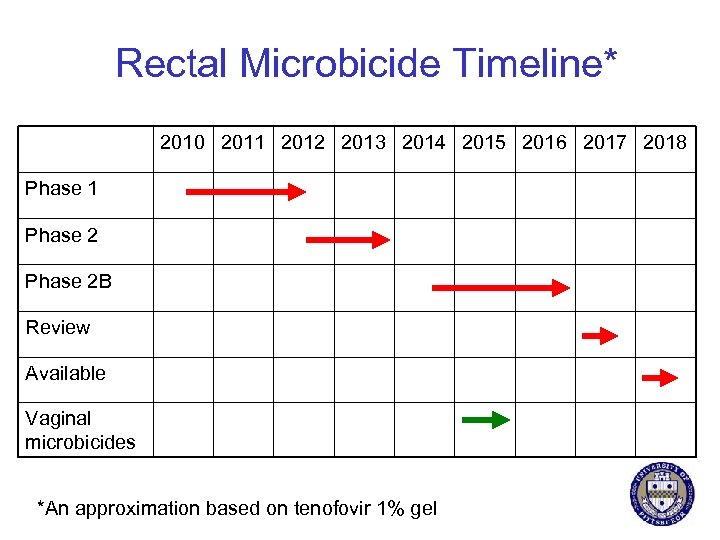
Rectal Microbicide Timeline* 2010 2011 2012 2013 2014 2015 2016 2017 2018 Phase 1 Phase 2 B Review Available Vaginal microbicides *An approximation based on tenofovir 1% gel

Summary § Rectal microbicides are needed for men and women in the developed and developing world who are at risk of HIV associated with unprotected RAI § RM development has moved from Phase 1 to Phase 2 § PK/PD models should increase likelihood of success in Phase 2 B/3 § Planning for an RM effectiveness study needs to start now.

Acknowledgements
9cb16ee225071bca19ab449d431fb2d4.ppt