4ddebdeef0dd6648be01539a38183c71.ppt
- Количество слайдов: 4
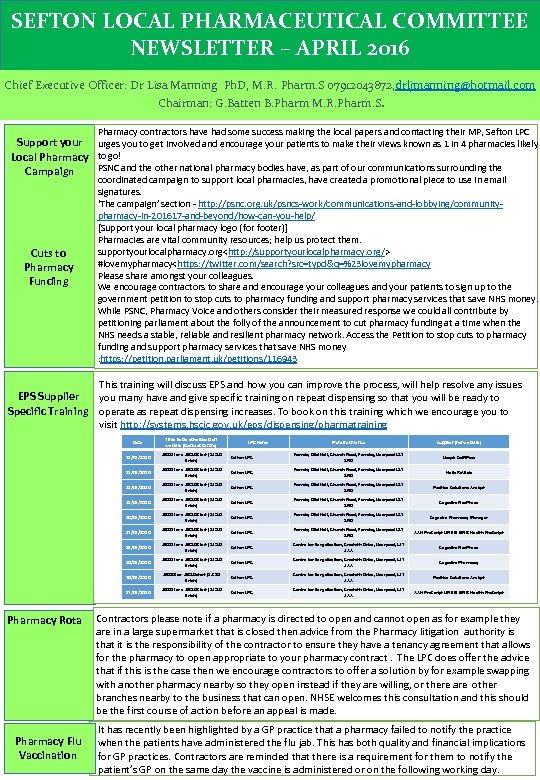 SEFTON LOCAL PHARMACEUTICAL COMMITTEE NEWSLETTER – APRIL 2016 Chief Executive Officer: Dr Lisa Manning Ph. D, M. R. Pharm. S 07912043872, drljmanning@hotmail. com Chairman: G. Batten B. Pharm M. R. Pharm. S. Pharmacy contractors have had some success making the local papers and contacting their MP, Sefton LPC Support your urges you to get involved and encourage your patients to make their views known as 1 in 4 pharmacies likely Local Pharmacy to go! PSNC and the other national pharmacy bodies have, as part of our communications surrounding the Campaign Cuts to Pharmacy Funding coordinated campaign to support local pharmacies, have created a promotional piece to use in email signatures. ‘The campaign’ section - http: //psnc. org. uk/psncs-work/communications-and-lobbying/communitypharmacy-in-201617 -and-beyond/how-can-you-help/ [Support your local pharmacy logo (for footer)] Pharmacies are vital community resources; help us protect them. supportyourlocalpharmacy. org
SEFTON LOCAL PHARMACEUTICAL COMMITTEE NEWSLETTER – APRIL 2016 Chief Executive Officer: Dr Lisa Manning Ph. D, M. R. Pharm. S 07912043872, drljmanning@hotmail. com Chairman: G. Batten B. Pharm M. R. Pharm. S. Pharmacy contractors have had some success making the local papers and contacting their MP, Sefton LPC Support your urges you to get involved and encourage your patients to make their views known as 1 in 4 pharmacies likely Local Pharmacy to go! PSNC and the other national pharmacy bodies have, as part of our communications surrounding the Campaign Cuts to Pharmacy Funding coordinated campaign to support local pharmacies, have created a promotional piece to use in email signatures. ‘The campaign’ section - http: //psnc. org. uk/psncs-work/communications-and-lobbying/communitypharmacy-in-201617 -and-beyond/how-can-you-help/ [Support your local pharmacy logo (for footer)] Pharmacies are vital community resources; help us protect them. supportyourlocalpharmacy. org
 Anti. Cholinergic Burden - Presentation Making health and social care information accessible GSK Inhaler Techniques and Devices Workshop Pharmacies with Anti-viral Stockholding Anti-Cholinergic Burden, reducing the risk within urinary incontinence- Date 20/4/16 The talk will commence at 07: 30 and will last for approximately 90 minutes including Q&A. A meal will be provided from 21: 00. Location: Left Bank Brasserie, 1 A The Beacon, Halsall Lane, Formby, Liverpool, L 37 3 NW The speaker will be Lynne Garforth and she will deliver a talk around the following topic; Anti-Cholinergic Burden, reducing the risk within urinary incontinence. To book a place contact Daniel Mc. Comish, Urology Account Representative, Astellas Pharma Ltd, 07770 552558 On 24. 06. 15 the new ‘accessible information standard’ was approved. All organisations that provide NHS or adult social care must follow the accessible information standard by law. Organisations must follow the standard in full by 31. 07. 16, and there are some things they must do before then. The aim of the accessible information standard is to make sure that people who have a disability, impairment or sensory loss get information that they can access and understand, and any communication support that they need. The standard tells organisations how they should make sure that patients and service users, and their carers and parents, can access and understand the information they are given. This includes making sure that people get information in different formats if they need it, for example in large print, braille, easy read or via email. The accessible information standard also tells organisations how they should make sure that people get any support with communication that they need, for example support from a British Sign Language (BSL) interpreter, deafblind manual interpreter or an advocate. What does the accessible information standard tell organisations to do? As part of the accessible information standard, organisations that provide NHS or adult social care must do five things. They must: 1. Ask people if they have any information or communication needs, and find out how to meet their needs. Pharmacy contractors should explore ways in which to meet the needs identified e. g. those people who have difficulty reading information in small print should be provided with the information in large print 2. Record those needs clearly and in a set way. For example - The contractor should document in the patients’ record (probably the patient medication record) that the patient has been asked if they have any information or communication needs and an entry note made that information or communication needs have been identified 3. Highlight or flag the person’s file or notes so it is clear that they have information or communication needs and how to meet those needs. 4. Share information about people’s information and communication needs with other providers of NHS and adult social care, when they have consent or permission to do so. Once it is established that the patient has an information or communication needs then the pharmacy contractor should ask the patient for their consent to share this information with other providers of NHS and adult social care services where required in the course of providing pharmaceutical services. An example of how this could operate in practice is where a pharmacy contractor undertakes a Medicines Use Review (MUR) on a patient taking multiple medicines, particularly those receiving medicines for long term conditions. During the course of the MUR consultation, the patient could mention to the pharmacist that they find it difficult to see the wording on the labelling on the medicines container. The pharmacist should then ask the patient for their consent for them to share this information with the patient’s GP. If they give consent then the pharmacist should ensure that the GP is aware and document what you have done. You may wish to suggest that to meet the communication need that large print documentation should be used for the patient. If the patient declines to give consent then the contractor should not share information about that patient’s information or communication needs. 5. Take steps to ensure that people receive information which they can access and understand, and receive communication support if they need it. There is more information about the accessible information standard, including the Specification and Implementation Guidance, on the NHS England website at www. england. nhs. uk/accessibleinfo Theguest speaker is John Bell. Date: Wednesday, 27 th April 2016 Time: 12: 00 to 16: 00 Venue: West Tower Hotel, Mill Lane, Aughton, Ormskirk L 39 7 HJTo Book a place contact: Tracey Halsall, Therapy Representative on 07876 391250 or email tracey. x. halsall@gsk. com The workshop will feature: a GSK portfolio update, Inhaler and Devices Workshop, a session on optimising outcomes in COPD and a question and answer session. Contractors are reminded that the 4 pharmacies listed carry a stock of anti-virals. This is to support patients out of normal trading days e. g during Bank Holidays or when their regular pharmacy may not be able to supply the medication in the timeframe required. Patients should be correctly signposted when appropriate. • Congleton Pharmacy, 29 -31 West Street, Congleton, CW 12 1 JP, 01260 277149 • Lloyds Pharmacy, Arrowe Park Hospital, Upton, CH 49 5 PE, 0151 6776449 • Stockton Heath Pharmacy, Warrington, WA 4 6 HJ, 01925 266753 • The Co-operative Pharmacy, Delamere St. Chester. , CH 1 4 DS, 01244 379268 SEFTON LOCAL PHARMACEUTICAL COMMITTEE http: //psnc. org. uk/sefton-lpc/
Anti. Cholinergic Burden - Presentation Making health and social care information accessible GSK Inhaler Techniques and Devices Workshop Pharmacies with Anti-viral Stockholding Anti-Cholinergic Burden, reducing the risk within urinary incontinence- Date 20/4/16 The talk will commence at 07: 30 and will last for approximately 90 minutes including Q&A. A meal will be provided from 21: 00. Location: Left Bank Brasserie, 1 A The Beacon, Halsall Lane, Formby, Liverpool, L 37 3 NW The speaker will be Lynne Garforth and she will deliver a talk around the following topic; Anti-Cholinergic Burden, reducing the risk within urinary incontinence. To book a place contact Daniel Mc. Comish, Urology Account Representative, Astellas Pharma Ltd, 07770 552558 On 24. 06. 15 the new ‘accessible information standard’ was approved. All organisations that provide NHS or adult social care must follow the accessible information standard by law. Organisations must follow the standard in full by 31. 07. 16, and there are some things they must do before then. The aim of the accessible information standard is to make sure that people who have a disability, impairment or sensory loss get information that they can access and understand, and any communication support that they need. The standard tells organisations how they should make sure that patients and service users, and their carers and parents, can access and understand the information they are given. This includes making sure that people get information in different formats if they need it, for example in large print, braille, easy read or via email. The accessible information standard also tells organisations how they should make sure that people get any support with communication that they need, for example support from a British Sign Language (BSL) interpreter, deafblind manual interpreter or an advocate. What does the accessible information standard tell organisations to do? As part of the accessible information standard, organisations that provide NHS or adult social care must do five things. They must: 1. Ask people if they have any information or communication needs, and find out how to meet their needs. Pharmacy contractors should explore ways in which to meet the needs identified e. g. those people who have difficulty reading information in small print should be provided with the information in large print 2. Record those needs clearly and in a set way. For example - The contractor should document in the patients’ record (probably the patient medication record) that the patient has been asked if they have any information or communication needs and an entry note made that information or communication needs have been identified 3. Highlight or flag the person’s file or notes so it is clear that they have information or communication needs and how to meet those needs. 4. Share information about people’s information and communication needs with other providers of NHS and adult social care, when they have consent or permission to do so. Once it is established that the patient has an information or communication needs then the pharmacy contractor should ask the patient for their consent to share this information with other providers of NHS and adult social care services where required in the course of providing pharmaceutical services. An example of how this could operate in practice is where a pharmacy contractor undertakes a Medicines Use Review (MUR) on a patient taking multiple medicines, particularly those receiving medicines for long term conditions. During the course of the MUR consultation, the patient could mention to the pharmacist that they find it difficult to see the wording on the labelling on the medicines container. The pharmacist should then ask the patient for their consent for them to share this information with the patient’s GP. If they give consent then the pharmacist should ensure that the GP is aware and document what you have done. You may wish to suggest that to meet the communication need that large print documentation should be used for the patient. If the patient declines to give consent then the contractor should not share information about that patient’s information or communication needs. 5. Take steps to ensure that people receive information which they can access and understand, and receive communication support if they need it. There is more information about the accessible information standard, including the Specification and Implementation Guidance, on the NHS England website at www. england. nhs. uk/accessibleinfo Theguest speaker is John Bell. Date: Wednesday, 27 th April 2016 Time: 12: 00 to 16: 00 Venue: West Tower Hotel, Mill Lane, Aughton, Ormskirk L 39 7 HJTo Book a place contact: Tracey Halsall, Therapy Representative on 07876 391250 or email tracey. x. halsall@gsk. com The workshop will feature: a GSK portfolio update, Inhaler and Devices Workshop, a session on optimising outcomes in COPD and a question and answer session. Contractors are reminded that the 4 pharmacies listed carry a stock of anti-virals. This is to support patients out of normal trading days e. g during Bank Holidays or when their regular pharmacy may not be able to supply the medication in the timeframe required. Patients should be correctly signposted when appropriate. • Congleton Pharmacy, 29 -31 West Street, Congleton, CW 12 1 JP, 01260 277149 • Lloyds Pharmacy, Arrowe Park Hospital, Upton, CH 49 5 PE, 0151 6776449 • Stockton Heath Pharmacy, Warrington, WA 4 6 HJ, 01925 266753 • The Co-operative Pharmacy, Delamere St. Chester. , CH 1 4 DS, 01244 379268 SEFTON LOCAL PHARMACEUTICAL COMMITTEE http: //psnc. org. uk/sefton-lpc/
 CLINICAL UPDATES OPIOID PRESCRIBING: As a reminder to prescribers and to re-iterate the Optimise. Rx intervention messages on Emis Web the following best practice guidance for non-palliative pain has been released: MORPHINE: • Opioid of first choice • Preferred brands of morphine are Zomorph capsules® and Morphgesic tablets® (not MST®) • Maximum 28 days supply OXYCODONE: • NOT opioid of ‘first’ choice - only an alternative to Morphine if: • intolerable side effects limit the dose escalation of morphine OR • in severe renal impairment (e. GFR < 30 ml/min) • 10 mg oral Morphine is approximately equivalent to 6. 6 mg Oxycodone • Preferred brands are Abtard® / Longtec® for the slow release tablets and Lynlor® / Shortec® for immediate release • Maximum 28 days supply FENTANYL: • Review licensed indications and dose conversions i. e. 25 microgram Fentanyl patch is approximately equivalent to 90 mg oral Morphine in 24 hours • Patients on 100 microgram Fentanyl or higher should be under a Pain Specialist BUPRENORPHINE: • Review licensed indications and dose conversions for Buprenorphine patches • Reserve the patches for those patients with stable chronic pain who have: • Difficulty swallowing • Unacceptable side effects with oral morphine or other opiates • Renal failure • Persistent nausea and vomiting • Gastrointestinal obstruction • Poor compliance with oral medication • Prescribe patches by brand – Transtec ®(4 day); Bu. Trans® or Bu. Tec® (7 day) • Maximum 28 days supply HIGH STRENGTH INSULIN PRODUCTS The Pan Mersey Area Prescribing Committee recommends prescribing of Humalog® Kwik. Pen® 200 units/m. L and Toujeo® Solo. Star® 300 units/m. L for specific patient groups, taking into consideration NICE guidance and key safety implications • Humalog® 200 units/m. L Kwik. Pen® (insulin lispro): No dose conversion is required when transferring a patient to a different strength of Humalog®. • Toujeo® 300 units/m. L Solo. Star® (insulin glargine): Toujeo® is not bioequivalent or directly interchangeable with Lantus® 100 units/m. L and dose adjustment is needed when switching from Lantus® or other basal insulins to Toujeo® and vice versa. Key safety considerations for health care professionals and patients: • Should always prescribed by brand to minimise risk of medication errors. • Always check formulation, strength and device prescribed are appropriate. Doses, if stated must be expressed in number of “units” to be administered and never abbreviated to “iu” or “u”. • Ensure patients know there is more than one strength of their insulin available. • Advise patients to check correct product has been supplied. • Ensure patients read and understand the product’s patient information leaflet, receive appropriate training on correct use and are either issued with an insulin passport, or their existing insulin passport is updated. • Close metabolic monitoring by the initiating specialist is essential during the switch and metabolic monitoring maintained until the patient is stabilised on the high strength insulin product. During this transition period prescribing and clinical responsibility must be maintained by the specialist. Patients will require a minimum of at least one clinical face-to-face follow-up appointment, prior to primary care taking over prescribing and clinical responsibility. • Encourage and support patients switching to a high strength insulin to monitor their blood glucose more closely to ensure good control is achieved. • For Humalog®, Lantus® and Toujeo® one dose step on the pen device is equivalent to one unit of insulin and also serves as a visual check of the dose to be administered. • Warn patients only to use insulin as they have been trained. Using it any other way may result in dangerous overdosing or underdosing. • NEVER draw up any high strength insulin solution from the prefilled pen device using an insulin syringe. Markings on the insulin syringe will not provide an accurate dose and there is a risk of serious overdosage and life threatening hypoglycaemia. SEFTON LOCAL PHARMACEUTICAL COMMITTEE http: //psnc. org. uk/sefton-lpc/
CLINICAL UPDATES OPIOID PRESCRIBING: As a reminder to prescribers and to re-iterate the Optimise. Rx intervention messages on Emis Web the following best practice guidance for non-palliative pain has been released: MORPHINE: • Opioid of first choice • Preferred brands of morphine are Zomorph capsules® and Morphgesic tablets® (not MST®) • Maximum 28 days supply OXYCODONE: • NOT opioid of ‘first’ choice - only an alternative to Morphine if: • intolerable side effects limit the dose escalation of morphine OR • in severe renal impairment (e. GFR < 30 ml/min) • 10 mg oral Morphine is approximately equivalent to 6. 6 mg Oxycodone • Preferred brands are Abtard® / Longtec® for the slow release tablets and Lynlor® / Shortec® for immediate release • Maximum 28 days supply FENTANYL: • Review licensed indications and dose conversions i. e. 25 microgram Fentanyl patch is approximately equivalent to 90 mg oral Morphine in 24 hours • Patients on 100 microgram Fentanyl or higher should be under a Pain Specialist BUPRENORPHINE: • Review licensed indications and dose conversions for Buprenorphine patches • Reserve the patches for those patients with stable chronic pain who have: • Difficulty swallowing • Unacceptable side effects with oral morphine or other opiates • Renal failure • Persistent nausea and vomiting • Gastrointestinal obstruction • Poor compliance with oral medication • Prescribe patches by brand – Transtec ®(4 day); Bu. Trans® or Bu. Tec® (7 day) • Maximum 28 days supply HIGH STRENGTH INSULIN PRODUCTS The Pan Mersey Area Prescribing Committee recommends prescribing of Humalog® Kwik. Pen® 200 units/m. L and Toujeo® Solo. Star® 300 units/m. L for specific patient groups, taking into consideration NICE guidance and key safety implications • Humalog® 200 units/m. L Kwik. Pen® (insulin lispro): No dose conversion is required when transferring a patient to a different strength of Humalog®. • Toujeo® 300 units/m. L Solo. Star® (insulin glargine): Toujeo® is not bioequivalent or directly interchangeable with Lantus® 100 units/m. L and dose adjustment is needed when switching from Lantus® or other basal insulins to Toujeo® and vice versa. Key safety considerations for health care professionals and patients: • Should always prescribed by brand to minimise risk of medication errors. • Always check formulation, strength and device prescribed are appropriate. Doses, if stated must be expressed in number of “units” to be administered and never abbreviated to “iu” or “u”. • Ensure patients know there is more than one strength of their insulin available. • Advise patients to check correct product has been supplied. • Ensure patients read and understand the product’s patient information leaflet, receive appropriate training on correct use and are either issued with an insulin passport, or their existing insulin passport is updated. • Close metabolic monitoring by the initiating specialist is essential during the switch and metabolic monitoring maintained until the patient is stabilised on the high strength insulin product. During this transition period prescribing and clinical responsibility must be maintained by the specialist. Patients will require a minimum of at least one clinical face-to-face follow-up appointment, prior to primary care taking over prescribing and clinical responsibility. • Encourage and support patients switching to a high strength insulin to monitor their blood glucose more closely to ensure good control is achieved. • For Humalog®, Lantus® and Toujeo® one dose step on the pen device is equivalent to one unit of insulin and also serves as a visual check of the dose to be administered. • Warn patients only to use insulin as they have been trained. Using it any other way may result in dangerous overdosing or underdosing. • NEVER draw up any high strength insulin solution from the prefilled pen device using an insulin syringe. Markings on the insulin syringe will not provide an accurate dose and there is a risk of serious overdosage and life threatening hypoglycaemia. SEFTON LOCAL PHARMACEUTICAL COMMITTEE http: //psnc. org. uk/sefton-lpc/
 INSULIN BIOSIMILAR PRODUCTS Patients should not be switched to a biosimilar if they are already established on insulin therapy and are well controlled. Biosimilar insulins are insulin therapy options when an analogue insulin is used in accordance with NICE guidance and the following criteria apply: • starting insulin treatment or • converting from NPH* isophane insulin to an insulin analogue for optimal control. Products are not interchangeable. Biosimilar and reference biological medicines that have the same international non-proprietary name (INN) are not presumed to be identical in the same way as generic non-biological medicines. Any insulin, including biosimilar, should only be initiated by a clinician who has undertaken relevant training and demonstrates ongoing competency in insulin initiation or switching. Key safety considerations for healthcare professionals and patients: • Should always prescribe by brand to minimise risk of medication errors. • Always check formulation, strength and device prescribed are appropriate. • Biosimilar products are NOT interchangeable and dose adjustment may be needed for some patients. • Doses, if stated must be expressed in number of “units” to be administered and never abbreviated to “IU” or “U”. • For Abasaglar▼ one dose step on the cartridge and pen device or prefilled pen is equivalent to one unit of insulin. The dose counter window of the pen device or prefilled pen displays the dose in units and also serves as a visual check of the dose to be administered. • Ensure patients know if there is more than one strength of their insulin available • Advise patients to check correct product has been supplied. • Ensure patients read and understand the product’s patient information leaflet, receive appropriate training on correct use and are either issued with an insulin passport, or their existing insulin passport is updated. • Close metabolic monitoring by the initiating clinician is essential during the switch and metabolic monitoring maintained until the patient is stabilised on the biosimilar insulin product. • Encourage and support patients switching to a biosimilar insulin to monitor their blood glucose more closely to ensure good control is achieved. • Warn patients only to use insulin as they have been trained. Using it any other way may result in dangerous overdosing or underdosing. • NEVER draw up any biosimilar strength insulin solution from the pen device or prefilled pen using an insulin syringe. Markings on the insulin syringe will not provide an accurate dose and there is a risk of serious overdosage and life threatening hypoglycaemia. • In accordance with the black triangle scheme ( ▼), report all suspected adverse reactions via the yellow card https: //yellowcard. mhra. gov. uk/
INSULIN BIOSIMILAR PRODUCTS Patients should not be switched to a biosimilar if they are already established on insulin therapy and are well controlled. Biosimilar insulins are insulin therapy options when an analogue insulin is used in accordance with NICE guidance and the following criteria apply: • starting insulin treatment or • converting from NPH* isophane insulin to an insulin analogue for optimal control. Products are not interchangeable. Biosimilar and reference biological medicines that have the same international non-proprietary name (INN) are not presumed to be identical in the same way as generic non-biological medicines. Any insulin, including biosimilar, should only be initiated by a clinician who has undertaken relevant training and demonstrates ongoing competency in insulin initiation or switching. Key safety considerations for healthcare professionals and patients: • Should always prescribe by brand to minimise risk of medication errors. • Always check formulation, strength and device prescribed are appropriate. • Biosimilar products are NOT interchangeable and dose adjustment may be needed for some patients. • Doses, if stated must be expressed in number of “units” to be administered and never abbreviated to “IU” or “U”. • For Abasaglar▼ one dose step on the cartridge and pen device or prefilled pen is equivalent to one unit of insulin. The dose counter window of the pen device or prefilled pen displays the dose in units and also serves as a visual check of the dose to be administered. • Ensure patients know if there is more than one strength of their insulin available • Advise patients to check correct product has been supplied. • Ensure patients read and understand the product’s patient information leaflet, receive appropriate training on correct use and are either issued with an insulin passport, or their existing insulin passport is updated. • Close metabolic monitoring by the initiating clinician is essential during the switch and metabolic monitoring maintained until the patient is stabilised on the biosimilar insulin product. • Encourage and support patients switching to a biosimilar insulin to monitor their blood glucose more closely to ensure good control is achieved. • Warn patients only to use insulin as they have been trained. Using it any other way may result in dangerous overdosing or underdosing. • NEVER draw up any biosimilar strength insulin solution from the pen device or prefilled pen using an insulin syringe. Markings on the insulin syringe will not provide an accurate dose and there is a risk of serious overdosage and life threatening hypoglycaemia. • In accordance with the black triangle scheme ( ▼), report all suspected adverse reactions via the yellow card https: //yellowcard. mhra. gov. uk/


