DOMMR Rheumatoid Arthritis.ppt
- Количество слайдов: 60
 Rheumatoid Arthritis DOM MR Week of 9/8/2008 Rozina Mithani
Rheumatoid Arthritis DOM MR Week of 9/8/2008 Rozina Mithani
 Joint Pain n most common symptom Pain (arthralgia) vs. Inflammation: n n (arthritis) heat, redness, pain, swelling, loss of function inflammatory arthritis (RA, SLE) vs. pain syndrome (fibromyalgia)
Joint Pain n most common symptom Pain (arthralgia) vs. Inflammation: n n (arthritis) heat, redness, pain, swelling, loss of function inflammatory arthritis (RA, SLE) vs. pain syndrome (fibromyalgia)
 n Number of Joints Affected n Inflammatory vs. Non-Inflammatory
n Number of Joints Affected n Inflammatory vs. Non-Inflammatory
 Number of Joints Affected n Monoarticular n n n Crystal-induced Infection Reactive Arthritis Hemarthrosis OA: joint effusions Autoimmune disease n Psoriasis, IBD, AS, Behçet's n Oligo/Polyarticular n n Monoarticular causes RA SLE Viral infection n n B 19 Acute Serum Sickness Untreated Crystal-induced Vasculidities
Number of Joints Affected n Monoarticular n n n Crystal-induced Infection Reactive Arthritis Hemarthrosis OA: joint effusions Autoimmune disease n Psoriasis, IBD, AS, Behçet's n Oligo/Polyarticular n n Monoarticular causes RA SLE Viral infection n n B 19 Acute Serum Sickness Untreated Crystal-induced Vasculidities
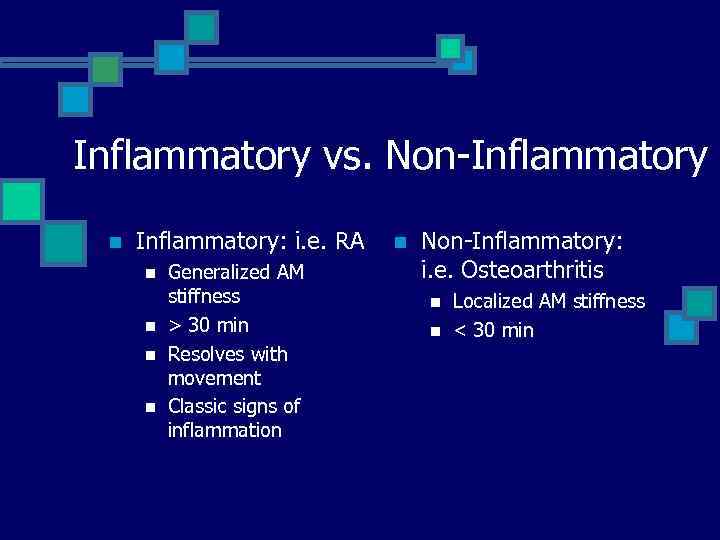 Inflammatory vs. Non-Inflammatory n Inflammatory: i. e. RA n n Generalized AM stiffness > 30 min Resolves with movement Classic signs of inflammation n Non-Inflammatory: i. e. Osteoarthritis n n Localized AM stiffness < 30 min
Inflammatory vs. Non-Inflammatory n Inflammatory: i. e. RA n n Generalized AM stiffness > 30 min Resolves with movement Classic signs of inflammation n Non-Inflammatory: i. e. Osteoarthritis n n Localized AM stiffness < 30 min
 Arthrocentesis n Confirm diagnoses Differentiate between inflammatory & noninflammatory Therapeutic/Adjunct to Antibiotics n Labs: n n n n cell count w/diff crystal analysis Gram stain & Culture WBC >2000/µL indicates inflammatory arthritis Arthroscopy n n n Evaluate ligamentous & cartilaginous integrity Biopsy Infectioun: aspirate thick or loculated fluid
Arthrocentesis n Confirm diagnoses Differentiate between inflammatory & noninflammatory Therapeutic/Adjunct to Antibiotics n Labs: n n n n cell count w/diff crystal analysis Gram stain & Culture WBC >2000/µL indicates inflammatory arthritis Arthroscopy n n n Evaluate ligamentous & cartilaginous integrity Biopsy Infectioun: aspirate thick or loculated fluid
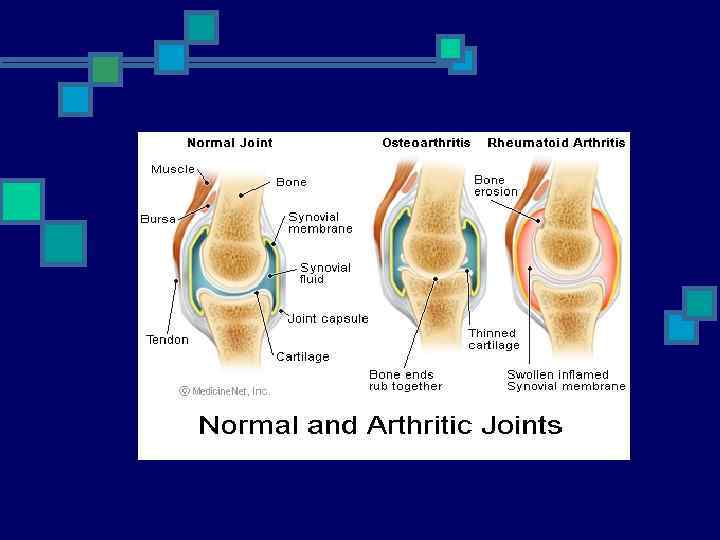
 Rheumatoid Arthritis
Rheumatoid Arthritis
 RA n n n Systemic inflammatory autoimmune disorder ~1% of population Onset: 52 years n n 40 -70 years of age <60 - 3 -5: 1 female predominance
RA n n n Systemic inflammatory autoimmune disorder ~1% of population Onset: 52 years n n 40 -70 years of age <60 - 3 -5: 1 female predominance
 Genetics n Increased incidence among Pima & Chippewa Native American tribes (5%) n n Genetic & Environmental HLA-DRB 1*0401 & HLA-DRB 1*0404 n n n Increased risk Increased joint damage Increased joint surgery
Genetics n Increased incidence among Pima & Chippewa Native American tribes (5%) n n Genetic & Environmental HLA-DRB 1*0401 & HLA-DRB 1*0404 n n n Increased risk Increased joint damage Increased joint surgery
 Pathophysiology
Pathophysiology
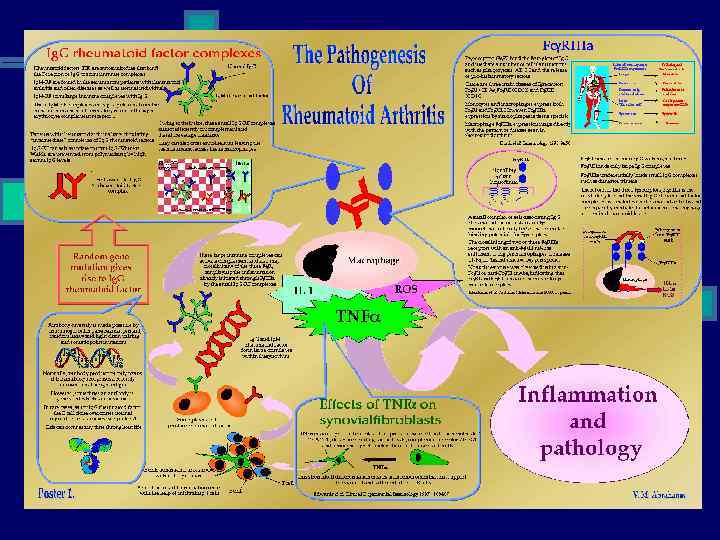
 Immunology n Macrophages: n n n Produce cytokines Cytokines (TNF-α) cause systemic features Release chemokines recruit PMNs into synovial fluid/membrane n TH-1 cells: n n n B cells: n n n TNF-α & IL-1: n n n Proliferation of T cells Activation of B cells Initiates proinflammatory/jointdamaging processes n Mediate disease processes Activate B cells Release cytokines Plasma cells that produce Ab Osteoclasts: n n Bone erosion Juxta-articular & Systemic osteoporosis
Immunology n Macrophages: n n n Produce cytokines Cytokines (TNF-α) cause systemic features Release chemokines recruit PMNs into synovial fluid/membrane n TH-1 cells: n n n B cells: n n n TNF-α & IL-1: n n n Proliferation of T cells Activation of B cells Initiates proinflammatory/jointdamaging processes n Mediate disease processes Activate B cells Release cytokines Plasma cells that produce Ab Osteoclasts: n n Bone erosion Juxta-articular & Systemic osteoporosis
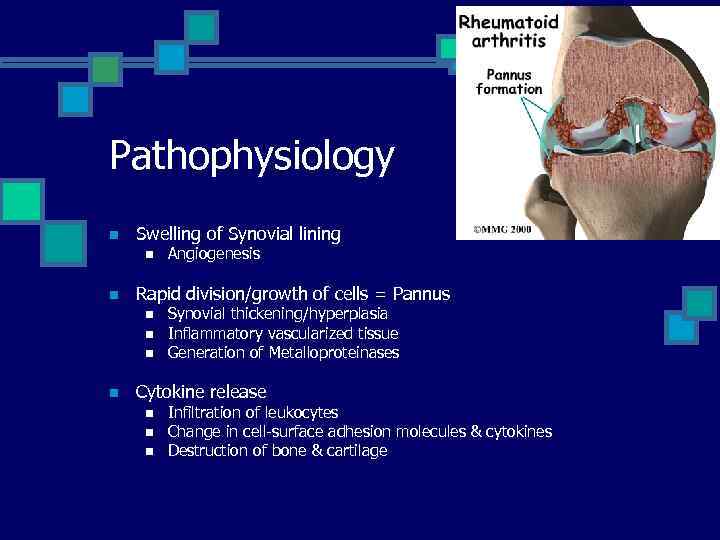 Pathophysiology n Swelling of Synovial lining n n Rapid division/growth of cells = Pannus n n Angiogenesis Synovial thickening/hyperplasia Inflammatory vascularized tissue Generation of Metalloproteinases Cytokine release n n n Infiltration of leukocytes Change in cell-surface adhesion molecules & cytokines Destruction of bone & cartilage
Pathophysiology n Swelling of Synovial lining n n Rapid division/growth of cells = Pannus n n Angiogenesis Synovial thickening/hyperplasia Inflammatory vascularized tissue Generation of Metalloproteinases Cytokine release n n n Infiltration of leukocytes Change in cell-surface adhesion molecules & cytokines Destruction of bone & cartilage
 Bottom Line n n n Proliferation Destruction of joints Disability
Bottom Line n n n Proliferation Destruction of joints Disability
 Disease Trigger n Subclinical vs. Viral trigger n n Lab manifestations up to 10 yrs before clinical RF & anti-CCP (anti–cyclic citrullinated peptide) Ab Increased CRP subclinical inflammatory disease ADLs: n n n > 50% of pts stop working w/i 5 -10 years of disease onset ~ 80% disabled to some degree > 20 years Life expectancy: decreased by 3 -18 years
Disease Trigger n Subclinical vs. Viral trigger n n Lab manifestations up to 10 yrs before clinical RF & anti-CCP (anti–cyclic citrullinated peptide) Ab Increased CRP subclinical inflammatory disease ADLs: n n n > 50% of pts stop working w/i 5 -10 years of disease onset ~ 80% disabled to some degree > 20 years Life expectancy: decreased by 3 -18 years
 Clinical Presentation n n Gradual onset Stiffness & Swelling Intermittent or Migratory involvement Extraarticular manifestations Myalgia, fatigue, low-grade fever, wt loss, depression
Clinical Presentation n n Gradual onset Stiffness & Swelling Intermittent or Migratory involvement Extraarticular manifestations Myalgia, fatigue, low-grade fever, wt loss, depression
 Stiffness & Swelling n n n Pain with pressure to joint Pain with movement of joint Swelling due to hypertrophy Effusion Heat Redness
Stiffness & Swelling n n n Pain with pressure to joint Pain with movement of joint Swelling due to hypertrophy Effusion Heat Redness
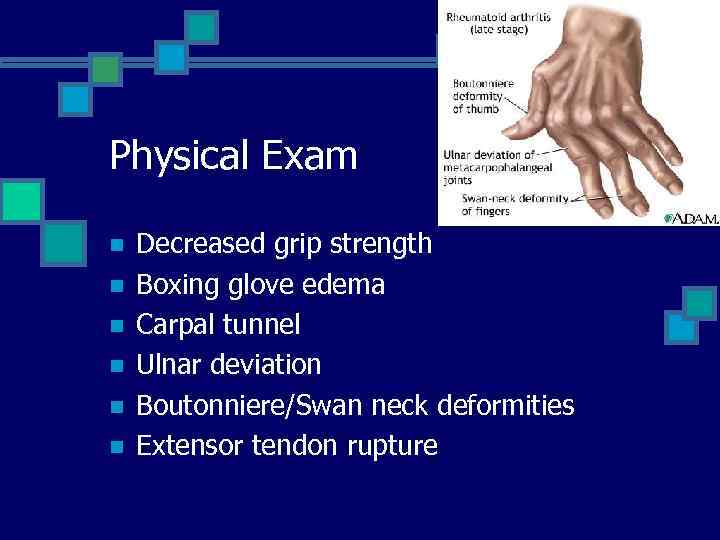 Physical Exam n n n Decreased grip strength Boxing glove edema Carpal tunnel Ulnar deviation Boutonniere/Swan neck deformities Extensor tendon rupture
Physical Exam n n n Decreased grip strength Boxing glove edema Carpal tunnel Ulnar deviation Boutonniere/Swan neck deformities Extensor tendon rupture
 Extraarticular Involvement n n Anemia Rheumatoid nodules Pleuropericarditis Neuropathy n n Episcleritis, Scleritis Splenomegaly Sjogren’s Vasculitis
Extraarticular Involvement n n Anemia Rheumatoid nodules Pleuropericarditis Neuropathy n n Episcleritis, Scleritis Splenomegaly Sjogren’s Vasculitis
 Differential n n n Seronegative polyarthritis n Erosive inflammatory OA Psoriatic arthritis n Reiter’s Crystal-induced n Enteropathic arthritis n Tophaceous gout n SLE n Pseudogout n Paraneoplastic syndrome
Differential n n n Seronegative polyarthritis n Erosive inflammatory OA Psoriatic arthritis n Reiter’s Crystal-induced n Enteropathic arthritis n Tophaceous gout n SLE n Pseudogout n Paraneoplastic syndrome
 Diagnostic Criteria
Diagnostic Criteria
 Diagnostic Criteria n n n Symmetric peripheral polyarthritis AM Stiffness >1 hour Rheumatoid nodules Laboratory features Radiographic bone erosions
Diagnostic Criteria n n n Symmetric peripheral polyarthritis AM Stiffness >1 hour Rheumatoid nodules Laboratory features Radiographic bone erosions
 Symmetric Peripheral Polyarthritis n 3 or more Joints for >6 weeks n Small Joints n n n Hands & Feet Peripheral to Proximal MCP and PIP Joints n SPARES DIP n MTP & Plantar subluxation n Leads to Deformity & Destruction of Joints n Erosion of cartilage and bone
Symmetric Peripheral Polyarthritis n 3 or more Joints for >6 weeks n Small Joints n n n Hands & Feet Peripheral to Proximal MCP and PIP Joints n SPARES DIP n MTP & Plantar subluxation n Leads to Deformity & Destruction of Joints n Erosion of cartilage and bone
 Stiffness n n AM or after Prolonged Inactivity Bilateral In/Around Joints > 1 hours n n n Reflects severe joint inflammation Better with movement Present >6 weeks
Stiffness n n AM or after Prolonged Inactivity Bilateral In/Around Joints > 1 hours n n n Reflects severe joint inflammation Better with movement Present >6 weeks
 Rheumatoid Nodules n Extensor surfaces n n elbows Very Specific Only occur in ~30% Late in Disease
Rheumatoid Nodules n Extensor surfaces n n elbows Very Specific Only occur in ~30% Late in Disease
 Laboratory Features n RF n n n Anti-Cyclic Citrulline Peptide (anti-CCP) n n 70 -80% of pts Overlap with HCV/Cryoglobulinemia Rare overlap with HCV Acute Phase reactants n ESR, CRP monitoring disease activity
Laboratory Features n RF n n n Anti-Cyclic Citrulline Peptide (anti-CCP) n n 70 -80% of pts Overlap with HCV/Cryoglobulinemia Rare overlap with HCV Acute Phase reactants n ESR, CRP monitoring disease activity
 Rheumatoid Factor n n n Ig. M against Ig. G Ig. M+ pts: more severe disease & poorer outcome Non-specific n SLE, Sjögren's, Sarcoidosis, Chronic infections
Rheumatoid Factor n n n Ig. M against Ig. G Ig. M+ pts: more severe disease & poorer outcome Non-specific n SLE, Sjögren's, Sarcoidosis, Chronic infections
 Anti-CCP n Ig. G against synovial membrane peptides damaged via inflammation n Value in Ig. M-RF negative n Sensitivity (65%) & Specificity (95%) n Predictive of Erosive Disease n n n Disease severity Radiologic progression Poor functional outcomes
Anti-CCP n Ig. G against synovial membrane peptides damaged via inflammation n Value in Ig. M-RF negative n Sensitivity (65%) & Specificity (95%) n Predictive of Erosive Disease n n n Disease severity Radiologic progression Poor functional outcomes
 Other Lab Abnormalities n n AOCD Thrombocytosis Leukocytosis ANA n n n 30 -40% Inflammatory synovial fluid Hypoalbuminemia
Other Lab Abnormalities n n AOCD Thrombocytosis Leukocytosis ANA n n n 30 -40% Inflammatory synovial fluid Hypoalbuminemia
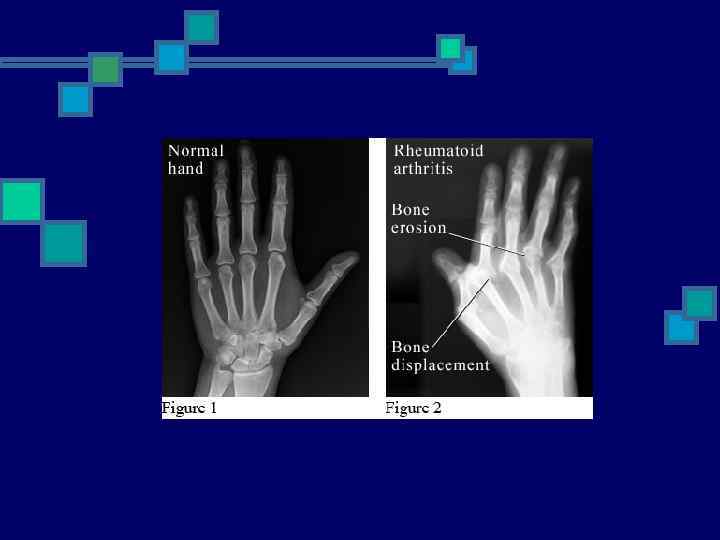
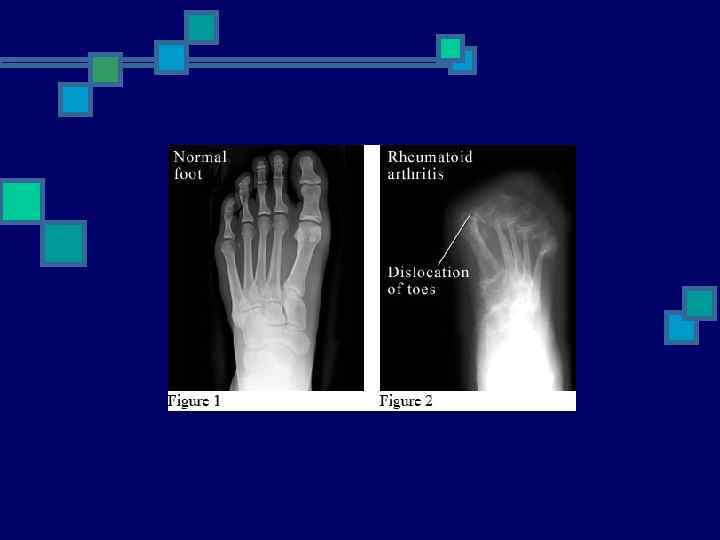
 Radiology n n Evaluate disease activity & joint damage Bony decalcification Baseline AP views Initiation of DMARDs
Radiology n n Evaluate disease activity & joint damage Bony decalcification Baseline AP views Initiation of DMARDs
 Radiological Studies n Plain Films n n n Bilateral hands & feet Only 25% of lesions Less expensive Through bone cortex around joint margins Color Doppler U/S & MRI n n Early signs of damage i. e. Erosions Bone Edema - even with normal findings on radiography
Radiological Studies n Plain Films n n n Bilateral hands & feet Only 25% of lesions Less expensive Through bone cortex around joint margins Color Doppler U/S & MRI n n Early signs of damage i. e. Erosions Bone Edema - even with normal findings on radiography
 Disease Severity
Disease Severity
 Mild Disease n n n Arthralgias >3 inflamed joints Mild functional limitation Minimally elevated ESR & CRP No erosions/cartilage loss No extraarticular disease i. e. anemia
Mild Disease n n n Arthralgias >3 inflamed joints Mild functional limitation Minimally elevated ESR & CRP No erosions/cartilage loss No extraarticular disease i. e. anemia
 Moderate Disease n n n 6 -20 Inflamed joints Moderate functional limitation Elevated ESR/CRP Radiographic evidence of inflammation No extraarticular disease
Moderate Disease n n n 6 -20 Inflamed joints Moderate functional limitation Elevated ESR/CRP Radiographic evidence of inflammation No extraarticular disease
 Severe Disease n n >20 persistently inflamed joints Rapid decline in functional capacity Radiographic evidence of rapid progession of bony erosions & loss of cartilage Extraarticular disease: n AOCD, Hypoalbuminemia
Severe Disease n n >20 persistently inflamed joints Rapid decline in functional capacity Radiographic evidence of rapid progession of bony erosions & loss of cartilage Extraarticular disease: n AOCD, Hypoalbuminemia
 Prognostic Features n n n n RF & Anti-CCP antibodies Early development of multiple inflamed joints and joint erosions Severe functional limitation Female HLA epitope presence Lower socioeconomic status & Less education Persistent joint inflammation for >12 weeks
Prognostic Features n n n n RF & Anti-CCP antibodies Early development of multiple inflamed joints and joint erosions Severe functional limitation Female HLA epitope presence Lower socioeconomic status & Less education Persistent joint inflammation for >12 weeks
 CV Disease n Leading cause of death ~50% n 2 x more likely to develop MI n n n chronic, inflammatory vascular burden premature atherosclerosis MTX: elevated homocysteine levels Control inflammatory process = Decreased atherosclerosis/morbidity n n n Lipid screening & treatment Control of obesity, Hyperhomocystinemia, DM, HTN ASA
CV Disease n Leading cause of death ~50% n 2 x more likely to develop MI n n n chronic, inflammatory vascular burden premature atherosclerosis MTX: elevated homocysteine levels Control inflammatory process = Decreased atherosclerosis/morbidity n n n Lipid screening & treatment Control of obesity, Hyperhomocystinemia, DM, HTN ASA
 Other diseases n 70% more likely to have a stroke n 70% higher risk for developing infection n 44 x more likely to develop NHL
Other diseases n 70% more likely to have a stroke n 70% higher risk for developing infection n 44 x more likely to develop NHL
 Staging n Early n n Established/Persistent n n <3 months 6 -12 months End-stage n n Significant joint destruction Functional disability
Staging n Early n n Established/Persistent n n <3 months 6 -12 months End-stage n n Significant joint destruction Functional disability
 Management n Early and aggressive disease control n Rheumatologist Referral n Early/Undiagnosed: NSAIDs, short course Corticosteroids n Late/Uncontrolled: DMARD therapy n n depends on the presence or absence of joint damage, functional limitation, presence of predictive factors for poorer prognosis Goals n n achieve NED & inflammation no treatment to resolve erosions once they occur
Management n Early and aggressive disease control n Rheumatologist Referral n Early/Undiagnosed: NSAIDs, short course Corticosteroids n Late/Uncontrolled: DMARD therapy n n depends on the presence or absence of joint damage, functional limitation, presence of predictive factors for poorer prognosis Goals n n achieve NED & inflammation no treatment to resolve erosions once they occur
 Treatment Strategies
Treatment Strategies
 Therapy n Non-Pharmacologic: n n n Referral to PT/OT Evaluate ADLs Assistive devices/splints Weight loss Smoking cessation n Pharmacologic: n n Anti-inflammatory Interrupt progression n n Development of erosions Joint space narrowing
Therapy n Non-Pharmacologic: n n n Referral to PT/OT Evaluate ADLs Assistive devices/splints Weight loss Smoking cessation n Pharmacologic: n n Anti-inflammatory Interrupt progression n n Development of erosions Joint space narrowing
 Pharmacologic Therapy n n n Analgesics NSAIDs Glucocorticoids SAARD/DMARD Anticytokine therapy
Pharmacologic Therapy n n n Analgesics NSAIDs Glucocorticoids SAARD/DMARD Anticytokine therapy
 Analgesics n Topical n Capsaicin n Diclofenac n Oral n Tylenol n Opiods
Analgesics n Topical n Capsaicin n Diclofenac n Oral n Tylenol n Opiods
 NSAIDs n n Analgesic, Antipyretic, Antiinflammatory n n n n Don’t alter disease progression n Ineffective in Erosive disease GI/Ulcers Hepatotoxicity Nephrotoxicity AIN Bleeding – antiplatelet Rash Aseptic meningitis
NSAIDs n n Analgesic, Antipyretic, Antiinflammatory n n n n Don’t alter disease progression n Ineffective in Erosive disease GI/Ulcers Hepatotoxicity Nephrotoxicity AIN Bleeding – antiplatelet Rash Aseptic meningitis
 Corticosteroids n Decrease cytokines n n n Slow Joint Inflammation n n Insomnia Emotional lability Fluid retention Weight gain HTN Hyperglycemia Osteoporosis n n n Bisphonates: >5 mg/d for >3 months Cataracts Avascular necrosis Myopathy Psychosis
Corticosteroids n Decrease cytokines n n n Slow Joint Inflammation n n Insomnia Emotional lability Fluid retention Weight gain HTN Hyperglycemia Osteoporosis n n n Bisphonates: >5 mg/d for >3 months Cataracts Avascular necrosis Myopathy Psychosis
 Disease modification n SAARD – slow acting antirheumatic drugs n DMARD – disease modifying antirheumatic drugs
Disease modification n SAARD – slow acting antirheumatic drugs n DMARD – disease modifying antirheumatic drugs
 Methotrexate Dihydrofolate reductase inhibitor n n n Well tolerated, Mono/Combo Onset: 6 -12 weeks Metabolism: Liver Clearance: Kidneys Monitoring: n n n Baseline: CXR, PFTs, HIV, HBV/HCV CBC, LFTs Q 4 -8 weeks Caution with CRI Nausea Mucosal ulcerations Fatigue & Flu-like symptoms BM Toxicity Hepatotoxicity n Treat with Folic acid, 1 mg/d n n
Methotrexate Dihydrofolate reductase inhibitor n n n Well tolerated, Mono/Combo Onset: 6 -12 weeks Metabolism: Liver Clearance: Kidneys Monitoring: n n n Baseline: CXR, PFTs, HIV, HBV/HCV CBC, LFTs Q 4 -8 weeks Caution with CRI Nausea Mucosal ulcerations Fatigue & Flu-like symptoms BM Toxicity Hepatotoxicity n Treat with Folic acid, 1 mg/d n n
 Leflunomide n n Inhibits dihydrooratate dehydrogenase n Dec. activated T-cells n Onset: rapid Efficacy: ≤ 6 weeks Monitoring: n CBC, LFTs n n Derm - rash, alopecia Diarrhea BM toxicity Hepatotoxicity
Leflunomide n n Inhibits dihydrooratate dehydrogenase n Dec. activated T-cells n Onset: rapid Efficacy: ≤ 6 weeks Monitoring: n CBC, LFTs n n Derm - rash, alopecia Diarrhea BM toxicity Hepatotoxicity
 Azathioprine n Corticosteroid-sparing n n n Monitoring: n n CBC Q 1 -2 months AST/ALT n n Infection BM Toxicity Hepatitis Malignancy
Azathioprine n Corticosteroid-sparing n n n Monitoring: n n CBC Q 1 -2 months AST/ALT n n Infection BM Toxicity Hepatitis Malignancy
 Cyclophosphamide n n Alkylating agent Monitoring: n n CBC, UA monthly Yearly UA +/- Cytology n n n n Alopecia Nausea Infection BM suppression pancytopenia Infertility – pretreat women with Leuprolide Renal: hemorrhagic cystitis, bladder malignancy – treat with acrolein Oral more toxic than IV
Cyclophosphamide n n Alkylating agent Monitoring: n n CBC, UA monthly Yearly UA +/- Cytology n n n n Alopecia Nausea Infection BM suppression pancytopenia Infertility – pretreat women with Leuprolide Renal: hemorrhagic cystitis, bladder malignancy – treat with acrolein Oral more toxic than IV
 Anticytokine therapy n Anti-TNF alpha agents n n Etanercept Infliximab Adalimumab IL-1 receptor antagonist (Anakinra)
Anticytokine therapy n Anti-TNF alpha agents n n Etanercept Infliximab Adalimumab IL-1 receptor antagonist (Anakinra)
 TNF-a Inhibitors n n n Anti-inflammatory Block TNF-α (proinflammatory cytokine) Etanercept, Adalimumab (SQ), Infliximab (IV) n n Very expensive: > $15, 000/patient Combo therapy with MTX n n Injection site reaction Infection Reactivated TB Infliximab n n n infusion reaction Pancytopenia Autoantibody/SLE-like Exacerbate CHF Malignancy – lymphoma
TNF-a Inhibitors n n n Anti-inflammatory Block TNF-α (proinflammatory cytokine) Etanercept, Adalimumab (SQ), Infliximab (IV) n n Very expensive: > $15, 000/patient Combo therapy with MTX n n Injection site reaction Infection Reactivated TB Infliximab n n n infusion reaction Pancytopenia Autoantibody/SLE-like Exacerbate CHF Malignancy – lymphoma
 n n n More aggressive approach Combo therapy Adjunctive therapy: TNF-α antagonist
n n n More aggressive approach Combo therapy Adjunctive therapy: TNF-α antagonist
 Disease Course n Long Remission n n Intermittent Disease n n 10% 15 -30% Progressive Disease
Disease Course n Long Remission n n Intermittent Disease n n 10% 15 -30% Progressive Disease
 Summary n Approach to Arthritis n n n Number of Joints Affected Inflammatory vs. Non-Inflammatory Rheumatoid Arthritis n n Diagnostic Criteria Pathophysiology Therapeutic Approach Disease Severity and Course
Summary n Approach to Arthritis n n n Number of Joints Affected Inflammatory vs. Non-Inflammatory Rheumatoid Arthritis n n Diagnostic Criteria Pathophysiology Therapeutic Approach Disease Severity and Course
 Questions?
Questions?


