8b92d611dfb8d2536ff6829f114a8dda.ppt
- Количество слайдов: 40
 Solar Thermal Fuel Production Christian Sattler 1, Hans Müller-Steinhagen 2, Martin Roeb 1, Dennis Thomey 1, Martina Neises 1 1 DLR Solar Research, Solar Chemical Engineering 2 Technical University of Dresden christian. sattler@dlr. de 1
Solar Thermal Fuel Production Christian Sattler 1, Hans Müller-Steinhagen 2, Martin Roeb 1, Dennis Thomey 1, Martina Neises 1 1 DLR Solar Research, Solar Chemical Engineering 2 Technical University of Dresden christian. sattler@dlr. de 1
 Overview Reasons for solar thermal fuel production Two examples SET-Plan Powertrains for Europe Concentrating Solar Systems Solar Fuels short and long term applications Processes Projects and existing pilot plants Summary and Outlook 2
Overview Reasons for solar thermal fuel production Two examples SET-Plan Powertrains for Europe Concentrating Solar Systems Solar Fuels short and long term applications Processes Projects and existing pilot plants Summary and Outlook 2
 Political view: SET-Plan (2007) European Strategic Plan for Energy Technology Development of energy technologies plays a crucial role for climate protection and the security of the global and European energy supply Goals of the EU until 2020 (20/20/20) 20% higher energy efficiency, 20% less GHG emission, , 20% renewable energy Actions in the field of energy efficiency, codes and standards, funding mechanisms, and the charging of carbon emissions necessary Significant research effort is necessary for the development of a new generation of CO 2 emission free energy technologies, like Offshore-Wind, Solar 2 nd generation Biomass Goal of the EU until 2050: 80% less CO 2 emissions than in 1990 3
Political view: SET-Plan (2007) European Strategic Plan for Energy Technology Development of energy technologies plays a crucial role for climate protection and the security of the global and European energy supply Goals of the EU until 2020 (20/20/20) 20% higher energy efficiency, 20% less GHG emission, , 20% renewable energy Actions in the field of energy efficiency, codes and standards, funding mechanisms, and the charging of carbon emissions necessary Significant research effort is necessary for the development of a new generation of CO 2 emission free energy technologies, like Offshore-Wind, Solar 2 nd generation Biomass Goal of the EU until 2050: 80% less CO 2 emissions than in 1990 3
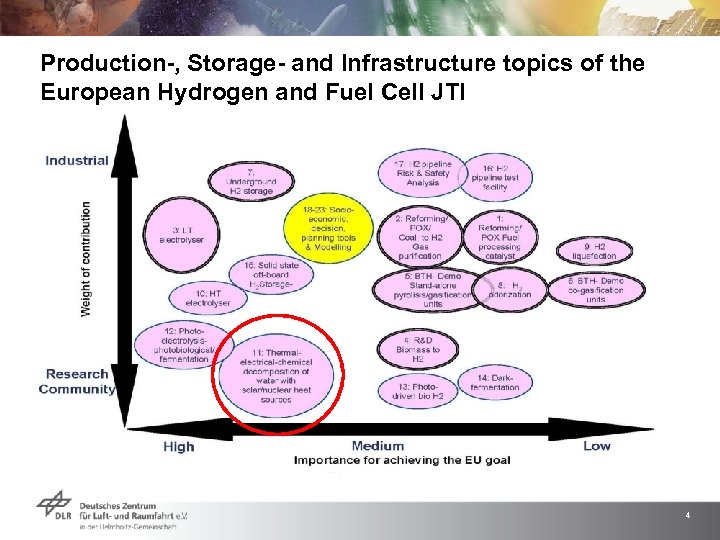 Production-, Storage- and Infrastructure topics of the European Hydrogen and Fuel Cell JTI 4
Production-, Storage- and Infrastructure topics of the European Hydrogen and Fuel Cell JTI 4
 Example for industrial view: „Powertrains for Europe“ 2010 fact based analysis on a portfolio of power-trains by Mc. Kinsey & Company for: Car manufacturers: BMW AG, Daimler AG, Ford, General Motors LLC, Honda R&D, Hyundai Motor Company, Kia Motors Corporation, Nissan, Renault, Toyota Motor Corporation, Volkswagen Oil and gas: ENI Refining and Marketing, Galp Energia, OMV Refining and Marketing Gmb. H, Shell Downstream Services International B. V. , Total Raffinage Marketing Utilities: En. BW Baden-Wuerttemberg AG, Vattenfall Industrial gas companies: Air Liquide, Air Products, The Linde Group Equipment car manufacturers: Intelligent Energy Holdings plc, Powertech Wind: Nordex Electrolyser companies: ELT Elektrolyse Technik, Hydrogenics, Hydrogen Technologies, Proton Energy Systems NGO: European Climate Foundation GOs: European Fuel Cells and Hydrogen Joint Undertaking, NOW Gmb. H Available online at: http: //ec. europa. eu/research/fch/index_en. cfm 5
Example for industrial view: „Powertrains for Europe“ 2010 fact based analysis on a portfolio of power-trains by Mc. Kinsey & Company for: Car manufacturers: BMW AG, Daimler AG, Ford, General Motors LLC, Honda R&D, Hyundai Motor Company, Kia Motors Corporation, Nissan, Renault, Toyota Motor Corporation, Volkswagen Oil and gas: ENI Refining and Marketing, Galp Energia, OMV Refining and Marketing Gmb. H, Shell Downstream Services International B. V. , Total Raffinage Marketing Utilities: En. BW Baden-Wuerttemberg AG, Vattenfall Industrial gas companies: Air Liquide, Air Products, The Linde Group Equipment car manufacturers: Intelligent Energy Holdings plc, Powertech Wind: Nordex Electrolyser companies: ELT Elektrolyse Technik, Hydrogenics, Hydrogen Technologies, Proton Energy Systems NGO: European Climate Foundation GOs: European Fuel Cells and Hydrogen Joint Undertaking, NOW Gmb. H Available online at: http: //ec. europa. eu/research/fch/index_en. cfm 5
![Development of EU GHG emissions [Gt CO 2 e] 6 Development of EU GHG emissions [Gt CO 2 e] 6](https://present5.com/presentation/8b92d611dfb8d2536ff6829f114a8dda/image-6.jpg) Development of EU GHG emissions [Gt CO 2 e] 6
Development of EU GHG emissions [Gt CO 2 e] 6
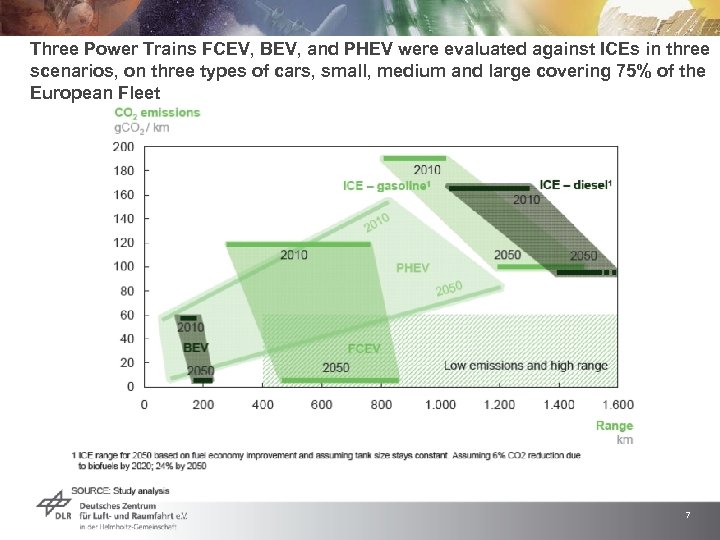 Three Power Trains FCEV, BEV, and PHEV were evaluated against ICEs in three scenarios, on three types of cars, small, medium and large covering 75% of the European Fleet 7
Three Power Trains FCEV, BEV, and PHEV were evaluated against ICEs in three scenarios, on three types of cars, small, medium and large covering 75% of the European Fleet 7
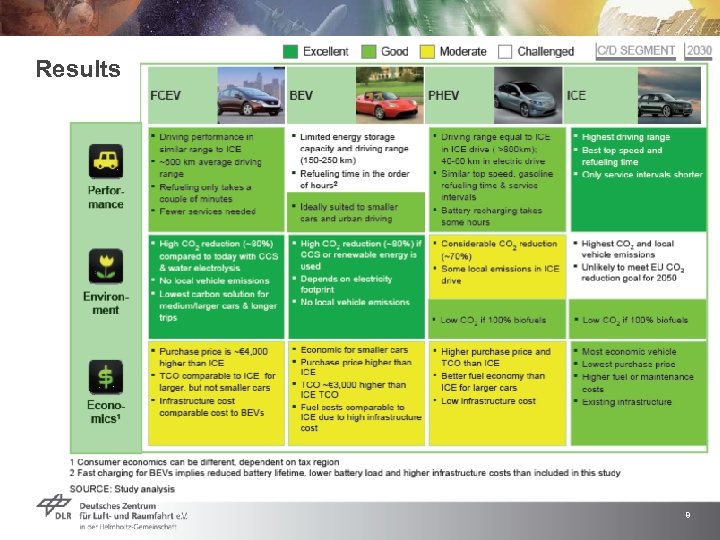 Results 8
Results 8
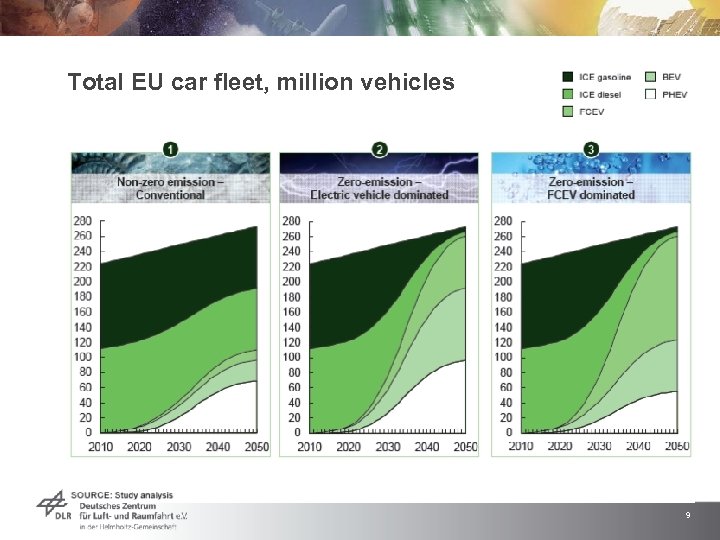 Total EU car fleet, million vehicles 9
Total EU car fleet, million vehicles 9
 Hydrogen production – benchmark processes for solar technologies 10
Hydrogen production – benchmark processes for solar technologies 10
 Concentrating Solar Technologies 11
Concentrating Solar Technologies 11
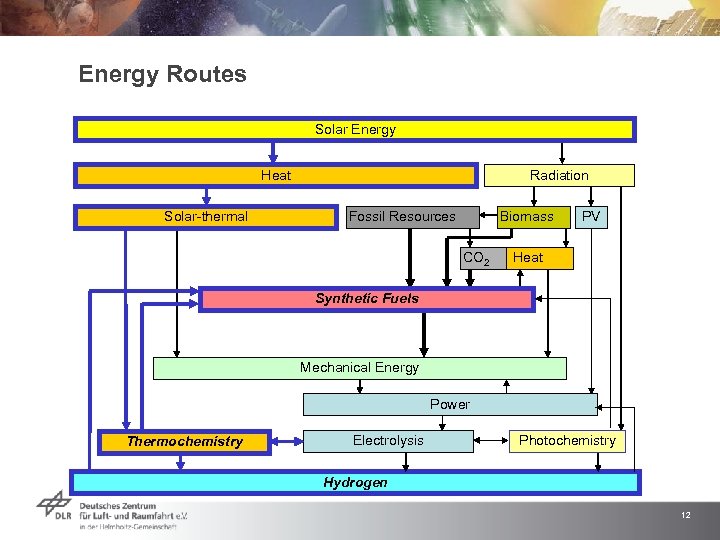 Energy Routes Solar Energy Heat Solar-thermal Radiation Fossil Resources Biomass PV CO 2 Heat Synthetic Fuels Mechanical Energy Power Thermochemistry Electrolysis Photochemistry Hydrogen 12
Energy Routes Solar Energy Heat Solar-thermal Radiation Fossil Resources Biomass PV CO 2 Heat Synthetic Fuels Mechanical Energy Power Thermochemistry Electrolysis Photochemistry Hydrogen 12
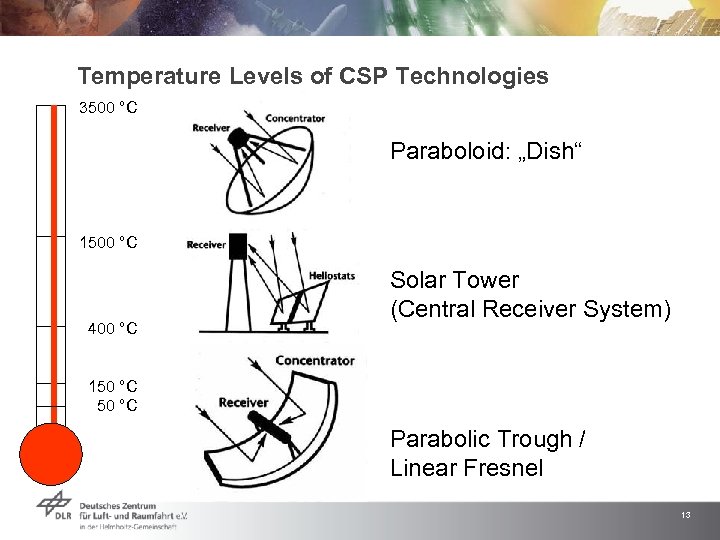 Temperature Levels of CSP Technologies 3500 °C Paraboloid: „Dish“ 1500 °C 400 °C Solar Tower (Central Receiver System) 150 °C Parabolic Trough / Linear Fresnel 13
Temperature Levels of CSP Technologies 3500 °C Paraboloid: „Dish“ 1500 °C 400 °C Solar Tower (Central Receiver System) 150 °C Parabolic Trough / Linear Fresnel 13
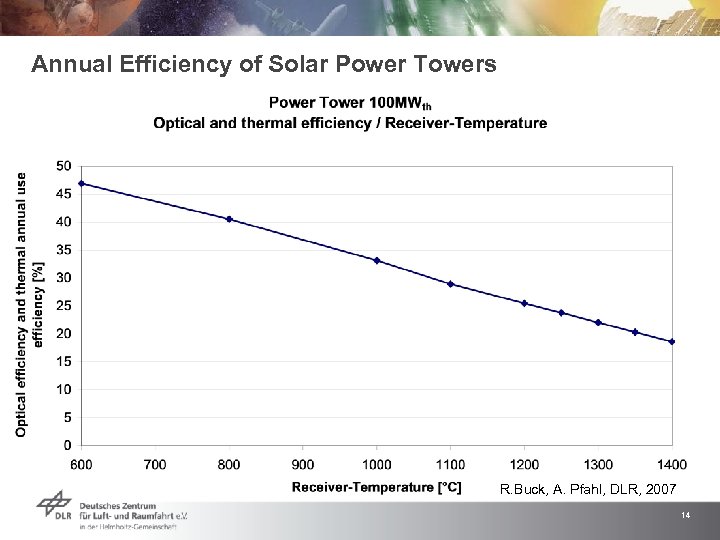 Annual Efficiency of Solar Power Towers R. Buck, A. Pfahl, DLR, 2007 14
Annual Efficiency of Solar Power Towers R. Buck, A. Pfahl, DLR, 2007 14
 Solar Towers, “Central Receiver Systems” PS 10+20, Sevilla, E PSA CESA-1, Almería, E Solar-Two, Daggett, USA Solarturm Jülich, D 15
Solar Towers, “Central Receiver Systems” PS 10+20, Sevilla, E PSA CESA-1, Almería, E Solar-Two, Daggett, USA Solarturm Jülich, D 15
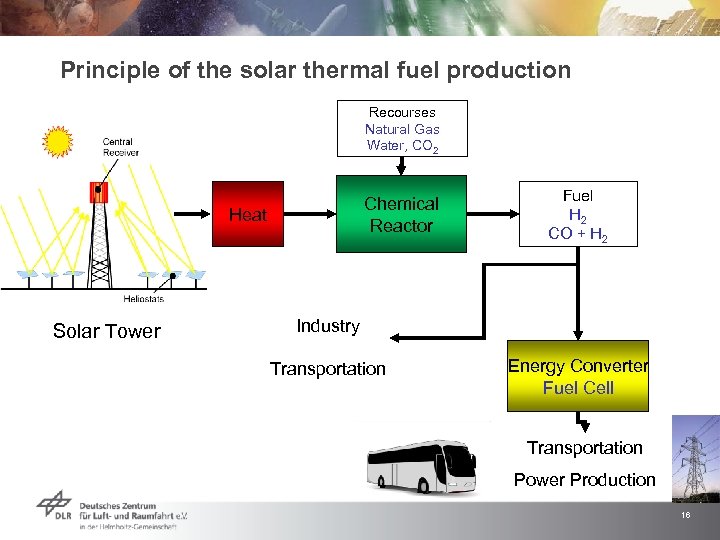 Principle of the solar thermal fuel production Recourses Natural Gas Water, CO 2 Chemical Reactor Heat Solar Tower Fuel H 2 CO + H 2 Industry Transportation Energy Converter Fuel Cell Transportation Power Production 16
Principle of the solar thermal fuel production Recourses Natural Gas Water, CO 2 Chemical Reactor Heat Solar Tower Fuel H 2 CO + H 2 Industry Transportation Energy Converter Fuel Cell Transportation Power Production 16
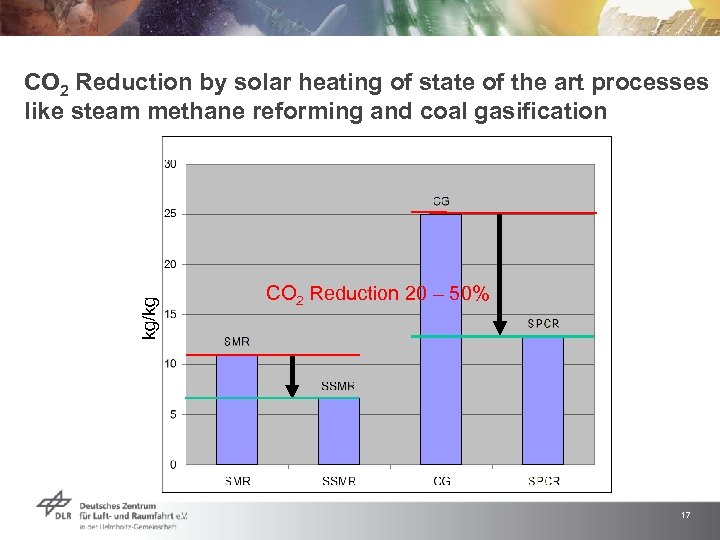 kg/kg CO 2 Reduction by solar heating of state of the art processes like steam methane reforming and coal gasification CO 2 Reduction 20 – 50% 17
kg/kg CO 2 Reduction by solar heating of state of the art processes like steam methane reforming and coal gasification CO 2 Reduction 20 – 50% 17
![Efficiency comparison for solar hydrogen production from water (SANDIA, 2008)* Process T [°C] Solar Efficiency comparison for solar hydrogen production from water (SANDIA, 2008)* Process T [°C] Solar](https://present5.com/presentation/8b92d611dfb8d2536ff6829f114a8dda/image-18.jpg) Efficiency comparison for solar hydrogen production from water (SANDIA, 2008)* Process T [°C] Solar plant Solarreceiver + power [MWth] η T/C (HHV) η Optical η Receiver η Annual Efficiency Solar – H 2 Elctrolysis (+solarthermal power) NA Actual Solar tower Molten Salt 700 30% 57% 83% 14% High temperature steam electrolysis 850 Future Solar tower Particle 700 45% 57% 76, 2% 20% Hybrid Sulfurprocess 850 Future Solar tower Particle 700 51% 57% 76% 22% Hybrid Copper Chlorine-process 600 Future Solar tower Molten Salt 700 49% 57% 83% 23% Nickel Manganese Ferrit Process 1800 Future Solar dish Rotating Disc < 1 52% 77% 62% 25% *G. J. Kolb, R. B. Diver SAND 2008 -1900 18
Efficiency comparison for solar hydrogen production from water (SANDIA, 2008)* Process T [°C] Solar plant Solarreceiver + power [MWth] η T/C (HHV) η Optical η Receiver η Annual Efficiency Solar – H 2 Elctrolysis (+solarthermal power) NA Actual Solar tower Molten Salt 700 30% 57% 83% 14% High temperature steam electrolysis 850 Future Solar tower Particle 700 45% 57% 76, 2% 20% Hybrid Sulfurprocess 850 Future Solar tower Particle 700 51% 57% 76% 22% Hybrid Copper Chlorine-process 600 Future Solar tower Molten Salt 700 49% 57% 83% 23% Nickel Manganese Ferrit Process 1800 Future Solar dish Rotating Disc < 1 52% 77% 62% 25% *G. J. Kolb, R. B. Diver SAND 2008 -1900 18
 Short-term CO 2 -Reduction: Solar Reforming 19
Short-term CO 2 -Reduction: Solar Reforming 19
 Steam and CO 2 -Reforming of Natural Gas Steam reforming: H 2 O + CH 4 3 H 2 + 1 CO CO 2 Reforming: CO 2 + CH 4 2 H 2 + 2 CO Reforming of mixtures of CO 2/H 2 O is possible and common Use of CO 2 for methanol production: e. g. 2 H 2 + CO CH 3 COH (Methanol) Both technologies can be driven by solar energy as shown in the projects: CAESAR, ASTERIX, SOLASYS, SOLREF… 20
Steam and CO 2 -Reforming of Natural Gas Steam reforming: H 2 O + CH 4 3 H 2 + 1 CO CO 2 Reforming: CO 2 + CH 4 2 H 2 + 2 CO Reforming of mixtures of CO 2/H 2 O is possible and common Use of CO 2 for methanol production: e. g. 2 H 2 + CO CH 3 COH (Methanol) Both technologies can be driven by solar energy as shown in the projects: CAESAR, ASTERIX, SOLASYS, SOLREF… 20
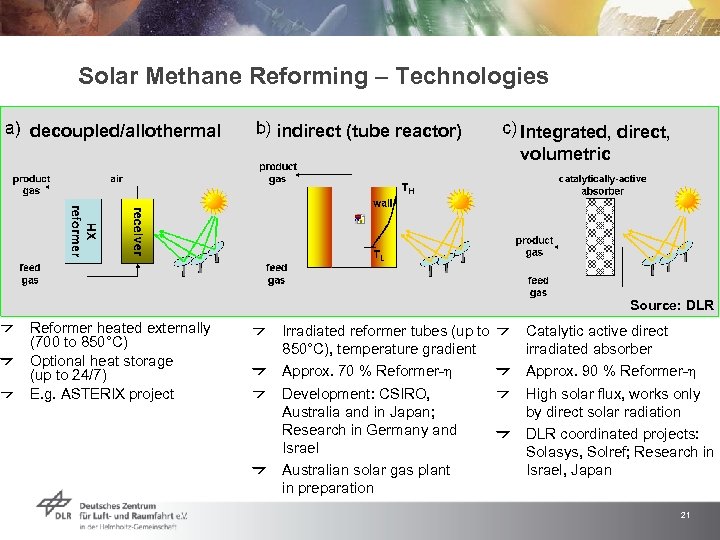 Solar Methane Reforming – Technologies decoupled/allothermal indirect (tube reactor) Integrated, direct, volumetric Source: DLR Reformer heated externally (700 to 850°C) Optional heat storage (up to 24/7) E. g. ASTERIX project Irradiated reformer tubes (up to 850°C), temperature gradient Approx. 70 % Reformer-h Development: CSIRO, Australia and in Japan; Research in Germany and Israel Australian solar gas plant in preparation Catalytic active direct irradiated absorber Approx. 90 % Reformer-h High solar flux, works only by direct solar radiation DLR coordinated projects: Solasys, Solref; Research in Israel, Japan 21
Solar Methane Reforming – Technologies decoupled/allothermal indirect (tube reactor) Integrated, direct, volumetric Source: DLR Reformer heated externally (700 to 850°C) Optional heat storage (up to 24/7) E. g. ASTERIX project Irradiated reformer tubes (up to 850°C), temperature gradient Approx. 70 % Reformer-h Development: CSIRO, Australia and in Japan; Research in Germany and Israel Australian solar gas plant in preparation Catalytic active direct irradiated absorber Approx. 90 % Reformer-h High solar flux, works only by direct solar radiation DLR coordinated projects: Solasys, Solref; Research in Israel, Japan 21
 Project Asterix: Allothermal Steam Reforming of Methan DLR, Steinmüller, CIEMAT 180 k. W plant at the Plataforma Solar de Almería, Spain (1990) Convective heated tube cracker as reformer Tubular receiver for air heating 22
Project Asterix: Allothermal Steam Reforming of Methan DLR, Steinmüller, CIEMAT 180 k. W plant at the Plataforma Solar de Almería, Spain (1990) Convective heated tube cracker as reformer Tubular receiver for air heating 22
 “Indirect heated“ tube receiver: CSIRO Solargas Indirect reactor technology Second tower at the CSIRO Solar Centre Newcastle, NSW, Australia Test facility for different Reactors One will be the volumetric SOLREF reactor Coordination by CSIRO, DLR is partner in an IPHE project 23
“Indirect heated“ tube receiver: CSIRO Solargas Indirect reactor technology Second tower at the CSIRO Solar Centre Newcastle, NSW, Australia Test facility for different Reactors One will be the volumetric SOLREF reactor Coordination by CSIRO, DLR is partner in an IPHE project 23
 Direct heated volumetric receivers: SOLASYS, SOLREF (EU FP 4, FP 6) Pressurised solar receiver, Developed by DLR Tested at the Weizmann Institute of Science, Israel Power coupled into the process gas: 220 k. Wth and 400 k. Wth Reforming temperature: between 765°C and 1000°C Pressure: SOLASYS 9 bar, SOLREF 15 bar Methane Conversion: max. 78 % (= theor. balance) DLR (D), WIS (IL), ETH (CH), Johnson Matthey (UK), APTL (GR), HYGEAR (NL), SHAP (I) 24
Direct heated volumetric receivers: SOLASYS, SOLREF (EU FP 4, FP 6) Pressurised solar receiver, Developed by DLR Tested at the Weizmann Institute of Science, Israel Power coupled into the process gas: 220 k. Wth and 400 k. Wth Reforming temperature: between 765°C and 1000°C Pressure: SOLASYS 9 bar, SOLREF 15 bar Methane Conversion: max. 78 % (= theor. balance) DLR (D), WIS (IL), ETH (CH), Johnson Matthey (UK), APTL (GR), HYGEAR (NL), SHAP (I) 24
 Pilot plant for solar pet-coke reformig - SYNPET 500 k. W SYNPET solar reactor Plataforma Solar de Almería Production: 100 -180 kg/h Synthesis gas CIEMAT (E), ETH (CH), PDVESA (VEN) T Denk et al. , CIEMAT, 2009 25
Pilot plant for solar pet-coke reformig - SYNPET 500 k. W SYNPET solar reactor Plataforma Solar de Almería Production: 100 -180 kg/h Synthesis gas CIEMAT (E), ETH (CH), PDVESA (VEN) T Denk et al. , CIEMAT, 2009 25
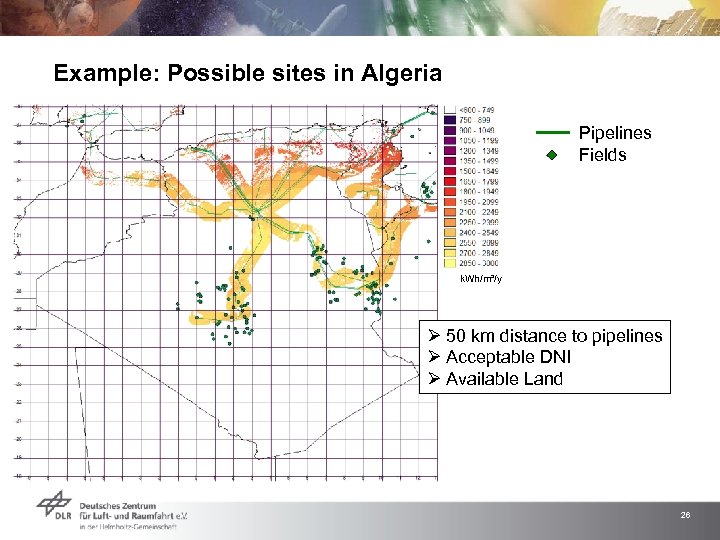 Example: Possible sites in Algeria Pipelines Fields k. Wh/m²/y Ø 50 km distance to pipelines Ø Acceptable DNI Ø Available Land 26
Example: Possible sites in Algeria Pipelines Fields k. Wh/m²/y Ø 50 km distance to pipelines Ø Acceptable DNI Ø Available Land 26
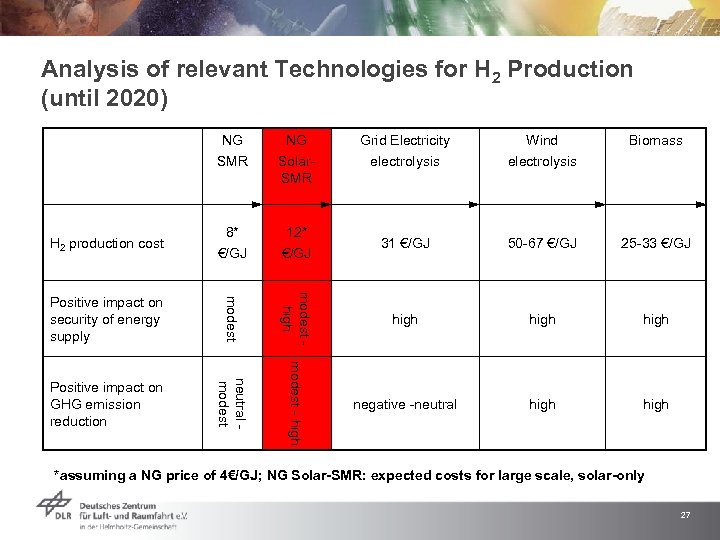 Analysis of relevant Technologies for H 2 Production (until 2020) NG NG Grid Electricity Wind SMR Solar. SMR electrolysis H 2 production cost 8* €/GJ 12* €/GJ 31 €/GJ 50 -67 €/GJ 25 -33 €/GJ Positive impact on security of energy supply modest high Positive impact on GHG emission reduction modest - high neutral - modest Biomass negative -neutral high *assuming a NG price of 4€/GJ; NG Solar-SMR: expected costs for large scale, solar-only 27
Analysis of relevant Technologies for H 2 Production (until 2020) NG NG Grid Electricity Wind SMR Solar. SMR electrolysis H 2 production cost 8* €/GJ 12* €/GJ 31 €/GJ 50 -67 €/GJ 25 -33 €/GJ Positive impact on security of energy supply modest high Positive impact on GHG emission reduction modest - high neutral - modest Biomass negative -neutral high *assuming a NG price of 4€/GJ; NG Solar-SMR: expected costs for large scale, solar-only 27
 Long-term: Water splitting processes 28
Long-term: Water splitting processes 28
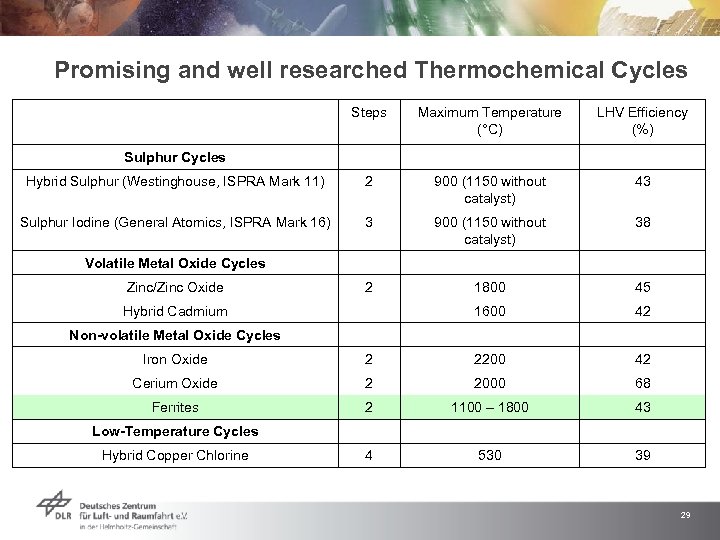 Promising and well researched Thermochemical Cycles Steps Maximum Temperature (°C) LHV Efficiency (%) Sulphur Cycles Hybrid Sulphur (Westinghouse, ISPRA Mark 11) 2 900 (1150 without catalyst) 43 Sulphur Iodine (General Atomics, ISPRA Mark 16) 3 900 (1150 without catalyst) 38 Volatile Metal Oxide Cycles Zinc/Zinc Oxide 2 1800 45 Hybrid Cadmium 1600 42 Non-volatile Metal Oxide Cycles Iron Oxide 2 2200 42 Cerium Oxide 2 2000 68 Ferrites 2 1100 – 1800 43 Low-Temperature Cycles Hybrid Copper Chlorine 4 530 39 29
Promising and well researched Thermochemical Cycles Steps Maximum Temperature (°C) LHV Efficiency (%) Sulphur Cycles Hybrid Sulphur (Westinghouse, ISPRA Mark 11) 2 900 (1150 without catalyst) 43 Sulphur Iodine (General Atomics, ISPRA Mark 16) 3 900 (1150 without catalyst) 38 Volatile Metal Oxide Cycles Zinc/Zinc Oxide 2 1800 45 Hybrid Cadmium 1600 42 Non-volatile Metal Oxide Cycles Iron Oxide 2 2200 42 Cerium Oxide 2 2000 68 Ferrites 2 1100 – 1800 43 Low-Temperature Cycles Hybrid Copper Chlorine 4 530 39 29
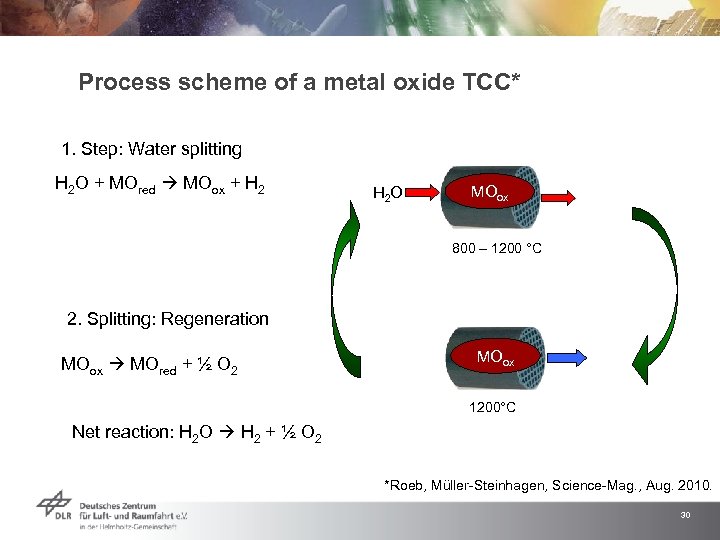 Process scheme of a metal oxide TCC* 1. Step: Water splitting H 2 O + MOred MOox + H 2 O MOred MOox H O 2 800 – 1200 °C 2. Splitting: Regeneration MOox MOred + ½ O 2 MOox O 2 MOred 1200°C Net reaction: H 2 O H 2 + ½ O 2 *Roeb, Müller-Steinhagen, Science-Mag. , Aug. 2010. 30
Process scheme of a metal oxide TCC* 1. Step: Water splitting H 2 O + MOred MOox + H 2 O MOred MOox H O 2 800 – 1200 °C 2. Splitting: Regeneration MOox MOred + ½ O 2 MOox O 2 MOred 1200°C Net reaction: H 2 O H 2 + ½ O 2 *Roeb, Müller-Steinhagen, Science-Mag. , Aug. 2010. 30
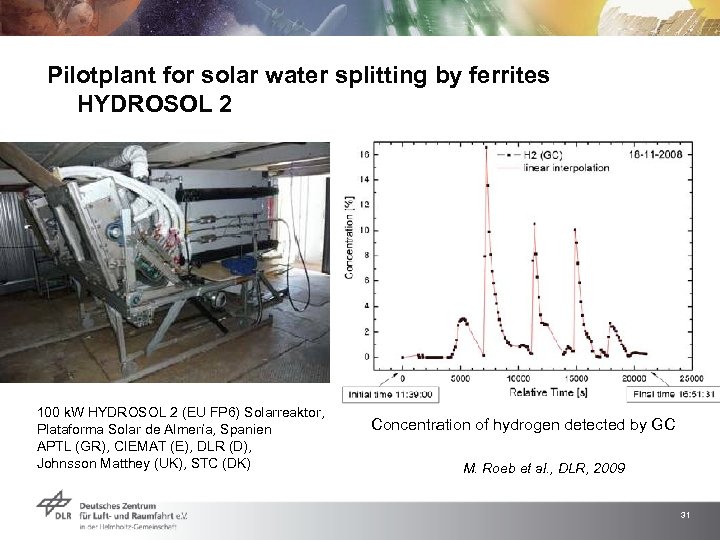 Pilotplant for solar water splitting by ferrites HYDROSOL 2 100 k. W HYDROSOL 2 (EU FP 6) Solarreaktor, Plataforma Solar de Almería, Spanien APTL (GR), CIEMAT (E), DLR (D), Johnsson Matthey (UK), STC (DK) Concentration of hydrogen detected by GC M. Roeb et al. , DLR, 2009 31
Pilotplant for solar water splitting by ferrites HYDROSOL 2 100 k. W HYDROSOL 2 (EU FP 6) Solarreaktor, Plataforma Solar de Almería, Spanien APTL (GR), CIEMAT (E), DLR (D), Johnsson Matthey (UK), STC (DK) Concentration of hydrogen detected by GC M. Roeb et al. , DLR, 2009 31
 Scale-up: 100 k. W-pilot-plant 32
Scale-up: 100 k. W-pilot-plant 32
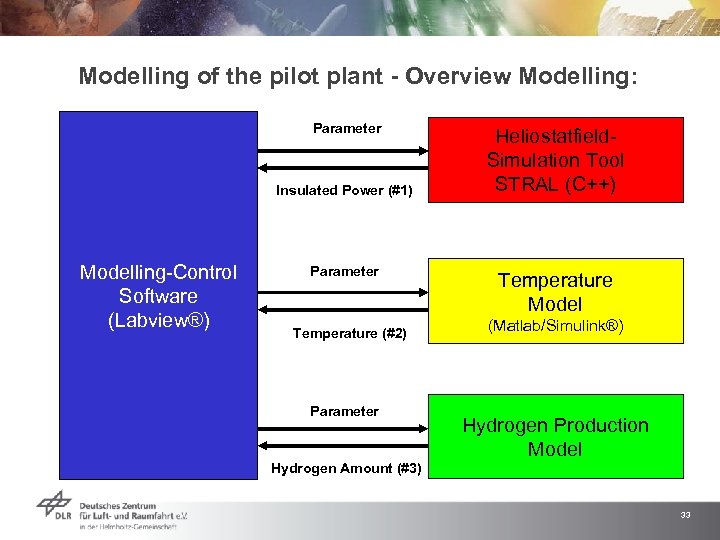 Modelling of the pilot plant - Overview Modelling: Parameter Insulated Power (#1) Modelling-Control Software (Labview®) Parameter Temperature (#2) Parameter Heliostatfield. Simulation Tool STRAL (C++) Temperature Model (Matlab/Simulink®) Hydrogen Production Model Hydrogen Amount (#3) 33
Modelling of the pilot plant - Overview Modelling: Parameter Insulated Power (#1) Modelling-Control Software (Labview®) Parameter Temperature (#2) Parameter Heliostatfield. Simulation Tool STRAL (C++) Temperature Model (Matlab/Simulink®) Hydrogen Production Model Hydrogen Amount (#3) 33
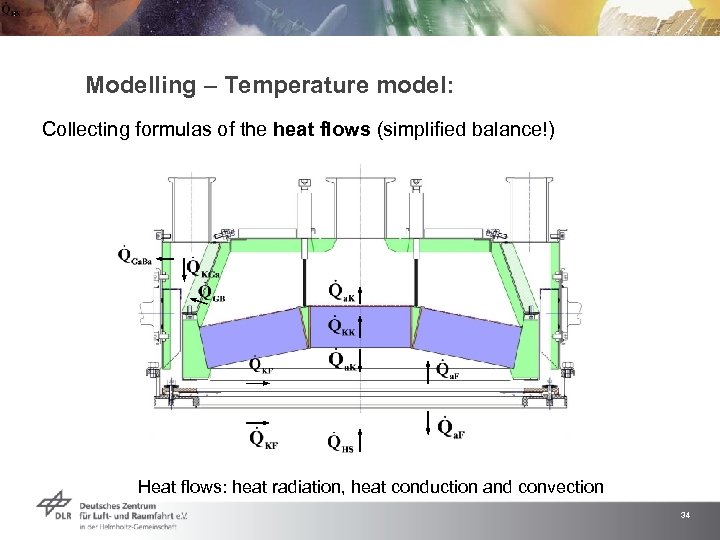 Modelling – Temperature model: Collecting formulas of the heat flows (simplified balance!) Heat flows: heat radiation, heat conduction and convection 34
Modelling – Temperature model: Collecting formulas of the heat flows (simplified balance!) Heat flows: heat radiation, heat conduction and convection 34
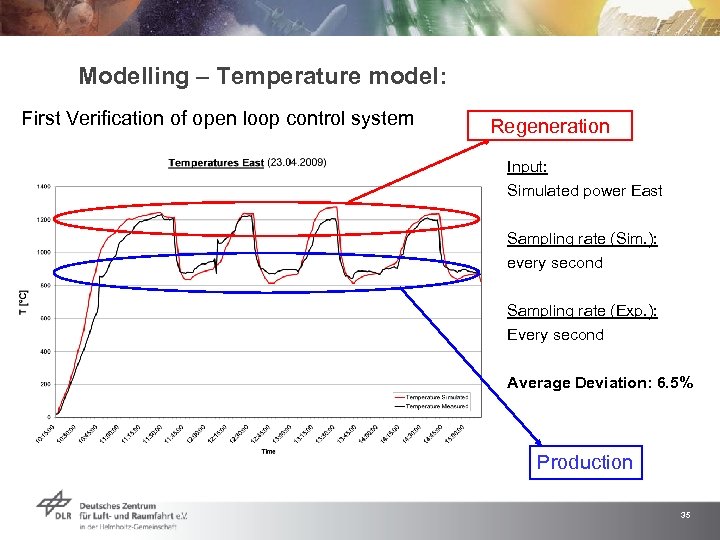 Modelling – Temperature model: First Verification of open loop control system Regeneration Input: Simulated power East Sampling rate (Sim. ): every second Sampling rate (Exp. ): Every second Average Deviation: 6. 5% Production 35
Modelling – Temperature model: First Verification of open loop control system Regeneration Input: Simulated power East Sampling rate (Sim. ): every second Sampling rate (Exp. ): Every second Average Deviation: 6. 5% Production 35
 Conclusion and Outlook 36
Conclusion and Outlook 36
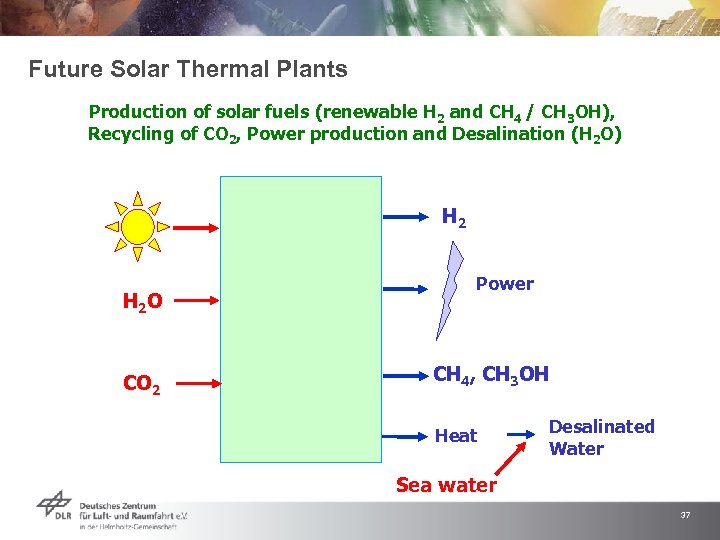 Future Solar Thermal Plants Production of solar fuels (renewable H 2 and CH 4 / CH 3 OH), Recycling of CO 2, Power production and Desalination (H 2 O) H 2 O CO 2 Power CH 4, CH 3 OH Heat Desalinated Water Sea water 37
Future Solar Thermal Plants Production of solar fuels (renewable H 2 and CH 4 / CH 3 OH), Recycling of CO 2, Power production and Desalination (H 2 O) H 2 O CO 2 Power CH 4, CH 3 OH Heat Desalinated Water Sea water 37
 Conclusion and Outlook CO 2 lean/free hydrogen is crucial for the energy economy no matter how the development will be To achieve the energy/emission goals for 2020 promising renewable technologies like solar thermal must be implemented now, at the right places Things to be done: Secure and enhance the know-how by strong co-operations of industry and R&D Close technological gaps Transfer of the technology to industry Provide technology for growing markets in solar regions 38
Conclusion and Outlook CO 2 lean/free hydrogen is crucial for the energy economy no matter how the development will be To achieve the energy/emission goals for 2020 promising renewable technologies like solar thermal must be implemented now, at the right places Things to be done: Secure and enhance the know-how by strong co-operations of industry and R&D Close technological gaps Transfer of the technology to industry Provide technology for growing markets in solar regions 38
 Acknowledgment The Projects HYDROSOL, HYDROSOL II; HYTHEC, HYCYCLES, Hi 2 H 2, INNOHYP-CA, SOLHYCARB and SOLREF were co-financed by the European Commission HYDROSOL 3 -D and ADEL are co-financed by the European Joint Technology Initiative on Hydrogen and Fuel Cells HYDROSOL was awarded Eco Tech Award Expo 2005, Tokyo IPHE Technical Achievement Award 2006 Descartes Research Price 2006 39
Acknowledgment The Projects HYDROSOL, HYDROSOL II; HYTHEC, HYCYCLES, Hi 2 H 2, INNOHYP-CA, SOLHYCARB and SOLREF were co-financed by the European Commission HYDROSOL 3 -D and ADEL are co-financed by the European Joint Technology Initiative on Hydrogen and Fuel Cells HYDROSOL was awarded Eco Tech Award Expo 2005, Tokyo IPHE Technical Achievement Award 2006 Descartes Research Price 2006 39
 Mahalo for your attention! 40
Mahalo for your attention! 40


