58ff40f948cd7cc26fb05e0d09bac9a2.ppt
- Количество слайдов: 36

Renin-Angiotensin-Aldosterone System Blockade: A Foundation for Antihypertensive Therapy George R. Aronoff, M. D. , FACP Professor of Medicine and Pharmacology Chief, Division of Nephrology University of Louisville School of Medicine

Renin-Angiotensin-Aldosterone System Blockade: A Foundation for Antihypertensive Therapy Educational Objectives – Review physiology of renin-angiotensin system – Establish renin-angiotensin system as major regulator of blood pressure – Review strategies for blocking reninangiotensin system

The Kidneys and Circulation Hypothesis – “A blood-pressure raising substance is formed in the kidneys and passed into the blood” Methods – Homogenized fresh rabbit kidney in saline – Centrifuged the material – Injected the supernatant fluid into other rabbits Tigerstedt R, Bergman PG: Niere und kreislauf. Scand Arch Physiol 8: 223, 1898
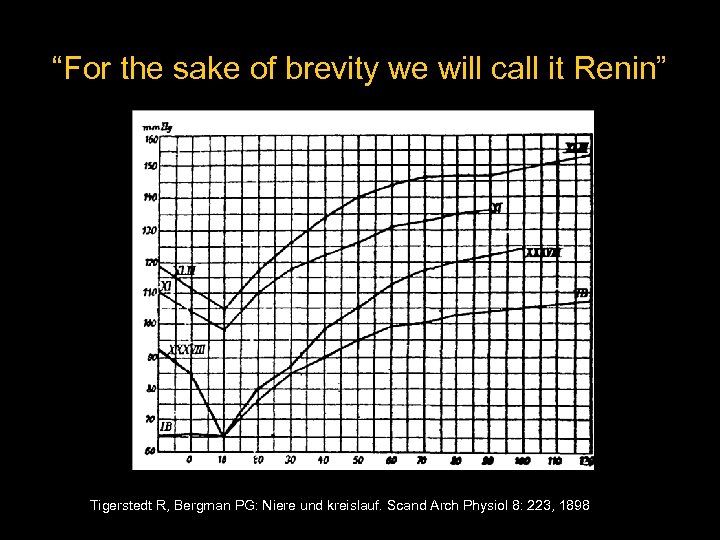
“For the sake of brevity we will call it Renin” Tigerstedt R, Bergman PG: Niere und kreislauf. Scand Arch Physiol 8: 223, 1898

Goldblatt Hypothesis – Decreased blood flow to the kidneys results in renal ischemia – Causes increased blood pressure Methods – Partial constriction of renal arteries of dogs using adjustable silver clamp
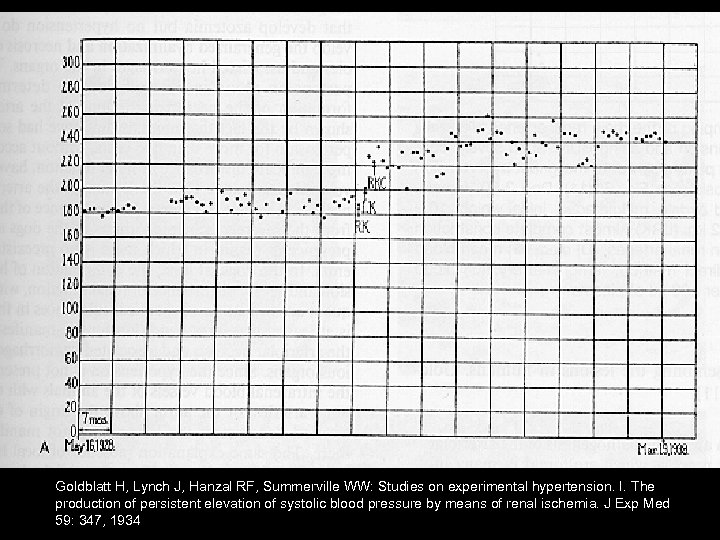
Goldblatt H, Lynch J, Hanzal RF, Summerville WW: Studies on experimental hypertension. I. The production of persistent elevation of systolic blood pressure by means of renal ischemia. J Exp Med 59: 347, 1934


Simultaneous Discoveries – Purified renin extract limited effect on blood pressure – Requires “renin substrate” – Results in formation of “hypertensin” or “angiotonin” Angiotensin Page, I. H. , and O. M. Helmer. 1939. Proc. Center Soc. Clin. Invest. 12: 17. Braun-Menendez, E. , and J. C. Fasciolo. 1939. Rev. Soc. Argent. Biol. 15: 420– 425.
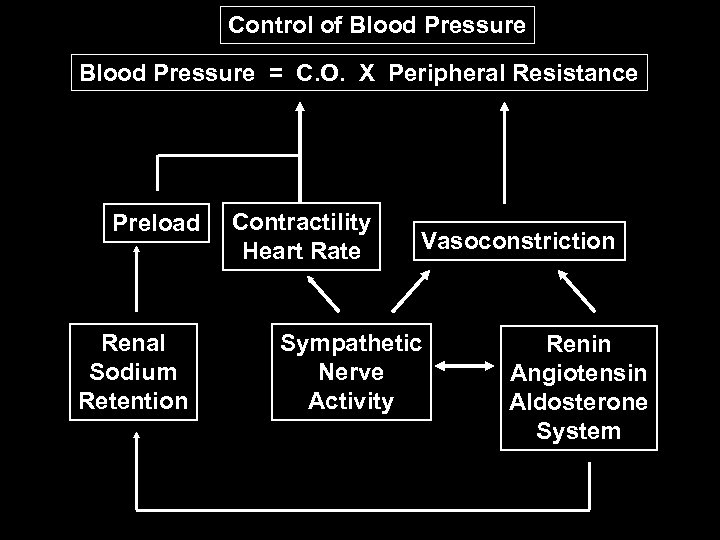
Control of Blood Pressure = C. O. X Peripheral Resistance Preload Renal Sodium Retention Contractility Heart Rate Vasoconstriction Sympathetic Nerve Activity Renin Angiotensin Aldosterone System

Angiotensin AT 1 Receptor Extracellular Intracellular Murphy TJ, Alexander RW, Griendling KK, Runge MSand Bernstein KE (1991) Isolation of a c. DNA encoding the vascular type-1 angiotensin II receptor. Nature (Lond) 16: 233– 236.
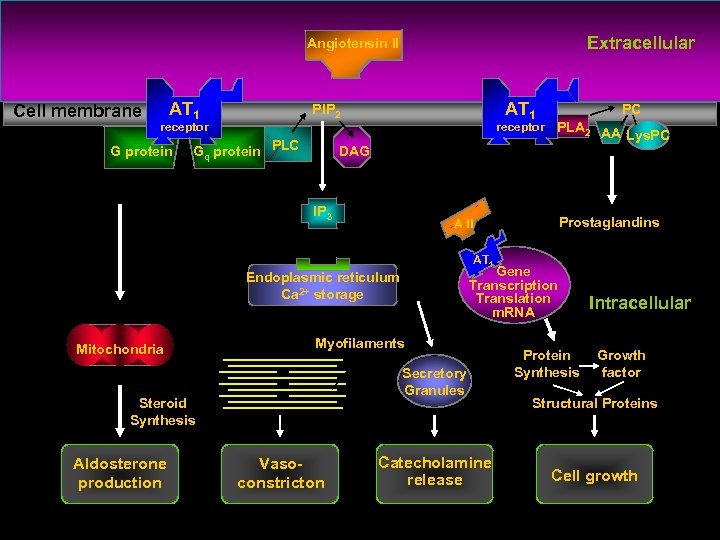
Extracellular Angiotensin II Cell membrane AT 1 receptor G protein AT 1 PIP 2 Gq protein PLC receptor Prostaglandins A II AT 1 Gene Transcription Translation m. RNA Ca 2+ Endoplasmic reticulum Ca 2+ storage Myofilaments Secretory Granules Steroid Synthesis Aldosterone production PLA 2 AA Lys. PC DAG IP 3 Mitochondria PC Vasoconstricton Catecholamine release Protein Synthesis Intracellular Growth factor Structural Proteins Cell growth
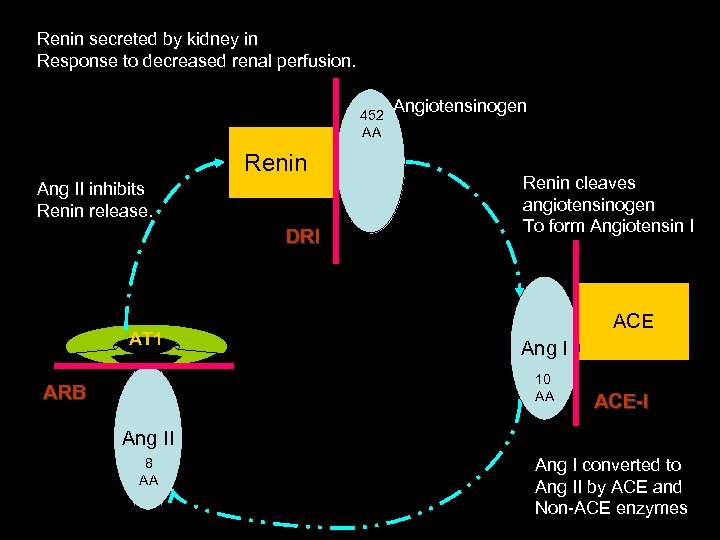
Renin secreted by kidney in Response to decreased renal perfusion. 452 AA Renin Ang II inhibits Renin release. DRI AT 1 Angiotensinogen Renin cleaves angiotensinogen To form Angiotensin I ACE Ang I 10 AA ARB ACE-I Ang II 8 AA Ang I converted to Ang II by ACE and Non-ACE enzymes
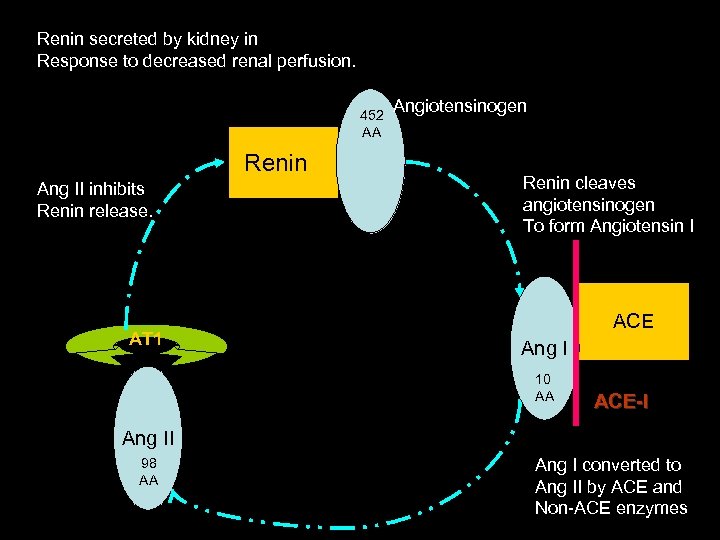
Renin secreted by kidney in Response to decreased renal perfusion. 452 AA Renin Ang II inhibits Renin release. AT 1 Angiotensinogen Renin cleaves angiotensinogen To form Angiotensin I ACE Ang I 10 AA ACE-I Ang II 98 AA Ang I converted to Ang II by ACE and Non-ACE enzymes
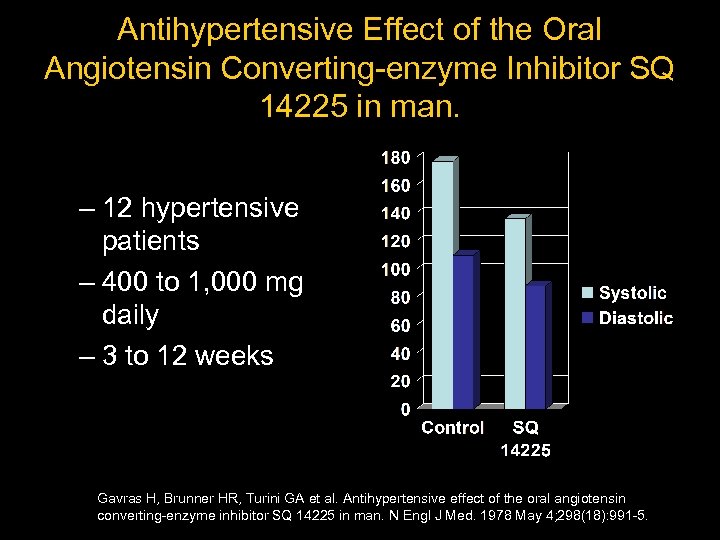
Antihypertensive Effect of the Oral Angiotensin Converting-enzyme Inhibitor SQ 14225 in man. – 12 hypertensive patients – 400 to 1, 000 mg daily – 3 to 12 weeks Gavras H, Brunner HR, Turini GA et al. Antihypertensive effect of the oral angiotensin converting-enzyme inhibitor SQ 14225 in man. N Engl J Med. 1978 May 4; 298(18): 991 -5.

The Effect of Angiotensin-Converting. Enzyme Inhibition on Diabetic Nephropathy 409 IDDM Patients Randomized – proteinuria more than 500 mg/day – creatinine less than 2. 5 mg/dl – 3 - 4 years follow-up – Captopril 25 mg tid or Placebo – Blood Pressure Equally Controlled – Doubling of Serum Creatinine Lewis, et al. NEJM 329: 1456 -62, 1993.
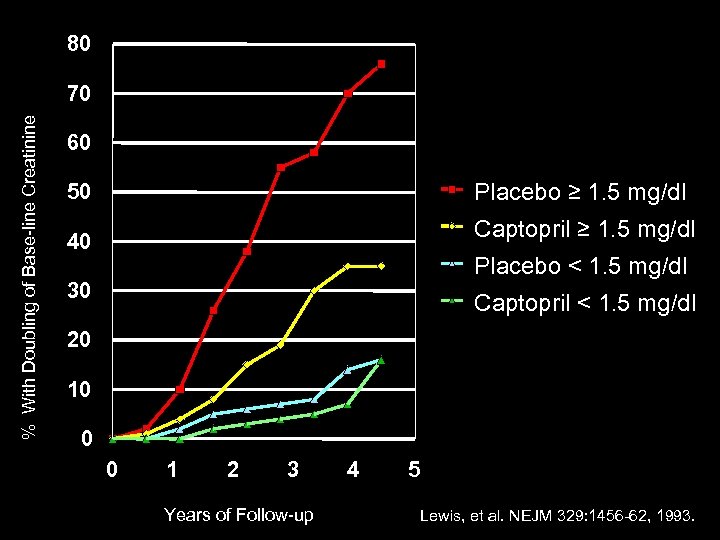
80 % With Doubling of Base-line Creatinine 70 60 Placebo ≥ 1. 5 mg/dl 50 Captopril ≥ 1. 5 mg/dl 40 Placebo < 1. 5 mg/dl 30 Captopril < 1. 5 mg/dl 20 10 0 0 1 2 3 Years of Follow-up 4 5 Lewis, et al. NEJM 329: 1456 -62, 1993.
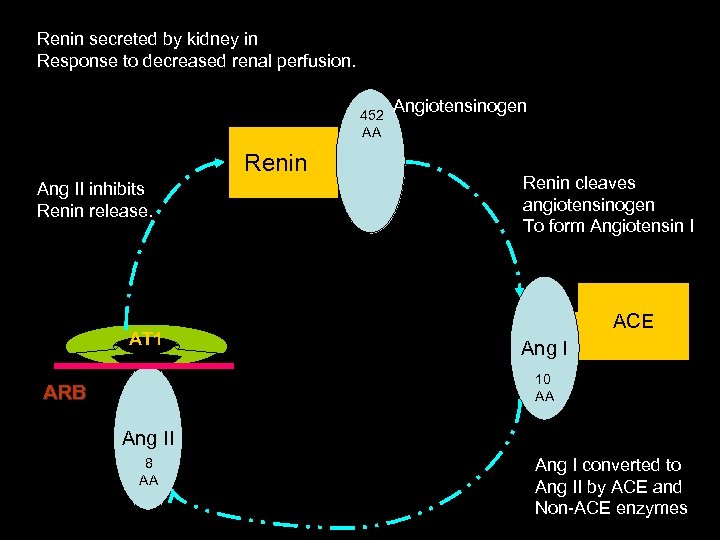
Renin secreted by kidney in Response to decreased renal perfusion. 452 AA Renin Ang II inhibits Renin release. AT 1 Angiotensinogen Renin cleaves angiotensinogen To form Angiotensin I ACE Ang I 10 AA ARB Ang II 8 AA Ang I converted to Ang II by ACE and Non-ACE enzymes
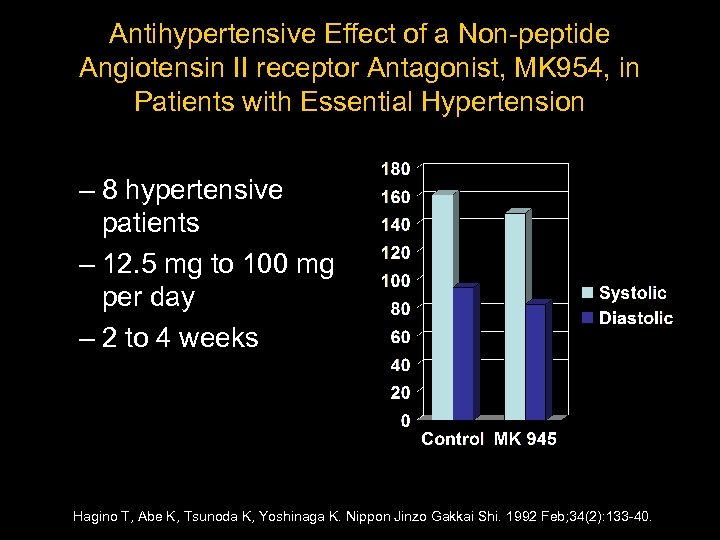
Antihypertensive Effect of a Non-peptide Angiotensin II receptor Antagonist, MK 954, in Patients with Essential Hypertension – 8 hypertensive patients – 12. 5 mg to 100 mg per day – 2 to 4 weeks Hagino T, Abe K, Tsunoda K, Yoshinaga K. Nippon Jinzo Gakkai Shi. 1992 Feb; 34(2): 133 -40.

RENAAL Reduction of Endpoints in Non-insulin Dependent Diabetes Mellitus With the Angiotensin II Antagonist Losartan – Multicenter – Randomized – Placebo controlled – 1, 513 type II diabetics Brenner et al. NEJM Sept. 2001
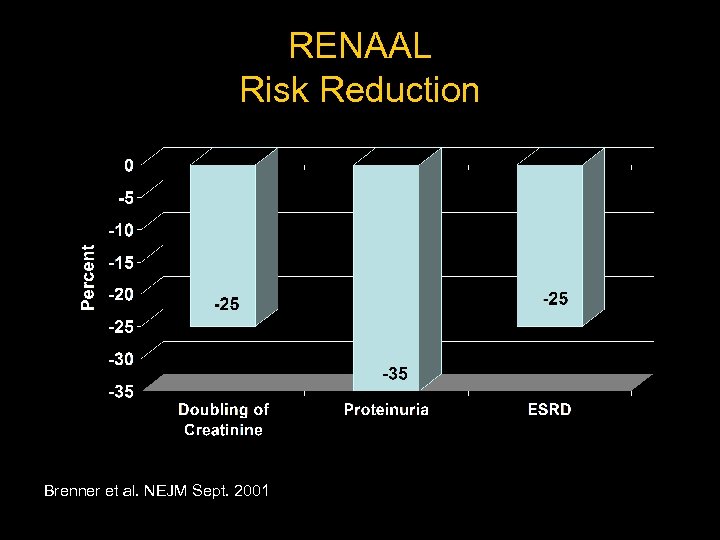
RENAAL Risk Reduction Brenner et al. NEJM Sept. 2001
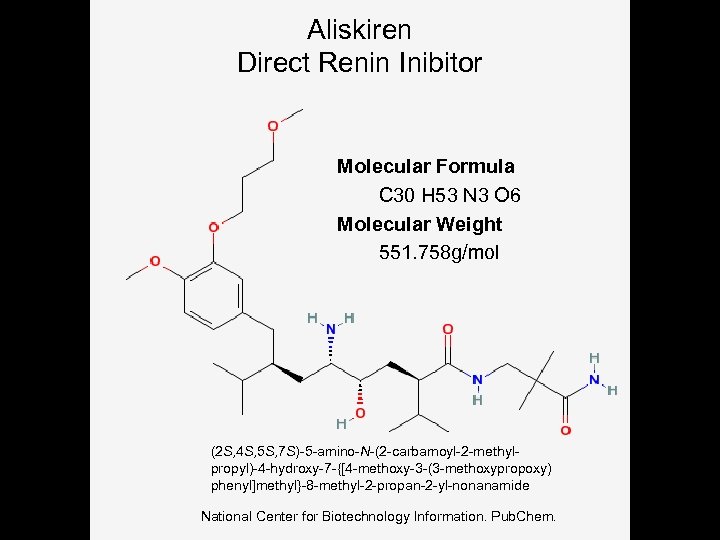
Aliskiren Direct Renin Inibitor Molecular Formula C 30 H 53 N 3 O 6 Molecular Weight 551. 758 g/mol (2 S, 4 S, 5 S, 7 S)-5 -amino-N-(2 -carbamoyl-2 -methylpropyl)-4 -hydroxy-7 -{[4 -methoxy-3 -(3 -methoxypropoxy) phenyl]methyl}-8 -methyl-2 -propan-2 -yl-nonanamide National Center for Biotechnology Information. Pub. Chem.

Alice Huxley The $880 Million Smile
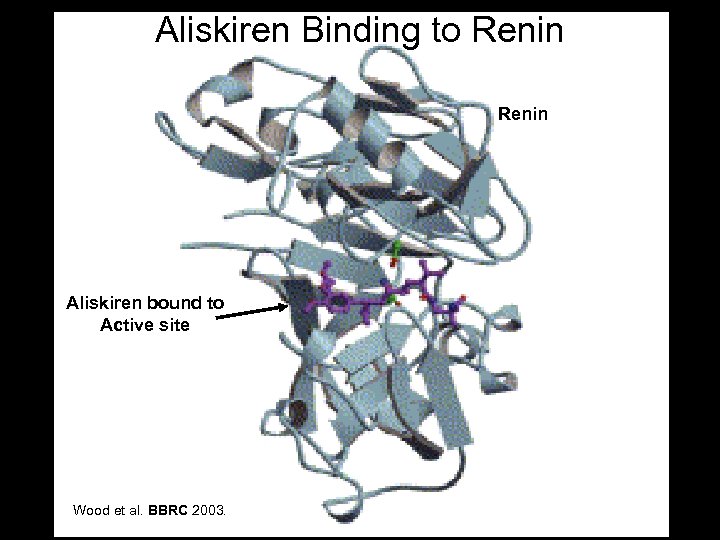
Aliskiren Binding to Renin Aliskiren bound to Active site Wood et al. BBRC 2003.

Aliskiren Direct renin inhibitor – 50 – 80 % decrease plasma renin activity Pharmacokinetics – Accumulation Half-life of ~ 24 hours – 7 – 8 days to achieve steady state levels – Elimination Half-life of ~ 48 hours – 25 % excreted by kidneys – Metabolized by CYP 450 -3 A 4 – Does not induce or suppress CYP 450 – No effect on QT interval

Aliskiren in Human Hypertension – 652 hypertensive patients – Randomized to: Placebo Aliskiren 150 mg, 300 mg, 600 mg Irbesartan 150 mg – Treated for 4 weeks Gradman, AH, Schmieder, RE, Lins, RL, et al. Circulation, Volume 111(8). March 1, 2005. 1012 -1018
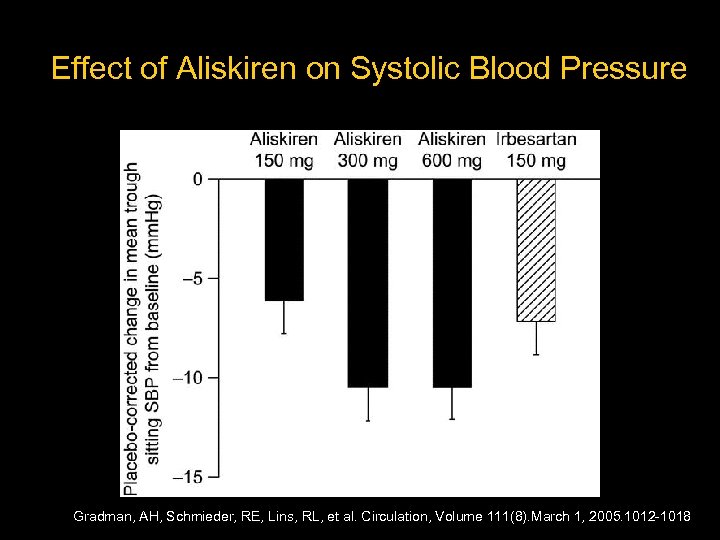
Effect of Aliskiren on Systolic Blood Pressure Gradman, AH, Schmieder, RE, Lins, RL, et al. Circulation, Volume 111(8). March 1, 2005. 1012 -1018
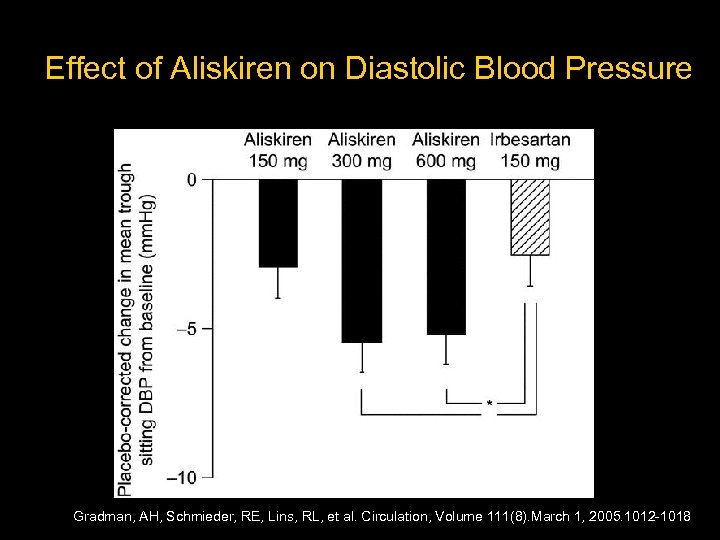
Effect of Aliskiren on Diastolic Blood Pressure Gradman, AH, Schmieder, RE, Lins, RL, et al. Circulation, Volume 111(8). March 1, 2005. 1012 -1018

Aliskiren Adverse Events 6, 460 patients Discontinuation – 2. 2% (3. 5% placebo) Cough – 1. 1% (0. 6% placebo) Angioedema – Less than 0. 06 % (2 cases reported with respiratory symptoms) Hyperkalemia – 0. 9% (0. 6% placebo) – 5. 5 % when in combination with ACE-I GI side effects – Diarrhea (2. 3 % with 1. 2% placebo) Do not use in pregnancy

Antiproteinuric Effects of Aliskiren – 15 Patients – Type 2 DM – Elevated urinary albumin/creatinine ratio – e. GFR ≥ 40 m. L/min – 4 week washout – Aliskiren 300 mg daily – 28 days Persson F, et al. Time course of the antiproteinuric and antihypertensive effects of direct renin inhibition in type 2 diabetes. Kidney Int. 2008 Mar 12
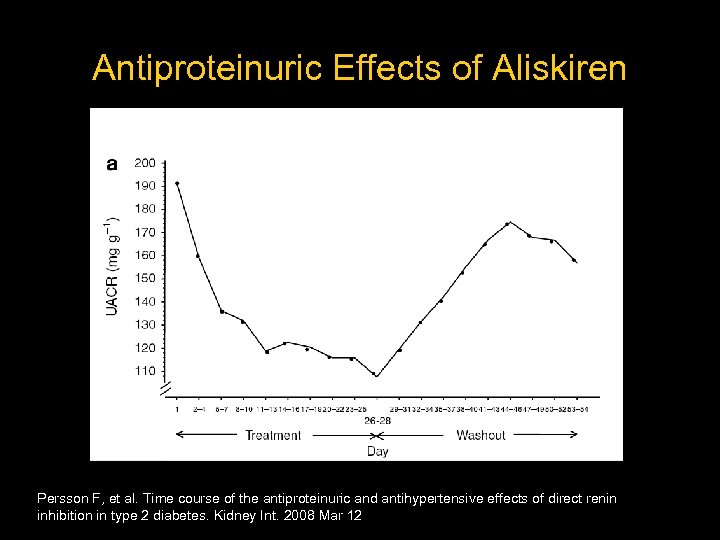
Antiproteinuric Effects of Aliskiren Persson F, et al. Time course of the antiproteinuric and antihypertensive effects of direct renin inhibition in type 2 diabetes. Kidney Int. 2008 Mar 12

Aliskiren Combined with Losartan in Type 2 Diabetes and Nephropathy – 599 patients – multinational, randomized, double-blind – UACR > 300 ≤ 3, 500 – All received 100 mg losartan – Aliskiren 150 mg for 3 mo, then 300 mg for 3 mo or placebo – Reduction in urine albumin/creatinine at 6 months Parving H-H, Persson F, Lewis, JB; for the AVOID Study Investigators. NEJM 358(23), 5 June 2008, p 2433– 2446.
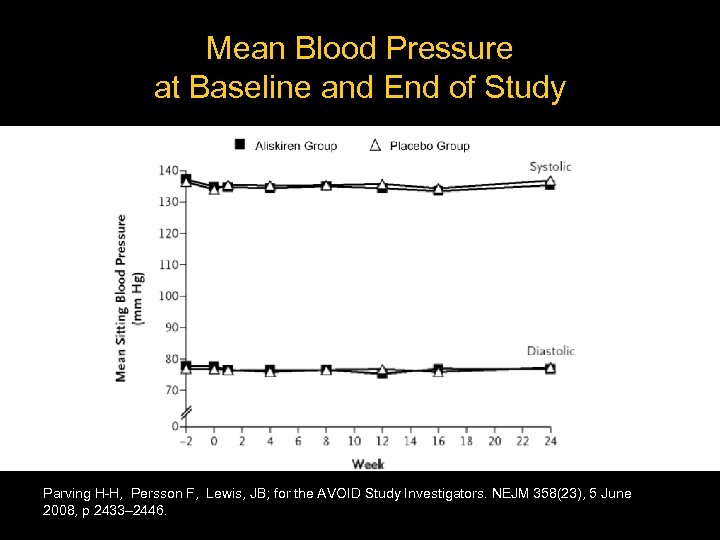
Mean Blood Pressure at Baseline and End of Study Parving H-H, Persson F, Lewis, JB; for the AVOID Study Investigators. NEJM 358(23), 5 June 2008, p 2433– 2446.
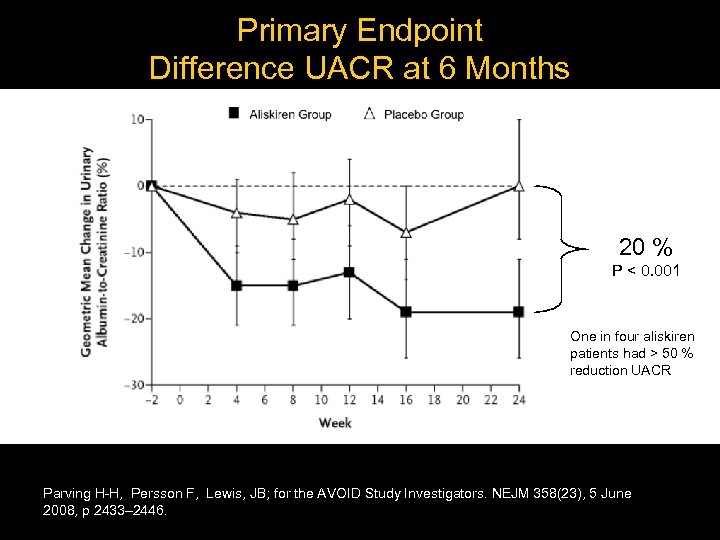
Primary Endpoint Difference UACR at 6 Months 20 % P < 0. 001 One in four aliskiren patients had > 50 % reduction UACR Parving H-H, Persson F, Lewis, JB; for the AVOID Study Investigators. NEJM 358(23), 5 June 2008, p 2433– 2446.

Effect of Aliskiren in Patients with Heart Failure 302 Patients – Stable heart failure – Hypertension – BNP > 100 pg/ml Randomized – Placebo or 150 mg aliskiren for 12 weeks – Standard therapy for HF (ACEI or ARB) Mc. Murray JJV, et al. Circ Heart Fail 2008; 1: 17 -24
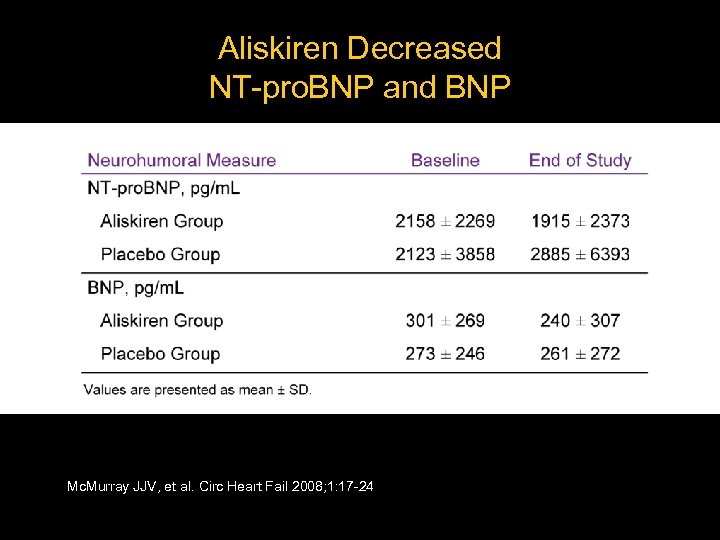
Aliskiren Decreased NT-pro. BNP and BNP Mc. Murray JJV, et al. Circ Heart Fail 2008; 1: 17 -24

Renin-Angiotensin-Aldosterone System Blockade: A Foundation for Antihypertensive Therapy Conclusions – Over a century of research has led elucidation of an important mechanism in the control of blood pressure in humans – RAAS is a target for the control of human hypertension – Downregulation of RAAS decreases the rate of sclerosing kidney disease
58ff40f948cd7cc26fb05e0d09bac9a2.ppt