5b59071a8e9b2a227d14eaff1ee53b51.ppt
- Количество слайдов: 63

SMTC Biofouling on Membranescauses, consequences, control Tony Fane et al. Singapore membrane technology centre, ntu unesco centre for membranes, unsw Tony Fane | Director | Singapore Membrane Technology Centre Nanyang Technological University 1 Singapore 639798| Phone: 65 -67905272 | Email: agfane@ntu. edu. sg

Et al. Acknowledgements Ziggy Chong Stani Suwarno Jinsong Zhang Scott Rice Diane Mc. Dougald Staffan Kjelleberg Yehuda Cohen Chen Xi Harvey Winters Greg Leslie SMTC

Special Acknowledgements SMTC Biofilm = cells immobilised at a substratum and frequently embedded in an organic polymer matrix of microbial origin. . . which is not necessarily uniform in time or space [Characklis and Marshall (eds. ) Biofilms ; a basis for an interdisciplinary approach, 1990] Professor Kevin Marshall (1933 -2010) Microbiology, UNSW
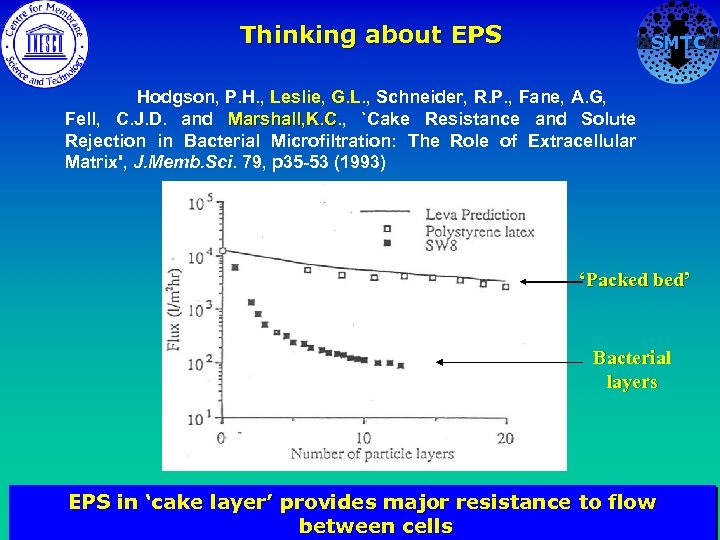
Thinking about EPS SMTC Hodgson, P. H. , Leslie, G. L. , Schneider, R. P. , Fane, A. G, Fell, C. J. D. and Marshall, K. C. , `Cake Resistance and Solute Marshall, K. C Rejection in Bacterial Microfiltration: The Role of Extracellular Matrix', J. Memb. Sci. 79, p 35 -53 (1993) ‘Packed bed’ Bacterial layers EPS in ‘cake layer’ provides major resistance to flow between cells
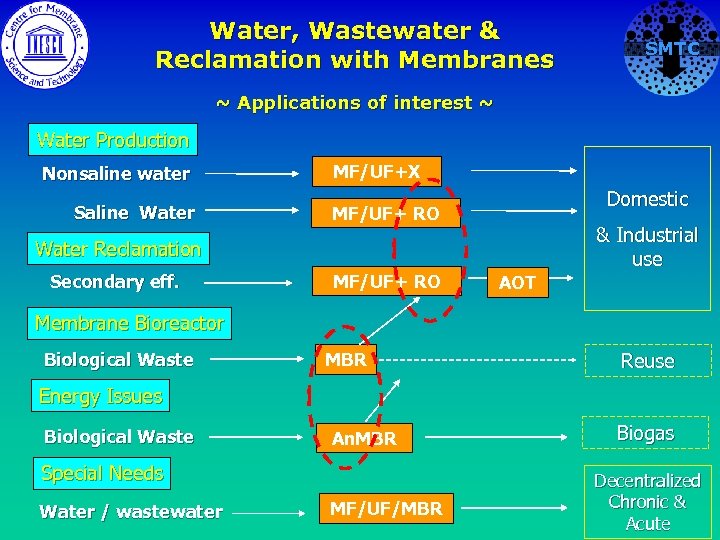
Water, Wastewater & Reclamation with Membranes SMTC ~ Applications of interest ~ Water Production Nonsaline water Saline Water MF/UF+X Domestic MF/UF+ RO & Industrial use Water Reclamation Secondary eff. MF/UF+ RO AOT Membrane Bioreactor Biological Waste MBR Reuse Energy Issues Biological Waste An. MBR Special Needs Water / wastewater MF/UF/MBR Biogas Decentralized Chronic & Acute

Biofouling in RO SMTC Ridgway Confocal image of biofilm on RO membrane Bacteria and EPS in biofilm – WF 21 Heavily fouled spiral wound membrane

Contents Introduction Consequences Fouling contributions (resistance & CP) Method for measurement Biofouling in RO – effect of flux Biofouling in RO – role of spacers Biofouling controls – examples Conclusions SMTC
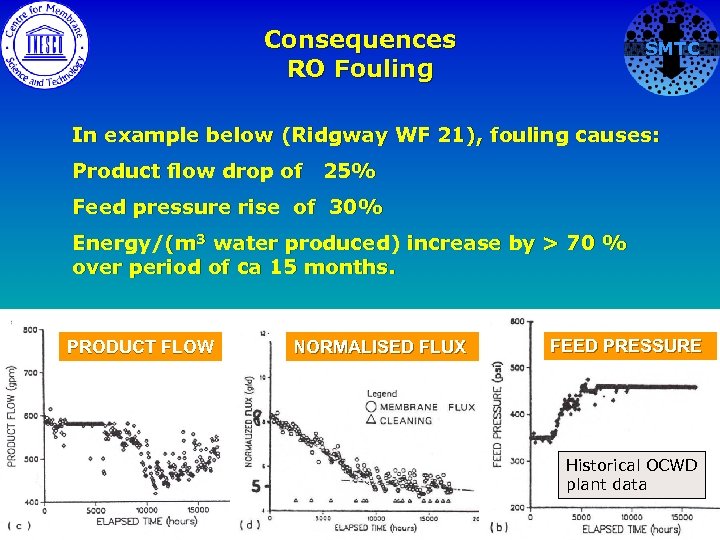
Consequences RO Fouling SMTC In example below (Ridgway WF 21), fouling causes: Product flow drop of 25% Feed pressure rise of 30% Energy/(m 3 water produced) increase by > 70 % over period of ca 15 months. PRODUCT FLOW NORMALISED FLUX FEED PRESSURE Historical OCWD plant data

Membrane Fouling Factors • The nature of the feed to the membrane • The membrane properties • The operating environment SMTC
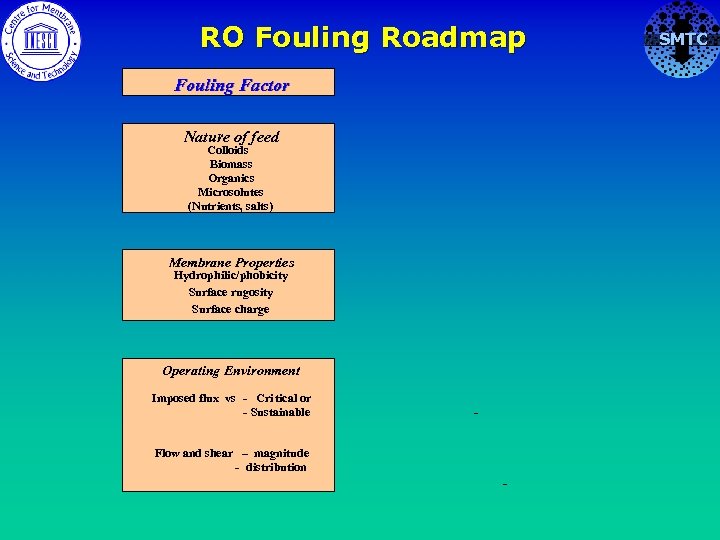
RO Fouling Roadmap Fouling Factor Nature of feed Colloids Biomass Organics Microsolutes (Nutrients, salts) Membrane Properties Hydrophilic/phobicity Surface rugosity Surface charge Operating Environment Imposed flux vs - Cri tical or - Sustainable - Flow and shear – magnitude - distribution - SMTC
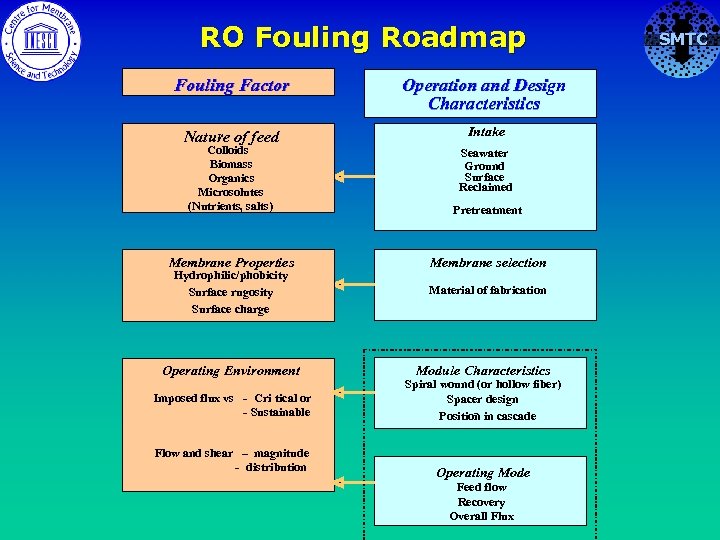
RO Fouling Roadmap Fouling Factor Operation and Design Characteristics Nature of feed Intake Colloids Biomass Organics Microsolutes (Nutrients, salts) Pretreatment Membrane Properties Membrane selection Hydrophilic/phobicity Surface rugosity Surface charge Operating Environment Seawater Ground Surface Reclaimed Material of fabrication Module Characteristics Imposed flux vs - Cri tical or - Sustainable Spiral wound (or hollow fiber) Spacer design Position in cascade Flow and shear – magnitude - distribution Operating Mode Feed flow Recovery Overall Flux SMTC
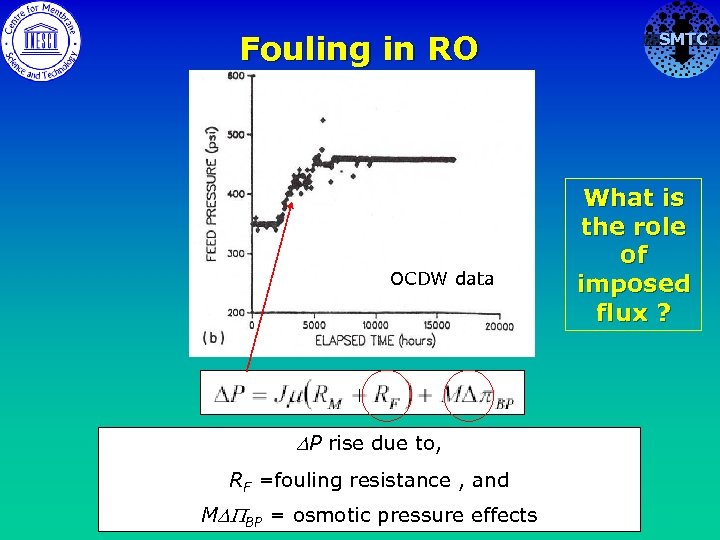
Fouling in RO OCDW data DP rise due to, RF =fouling resistance , and MDPBP = osmotic pressure effects SMTC What is the role of imposed flux ?
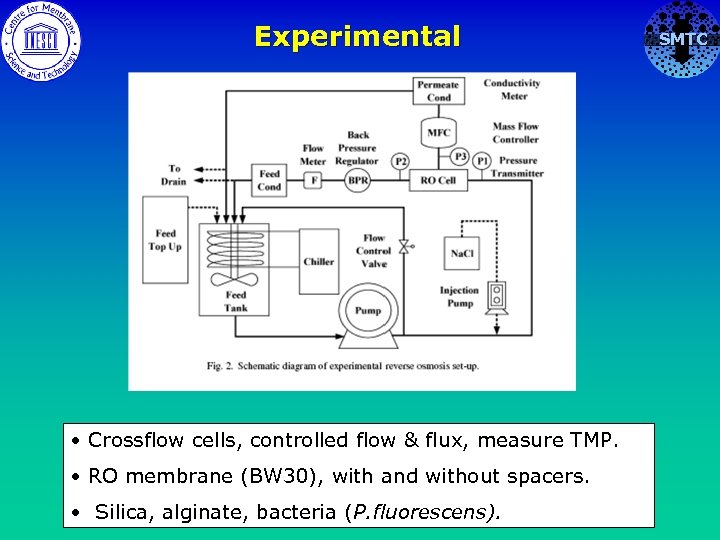
Experimental • Crossflow cells, controlled flow & flux, measure TMP. • RO membrane (BW 30), with and without spacers. • Silica, alginate, bacteria (P. fluorescens). SMTC
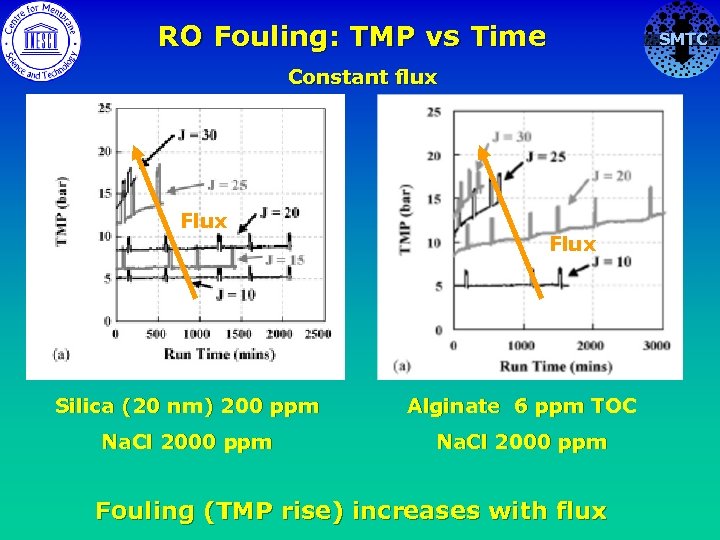
RO Fouling: TMP vs Time Constant flux Flux Silica (20 nm) 200 ppm Alginate 6 ppm TOC Na. Cl 2000 ppm Fouling (TMP rise) increases with flux SMTC
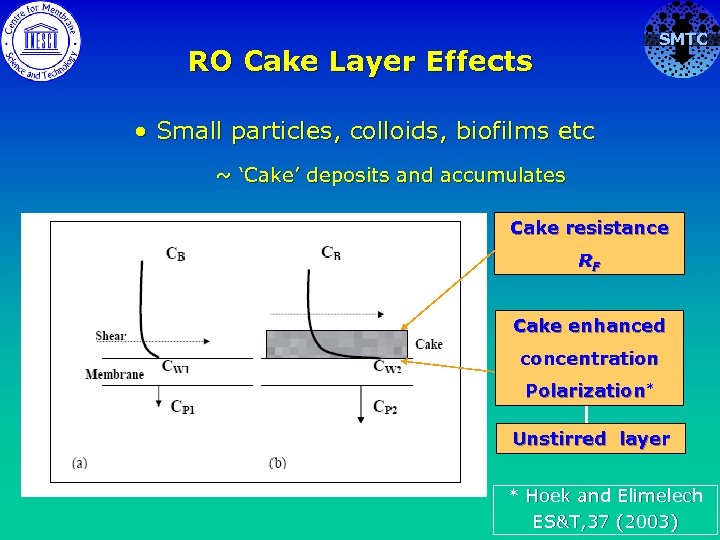
SMTC RO Cake Layer Effects • Small particles, colloids, biofilms etc ~ ‘Cake’ deposits and accumulates Cake resistance RF Cake enhanced concentration Polarization* Unstirred layer * Hoek and Elimelech ES&T, 37 (2003)

Basic Theory ~ effect of fouling SMTC Basic relationship TMP required Pressure increases due to Rf + osmotic pressure Polarization increases with flux & decreases with mass transfer, k Mass transfer in deposit layer is << k in the boundary layer Cake height

TMP at fixed flux Cake enhanced osmotic Pressure (CEOP) Unfouled M = 1. 2 Fouled cake (dc is 15. 0 mm, e = 0. 3) M = 2. 4 CEOP increases DP by 2. 0 x Depends on deposit thickness and voidage, not ‘particle’ size Fouling resistance and CEOP can both have major impact in RO SMTC
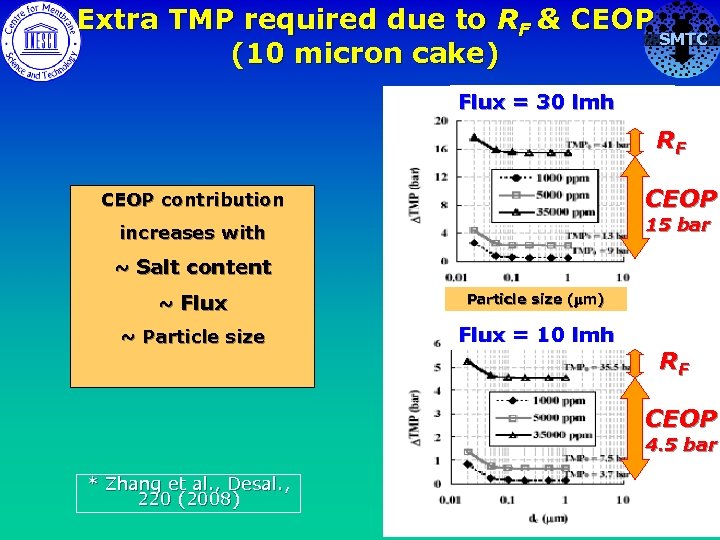
Extra TMP required due to RF & CEOP SMTC (10 micron cake) Flux = 30 lmh RF CEOP contribution CEOP increases with 15 bar ~ Salt content ~ Flux Particle size (mm) ~ Particle size Flux = 10 lmh RF CEOP 4. 5 bar * Zhang et al. , Desal. , 220 (2008)
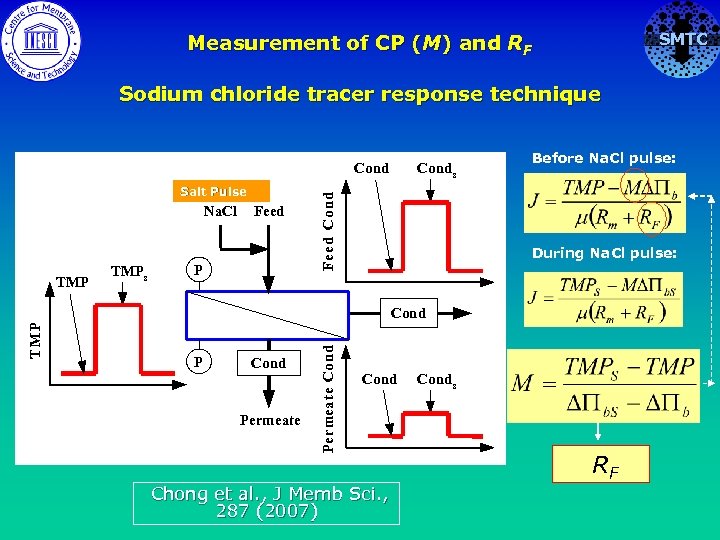
SMTC Measurement of CP (M) and RF Sodium chloride tracer response technique Salt Pulse Na. Cl TMP s Feed P Cond s Fe ed Cond Before Na. Cl pulse: During Na. Cl pulse: P Cond Permeate Cond T MP Cond Chong et al. , J Memb Sci. , 287 (2007) Cond s RF
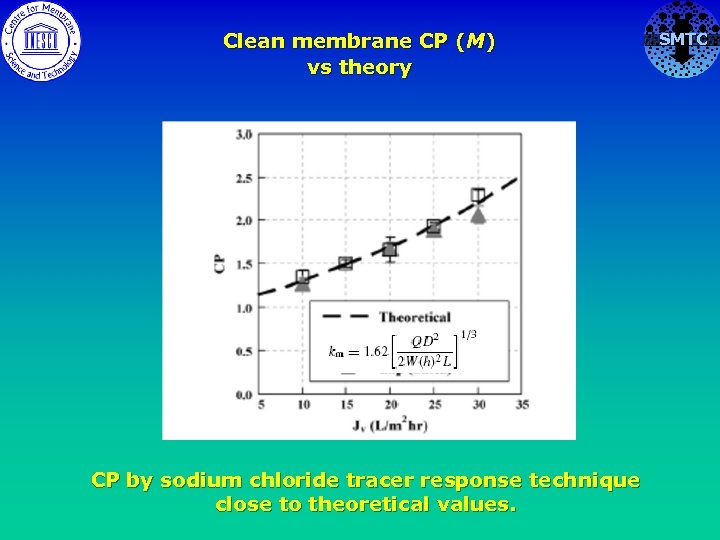
Clean membrane CP (M) vs theory CP by sodium chloride tracer response technique close to theoretical values. SMTC
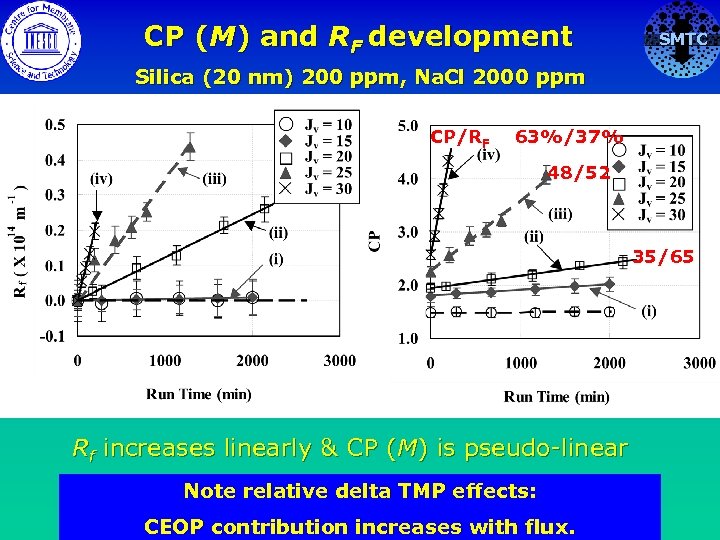
CP (M) and RF development SMTC Silica (20 nm) 200 ppm, Na. Cl 2000 ppm CP/RF 63%/37% 48/52 35/65 Rf increases linearly & CP (M) is pseudo-linear Note relative delta TMP effects: CEOP contribution increases with flux.
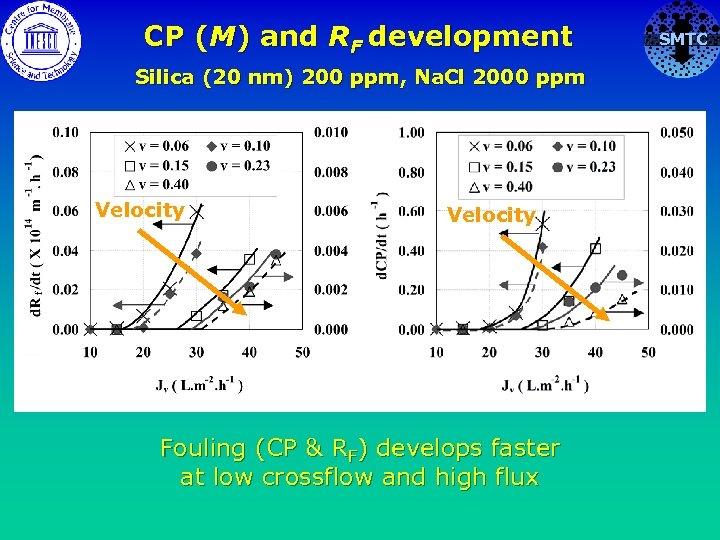
CP (M) and RF development Silica (20 nm) 200 ppm, Na. Cl 2000 ppm Velocity Fouling (CP & RF) develops faster at low crossflow and high flux SMTC
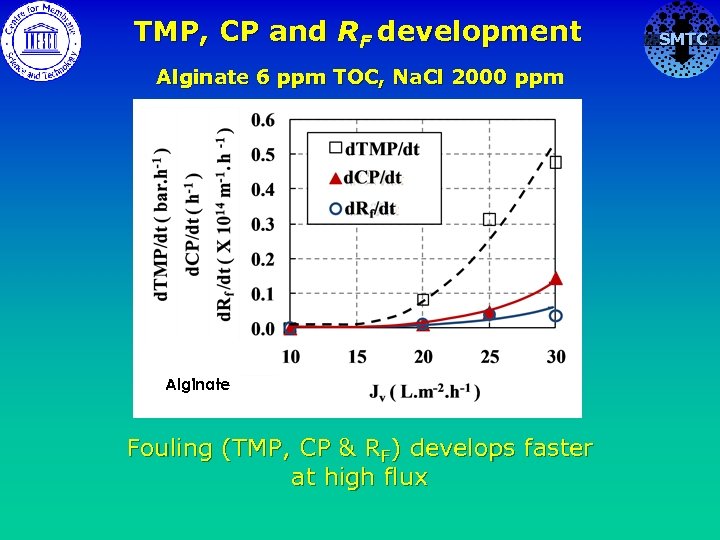
TMP, CP and RF development Alginate 6 ppm TOC, Na. Cl 2000 ppm Alginate Fouling (TMP, CP & RF) develops faster at high flux SMTC

Summary SMTC q At fixed flux fouling manifests as rise in required TMP. q Cause of this rise is hydraulic resistance (RF) and enhanced CP (leading to higher DP ) (CEOP). q CEOP contribution can be significant and is measurable (in the lab). q RO fouling effect increases (exponentially) with imposed flux, and decreased crossflow.
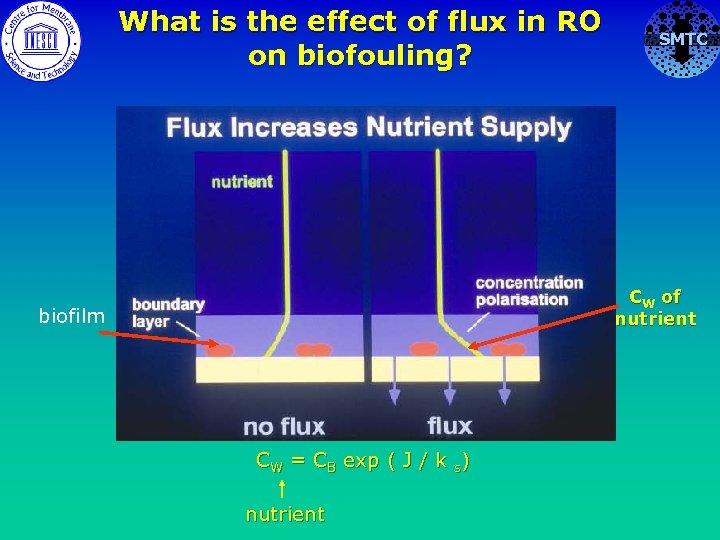
What is the effect of flux in RO on biofouling? SMTC CW of nutrient biofilm CW = CB exp ( J / k s) nutrient
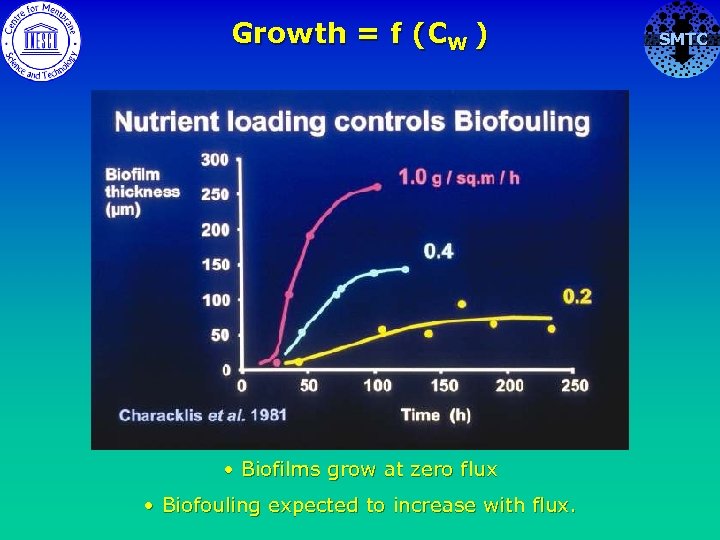
Growth = f (CW ) • Biofilms grow at zero flux • Biofouling expected to increase with flux. SMTC

Biofouling vs flux q q q Is biofouling flux dependent? Does biofilm present as a ‘resistance’ or an enhanced concentration polarization effect? Controlled flux tests using p. fluorescens and model feed (Na. Cl + nutrient). Chong et al. , J Memb Sci. , 325 (2008) SMTC

Experimental • Crossflow cells, controlled flow & flux, measure TMP. • RO membrane (BW 30), without spacers. • P. fluorescens at ca. 2 x 105 CFU/ml continuous feed. • 2000 ppm Nacl, TOC ~ 6 ppm SMTC
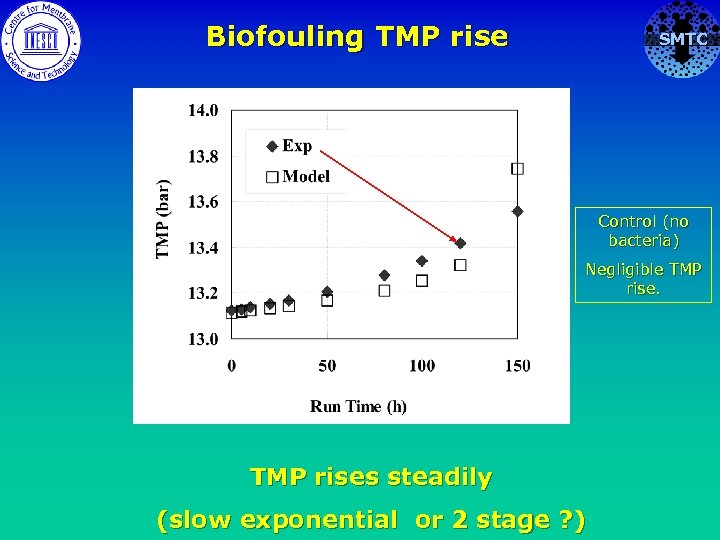
Biofouling TMP rise SMTC Control (no bacteria) Negligible TMP rises steadily (slow exponential or 2 stage ? )
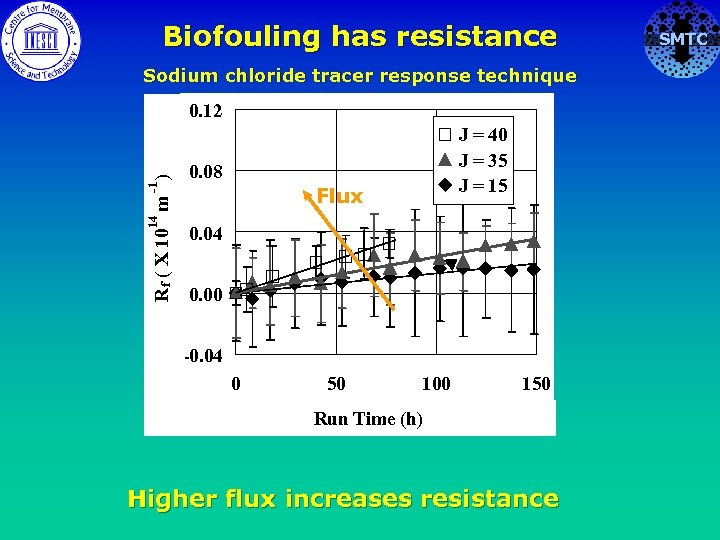
Biofouling has resistance Sodium chloride tracer response technique 14 -1 R f ( X 10 m ) 0. 12 J = 40 J = 35 J = 15 0. 08 Flux 0. 04 0. 00 -0. 04 0 50 100 150 Run Time (h) Higher flux increases resistance SMTC
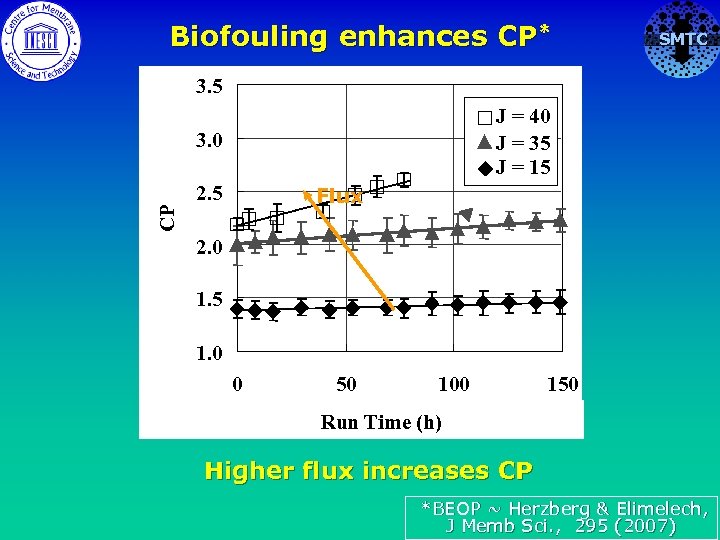
Biofouling enhances CP* SMTC 3. 5 J = 40 J = 35 J = 15 CP 3. 0 2. 5 Flux 2. 0 1. 5 1. 0 0 50 100 150 Run Time (h) Higher flux increases CP *BEOP ~ Herzberg & Elimelech, J Memb Sci. , 295 (2007)
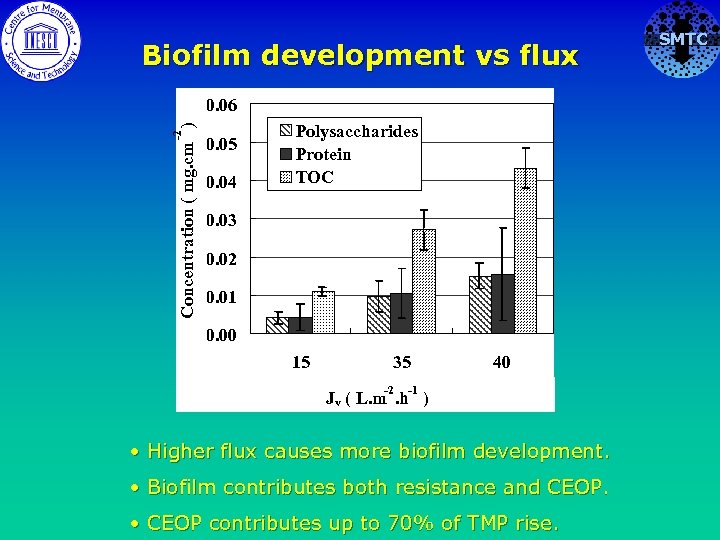
Biofilm development vs flux -2 Concentration ( mg. cm ) 0. 06 0. 05 0. 04 Polysaccharides Protein TOC 0. 03 0. 02 0. 01 0. 00 15 35 -2 40 -1 Jv ( L. m. h ) • Higher flux causes more biofilm development. • Biofilm contributes both resistance and CEOP. • CEOP contributes up to 70% of TMP rise. SMTC
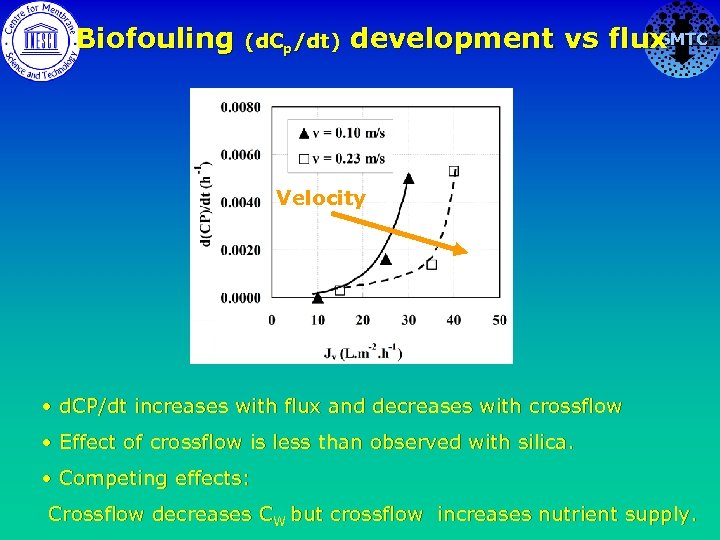
Biofouling (d. Cp/dt) SMTC development vs flux Velocity • d. CP/dt increases with flux and decreases with crossflow • Effect of crossflow is less than observed with silica. • Competing effects: Crossflow decreases CW but crossflow increases nutrient supply.
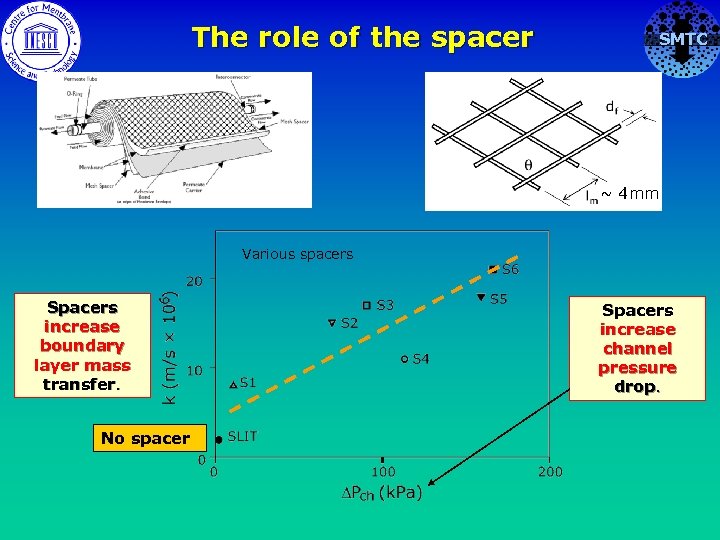
The role of the spacer SMTC ~ 4 mm Various spacers Spacers increase boundary layer mass transfer. No spacer Spacers increase channel pressure drop.
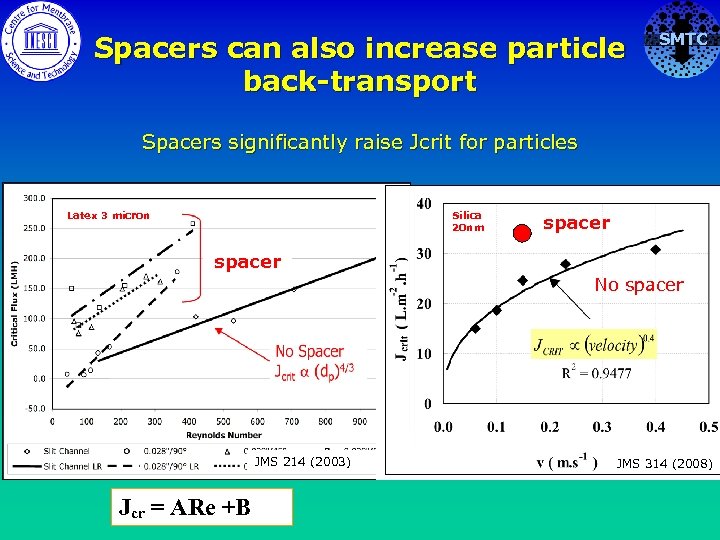
Spacers can also increase particle back-transport SMTC Spacers significantly raise Jcrit for particles Latex 3 micron Silica 20 nm spacer No spacer JMS 214 (2003) Jcr = ARe +B JMS 314 (2008)

Spacers can also promote localized deposition Diamond spacer Ladder spacer SMTC Local variations in Jcrit due to local variations in surface shear ~ causes patchy deposition
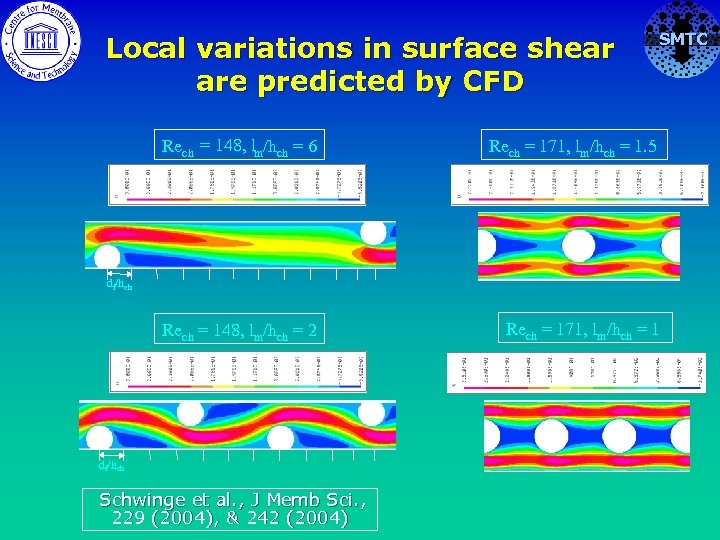
Local variations in surface shear are predicted by CFD SMTC Rech = 148, lm/hch = 6 Rech = 171, lm/hch = 1. 5 Rech = 148, lm/hch = 2 Rech = 171, lm/hch = 1 df/hch Schwinge et al. , J Memb Sci. , 229 (2004), & 242 (2004)

Biofouling with spacers • P. aeruginosa at ca. 2 x 105 CFU/ml continuous feed. • 2000 ppm Nacl, TOC ~ 6 ppm • Constant flux 35 lmh, • Cross flow velocity ~ 0. 2 m/s • Monitoring TMP • Some autopsies after experimental run • Biofilm removal tests SMTC
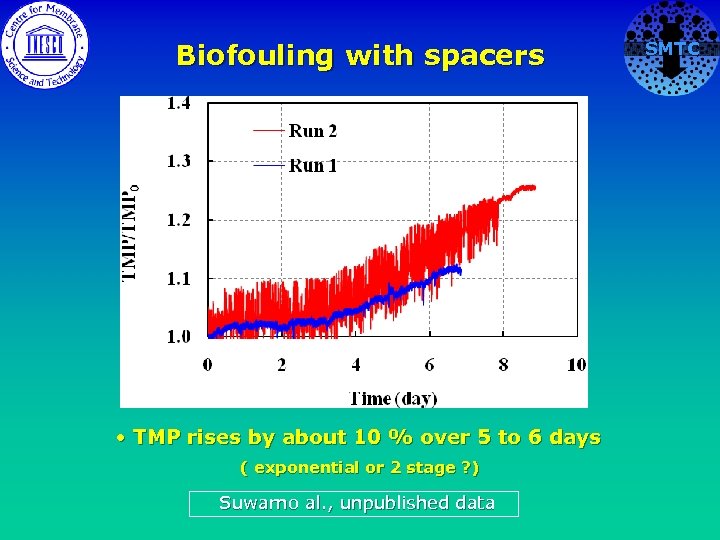
Biofouling with spacers • TMP rises by about 10 % over 5 to 6 days ( exponential or 2 stage ? ) Suwarno al. , unpublished data SMTC
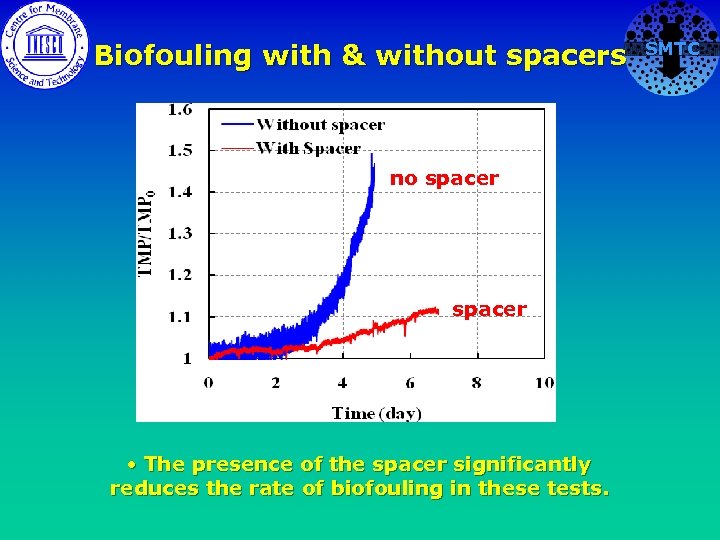
Biofouling with & without spacers no spacer • The presence of the spacer significantly reduces the rate of biofouling in these tests. SMTC
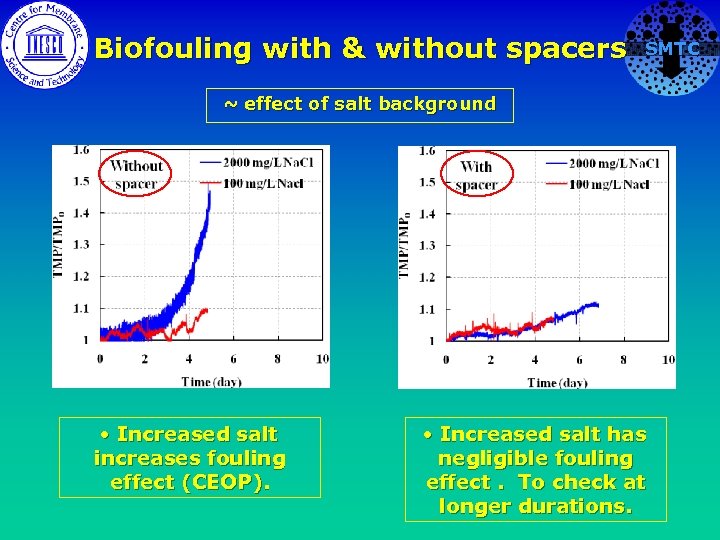
Biofouling with & without spacers SMTC ~ effect of salt background • Increased salt increases fouling effect (CEOP). • Increased salt has negligible fouling effect. To check at longer durations.
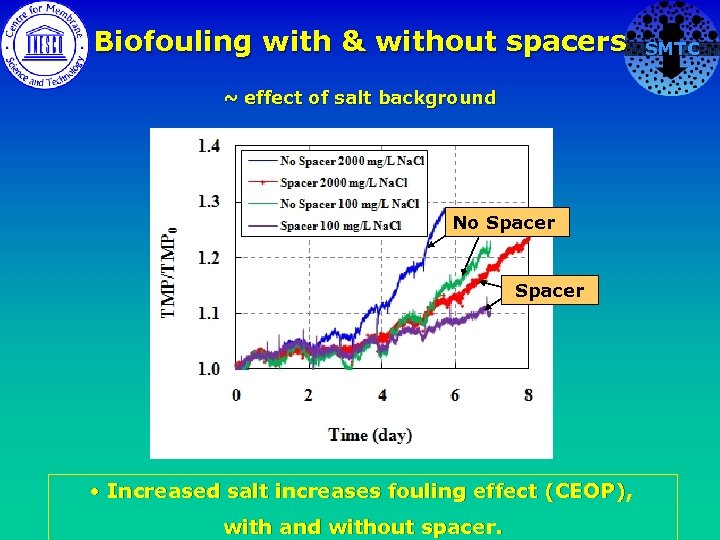
Biofouling with & without spacers ~ effect of salt background No Spacer • Increased salt increases fouling effect (CEOP), with and without spacer. SMTC
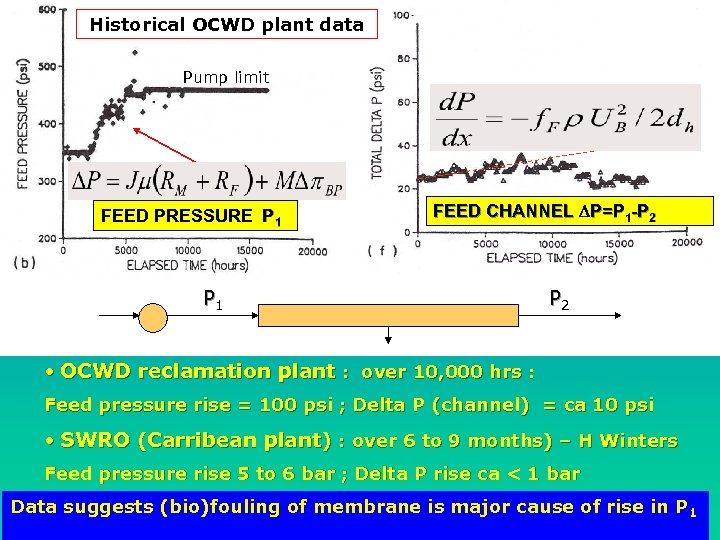
Historical OCWD plant data SMTC Pump limit FEED PRESSURE P 1 FEED CHANNEL DP=P 1 -P 2 P 1 P 2 • OCWD reclamation plant : over 10, 000 hrs : Feed pressure rise = 100 psi ; Delta P (channel) = ca 10 psi • SWRO (Carribean plant) : over 6 to 9 months) – H Winters Feed pressure rise 5 to 6 bar ; Delta P rise ca < 1 bar Data suggests (bio)fouling of membrane is major cause of rise in P 1

Biofilm development - The role of the spacer ? SMTC • Evidence for biofouling on spacers and membrane. • Spacer accounts for delta P rise in biofouled modules. • Does not explain why biofouling increases with flux. • Does not explain why P 1 rise >> delta P • Possible mechanism; Biofouling 1. Biofouling initiates on spacer Spacer biofouling causes delta rise causes delta PP rise Biofilm spreads to 2. Biofilm spreads to ‘nutrient rich’ membrane surface Membrane biofouling causes rise – causes TMPTMP rise – magnitude depends on CEOP (more effect at higher salinity)
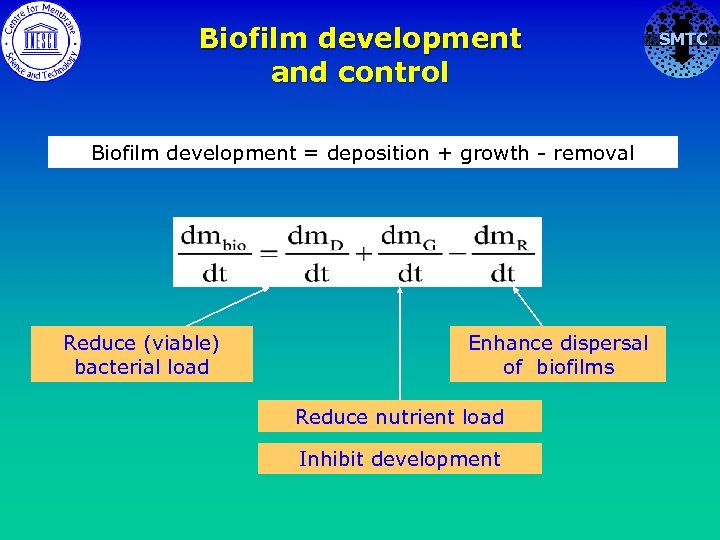
Biofilm development and control Biofilm development = deposition + growth - removal Reduce (viable) bacterial load Enhance dispersal of biofilms Reduce nutrient load Inhibit development SMTC
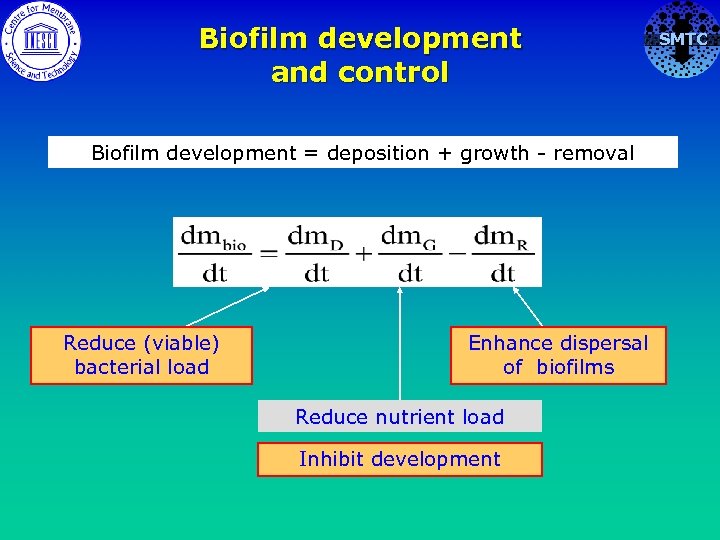
Biofilm development and control Biofilm development = deposition + growth - removal Reduce (viable) bacterial load Enhance dispersal of biofilms Reduce nutrient load Inhibit development SMTC

RO Biofouling Control • UV to inactivate bacteria • Biomimicry control SMTC
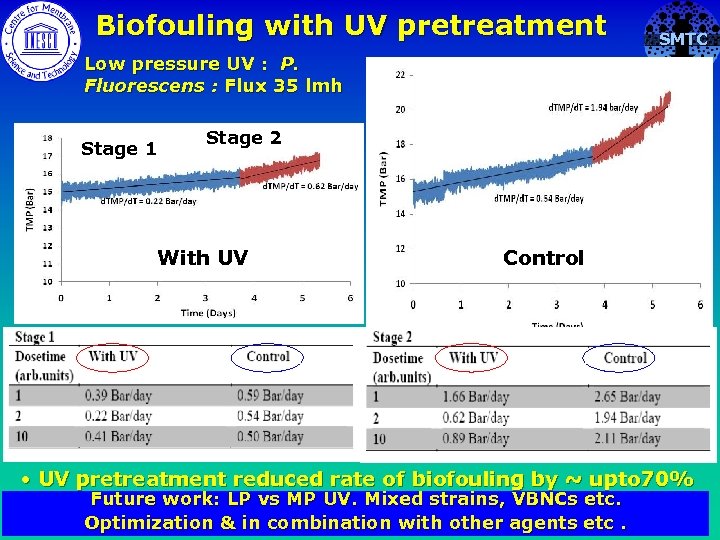
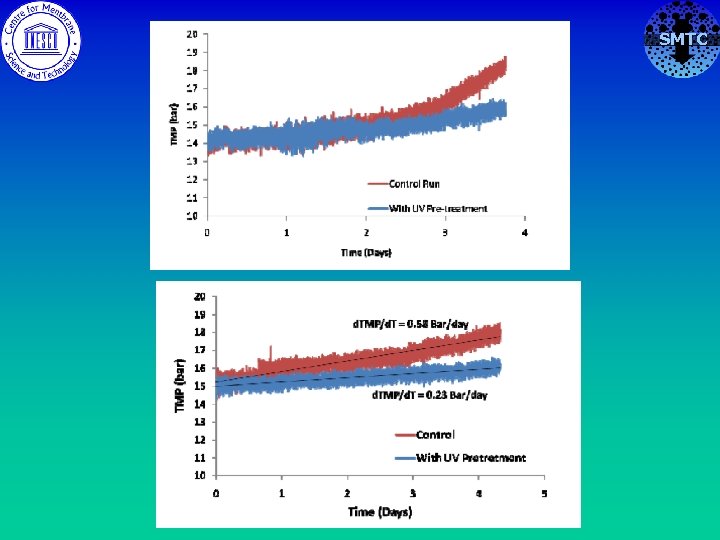
Biofouling with UV pretreatment SMTC Low pressure UV : P. Fluorescens : Flux 35 lmh Stage 1 Stage 2 With UV Control • UV pretreatment reduced rate of biofouling by ~ upto 70% Future work: LP vs MP UV. . Mixed strains, VBNCs etc. Optimization & in combination with other agents etc.

RO Biofouling Control SMTC • UV to inactivate bacteria • Biomimicry control ~ Dispersal agents (NO) ~ Q Sensing interference (Furanones)

Nitric Oxide as a Biofilm dispersal agent Staffan Kjelleberg, Scott Rice et al. SMTC
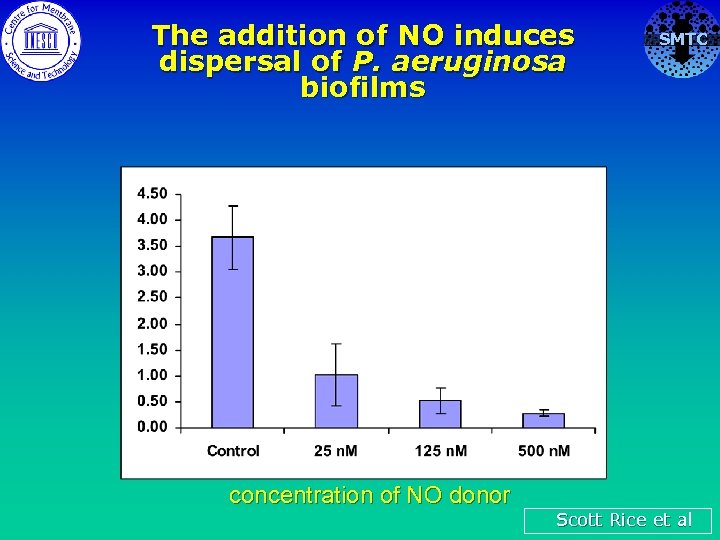
The addition of NO induces dispersal of P. aeruginosa biofilms concentration of NO donor SMTC Scott Rice et al

NO donors work synergistically SMTC with traditional biocides NO is effective for biofilms formed in recycled water systems Scott Rice et al

Nitric oxide reduces biofilms on RO membranes n RO membrane parameters: Operation time 7 days Starting TMP : 14. 6372 bar Ending TMP : 19. 8176 bar Flux : 35 LMH Cross-flow velocity : 0. 17 m/s Na. Cl Concentration : 4000 mg/L NB Concentration : 20 mg/L Membrane operation with Spacers NO treatment: 2 m. M SNP donor for 2 h in batch n Stain with viability stains, EPS stains, total cell stains CFU counts from the membrane

NO treatment reduces biofilms on RO membranes • Preliminary data, based on CFU and staining indicate that NO treatment reduces pre-established biofilms • The percentage of biofilm reduction was concentration dependent: • 2 m. M , 1 m. M and 0. 5 m. M 95 %, 70 % and 25% reduction in CFU counts. -NO +NO Future work: In situ application during RO operation. How ? When ? In combination with other agents etc ?

SMTC Biofouling in RO • Flux drives biofouling in RO. • Fouling presents as a resistance and enhanced CP. • Fouling can be reduced by decreasing flux or increasing crossflow velocity (trade-off in capital & energy costs). • Other strategies, - membrane & spacer developments, - improved pretreatment (UV) or novel additives (dispersing agents; QS inhibitors)

ACKNOWLEDGEMENTS Colleagues and students at NTU and UNSW. ASTAR & EWI Singapore ARC Australia PUB Singapore & Trojan Technologies SMTC

Materials and Methods • Model foulants – (i) colloidal silica (20 nm, 200 mg/L), (ii) alginic acid (6 mg/L as TOC), (iii) Pseudomonas fluorescens (ATCC 700830, 105 CFU/m. L, 20 mg/L Nutrient Broth) with background salinity of 2000 mg/L Na. Cl • Experimental protocols – Equilibrate with Milli-Q water for 12 hours (membrane compaction) – Add in Na. Cl as background ions solution (and add in Nutrient Broth for biofouling studies) and equilibrate for 2 hours – Add in model foulants or continuous injection of bacteria solution to initiate fouling process – Monitor TMP and membrane autopsy at the end of experiment • Test conditions – Fixed flux operation – No feed spacer used (to accelerate fouling process) – Vary flux level, crossflow velocity, feed pressure IDA WORLD CONGRESS 2009 Paper DB 09 -128: The Effect of Flux and Pressure on Fouling in RO Desalination 57
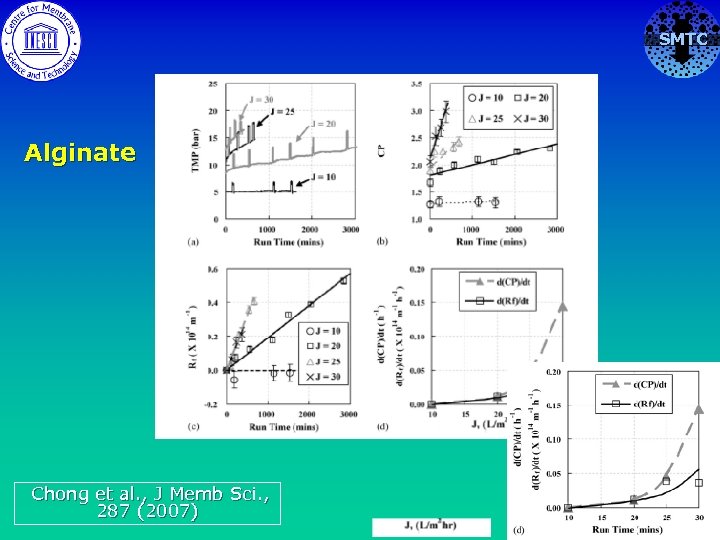
SMTC Alginate Chong et al. , J Memb Sci. , 287 (2007)
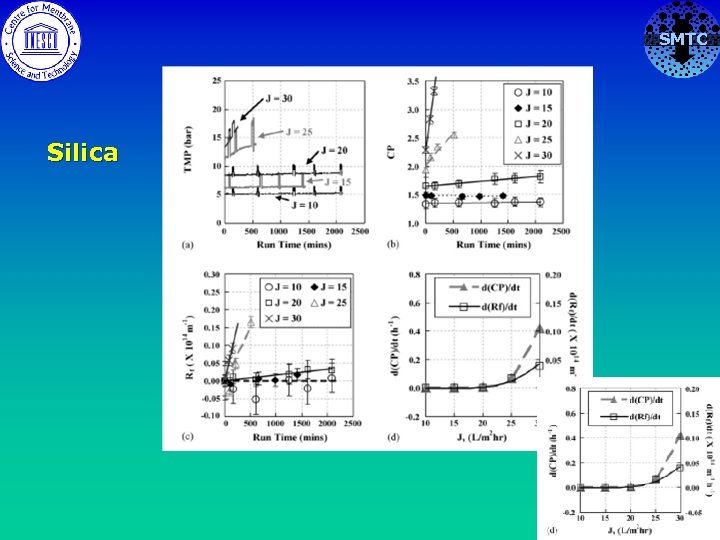
SMTC Silica
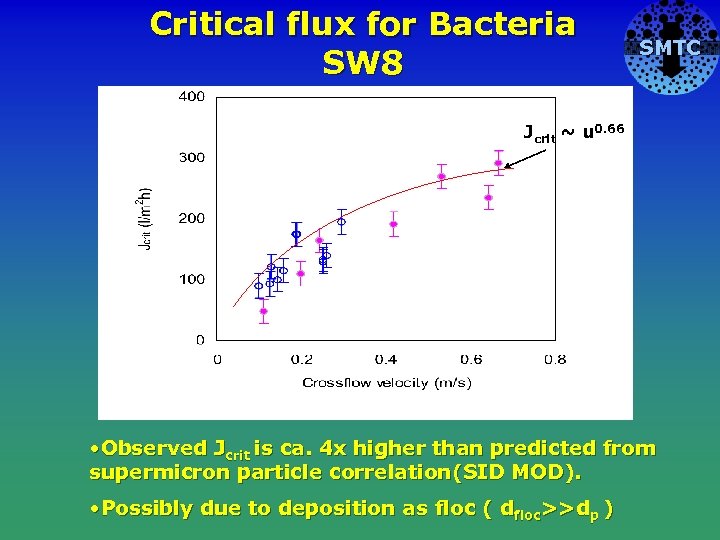
Critical flux for Bacteria SW 8 SMTC Jcrit ~ u 0. 66 Cake No cake • Observed Jcrit is ca. 4 x higher than predicted from supermicron particle correlation(SID MOD). • Possibly due to deposition as floc ( dfloc>>dp )
SMTC
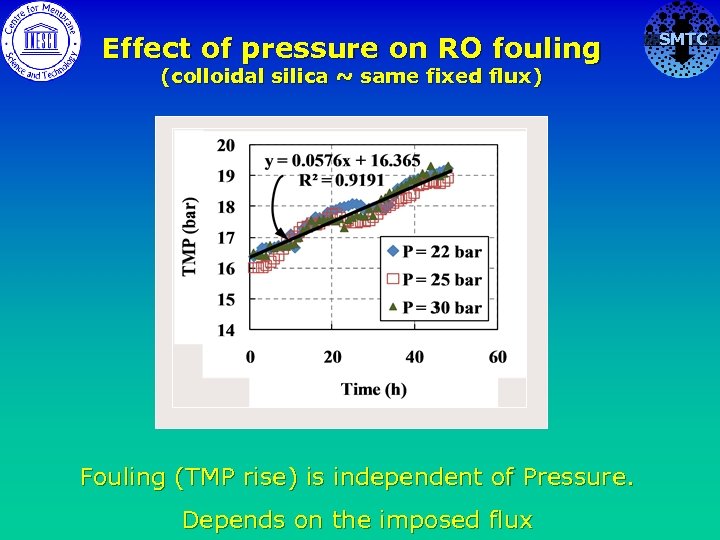
Effect of pressure on RO fouling (colloidal silica ~ same fixed flux) Fouling (TMP rise) is independent of Pressure. Depends on the imposed flux SMTC
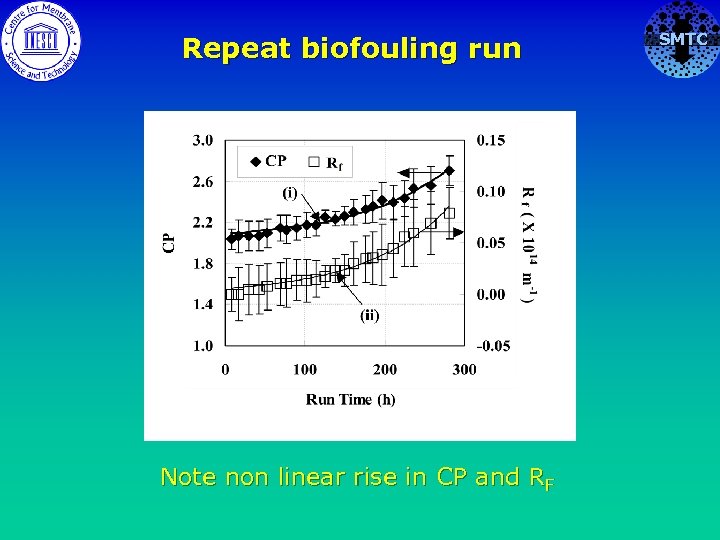
Repeat biofouling run Note non linear rise in CP and RF SMTC
5b59071a8e9b2a227d14eaff1ee53b51.ppt