выступление по английскому шилова.pptx
- Количество слайдов: 17

SHILOVA ANASTASIYA SYNTHESIS OF POLYLACTIDE BY VARIOUS CATALYSTS AND TEMPERATURES



ADVANTAGES OF BIODEGRADABLE POLYMERS - Treatment and education of their final product produced on standard equipment; - Provide good protection against water vapor and oxygen; - Rapid and complete degradability under the influence of natural environmental factors to form water and carbon dioxide; - Independence from petrochemical raw materials; - The production of renewable raw materials (corn, potatoes, wood or beet sugar).

DISADVANTAGES OF BIODEGRADABLE POLYMERS: - Limited opportunities for largescale production; - - High cost (as long as an average of 2 - 5 euros per kg).

THE METHODOLOGY OF THE EXPERIMENT • Temperature 150 ° C, different catalysts, argon, time - 18 hours • Temperature 100 -150 ° C, the catalyst K 2 CO 3, oxygen environment, time - 18 hours

CATALYSTS: • potassium carbonate • potassium sulfate • potassium chloride • potassium nitrate • sodium carbonate • lithium carbonate
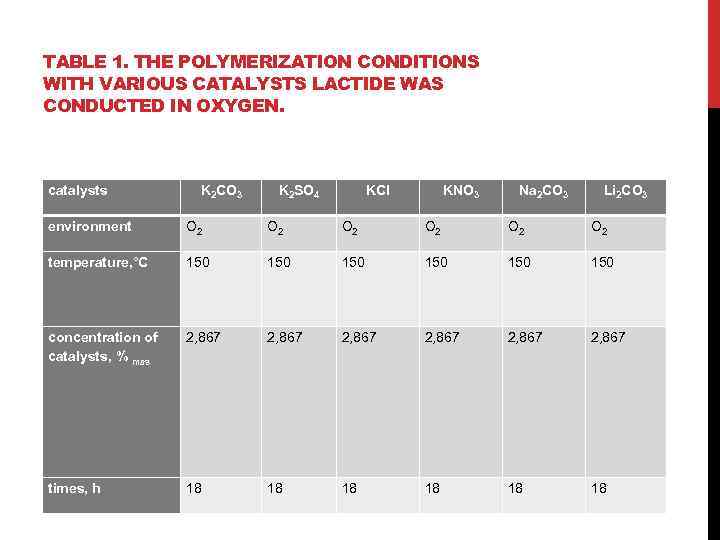
TABLE 1. THE POLYMERIZATION CONDITIONS WITH VARIOUS CATALYSTS LACTIDE WAS CONDUCTED IN OXYGEN. catalysts K 2 CO 3 K 2 SO 4 KCl KNO 3 Na 2 CO 3 Li 2 CO 3 environment О 2 О 2 О 2 temperature, °С 150 150 150 concentration of catalysts, % mas 2, 867 2, 867 times, h 18 18 18
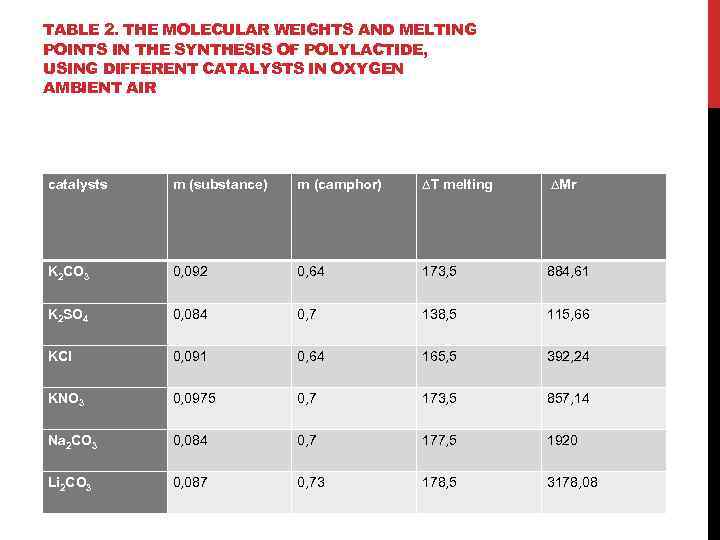
TABLE 2. THE MOLECULAR WEIGHTS AND MELTING POINTS IN THE SYNTHESIS OF POLYLACTIDE, USING DIFFERENT CATALYSTS IN OXYGEN AMBIENT AIR catalysts m (substance) m (camphor) ∆Т melting ∆Mr K 2 CO 3 0, 092 0, 64 173, 5 884, 61 K 2 SO 4 0, 084 0, 7 138, 5 115, 66 KCl 0, 091 0, 64 165, 5 392, 24 KNO 3 0, 0975 0, 7 173, 5 857, 14 Na 2 CO 3 0, 084 0, 7 177, 5 1920 Li 2 CO 3 0, 087 0, 73 178, 5 3178, 08
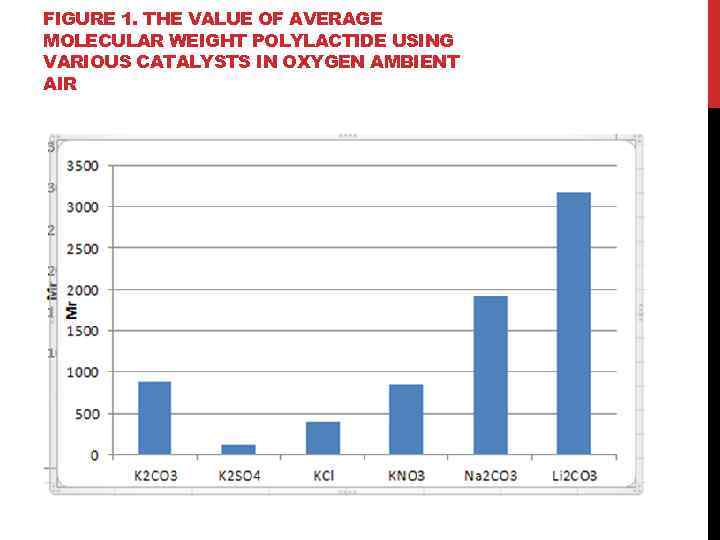
FIGURE 1. THE VALUE OF AVERAGE MOLECULAR WEIGHT POLYLACTIDE USING VARIOUS CATALYSTS IN OXYGEN AMBIENT AIR
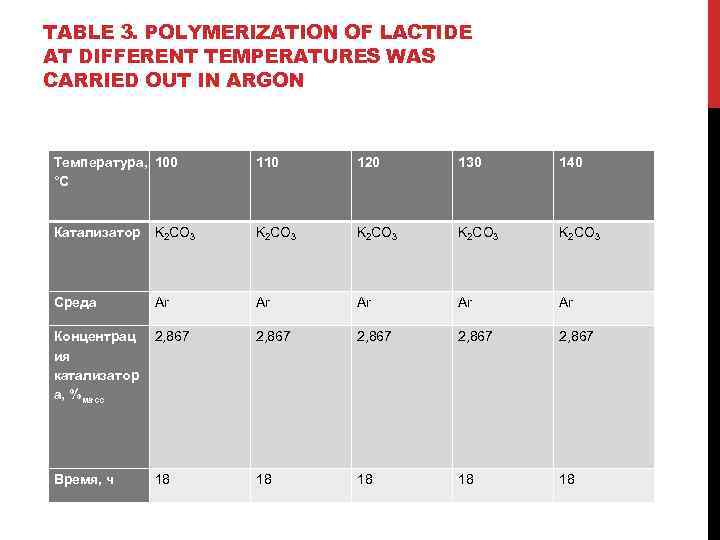
TABLE 3. POLYMERIZATION OF LACTIDE AT DIFFERENT TEMPERATURES WAS CARRIED OUT IN ARGON Температура, 100 °С 110 120 130 140 Катализатор K 2 CO 3 K 2 CO 3 Среда Ar Ar Ar Концентрац ия катализатор а, %масс 2, 867 2, 867 Время, ч 18 18 18
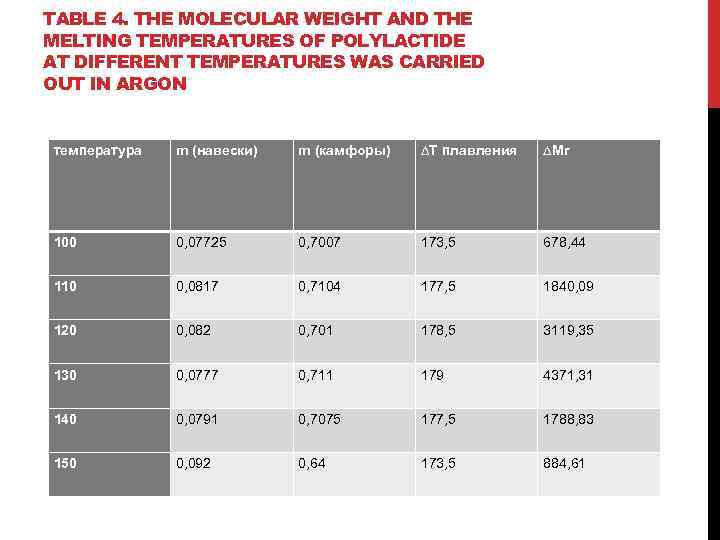
TABLE 4. THE MOLECULAR WEIGHT AND THE MELTING TEMPERATURES OF POLYLACTIDE AT DIFFERENT TEMPERATURES WAS CARRIED OUT IN ARGON температура m (навески) m (камфоры) ∆Т плавления ∆Mr 100 0, 07725 0, 7007 173, 5 678, 44 110 0, 0817 0, 7104 177, 5 1840, 09 120 0, 082 0, 701 178, 5 3119, 35 130 0, 0777 0, 711 179 4371, 31 140 0, 0791 0, 7075 177, 5 1788, 83 150 0, 092 0, 64 173, 5 884, 61
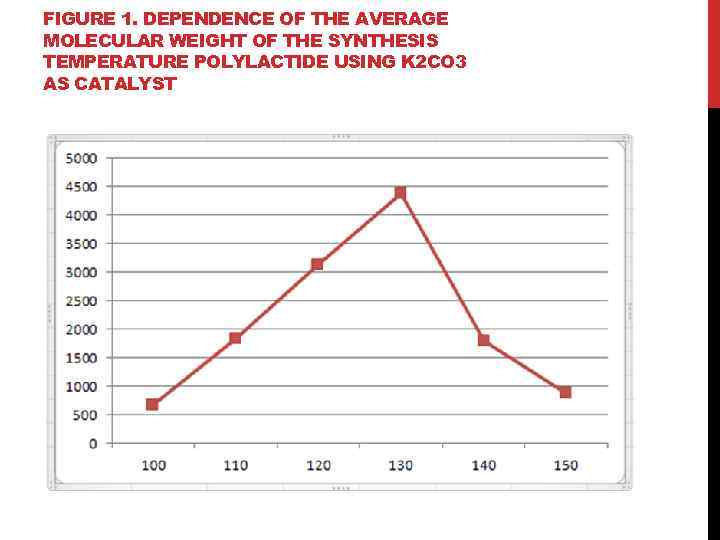
FIGURE 1. DEPENDENCE OF THE AVERAGE MOLECULAR WEIGHT OF THE SYNTHESIS TEMPERATURE POLYLACTIDE USING K 2 CO 3 AS CATALYST


FINDINGS: 1. It is shown that the synthesis of polylactide using K 2 CO 3 temperature of 130 ° C is optimal for increasing the degree of polymerization in a temperature range from 100 to 150 ° C. 2. It was found that the number of alkali metal carbonate, lithium carbonate, has the strongest influence on the synthesis of polylactide, at a temperature of 150°C 3. The effect of potassium on the synthesis of polylactide at 150˚C; shows that the highest molecular weight forms in the presence of potassium carbonate (Mr = 884, 61). 4. It has been established that the optimum conditions for the synthesis of polylactide, wherein the catalyst used is lithium carbonate: temperature 150° C and the presence of oxygen in the air environment, and the application of potassium carbonate corresponds to the optimum temperature of 130 ° C and the reaction takes place under an argon atmosphere. 5. It is shown that by using a potassium salt as a catalyst, highest molecular weight polylactide is formed by adding potassium carbonate.

THANK YOU FOR YOUR ATTENTION
выступление по английскому шилова.pptx