559d057ce893740900f3e9142f62b9fc.ppt
- Количество слайдов: 18
 Siliceous Mesocellular Foam (MCF) for Green Chemistry Jaehong Lim, Su Seong Lee and Jackie Y. Ying Institute of Bioengineering and Nanotechnology, Singapore 12 th Annual Green Chemistry and Engineering Conference June 25, 2008 Washington, DC
Siliceous Mesocellular Foam (MCF) for Green Chemistry Jaehong Lim, Su Seong Lee and Jackie Y. Ying Institute of Bioengineering and Nanotechnology, Singapore 12 th Annual Green Chemistry and Engineering Conference June 25, 2008 Washington, DC
 IBN, Biopolis, Singapore IBN @ The Nanos Helios Chromos Nanos Centros Matrix Proteos Genome
IBN, Biopolis, Singapore IBN @ The Nanos Helios Chromos Nanos Centros Matrix Proteos Genome
 Heterogenization of Catalysts Shortcomings of Homogeneous Catalysts • High cost • Low stability • Difficulties in reuse • Toxic residues in product Benefits of Heterogenization • Cost-effective • Good stability • Easy purification • Reduced toxicity in product • Recycling use of expensive material • Feasible for scale-up • Potential application as a continuous flow process
Heterogenization of Catalysts Shortcomings of Homogeneous Catalysts • High cost • Low stability • Difficulties in reuse • Toxic residues in product Benefits of Heterogenization • Cost-effective • Good stability • Easy purification • Reduced toxicity in product • Recycling use of expensive material • Feasible for scale-up • Potential application as a continuous flow process
 Spherical Mesocellular Foam (MCF) Conventional MCF Spherical MCF - Effective for catalysis and separation - Easy to prepare - Robust - Uniform particle size - Large pores - Large surface area - Potential scale-up Particle size: ~5 mm Pore size: ~25 nm Window size: ~15 nm Wall width: ~5 nm Pore volume: 2. 0 -2. 5 cm 3/g BET surface area: 500 -600 m 2/g
Spherical Mesocellular Foam (MCF) Conventional MCF Spherical MCF - Effective for catalysis and separation - Easy to prepare - Robust - Uniform particle size - Large pores - Large surface area - Potential scale-up Particle size: ~5 mm Pore size: ~25 nm Window size: ~15 nm Wall width: ~5 nm Pore volume: 2. 0 -2. 5 cm 3/g BET surface area: 500 -600 m 2/g
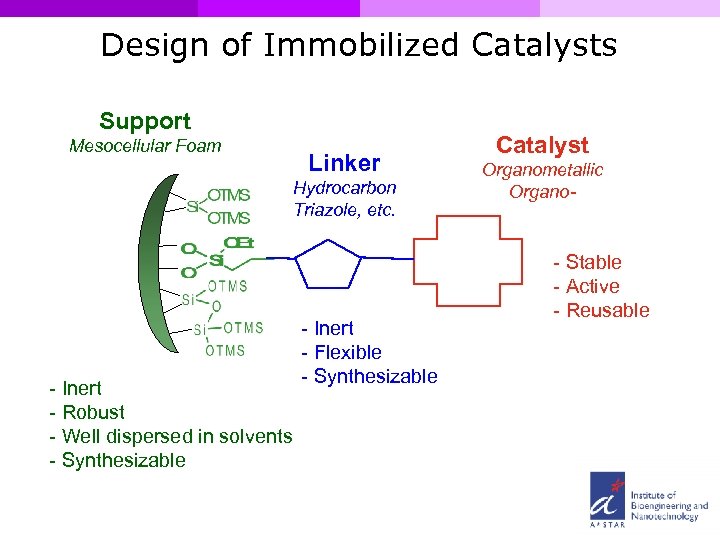 Design of Immobilized Catalysts Support Mesocellular Foam Linker Hydrocarbon Triazole, etc. - Inert - Robust - Well dispersed in solvents - Synthesizable - Inert - Flexible - Synthesizable Catalyst Organometallic Organo- - Stable - Active - Reusable
Design of Immobilized Catalysts Support Mesocellular Foam Linker Hydrocarbon Triazole, etc. - Inert - Robust - Well dispersed in solvents - Synthesizable - Inert - Flexible - Synthesizable Catalyst Organometallic Organo- - Stable - Active - Reusable
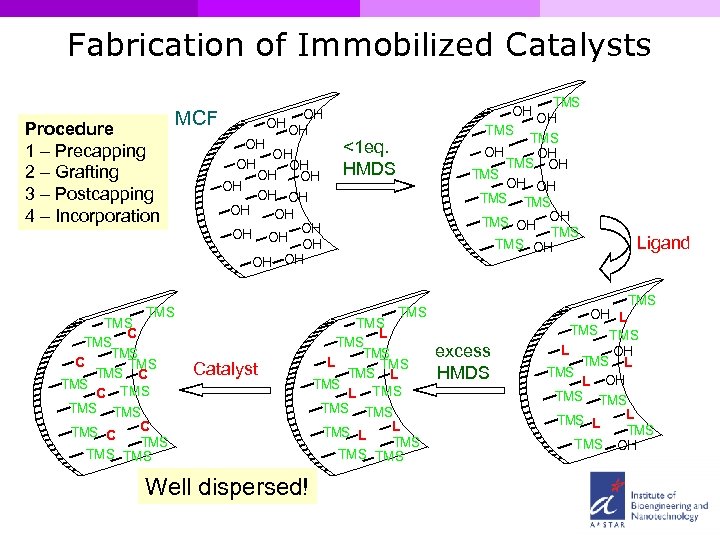 Fabrication of Immobilized Catalysts Procedure 1 – Precapping 2 – Grafting 3 – Postcapping 4 – Incorporation MCF OH OH OH OH OH TMS TMS C TMS C TMS TMS TMS OH OH TMS OH OH TMS TMS OH <1 eq. HMDS TMS Catalyst Well dispersed! TMS L TMS L TMS TMS TMS excess HMDS Ligand TMS OH L TMS L OH TMS L TMS TMS OH
Fabrication of Immobilized Catalysts Procedure 1 – Precapping 2 – Grafting 3 – Postcapping 4 – Incorporation MCF OH OH OH OH OH TMS TMS C TMS C TMS TMS TMS OH OH TMS OH OH TMS TMS OH <1 eq. HMDS TMS Catalyst Well dispersed! TMS L TMS L TMS TMS TMS excess HMDS Ligand TMS OH L TMS L OH TMS L TMS TMS OH
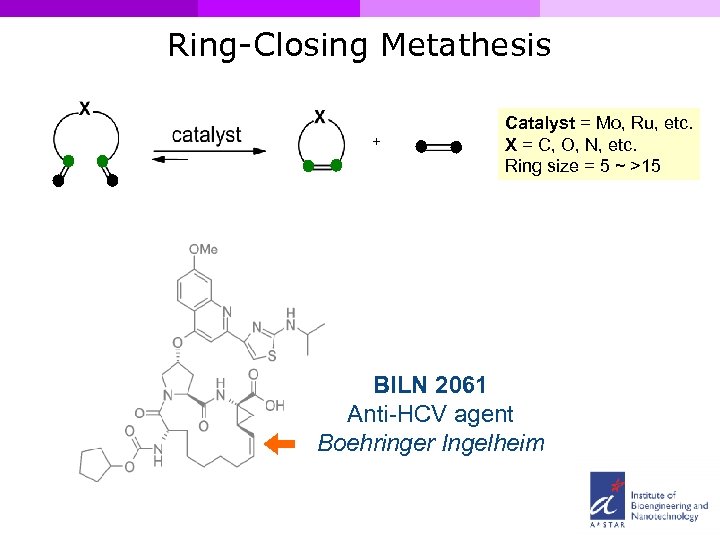 Ring-Closing Metathesis + Catalyst = Mo, Ru, etc. X = C, O, N, etc. Ring size = 5 ~ >15 BILN 2061 Anti-HCV agent Boehringer Ingelheim
Ring-Closing Metathesis + Catalyst = Mo, Ru, etc. X = C, O, N, etc. Ring size = 5 ~ >15 BILN 2061 Anti-HCV agent Boehringer Ingelheim
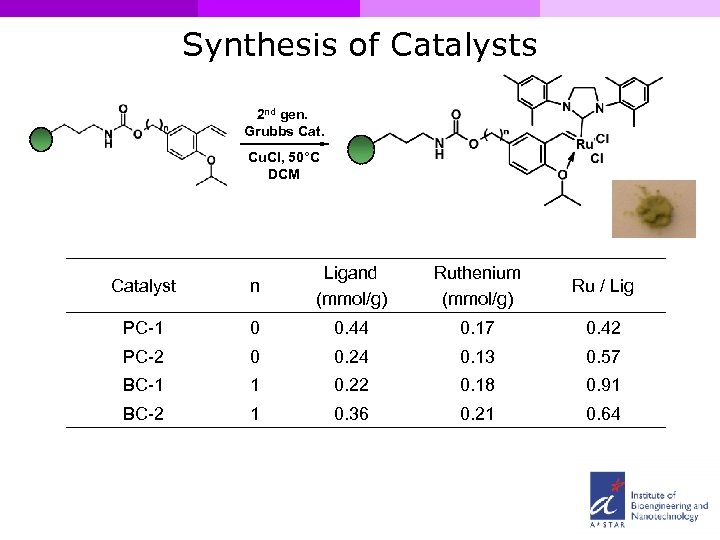 Synthesis of Catalysts 2 nd gen. Grubbs Cat. Cu. Cl, 50°C DCM Catalyst n Ligand (mmol/g) Ruthenium (mmol/g) Ru / Lig PC-1 0 0. 44 0. 17 0. 42 PC-2 0 0. 24 0. 13 0. 57 BC-1 1 0. 22 0. 18 0. 91 BC-2 1 0. 36 0. 21 0. 64
Synthesis of Catalysts 2 nd gen. Grubbs Cat. Cu. Cl, 50°C DCM Catalyst n Ligand (mmol/g) Ruthenium (mmol/g) Ru / Lig PC-1 0 0. 44 0. 17 0. 42 PC-2 0 0. 24 0. 13 0. 57 BC-1 1 0. 22 0. 18 0. 91 BC-2 1 0. 36 0. 21 0. 64
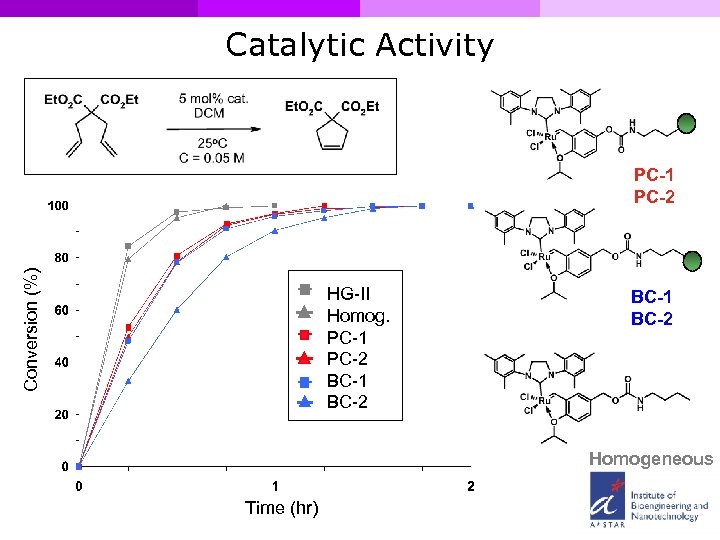 Catalytic Activity Conversion (%) PC-1 PC-2 HG-II Homog. PC-1 PC-2 BC-1 BC-2 Homogeneous Time (hr)
Catalytic Activity Conversion (%) PC-1 PC-2 HG-II Homog. PC-1 PC-2 BC-1 BC-2 Homogeneous Time (hr)
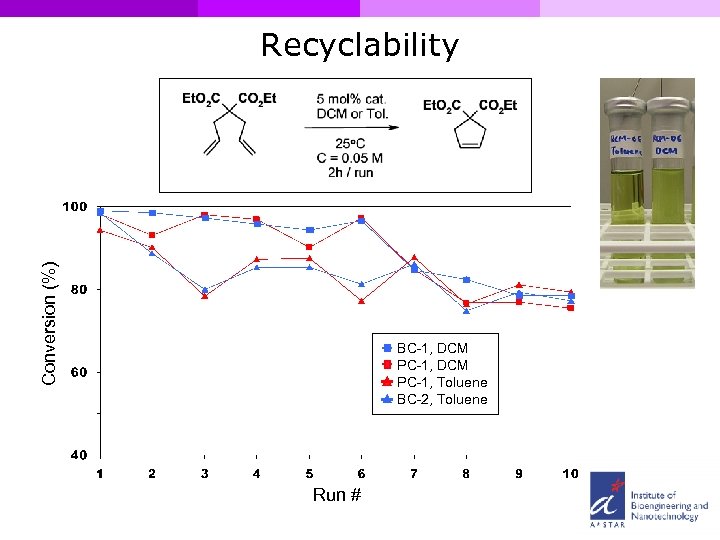 Conversion (%) Recyclability BC-1, DCM PC-1, Toluene BC-2, Toluene Run #
Conversion (%) Recyclability BC-1, DCM PC-1, Toluene BC-2, Toluene Run #
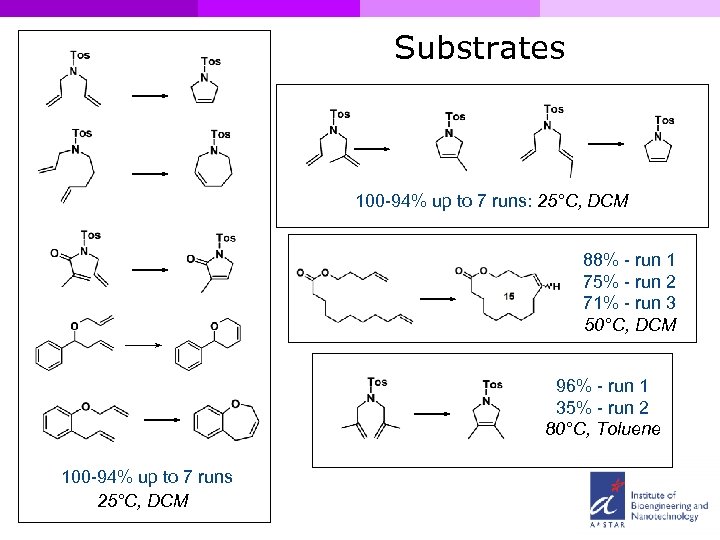 Substrates 100 -94% up to 7 runs: 25°C, DCM 88% - run 1 75% - run 2 71% - run 3 50°C, DCM 96% - run 1 35% - run 2 80°C, Toluene 100 -94% up to 7 runs 25°C, DCM
Substrates 100 -94% up to 7 runs: 25°C, DCM 88% - run 1 75% - run 2 71% - run 3 50°C, DCM 96% - run 1 35% - run 2 80°C, Toluene 100 -94% up to 7 runs 25°C, DCM
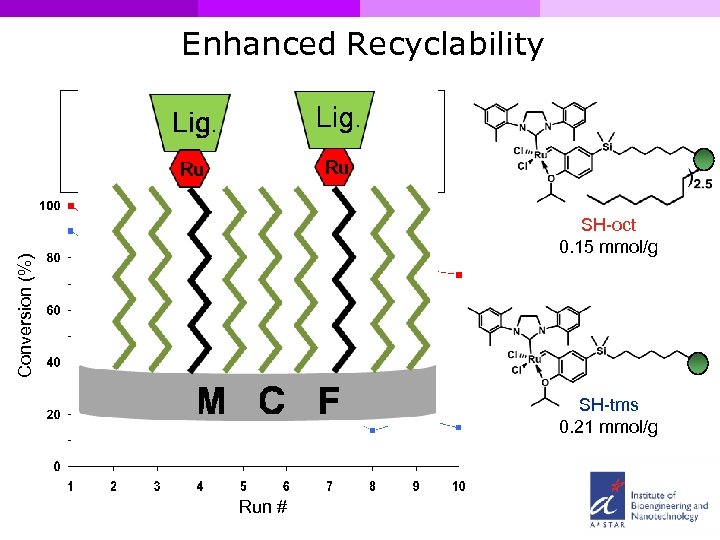 Enhanced Recyclability Conversion (%) SH-oct 0. 15 mmol/g SH-oct SH-tms 0. 21 mmol/g Run #
Enhanced Recyclability Conversion (%) SH-oct 0. 15 mmol/g SH-oct SH-tms 0. 21 mmol/g Run #
![Leaching Study Recyclability ICP-MS Conversion (%) Temp. pre-BC-1 25°C Ru residue [ppm] Run 5 Leaching Study Recyclability ICP-MS Conversion (%) Temp. pre-BC-1 25°C Ru residue [ppm] Run 5](https://present5.com/presentation/559d057ce893740900f3e9142f62b9fc/image-13.jpg) Leaching Study Recyclability ICP-MS Conversion (%) Temp. pre-BC-1 25°C Ru residue [ppm] Run 5 - 35 30 20 25°C 5 mol% 23 18 15 - 13 9 7 50°C Run # Run 3 50°C 25°C, 1 hr / run 50°C, 0. 5 hr / run 25°C, 5 mol% pre-BC-1, 2 hr / run 50°C, 5 mol% pre-BC-1, 1 hr / run Run 1 5 mol% 10 9 9
Leaching Study Recyclability ICP-MS Conversion (%) Temp. pre-BC-1 25°C Ru residue [ppm] Run 5 - 35 30 20 25°C 5 mol% 23 18 15 - 13 9 7 50°C Run # Run 3 50°C 25°C, 1 hr / run 50°C, 0. 5 hr / run 25°C, 5 mol% pre-BC-1, 2 hr / run 50°C, 5 mol% pre-BC-1, 1 hr / run Run 1 5 mol% 10 9 9
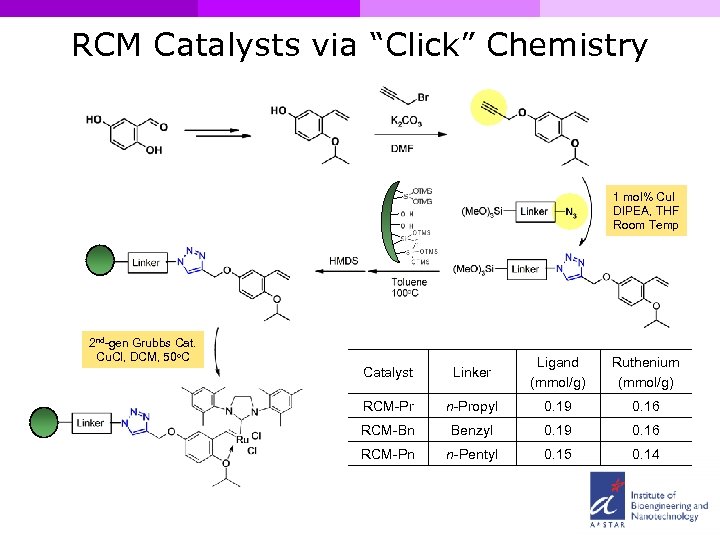 RCM Catalysts via “Click” Chemistry 1 mol% Cu. I DIPEA, THF Room Temp 2 nd-gen Grubbs Cat. Cu. Cl, DCM, 50 o. C Catalyst Linker Ligand (mmol/g) Ruthenium (mmol/g) RCM-Pr n-Propyl 0. 19 0. 16 RCM-Bn Benzyl 0. 19 0. 16 RCM-Pn n-Pentyl 0. 15 0. 14
RCM Catalysts via “Click” Chemistry 1 mol% Cu. I DIPEA, THF Room Temp 2 nd-gen Grubbs Cat. Cu. Cl, DCM, 50 o. C Catalyst Linker Ligand (mmol/g) Ruthenium (mmol/g) RCM-Pr n-Propyl 0. 19 0. 16 RCM-Bn Benzyl 0. 19 0. 16 RCM-Pn n-Pentyl 0. 15 0. 14
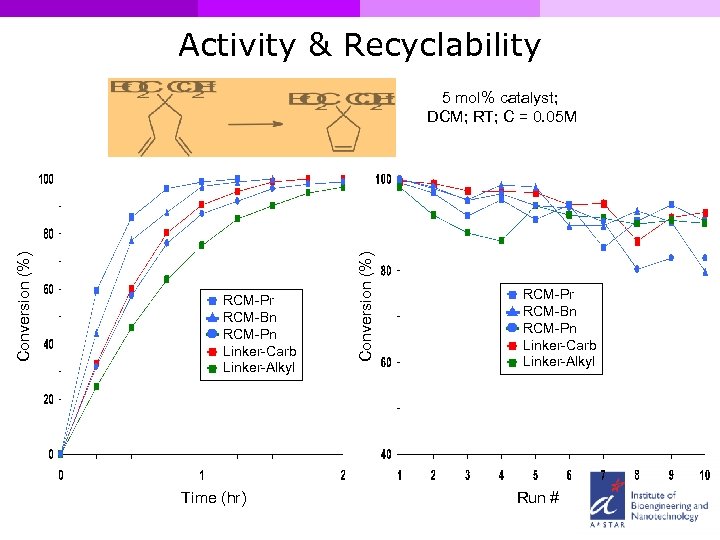 Activity & Recyclability RCM-Pr RCM-Bn RCM-Pn Linker-Carb Linker-Alkyl Time (hr) Conversion (%) 5 mol% catalyst; DCM; RT; C = 0. 05 M RCM-Pr RCM-Bn RCM-Pn Linker-Carb Linker-Alkyl Run #
Activity & Recyclability RCM-Pr RCM-Bn RCM-Pn Linker-Carb Linker-Alkyl Time (hr) Conversion (%) 5 mol% catalyst; DCM; RT; C = 0. 05 M RCM-Pr RCM-Bn RCM-Pn Linker-Carb Linker-Alkyl Run #
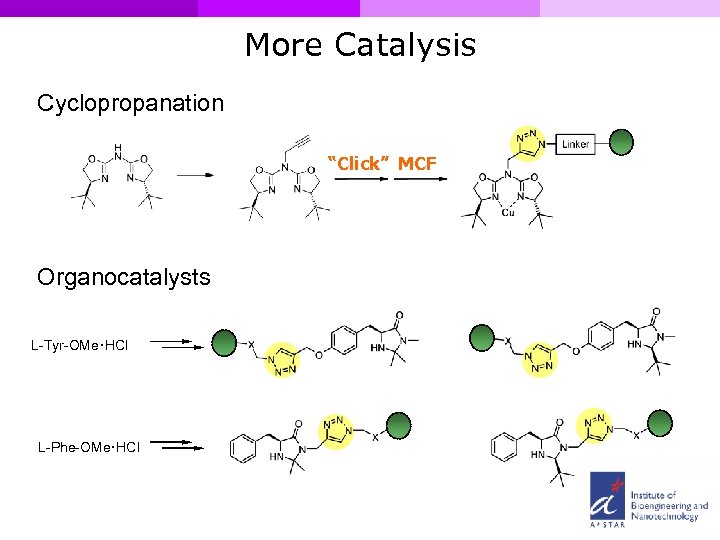 More Catalysis Cyclopropanation “Click” MCF Organocatalysts L-Tyr-OMe. HCl L-Phe-OMe. HCl
More Catalysis Cyclopropanation “Click” MCF Organocatalysts L-Tyr-OMe. HCl L-Phe-OMe. HCl
 Magnetic Micro-catalyst for RCM Synthesis Si. O 2 Fe 2 O 3 Easy recycle Images 10 nm 200 nm
Magnetic Micro-catalyst for RCM Synthesis Si. O 2 Fe 2 O 3 Easy recycle Images 10 nm 200 nm
 Acknowledgments Prof. Jackie Y. Ying and Ms. Noreena Abu. Bakar Dr. Su Seong Lee Ms. Siti Nurhanna Riduan IBN, BMRC and A*STAR, Singapore
Acknowledgments Prof. Jackie Y. Ying and Ms. Noreena Abu. Bakar Dr. Su Seong Lee Ms. Siti Nurhanna Riduan IBN, BMRC and A*STAR, Singapore


