35d4faf2f4a85e6c1c4ae0c0bbb82c76.ppt
- Количество слайдов: 117


Social Media “The use of web-based technology to facilitate interaction with others. ”

ARS Question: With the exception of e-mail, how often do you interact with peers online through social networking sites like Facebook & Twitter? 1) Always 2) Very Often 3) Sometimes 4) Rarely 5) Never

ARS Question: How likely are you to use social media to help increase public awareness of chronic hepatitis C? 1) Very likely 2) Likely 3) Maybe 4) Probably not 5) Never

ARS Question: In patients who present with non liver related complaints, how often do you evaluate a patient’s risk factors for chronic hepatitis C infection? 1) Always 2) Very Often 3) Sometimes 4) Rarely 5) Never

ARS Question: Among all your genotype 1 patients who are eligible for current therapy, how often are you withholding therapy while waiting for novel drugs? 1) Always 2) Very Often 3) Sometimes 4) Rarely 5) Never

ARS Question: Among your genotype 1 patients, how frequently will you be incorporating novel agents once they are approved? 1) Always 2) Very Often 3) Sometimes 4) Rarely 5) Never

How Will Social Media Impact Your Practice? Bryan S. Vartabedian, MD, FAAP Assistant Professor of Pediatrics Section of Gastroenterology, Hepatology, & Nutrition Baylor College of Medicine Houston, Texas

How Health Information Was Shared MD Patient Graphic courtesy of Dr. Bryan Vartabedian.

Evolution of Social Health Now Patients find each other 2000 s Information finds patients 1990 s Patients find information Long ago Patients depend on what they’re told Graphic courtesy of Dr. Bryan Vartabedian.
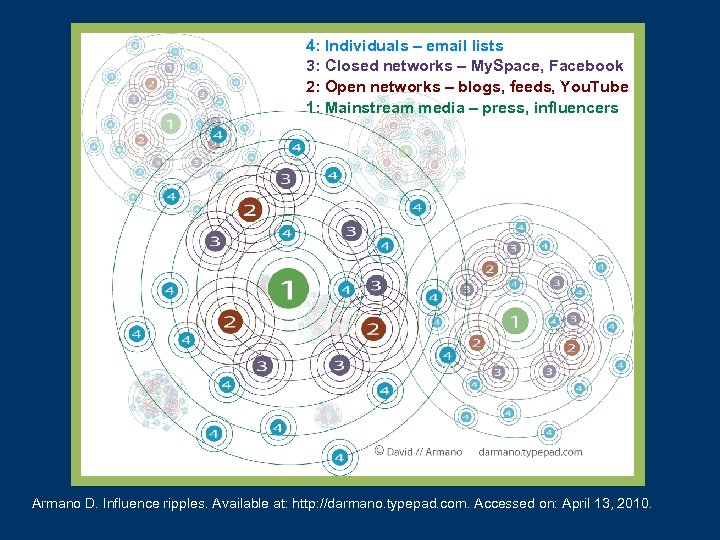
4: Individuals – email lists 3: Closed networks – My. Space, Facebook 2: Open networks – blogs, feeds, You. Tube 1: Mainstream media – press, influencers Armano D. Influence ripples. Available at: http: //darmano. typepad. com. Accessed on: April 13, 2010.

“You don’t need to go any further doctor. Just spell ‘eosinophil’ if you would. ”

Cure. Together Available at: http: //www. curetogether. com. Accessed on: April 13, 2010.

Cure. Together • Patient to patient • Crowdsourced data • “Clinical trials” Available at: http: //www. curetogether. com/Irritable-Bowel-Syndrome/treatments/. Accessed on: April 13, 2010.

Participatory Medicine • User-generated healthcare is shifting the balance of power from doctor to patient • The physician encounter is evolving as a narrow, more defined element in a patient’s quest to understand what’s wrong with them

What Is Social Media? • Content created by people using scalable online publishing technologies intended to facilitate communication and interaction • Most often refers to activities that integrate technology, telecommunications, and social interaction, with the construction of words, pictures, videos, and audio
With permission from Solis B, et al. The conversation prism. Available at: http: //www. theconversationprism. com. Accessed on: April 13, 2010.
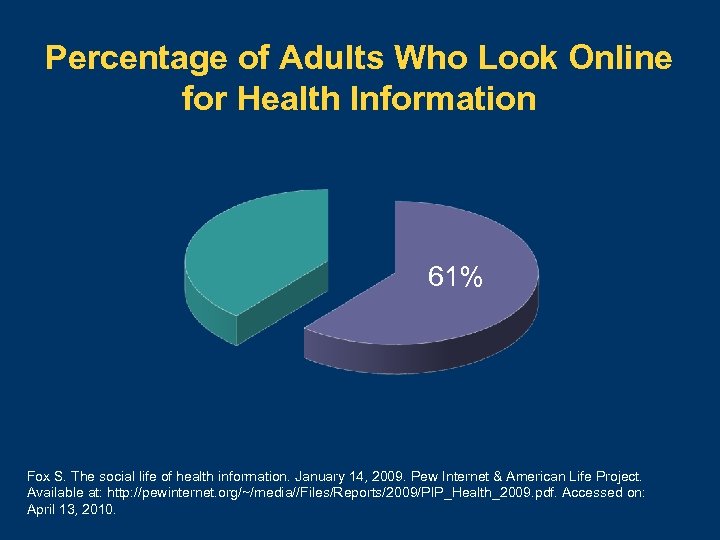
Percentage of Adults Who Look Online for Health Information 61% Fox S. The social life of health information. January 14, 2009. Pew Internet & American Life Project. Available at: http: //pewinternet. org/~/media//Files/Reports/2009/PIP_Health_2009. pdf. Accessed on: April 13, 2010.
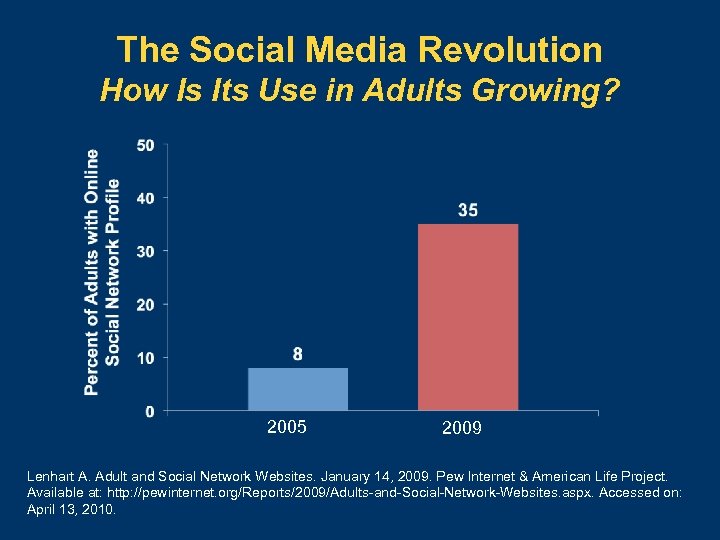
The Social Media Revolution How Is Its Use in Adults Growing? 2005 2009 Lenhart A. Adult and Social Network Websites. January 14, 2009. Pew Internet & American Life Project. Available at: http: //pewinternet. org/Reports/2009/Adults-and-Social-Network-Websites. aspx. Accessed on: April 13, 2010.
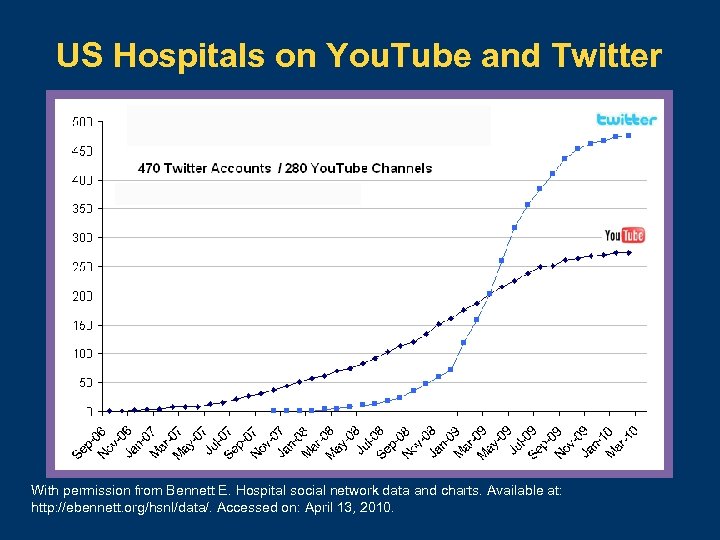
US Hospitals on You. Tube and Twitter With permission from Bennett E. Hospital social network data and charts. Available at: http: //ebennett. org/hsnl/data/. Accessed on: April 13, 2010.

Where Are the Doctors? The Absence of MDs in Social Media Space • Late adopters • Time/impatience • Concerns over privacy, liability, and image

What Can You Do on Social Networks? • • • Educate patients Influence behavior Promote awareness of yourself or your hospital Build relationships Filter information
Armano D. Human feed. Available at: http: //darmano. typepad. com. Accessed on: April 13, 2010.
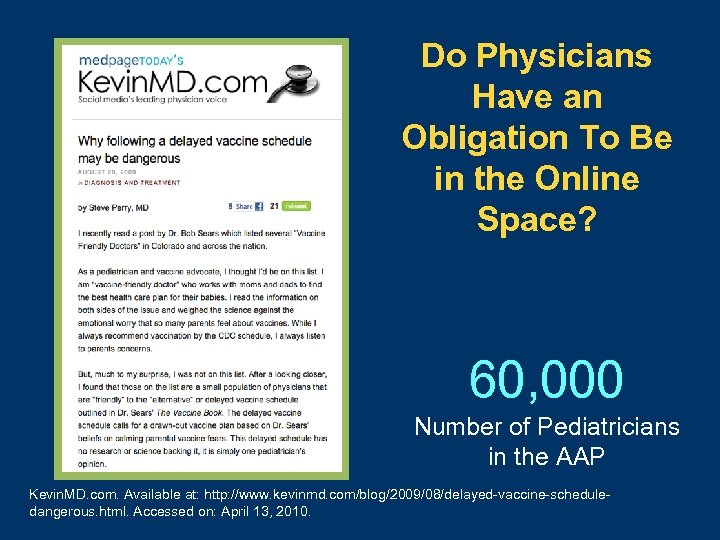
Do Physicians Have an Obligation To Be in the Online Space? 60, 000 Number of Pediatricians in the AAP Kevin. MD. com. Available at: http: //www. kevinmd. com/blog/2009/08/delayed-vaccine-scheduledangerous. html. Accessed on: April 13, 2010.

DDW’s 4 Sponsoring Societies 16, 000 3000 Number of Members in the AGA Number of Members in the AASLD 10, 000 2500 Number of Members in the ASGE Number of Members in the SSAT Abbreviations: AASLD, American Society for the Study of Liver Diseases; AGA, American Gastroenterological Association; ASGE, American Society for Gastrointestinal Endoscopy; DDW, Digestive Disease Week; SSAT, The Society for Surgery of the Alimentary Tract. DDW 2010. FAQs. Available at: http: //www. ddw. org/wmspage. cfm? parm 1=710. Accessed on: April 13, 2010.

How Should Physicians Handle Patient Encounters in the Social Media Space? They Shouldn’t

Staying Safe on Social Networks • • Never discuss patients Patients, boss, future employer will read everything you write Be nice Don’t be anonymous

Blog Put a stake in the ground Graphic courtesy of Dr. Bryan Vartabedian.
Blog Graphic courtesy of Dr. Bryan Vartabedian.

Visibility in Action Left graphic: Kaplan Publishing. Spring 2009 Catalog. Available at: http: //www. kaptest. com/pdf_files/publishing/seasonalcatalogs/KPSpring 09 Catalog. pdf. Accessed on: April 13, 2010. Upper right graphic: Courtesy of Dr. Bryan Vartabedian. 33 Charts blog. Available at: http: //www. 33 charts. com. Lower graphic: Courtesy of Dr. Bryan Vartabedian. Personal Correspondence. 2010.
Armano D. Conversion. Available at: http: //darmano. typepad. com. Accessed on: April 13, 2010.

The Low Rates of Chronic HCV Diagnosis and Treatment: Why and What Can We Do? Hashem B. El-Serag, MD, MPH Professor of Medicine Chief, Section of Gastroenterology and Hepatology Baylor College of Medicine Michael E. De. Bakey VA Medical Center Houston, Texas
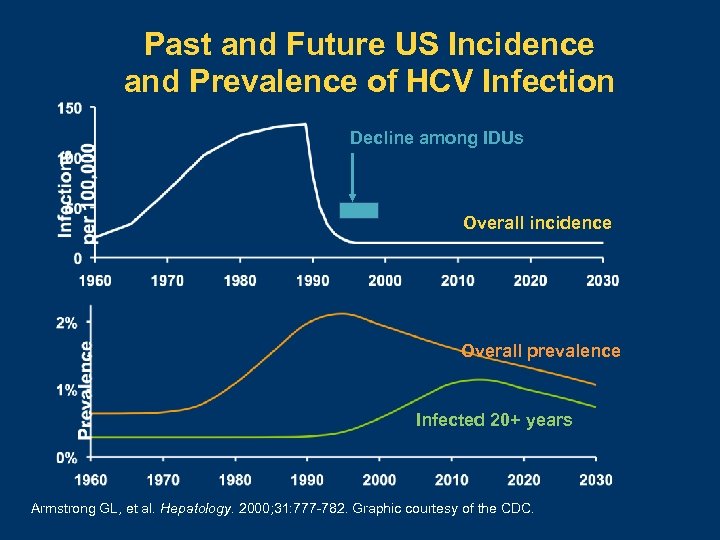
Past and Future US Incidence and Prevalence of HCV Infection Decline among IDUs Overall incidence Overall prevalence Infected 20+ years Armstrong GL, et al. Hepatology. 2000; 31: 777 -782. Graphic courtesy of the CDC.
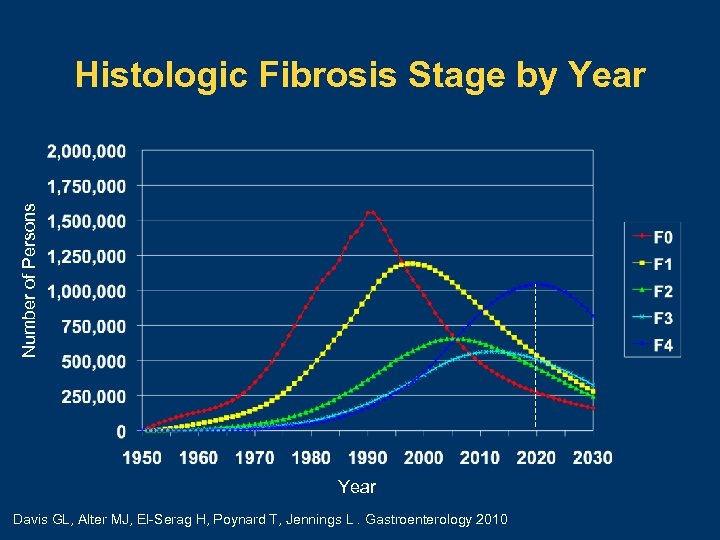
Number of Persons Histologic Fibrosis Stage by Year Davis GL, Alter MJ, El-Serag H, Poynard T, Jennings L. Gastroenterology 2010

Slide Not Available
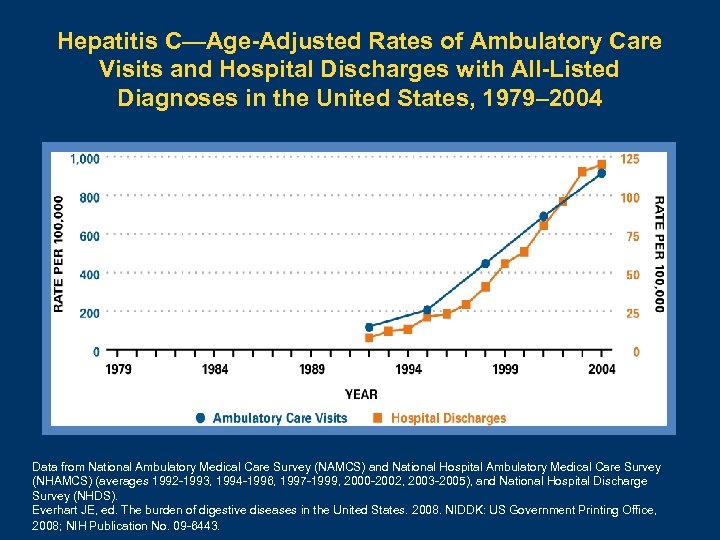
Hepatitis C—Age-Adjusted Rates of Ambulatory Care Visits and Hospital Discharges with All-Listed Diagnoses in the United States, 1979– 2004 Data from National Ambulatory Medical Care Survey (NAMCS) and National Hospital Ambulatory Medical Care Survey (NHAMCS) (averages 1992 -1993, 1994 -1996, 1997 -1999, 2000 -2002, 2003 -2005), and National Hospital Discharge Survey (NHDS). Everhart JE, ed. The burden of digestive diseases in the United States. 2008. NIDDK: US Government Printing Office, 2008; NIH Publication No. 09 -6443.
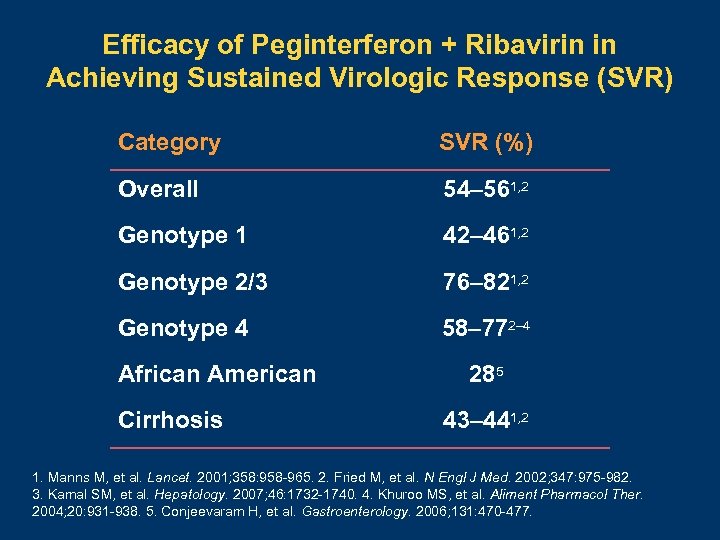
Efficacy of Peginterferon + Ribavirin in Achieving Sustained Virologic Response (SVR) Category SVR (%) Overall 54– 561, 2 Genotype 1 42– 461, 2 Genotype 2/3 76– 821, 2 Genotype 4 58– 772– 4 African American Cirrhosis 285 43– 441, 2 1. Manns M, et al. Lancet. 2001; 358: 958 -965. 2. Fried M, et al. N Engl J Med. 2002; 347: 975 -982. 3. Kamal SM, et al. Hepatology. 2007; 46: 1732 -1740. 4. Khuroo MS, et al. Aliment Pharmacol Ther. 2004; 20: 931 -938. 5. Conjeevaram H, et al. Gastroenterology. 2006; 131: 470 -477.
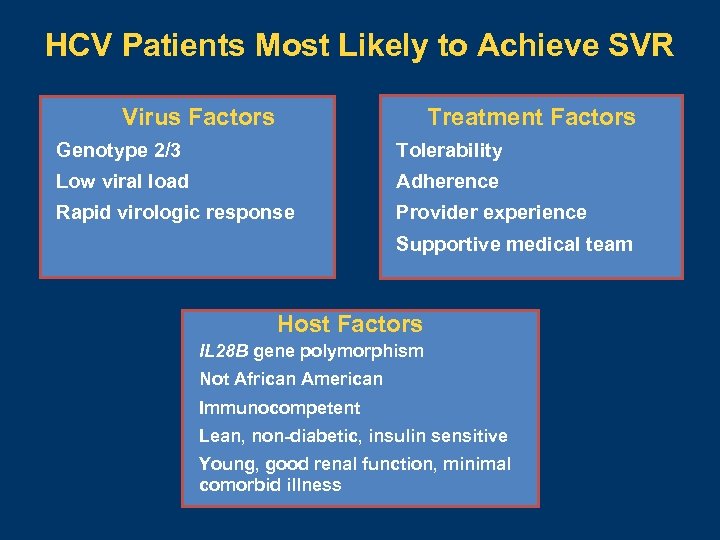
HCV Patients Most Likely to Achieve SVR Virus Factors Treatment Factors Genotype 2/3 Tolerability Low viral load Adherence Rapid virologic response Provider experience Supportive medical team Host Factors IL 28 B gene polymorphism Not African American Immunocompetent Lean, non-diabetic, insulin sensitive Young, good renal function, minimal comorbid illness

Treatment of HCV Efficacy in clinical trials and research centers Effectiveness in community practice Efficacy x Access x Correct Diagnosis x Recommendation x Acceptance x Adherence El-Serag HB. Gastroenterology. 2007; 132: 8 -10.
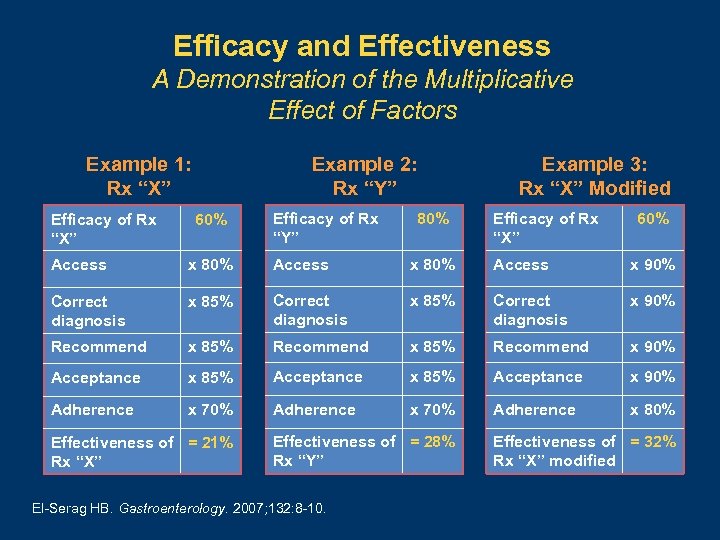
Efficacy and Effectiveness A Demonstration of the Multiplicative Effect of Factors Example 1: Rx “X” Efficacy of Rx “X” Example 2: Rx “Y” 60% Efficacy of Rx “Y” 80% Example 3: Rx “X” Modified Efficacy of Rx “X” 60% Access x 80% Access x 90% Correct diagnosis x 85% Correct diagnosis x 90% Recommend x 85% Recommend x 90% Acceptance x 85% Acceptance x 90% Adherence x 70% Adherence x 80% Effectiveness of = 21% Rx “X” Effectiveness of = 28% Rx “Y” El-Serag HB. Gastroenterology. 2007; 132: 8 -10. Effectiveness of = 32% Rx “X” modified
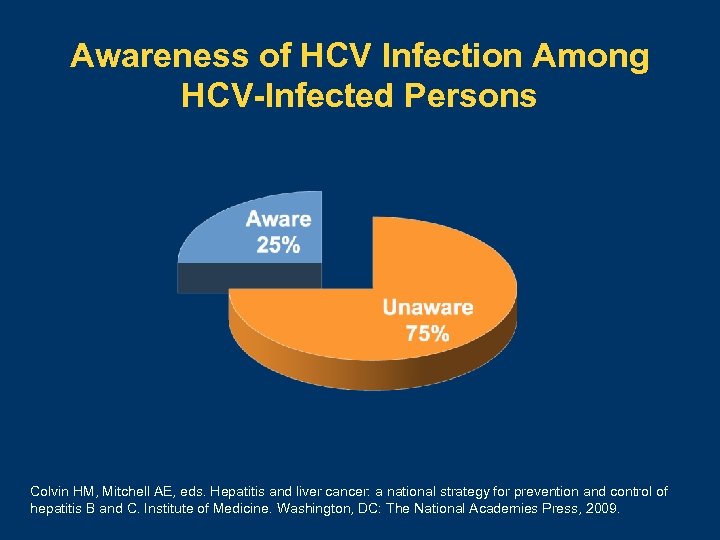
Awareness of HCV Infection Among HCV-Infected Persons Colvin HM, Mitchell AE, eds. Hepatitis and liver cancer: a national strategy for prevention and control of hepatitis B and C. Institute of Medicine. Washington, DC: The National Academies Press, 2009.
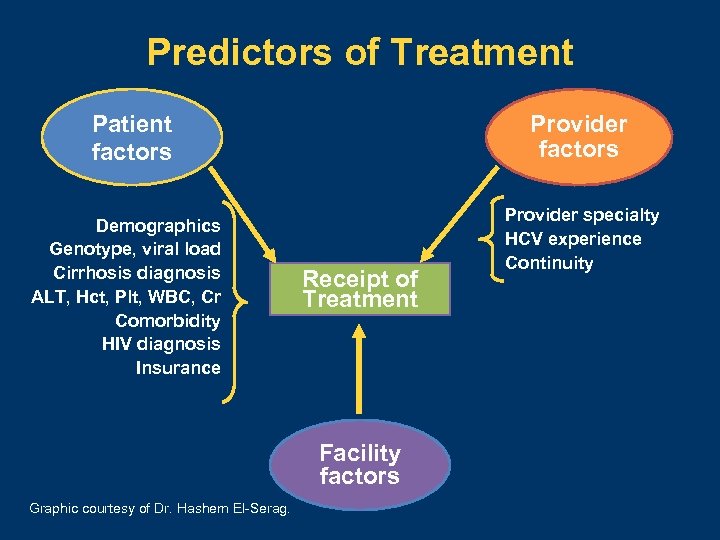
Predictors of Treatment Patient factors Demographics Genotype, viral load Cirrhosis diagnosis ALT, Hct, Plt, WBC, Cr Comorbidity HIV diagnosis Insurance Provider factors Receipt of Treatment Facility factors Graphic courtesy of Dr. Hashem El-Serag. Provider specialty HCV experience Continuity

Eligibility and Acceptability of HCV Treatment • 4084 HCV+ patients in VA Multicenter Study 12/99– 12/00 • Eligibility – 32% by standard criteria, 41% by treating physician – Predictors of noneligibility n n • OR 17. 68 OR 9. 62 OR 9. 45 OR 8. 43 Acceptability – 76% of eligible patients – Reasons for nonacceptance n n • Ongoing substance abuse Comorbid medical disease Psychiatric disease Advanced liver disease Defer Rx until better therapies Concerns regarding side effects 50% 22% Treatment completion rates – ~50% of those treated (~8% of all patients) Bini E, et al. Am J Gastroenterol. 2005; 100: 1772 -1779.
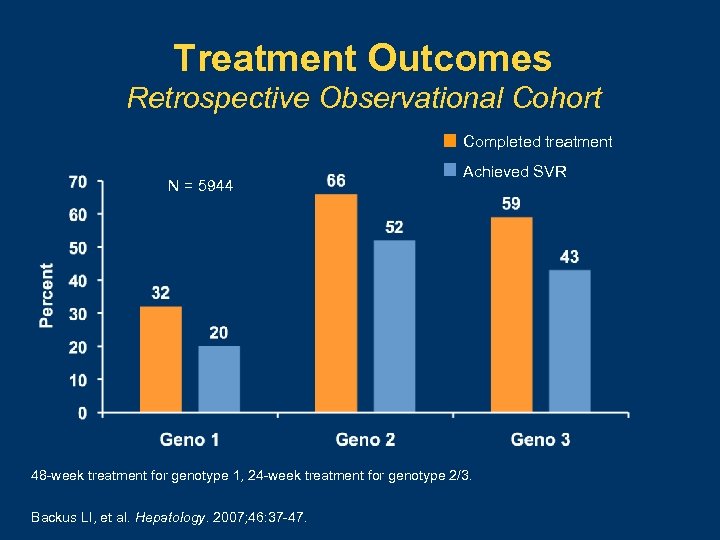
Treatment Outcomes Retrospective Observational Cohort Completed treatment N = 5944 Achieved SVR 48 -week treatment for genotype 1, 24 -week treatment for genotype 2/3. Backus LI, et al. Hepatology. 2007; 46: 37 -47.
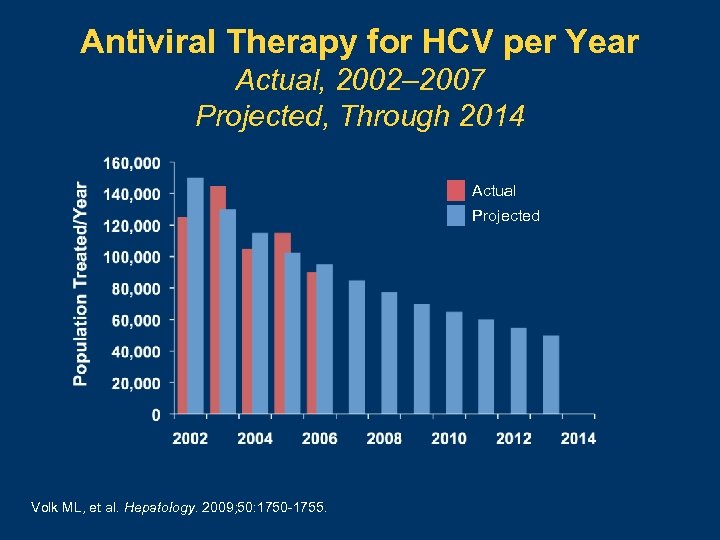
Antiviral Therapy for HCV per Year Actual, 2002– 2007 Projected, Through 2014 Actual Projected Volk ML, et al. Hepatology. 2009; 50: 1750 -1755.
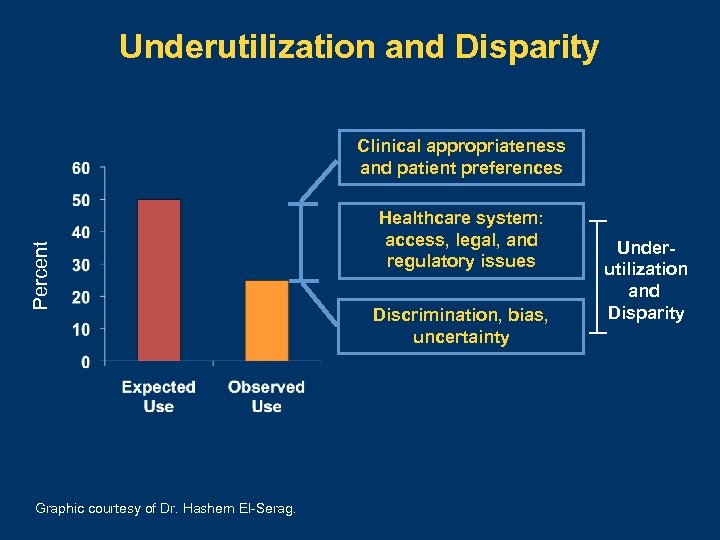
Underutilization and Disparity Percent Clinical appropriateness and patient preferences Graphic courtesy of Dr. Hashem El-Serag. Healthcare system: access, legal, and regulatory issues Discrimination, bias, uncertainty Underutilization and Disparity
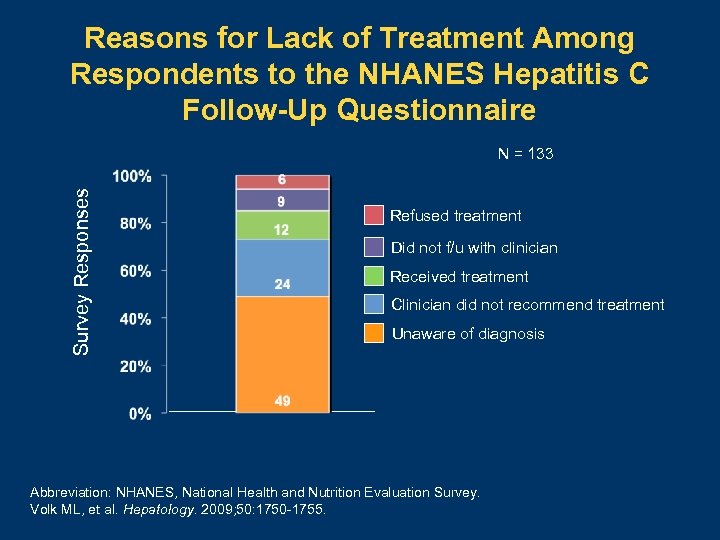
Reasons for Lack of Treatment Among Respondents to the NHANES Hepatitis C Follow-Up Questionnaire Survey Responses N = 133 Refused treatment Did not f/u with clinician Received treatment Clinician did not recommend treatment Unaware of diagnosis Abbreviation: NHANES, National Health and Nutrition Evaluation Survey. Volk ML, et al. Hepatology. 2009; 50: 1750 -1755.

Summary • HCV prevalence peaked in 2001 at 3. 6 million persons • Number and proportion of HCV patients with cirrhosis, decompensation, and HCC will increase for at least another 10– 15 years • Age of persons with complications will rise • Will antiviral therapy change the future? – Eradication of HCV stops progression, eliminates the risk of liver failure, and reduces HCC risk n Requires therapeutic intervention before onset of advanced fibrosis n Requires identification of infected cases

Summary • Clinical effectiveness is dependent on several factors in addition to clinical efficacy – HCV diagnosis – Referral to specialists – Treatment of comorbidities – Adherence to treatment

Institute of Medicine Report Underlying Factors • Lack of knowledge and awareness about chronic viral hepatitis among Healthcare and social service providers At-risk populations Policy makers and the public • Insufficient understanding about the extent and seriousness of this public health problem Colvin HM, Mitchell AE, eds. Hepatitis and liver cancer: a national strategy for prevention and control of hepatitis B and C. Institute of Medicine. Washington, DC: The National Academies Press, 2009.

Institute of Medicine Report Consequences Inadequate surveillance systems underreport acute and chronic infections At-risk people do not know they are at risk or how to avoid being infected At-risk people may not have access to preventive services People with chronic HCV infection do not know they are infected Many clinicians do not know how to screen people for risk factors or manage those who are infected HCV-infected people often have inadequate access to testing, social support, and medical management services Colvin HM, Mitchell AE, eds. Hepatitis and liver cancer: a national strategy for prevention and control of hepatitis B and C. Institute of Medicine. Washington, DC: The National Academies Press, 2009.

Institute of Medicine Report Recommendations Surveillance The CDC should: Evaluate national HCV public health surveillance system Develop agreements with state health departments to support core HCV surveillance Support targeted surveillance Knowledge & Awareness The CDC should work with key stakeholders to: Develop HCV educational programs for providers Develop and evaluate innovative and effective outreach programs to 1) target at-risk populations, and 2) increase public awareness Viral Hepatitis Services Federally funded health insurance programs (eg, Medicare) should: Incorporate guidelines for riskfactor screening as required component of preventive care Colvin HM, Mitchell AE, eds. Hepatitis and liver cancer: a national strategy for prevention and control of hepatitis B and C. Institute of Medicine. Washington, DC: The National Academies Press, 2009.

ARS Question: How likely are you to use social media to help increase public awareness of chronic hepatitis C? 1) Very likely 2) Likely 3) Maybe 4) Probably not 5) Never

ARS Question: In patients who present with non liver related complaints, how often do you evaluate a patient’s risk factors for chronic hepatitis C infection? 1) Always 2) Very Often 3) Sometimes 4) Rarely 5) Never

Optimization of Interferon. Based Therapies in Current HCV Patients– Can We Do Better? Donald M. Jensen, MD Professor of Medicine Director, Center for Liver Diseases University of Chicago Medical Center Chicago, Illinois

Why Do Current Drugs Fail? • Viral factors 1 – Viral protein products as inhibitors of innate immunity – Viral proteins as inhibitors of adaptive responses • Host factors – Fixed: race, genetics, age, gender – Modifiable: body mass index, insulin resistance, steatosis 1 • Treatment factors – Dose, duration, adherence, tolerability 1. Tai AW, et al. J Hepatol. 2009; 50: 412 -420.

Baseline Predictors • Host genetic makeup – IL 28 B polymorphism 1 n Allele on chromosome 19, rs 12979860 n Resides 3 kilobases upstream from the IL 28 B gene encoding IFN-gamma-3 n Strongly associated with rate of sustained virologic response – Other racial and genetic factors • Viral factors – Genotype – Viral load 1. Ge D, et al. Nature. 2009; 461: 399 -401.

Importance of IL 28 B Polymorphisms • C/C genotype is more common in racial groups with a higher sustained virologic response (SVR) • C/C genotype is associated with a higher SVR compared with T/C or T/T • IL 28 B polymorphisms may partially explain racial differences in antiviral response and may be a useful pretherapy tool Ge D, et al. Nature. 2009; 461: 399 -401.
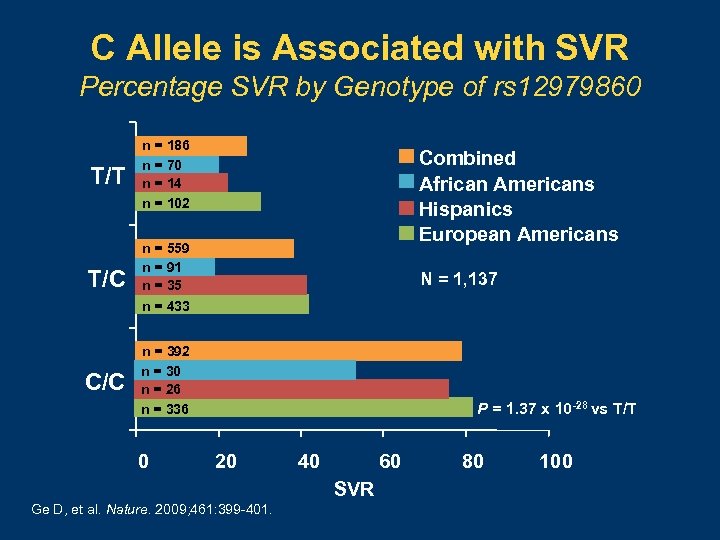
C Allele is Associated with SVR Percentage SVR by Genotype of rs 12979860 T/T n = 186 n = 70 n = 14 n = 102 T/C n = 559 n = 91 n = 35 n = 433 C/C n = 392 n = 30 n = 26 n = 336 0 Combined African Americans Hispanics European Americans N = 1, 137 P = 1. 37 x 10 -28 vs T/T 20 40 60 SVR Ge D, et al. Nature. 2009; 461: 399 -401. 80 100
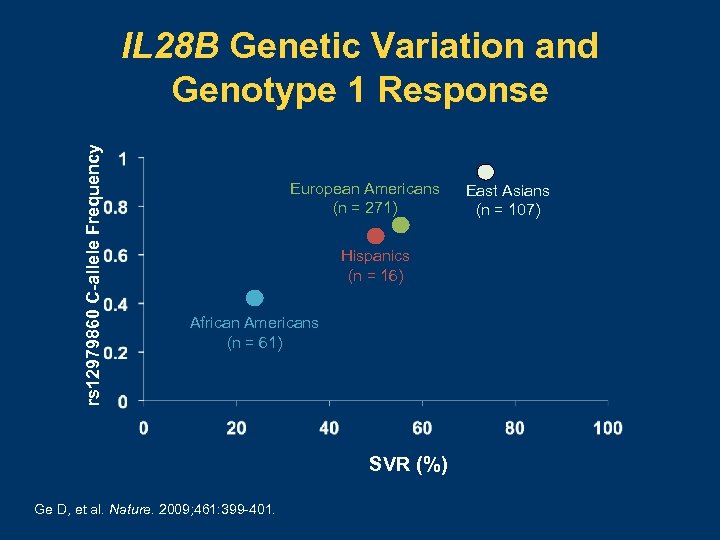
rs 12979860 C-allele Frequency IL 28 B Genetic Variation and Genotype 1 Response European Americans (n = 271) Hispanics (n = 16) African Americans (n = 61) SVR (%) Ge D, et al. Nature. 2009; 461: 399 -401. East Asians (n = 107)
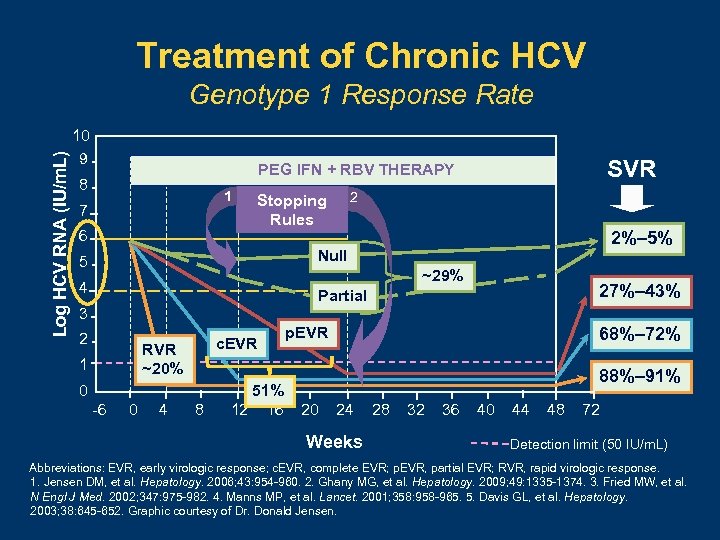
Treatment of Chronic HCV Log HCV RNA (IU/m. L) Genotype 1 Response Rate 10 9 SVR PEG IFN + RBV THERAPY 8 1 7 6 2 Stopping Rules 2%– 5% Null 5 ~29% 4 27%– 43% Partial 3 2 c. EVR RVR ~20% 1 0 -6 0 4 8 51% 12 16 p. EVR 68%– 72% 88%– 91% 20 24 Weeks 28 32 36 40 44 48 72 Detection limit (50 IU/m. L) Abbreviations: EVR, early virologic response; c. EVR, complete EVR; p. EVR, partial EVR; RVR, rapid virologic response. 1. Jensen DM, et al. Hepatology. 2006; 43: 954 -960. 2. Ghany MG, et al. Hepatology. 2009; 49: 1335 -1374. 3. Fried MW, et al. N Engl J Med. 2002; 347: 975 -982. 4. Manns MP, et al. Lancet. 2001; 358: 958 -965. 5. Davis GL, et al. Hepatology. 2003; 38: 645 -652. Graphic courtesy of Dr. Donald Jensen.

Improving Current Therapy 2 Goals • Increase SVR rate – Decrease risk of hepatocellular carcinoma (HCC)1, 2 – Decrease risk of decompensation 3, 4 – Improve fibrosis/reversal of cirrhosis 4 – Improve quality of life 5 • Improve adherence and tolerability – Shorten treatment duration if RVR – Minimize treatment-related adverse events 1. Craxi A, et al. Clin Liver Dis. 2005; 9: 329 -346. 2. Shiratori Y, et al. Ann Intern Med. 2005; 142: 105 -114. 3. Shiratori Y, et al. Ann Intern Med. 2000; 132: 517 -524. 4. Poynard T, et al. Gastroenterology. 2002; 122: 1303 -1313. 5. Foster GR, et al. J Viral Hepat. 2009; 16: 605 -611.

Goal—Increase SVR Rate Current options • Increase interferon and/or ribavirin dose • Increase adherence • Increase treatment duration Is there evidence that these work?
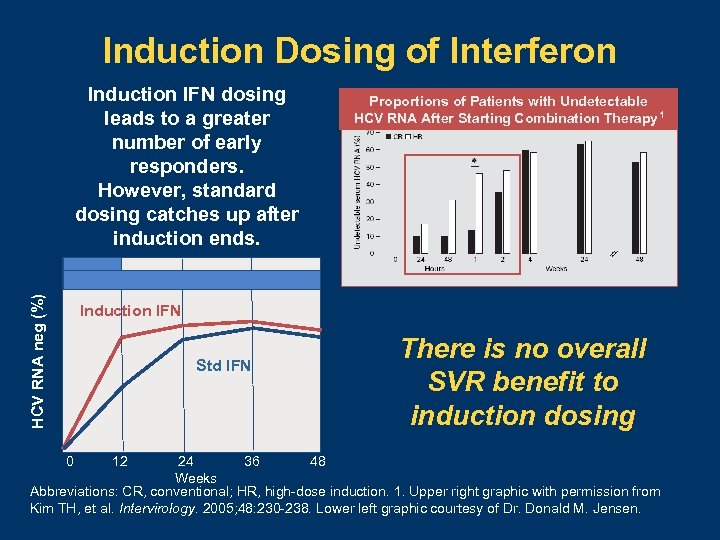
Induction Dosing of Interferon HCV RNA neg (%) Induction IFN dosing leads to a greater number of early responders. However, standard dosing catches up after induction ends. Proportions of Patients with Undetectable HCV RNA After Starting Combination Therapy 1 Induction IFN Std IFN There is no overall SVR benefit to induction dosing 0 12 24 36 48 Weeks Abbreviations: CR, conventional; HR, high-dose induction. 1. Upper right graphic with permission from Kim TH, et al. Intervirology. 2005; 48: 230 -238. Lower left graphic courtesy of Dr. Donald M. Jensen.
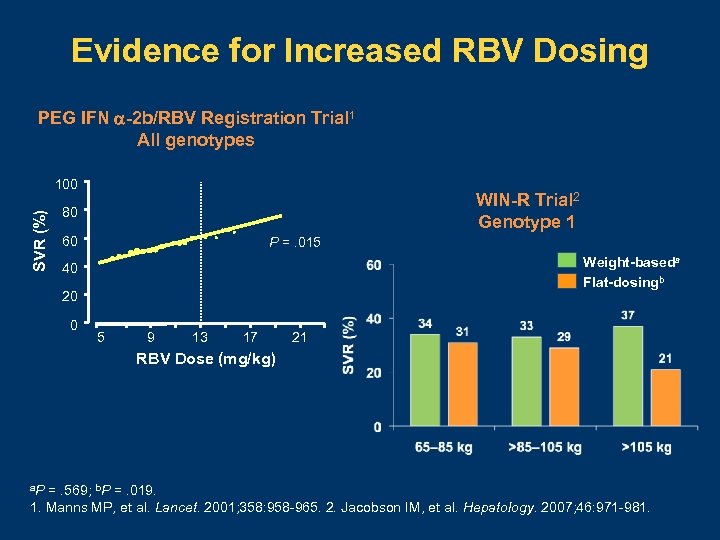
Evidence for Increased RBV Dosing PEG IFN -2 b/RBV Registration Trial 1 All genotypes SVR (%) 100 WIN-R Trial 2 Genotype 1 80 60 P =. 015 Weight-baseda Flat-dosingb 40 20 0 5 9 13 17 21 RBV Dose (mg/kg) a. P =. 569; b. P =. 019. 1. Manns MP, et al. Lancet. 2001; 358: 958 -965. 2. Jacobson IM, et al. Hepatology. 2007; 46: 971 -981.
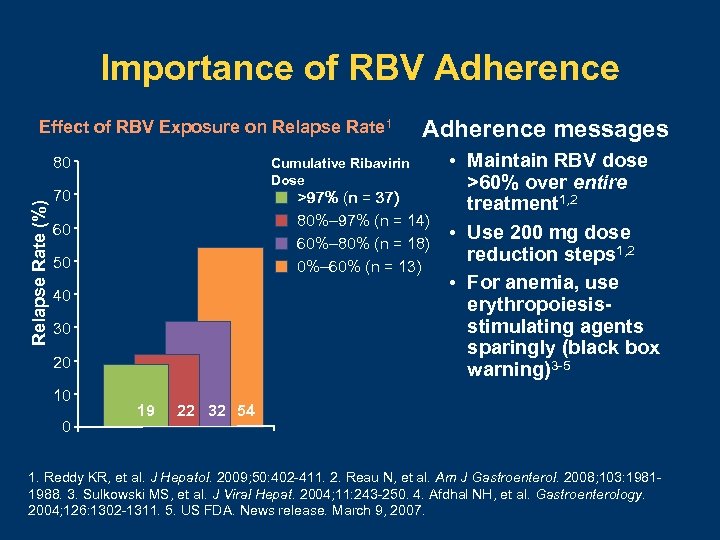
Importance of RBV Adherence Effect of RBV Exposure on Relapse Rate 1 Relapse Rate (%) 80 Cumulative Ribavirin Dose 70 >97% (n = 37) 80%– 97% (n = 14) 60%– 80% (n = 18) 0%– 60% (n = 13) 60 50 40 30 20 10 0 Adherence messages 19 • Maintain RBV dose >60% over entire treatment 1, 2 • Use 200 mg dose reduction steps 1, 2 • For anemia, use erythropoiesisstimulating agents sparingly (black box warning)3 -5 22 32 54 1. Reddy KR, et al. J Hepatol. 2009; 50: 402 -411. 2. Reau N, et al. Am J Gastroenterol. 2008; 103: 19811988. 3. Sulkowski MS, et al. J Viral Hepat. 2004; 11: 243 -250. 4. Afdhal NH, et al. Gastroenterology. 2004; 126: 1302 -1311. 5. US FDA. News release. March 9, 2007.
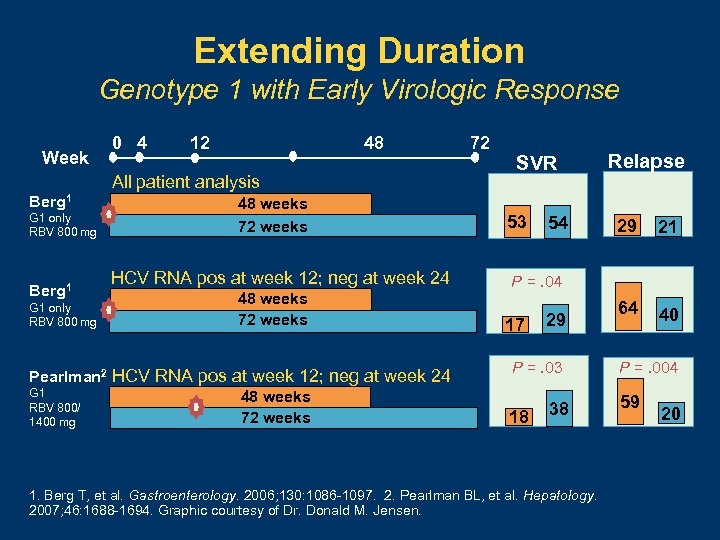
Extending Duration Genotype 1 with Early Virologic Response Week 0 4 12 48 All patient analysis Berg 1 G 1 only RBV 800 mg 48 weeks 72 weeks HCV RNA pos at week 12; neg at week 24 48 weeks 72 weeks Pearlman 2 HCV RNA pos at week 12; neg at week 24 G 1 48 weeks RBV 800/ 72 weeks 1400 mg 72 SVR 53 54 Relapse 29 21 64 40 P =. 04 17 29 P =. 03 18 38 1. Berg T, et al. Gastroenterology. 2006; 130: 1086 -1097. 2. Pearlman BL, et al. Hepatology. 2007; 46: 1688 -1694. Graphic courtesy of Dr. Donald M. Jensen. P =. 004 59 20

Can We Shorten Therapy Based on an RVR to Improve Adherence and Minimize Side Effects?
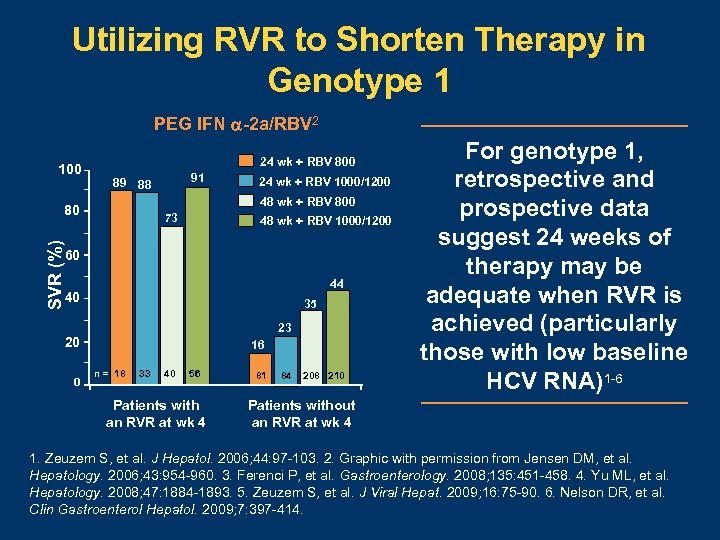
Utilizing RVR to Shorten Therapy in Genotype 1 PEG IFN -2 a/RBV 2 100 24 wk + RBV 800 91 89 88 24 wk + RBV 1000/1200 48 wk + RBV 800 80 73 SVR (%) 48 wk + RBV 1000/1200 60 44 40 35 23 20 0 16 n = 18 33 40 56 Patients with an RVR at wk 4 81 84 208 210 For genotype 1, retrospective and prospective data suggest 24 weeks of therapy may be adequate when RVR is achieved (particularly those with low baseline HCV RNA)1 -6 Patients without an RVR at wk 4 1. Zeuzem S, et al. J Hepatol. 2006; 44: 97 -103. 2. Graphic with permission from Jensen DM, et al. Hepatology. 2006; 43: 954 -960. 3. Ferenci P, et al. Gastroenterology. 2008; 135: 451 -458. 4. Yu ML, et al. Hepatology. 2008; 47: 1884 -1893. 5. Zeuzem S, et al. J Viral Hepat. 2009; 16: 75 -90. 6. Nelson DR, et al. Clin Gastroenterol Hepatol. 2009; 7: 397 -414.
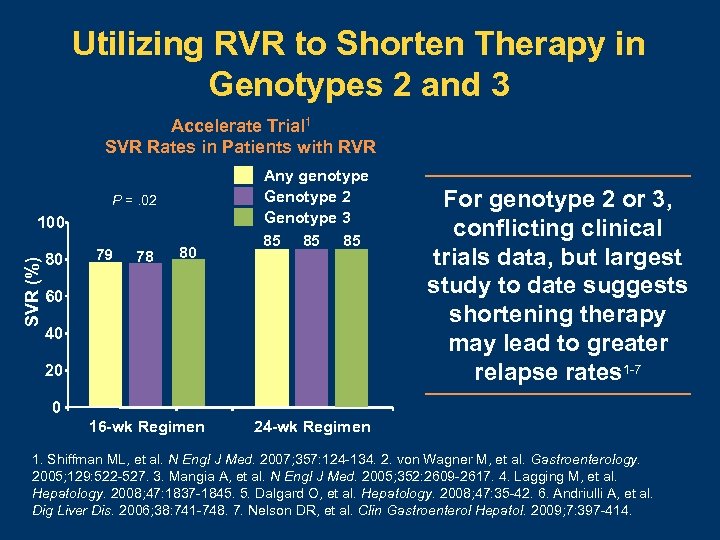
Utilizing RVR to Shorten Therapy in Genotypes 2 and 3 Accelerate Trial 1 SVR Rates in Patients with RVR P =. 02 SVR (%) 100 80 79 78 80 Any genotype Genotype 2 Genotype 3 85 85 85 60 40 20 For genotype 2 or 3, conflicting clinical trials data, but largest study to date suggests shortening therapy may lead to greater relapse rates 1 -7 0 16 -wk Regimen 24 -wk Regimen 1. Shiffman ML, et al. N Engl J Med. 2007; 357: 124 -134. 2. von Wagner M, et al. Gastroenterology. 2005; 129: 522 -527. 3. Mangia A, et al. N Engl J Med. 2005; 352: 2609 -2617. 4. Lagging M, et al. Hepatology. 2008; 47: 1837 -1845. 5. Dalgard O, et al. Hepatology. 2008; 47: 35 -42. 6. Andriulli A, et al. Dig Liver Dis. 2006; 38: 741 -748. 7. Nelson DR, et al. Clin Gastroenterol Hepatol. 2009; 7: 397 -414.

Response-Guided Therapy A New Paradigm? Individualized treatment based on viral kinetic measurements should…. • Improve overall cost-benefit • Improve tolerability for those with RVR • Decrease relapse rate

Nonresponders

PEG IFN/RBV Nonresponder Clinical Trials EPIC 31 476 NRs PEG IFN -2 b 1. 5 mcg/kg + RBV 800– SVR: 1400 mg/d x 48 wk 6% overall 4% geno-1 REPEAT 2 942 Geno-1 NRs PEG IFN -2 a 180 or 360/180 g/wk + SVR: RBV 1000– 1200 mg/d x 48 or 72 wk 7%– 16% DIRECT 3 659 Geno-1 NRs c. IFN 9 or 15 g/d + RBV 1000– 1200 x SVR: 48 weeks 7%– 11% Abbreviation: c. IFN, consensus interferon. 1. Poynard T, et al. Gastroenterology. 2009; 136: 1618 -1628. 2. Jensen DM, et al. Ann Intern Med. 2009; 150: 528 -540. 3. Bacon BR, et al. Hepatology. 2009; 49: 1838 -1846.

Maintenance Therapy

Maintenance PEG IFN in Nonresponders 3 Randomized Controlled Trials HALT-C COPILOT Bruix, et al No difference in clinical outcomes 1 Subjects with In primary baseline analysis, portal maintenance hypertension was not had reduced superior to Subgroup outcomes observational with >4 -log (variceal control in reduction in bleeding) preventing HCV RNA had with PEG IFN 3 occurrence of reduced clinical 2 outcomes events 4 1. Di Bisceglie AM, et al. Hepatology. 2007; 46: LB 1. 2. Shiffman ML, et al. J Hepatol. 2008; 48: S 62. 3. Afdhal NH, et al. J Hepatol. 2008; 48: S 4. 4. Bruix J, et al. J Hepatol. 2009; 50(suppl 1): S 22.

Treat Now or Wait Until STAT-C? Treat Now • Advanced fibrosis • Symptomatic • Genotype 2, 3, or 4 • IL 28 B C/C genotype (? ) May Consider Waiting • Genotype 1 with mild histology • PEG IFN/RBV prior nonresponders • IL 28 B T/T or T/C genotype Interferon and ribavirin will be required in combination with all new STAT-C agents for several years

Conclusions • • Response-guided therapy is important theme now for optimizing existing interferon/ribavirin-based therapies Improvements in current standard of care can be demonstrated by – Identifying likely responder populations – Shortening treatment duration in patients with RVR and poor tolerability – Extending treatment duration in patients with slow partial response


ARS Question: Among all your genotype 1 patients who are eligible for current therapy, how often are you withholding therapy while waiting for novel drugs? 1) Always 2) Very Often 3) Sometimes 4) Rarely 5) Never

How Will STAT-C Therapies Affect Future Anti-HCV Treatment Paradigms? Ira M. Jacobson, MD Vincent Astor Professor of Medicine Chief, Division of Gastroenterology and Hepatology Medical Director of the Center for the Study of Hepatitis C Weill Medical College of Cornell University New York, New York
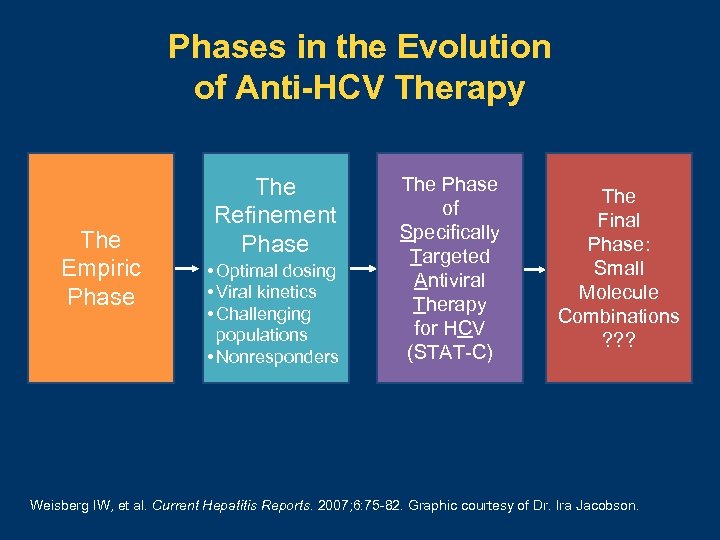
Phases in the Evolution of Anti-HCV Therapy The Empiric Phase The Refinement Phase • Optimal dosing • Viral kinetics • Challenging populations • Nonresponders The Phase of Specifically Targeted Antiviral Therapy for HCV (STAT-C) The Final Phase: Small Molecule Combinations ? ? ? Weisberg IW, et al. Current Hepatitis Reports. 2007; 6: 75 -82. Graphic courtesy of Dr. Ira Jacobson.
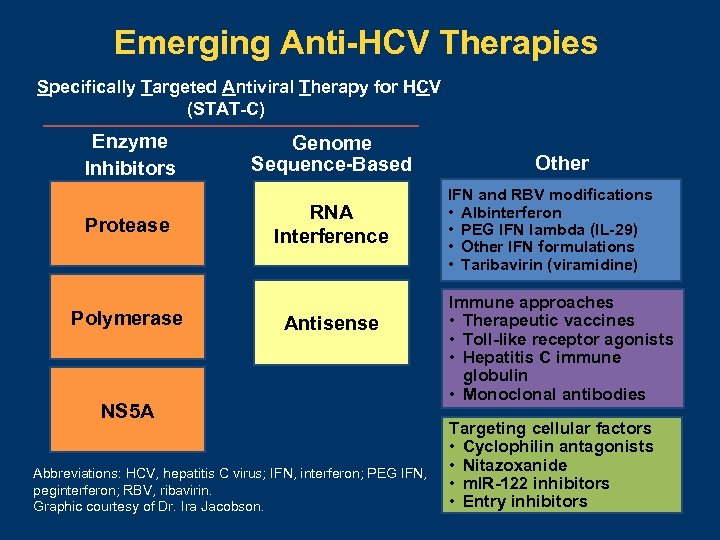
Emerging Anti-HCV Therapies Specifically Targeted Antiviral Therapy for HCV (STAT-C) Enzyme Inhibitors Protease Polymerase Genome Sequence-Based RNA Interference Antisense NS 5 A Abbreviations: HCV, hepatitis C virus; IFN, interferon; PEG IFN, peginterferon; RBV, ribavirin. Graphic courtesy of Dr. Ira Jacobson. Other IFN and RBV modifications • Albinterferon • PEG IFN lambda (IL-29) • Other IFN formulations • Taribavirin (viramidine) Immune approaches • Therapeutic vaccines • Toll-like receptor agonists • Hepatitis C immune globulin • Monoclonal antibodies Targeting cellular factors • Cyclophilin antagonists • Nitazoxanide • m. IR-122 inhibitors • Entry inhibitors
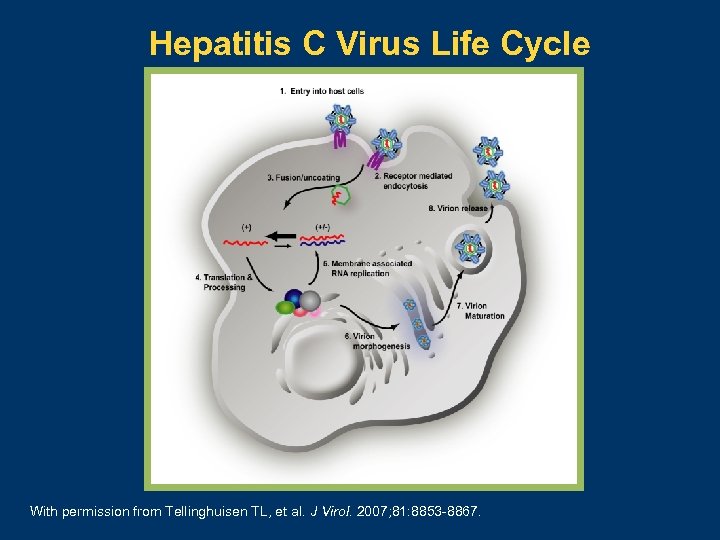
Hepatitis C Virus Life Cycle With permission from Tellinghuisen TL, et al. J Virol. 2007; 81: 8853 -8867.
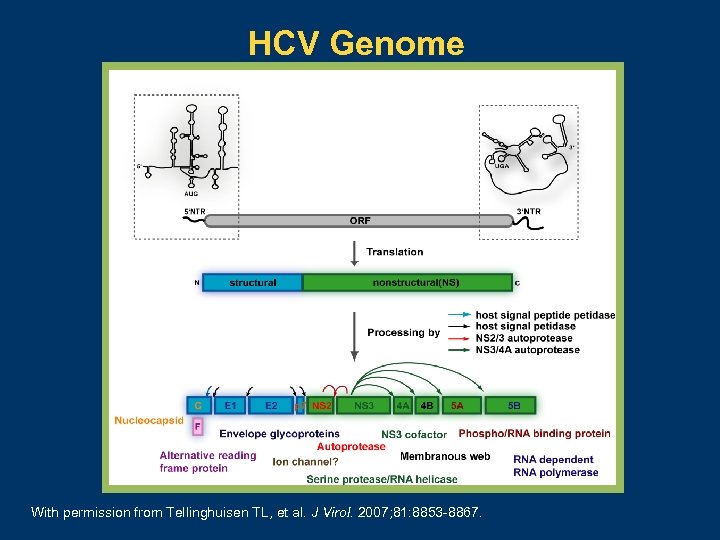
HCV Genome With permission from Tellinghuisen TL, et al. J Virol. 2007; 81: 8853 -8867.
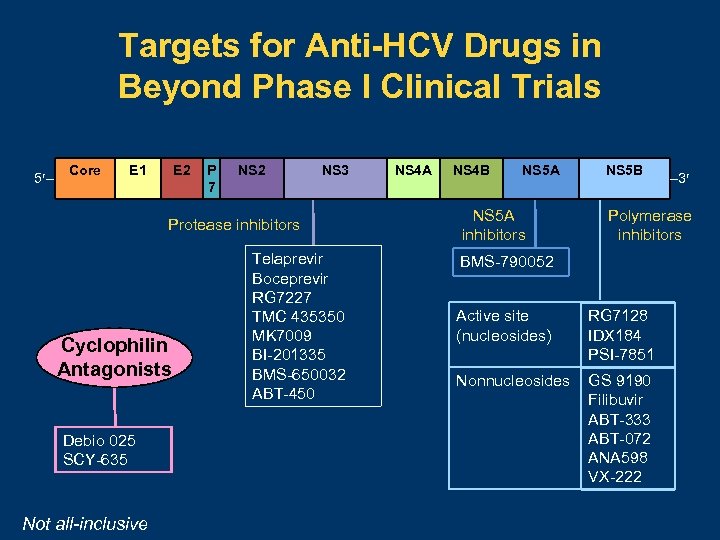
Targets for Anti-HCV Drugs in Beyond Phase I Clinical Trials 5 – Core E 1 E 2 P 7 NS 2 NS 3 Protease inhibitors Cyclophilin Antagonists Debio 025 SCY-635 Not all-inclusive Telaprevir Boceprevir RG 7227 TMC 435350 MK 7009 BI-201335 BMS-650032 ABT-450 NS 4 A NS 4 B NS 5 A inhibitors NS 5 B – 3 Polymerase inhibitors BMS-790052 Active site (nucleosides) RG 7128 IDX 184 PSI-7851 Nonnucleosides GS 9190 Filibuvir ABT-333 ABT-072 ANA 598 VX-222
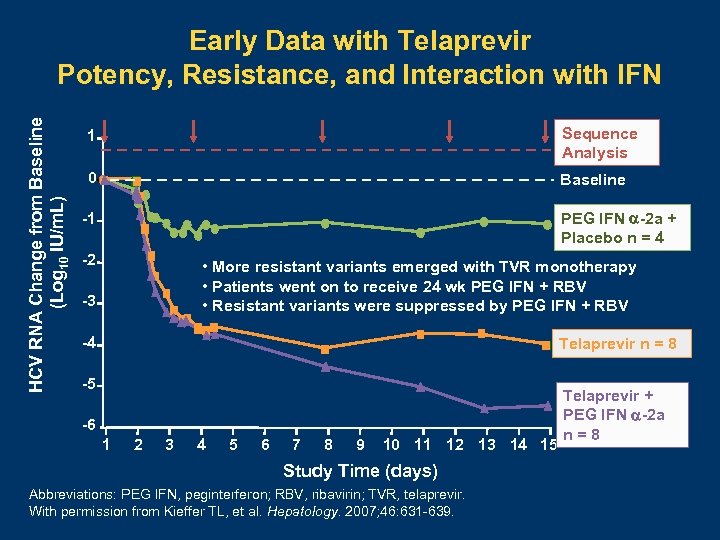
HCV RNA Change from Baseline (Log 10 IU/m. L) Early Data with Telaprevir Potency, Resistance, and Interaction with IFN 1 Sequence Analysis 0 Baseline PEG IFN -2 a + Placebo n = 4 -1 -2 • More resistant variants emerged with TVR monotherapy • Patients went on to receive 24 wk PEG IFN + RBV • Resistant variants were suppressed by PEG IFN + RBV -3 -4 Telaprevir n = 8 -5 -6 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Study Time (days) Abbreviations: PEG IFN, peginterferon; RBV, ribavirin; TVR, telaprevir. With permission from Kieffer TL, et al. Hepatology. 2007; 46: 631 -639. Telaprevir + PEG IFN -2 a n=8
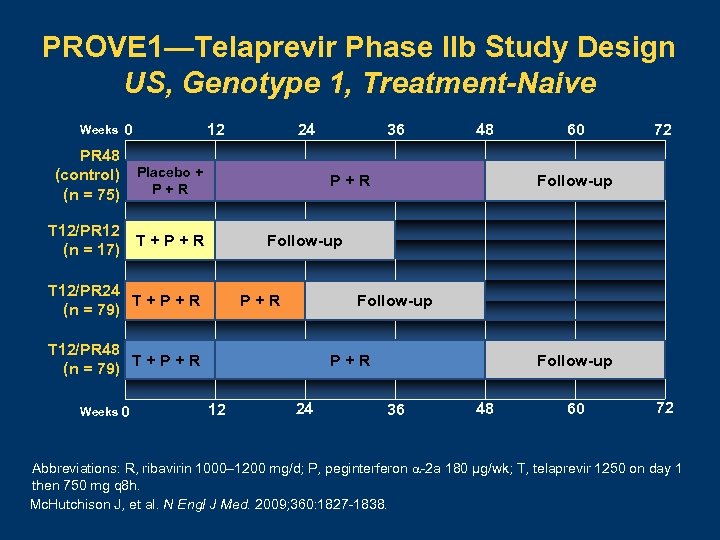
PROVE 1—Telaprevir Phase IIb Study Design US, Genotype 1, Treatment-Naive Weeks 0 PR 48 (control) (n = 75) 12 24 Placebo + P+R 48 P+R T 12/PR 12 T+P+R (n = 17) 60 72 Follow-up T 12/PR 24 T+P+R (n = 79) P+R Follow-up T 12/PR 48 T+P+R (n = 79) Weeks 0 36 P+R 12 24 Follow-up 36 48 60 72 Abbreviations: R, ribavirin 1000– 1200 mg/d; P, peginterferon -2 a 180 µg/wk; T, telaprevir 1250 on day 1 then 750 mg q 8 h. Mc. Hutchison J, et al. N Engl J Med. 2009; 360: 1827 -1838.
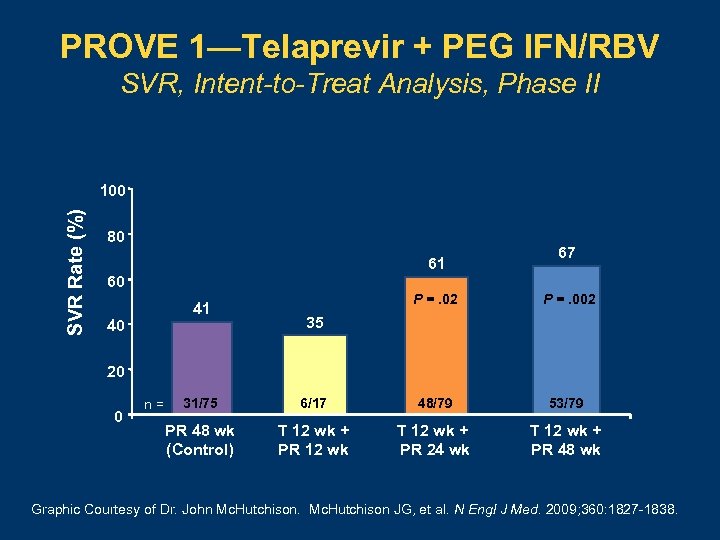
PROVE 1—Telaprevir + PEG IFN/RBV SVR, Intent-to-Treat Analysis, Phase II SVR Rate (%) 100 80 61 60 41 40 67 P =. 02 P =. 002 35 20 0 31/75 6/17 48/79 53/79 PR 48 wk (Control) T 12 wk + PR 12 wk T 12 wk + PR 24 wk T 12 wk + PR 48 wk n = Graphic Courtesy of Dr. John Mc. Hutchison JG, et al. N Engl J Med. 2009; 360: 1827 -1838.
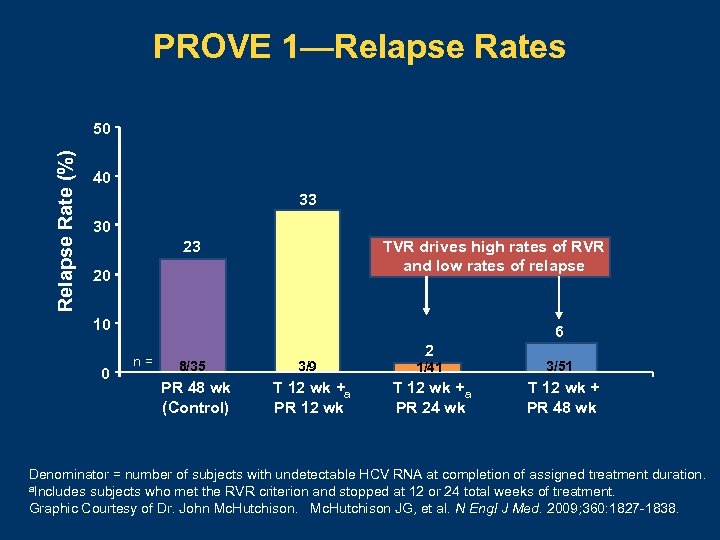
PROVE 1—Relapse Rates Relapse Rate (%) 50 40 33 30 23 TVR drives high rates of RVR and low rates of relapse 20 10 0 6 n = 8/35 PR 48 wk (Control) 3/9 T 12 wk +a PR 12 wk 2 1/41 3/51 T 12 wk + a PR 24 wk T 12 wk + PR 48 wk Denominator = number of subjects with undetectable HCV RNA at completion of assigned treatment duration. a. Includes subjects who met the RVR criterion and stopped at 12 or 24 total weeks of treatment. Graphic Courtesy of Dr. John Mc. Hutchison JG, et al. N Engl J Med. 2009; 360: 1827 -1838.

PROVE 1—Viral Breakthrough by Week 12 • 12/175 (7%) of telaprevir-treated patients had breakthrough – – – • 9/12 occurred in first 4 weeks 3/12 occurred between weeks 5 and 12 Only 2 breakthroughs occurred in subjects after HCV RNA became undetectable Telaprevir-resistant viral variants – – – V 36 M and R 155 K (10 patients with genotype 1 a) A 156 T (1 patient with genotype 1 b) Wild type (1 patient who missed several days) Mc. Hutchison JG, et al. N Engl J Med. 2009; 360: 1827 -1838.
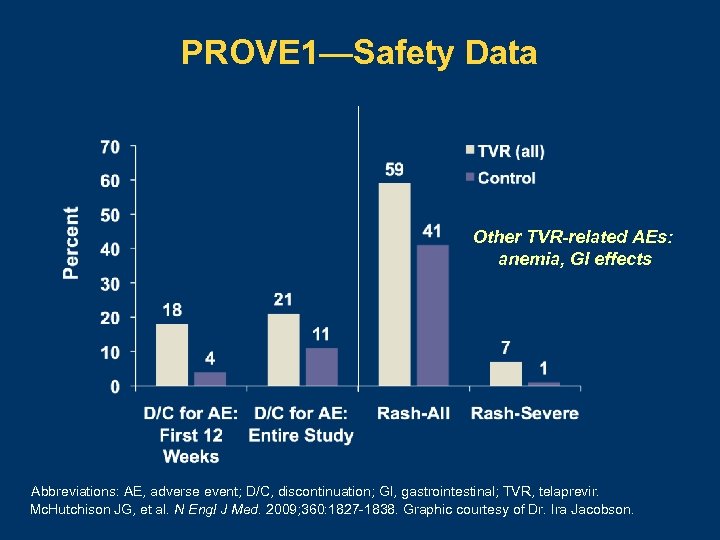
PROVE 1—Safety Data Other TVR-related AEs: anemia, GI effects Abbreviations: AE, adverse event; D/C, discontinuation; GI, gastrointestinal; TVR, telaprevir. Mc. Hutchison JG, et al. N Engl J Med. 2009; 360: 1827 -1838. Graphic courtesy of Dr. Ira Jacobson.
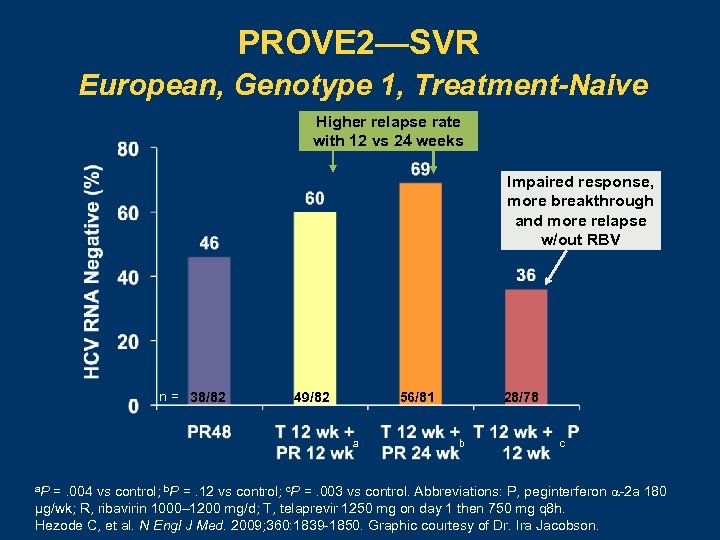
PROVE 2—SVR European, Genotype 1, Treatment-Naive Higher relapse rate with 12 vs 24 weeks Impaired response, more breakthrough and more relapse w/out RBV n = 38/82 49/82 56/81 a 28/78 b c a. P =. 004 vs control; b. P =. 12 vs control; c. P =. 003 vs control. Abbreviations: P, peginterferon -2 a 180 µg/wk; R, ribavirin 1000– 1200 mg/d; T, telaprevir 1250 mg on day 1 then 750 mg q 8 h. Hezode C, et al. N Engl J Med. 2009; 360: 1839 -1850. Graphic courtesy of Dr. Ira Jacobson.
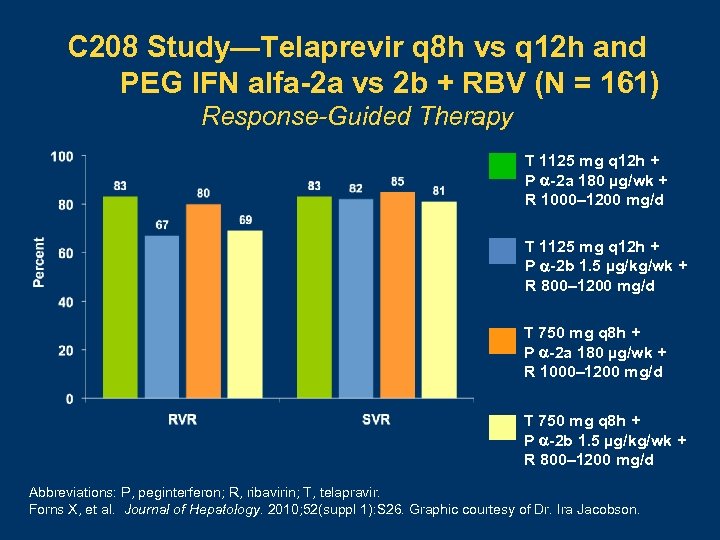
C 208 Study—Telaprevir q 8 h vs q 12 h and PEG IFN alfa-2 a vs 2 b + RBV (N = 161) Response-Guided Therapy T 1125 mg q 12 h + P -2 a 180 µg/wk + R 1000– 1200 mg/d T 1125 mg q 12 h + P -2 b 1. 5 µg/kg/wk + R 800– 1200 mg/d T 750 mg q 8 h + P -2 a 180 µg/wk + R 1000– 1200 mg/d T 750 mg q 8 h + P -2 b 1. 5 µg/kg/wk + R 800– 1200 mg/d Abbreviations: P, peginterferon; R, ribavirin; T, telapravir. Forns X, et al. Journal of Hepatology. 2010; 52(suppl 1): S 26. Graphic courtesy of Dr. Ira Jacobson.
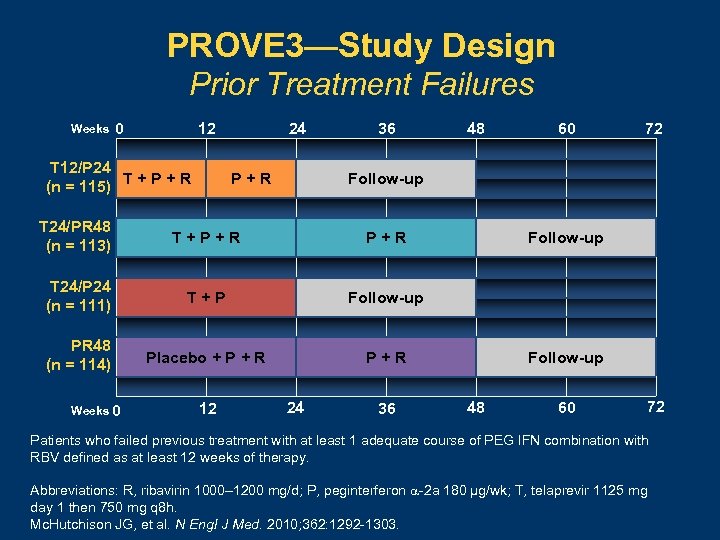
PROVE 3—Study Design Prior Treatment Failures Weeks 0 12 T 12/P 24 T+P+R (n = 115) 24 P+R 36 48 60 72 Follow-up T 24/PR 48 (n = 113) T+P+R T 24/P 24 (n = 111) T+P Follow-up PR 48 (n = 114) Placebo + P + R P+R Weeks 0 12 24 36 Follow-up 48 60 72 Patients who failed previous treatment with at least 1 adequate course of PEG IFN combination with RBV defined as at least 12 weeks of therapy. Abbreviations: R, ribavirin 1000– 1200 mg/d; P, peginterferon -2 a 180 µg/wk; T, telaprevir 1125 mg day 1 then 750 mg q 8 h. Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -1303.
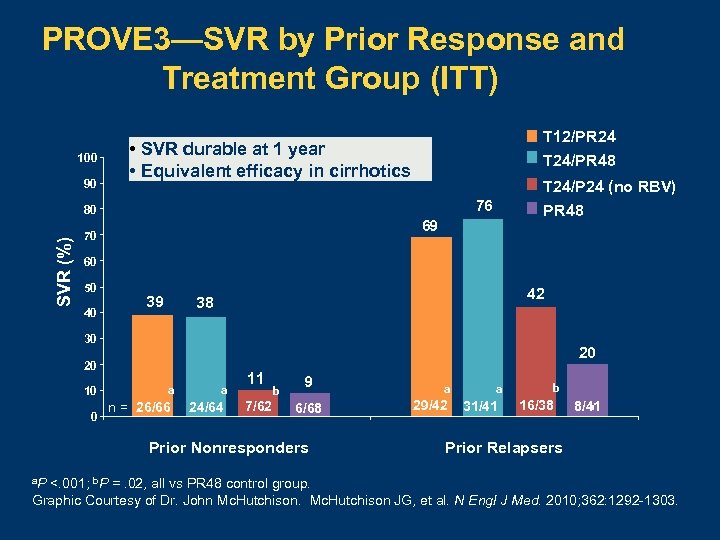
PROVE 3—SVR by Prior Response and Treatment Group (ITT) 100 90 T 12/PR 24 T 24/PR 48 • SVR durable at 1 year • Equivalent efficacy in cirrhotics 76 80 SVR (%) T 24/P 24 (no RBV) PR 48 69 70 60 50 40 39 42 38 30 20 20 10 0 a n = 26/66 a 24/64 11 b 7/62 9 6/68 Prior Nonresponders a. P a 29/42 a 31/41 b 16/38 8/41 Prior Relapsers <. 001; b. P =. 02, all vs PR 48 control group. Graphic Courtesy of Dr. John Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -1303.
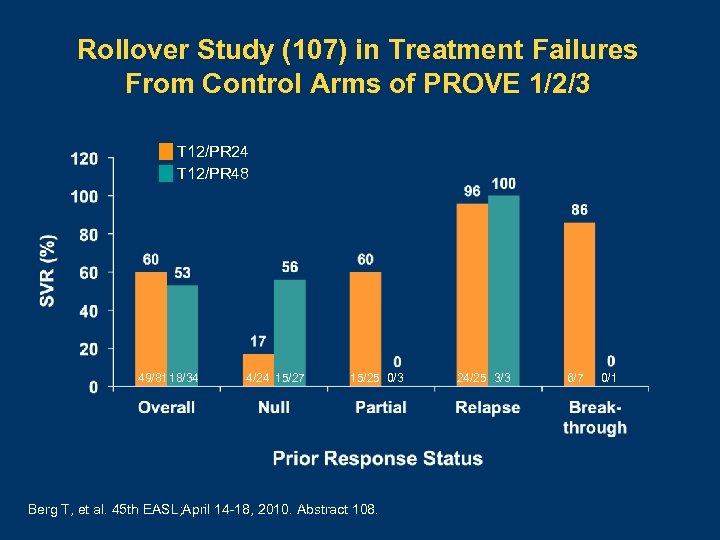
Rollover Study (107) in Treatment Failures From Control Arms of PROVE 1/2/3 T 12/PR 24 T 12/PR 48 49/8118/34 4/24 15/27 15/25 0/3 Berg T, et al. 45 th EASL; April 14 -18, 2010. Abstract 108. 24/25 3/3 6/7 0/1
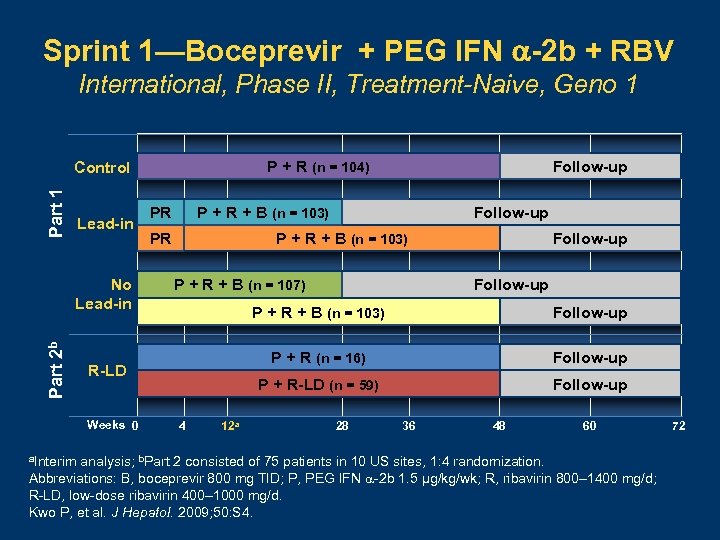
Sprint 1—Boceprevir + PEG IFN -2 b + RBV International, Phase II, Treatment-Naive, Geno 1 P + R (n = 104) Part 1 Control Lead-in Part 2 b No Lead-in PR P + R + B (n = 103) PR Follow-up P + R + B (n = 103) P + R + B (n = 107) Follow-up P + R + B (n = 103) 12 a Follow-up P + R-LD (n = 59) 4 Follow-up P + R (n = 16) R-LD Weeks 0 Follow-up 28 36 48 60 a. Interim analysis; b. Part 2 consisted of 75 patients in 10 US sites, 1: 4 randomization. Abbreviations: B, boceprevir 800 mg TID; P, PEG IFN -2 b 1. 5 µg/kg/wk; R, ribavirin 800– 1400 mg/d; R-LD, low-dose ribavirin 400– 1000 mg/d. Kwo P, et al. J Hepatol. 2009; 50: S 4. 72
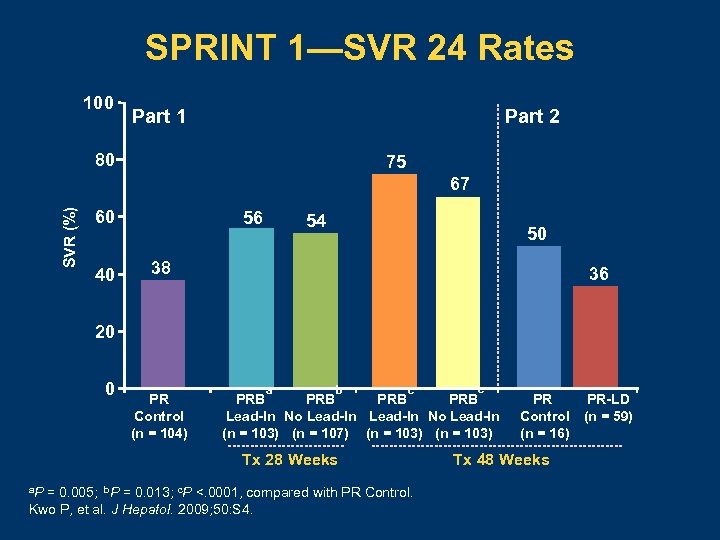
SPRINT 1—SVR 24 Rates 100 Part 1 Part 2 80 75 SVR (%) 67 60 40 56 54 50 38 36 20 0 PR Control (n = 104) a b c Tx 28 Weeks a. P c PRB PRB Lead-In No Lead-In (n = 103) (n = 107) (n = 103) = 0. 005; b. P = 0. 013; c. P <. 0001, compared with PR Control. Kwo P, et al. J Hepatol. 2009; 50: S 4. PR Control (n = 16) Tx 48 Weeks PR-LD (n = 59)
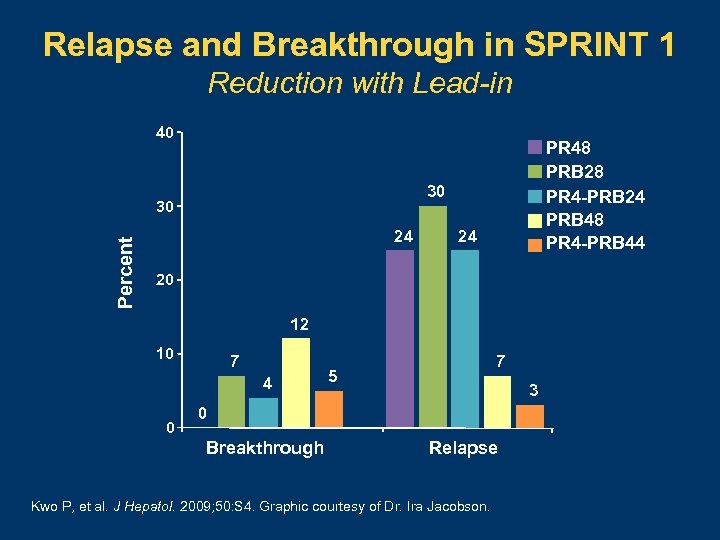
Relapse and Breakthrough in SPRINT 1 Reduction with Lead-in 40 30 30 Percent PR 48 PRB 28 PR 4 -PRB 24 PRB 48 PR 4 -PRB 44 24 24 20 12 10 7 4 0 7 5 3 0 Breakthrough Relapse Kwo P, et al. J Hepatol. 2009; 50: S 4. Graphic courtesy of Dr. Ira Jacobson.
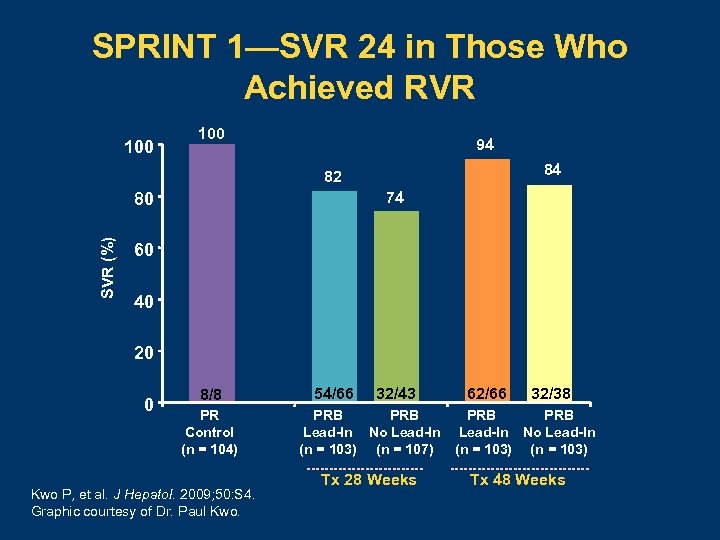
SPRINT 1—SVR 24 in Those Who Achieved RVR 100 94 84 82 74 SVR (%) 80 60 40 20 0 8/8 PR Control (n = 104) Kwo P, et al. J Hepatol. 2009; 50: S 4. Graphic courtesy of Dr. Paul Kwo. 54/66 32/43 PRB Lead-In No Lead-In (n = 103) (n = 107) Tx 28 Weeks 62/66 32/38 PRB Lead-In No Lead-In (n = 103) Tx 48 Weeks
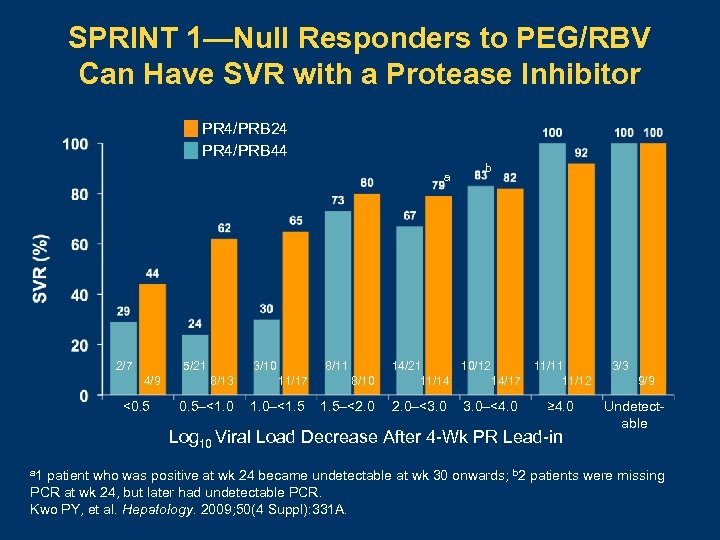
SPRINT 1—Null Responders to PEG/RBV Can Have SVR with a Protease Inhibitor PR 4/PRB 24 PR 4/PRB 44 a 2/7 5/21 4/9 <0. 5 3/10 8/11 8/13 11/17 8/10 0. 5–<1. 0–<1. 5–<2. 0 b 14/21 10/12 11/14 14/17 2. 0–<3. 0–<4. 0 11/11 11/12 ≥ 4. 0 Log 10 Viral Load Decrease After 4 -Wk PR Lead-in 3/3 9/9 Undetectable a 1 patient who was positive at wk 24 became undetectable at wk 30 onwards; b 2 patients were missing PCR at wk 24, but later had undetectable PCR. Kwo PY, et al. Hepatology. 2009; 50(4 Suppl): 331 A.
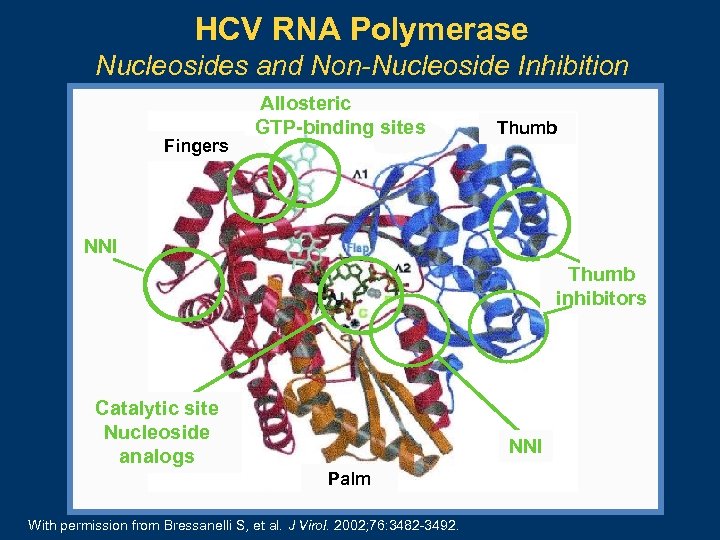
HCV RNA Polymerase Nucleosides and Non-Nucleoside Inhibition Fingers Allosteric GTP-binding sites Thumb NNI Thumb inhibitors Catalytic site Nucleoside analogs NNI Palm With permission from Bressanelli S, et al. J Virol. 2002; 76: 3482 -3492.

NS 5 B Polymerase Inhibitor Overview Efficacy l l l Many agents with antiviral activity, increased with PEG IFN + RBV Nucleosides appear active across genotypes Some non-nucleosides have potency gradient across genotypes and even G 1 subtypes

NS 5 B Polymerase Inhibitor Overview Efficacy • Phase II trials – Nucleos(t)ide polymerase inhibitors (RG 7128 + PEG FN alfa + RBV) n RVR in 85% with RG 7128 + SOC vs 10% with SOC alone 1 – Nonnucleos(t)ide polymerase inhibitors (filibuvir, ANA 598, GS 9190, ABT-333) + PEG IFN alfa + RBV n Higher rates of RVR (40%– 75%) vs SOC 2, 3 1. Lalezari J, et al. J Hepatol. 2008; 48: S 29. 2. Jacobson I, et al. J Hepatol. 2009; 48: S 382 -S 383. 3. Rodriguez-Torres M, et al. Hepatology. 2009; 50(4 suppl): LB 6.

NS 5 B Polymerase Inhibitor Overview Resistance and Adverse Effects • Differences in genetic barrier to resistance class: nucleosides have high barrier • Resistance can be minimized by combining with PEG IFN + RBV • Different resistance mutation patterns suggest potential for combinations of polymerase inhibitors • Toxicity issues with earlier agents: GI effects, hepatotoxicity, hematologic toxicity, and visual disturbance 1. Shi ST, et al. Antimicrob Agents Chemother. 2008; 52: 675 -683. 2. Lawitz E, et al. J Hepatol. 2009; 50(suppl 1): S 37. 3. Nelson D, et al. J Hepatol. 2008; 48: S 371. 4. Mc. Cown MF, et al. Antimicrob Agents Chemother. 2008; 52: 1604 -1612. 5. Lalezari J, et al. J Hepatol. 2008; 48: S 29. 6. Bavisotto L, et al. Hepatology. 2007; 46 (suppl 1): 255 A. 7. Cooper C, et al. Hepatology. 2007; 46(suppl 1): LB 11.

Highlights from EASL 2010 Protease Inhibitors • Good activity for telaprevir against genotype 21 – 3. 7 -log decline with 2 weeks of telaprevir monotherapy • Ritonavir boosting lowers dose needed for RG 7227, a protease inhibitor 2 • Development of a protease inhibitor (MK-5172) with in vitro activity against resistant variants R 155 K, A 156 T, D 168 V 3 • Excellent activity of a protease inhibitor (BI 201335) in nonresponders to PEG IFN + RBV with low breakthrough rates 4 – RVR 62%– 69% with 240 mg QD or BID, with or without 3 -day lead-in – Viral rebound at 12 weeks 16%– 22% 1. Foster GR, et al. 45 th EASL; April 14 -18, 2010. Abstract 206. 2. Gane E, et al. 45 th EASL; April 14 -18, 2010. Abstract 144. 3. Carroll S, et al. 45 th EASL; April 14 -18, 2010. Abstract 128. 4. Sulkowski M, et al. 45 th EASL; April 14 -18, 2010. Abstract 298.

Highlights from EASL 2010 NS 5 B and NS 5 A Inhibitors • SVR data for a nucleoside (RG 7128) in genotypes 2, 31 – 13/20 (65%) prior treatment failures had SVR Robust nonnucleoside with high barrier to resistance (VX-222)2 – >3 -log decline in HCV RNA with 3 days of dosing • Promise for NS 5 A inhibitors 4 • – Up to 92% RVR with BMS-790052 in dose ranging study with Peg IFN + RBV 1. Gane EJ, et al. 45 th EASL; April 14 -18, 2010. Abstract 143. 2. Rodriguez-Torres M, et al. 45 th EASL; April 14 -18, 2010. Abstract 137. 3. Jacobson I, et al. 45 th EASL; April 14 -18, 2010. Abstract 5. 4. Pol S, et al. 45 th EASL; April 14 -18, 2010. Abstract 297.

Highlights from EASL 2010 Non. STAT-C Drugs • Intravenous silibin for partial responders 1 • Potential role for immune stimulation (GI 5005, TLR-9 agonist)2, 3 • Increased SVR with vitamin D (86% vs 41%, n = 27)4 • Nitazoxanide in nonresponders (SVR 7%)5 • Cyclophilin Inhibitor Debio 025 can induce SVR ± Peg IFN after a four week course (four patients) 1. Biermer M, et al. 45 th EASL; April 14 -18, 2010. Abstract 142. 2. Jacobson I, et al. 45 th EASL; April 14 -18, 2010. Abstract 6. 3. Muir A, et al. 45 th EASL; April 14 -18, 2010. Abstract 138. 4. Mouch SA, et al. 45 th EASL; April 14 -18, 2010. Abstract 204. 5. Shiffman ML, et al. 45 th EASL; April 14 -18, 2010. Abstract 296.
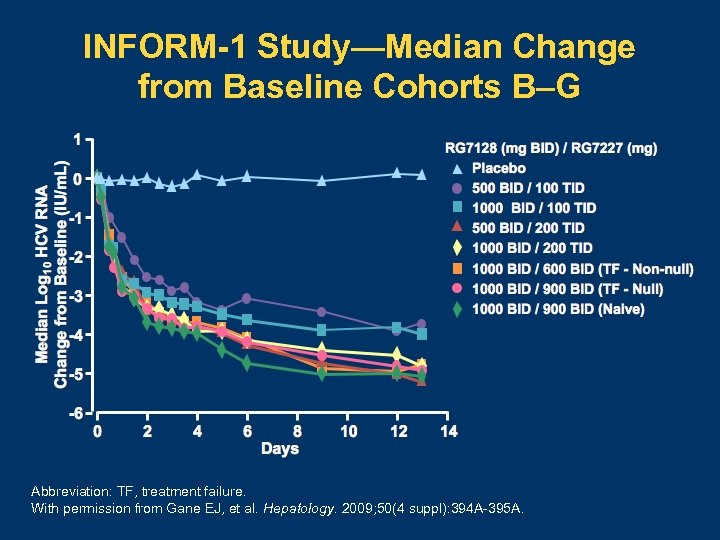
INFORM-1 Study—Median Change from Baseline Cohorts B–G Abbreviation: TF, treatment failure. With permission from Gane EJ, et al. Hepatology. 2009; 50(4 suppl): 394 A-395 A.
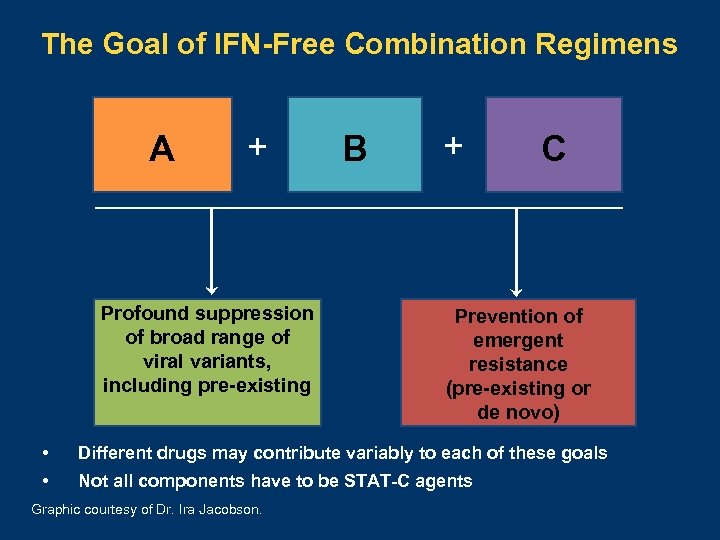
The Goal of IFN-Free Combination Regimens A A + Profound suppression of broad range of viral variants, including pre-existing B + C Prevention of emergent resistance (pre-existing or de novo) • Different drugs may contribute variably to each of these goals • Not all components have to be STAT-C agents Graphic courtesy of Dr. Ira Jacobson.

New Anti-HCV Therapies Anticipated Developments • • • Approval of telaprevir/boceprevir seems likely in 2011 Other direct antivirals and compounds with novel mechanisms of action in development Response-guided therapy Incorporation of genetic testing Accelerated development of interferon-free combinations The question for clinicians: to wait or not to wait?

ARS Question: Among your genotype 1 patients, how frequently will you be incorporating novel agents once they are approved? 1) Always 2) Very Often 3) Sometimes 4) Rarely 5) Never
35d4faf2f4a85e6c1c4ae0c0bbb82c76.ppt