fe2fd358d6054c94b86fcb061bf92365.ppt
- Количество слайдов: 29
 Regulatory tennis games across the Atlantic Comparative Risk Regulation workshop Berkeley, 12 -14 December 2012 Frederic Bouder
Regulatory tennis games across the Atlantic Comparative Risk Regulation workshop Berkeley, 12 -14 December 2012 Frederic Bouder
 The purpose of this presentation 1. Introduce the concept of transatlantic “regulatory tennis games” 2. Present 2 case studies from the pharmaceutical area 3. How prevalent? First observed in the pharma sector through comparative research. Can we see the same phenomenon in other sectors?
The purpose of this presentation 1. Introduce the concept of transatlantic “regulatory tennis games” 2. Present 2 case studies from the pharmaceutical area 3. How prevalent? First observed in the pharma sector through comparative research. Can we see the same phenomenon in other sectors?
 Transatlantic debate on risk and precaution Europe on the whole more precautionary than the United States (Kempton and Craig, 1993) Catch-up: European 30 behind, more time needed (Majone, 1996, 2001 l Löfstedt 2004). Flip-flop : Americans being excessively precautionary to now the Europeans being so (Löfstedt and Vogel, 2001; Vogel 2012) Reality of Precaution: both trade blocks precautionary at some stages and less precautionary at other times (Wiener and Rogers 2002; Wiener et al. 2011)
Transatlantic debate on risk and precaution Europe on the whole more precautionary than the United States (Kempton and Craig, 1993) Catch-up: European 30 behind, more time needed (Majone, 1996, 2001 l Löfstedt 2004). Flip-flop : Americans being excessively precautionary to now the Europeans being so (Löfstedt and Vogel, 2001; Vogel 2012) Reality of Precaution: both trade blocks precautionary at some stages and less precautionary at other times (Wiener and Rogers 2002; Wiener et al. 2011)
 The problem with the nondivergent These various hypotheses also point to the limits of binary oppositions in specific situations, especially in globalised areas of regulation – e. g. pharmaceuticals Often put under the unspecified label of “convergence”.
The problem with the nondivergent These various hypotheses also point to the limits of binary oppositions in specific situations, especially in globalised areas of regulation – e. g. pharmaceuticals Often put under the unspecified label of “convergence”.
 Transatlantic tennis Games : what is it? More complex than convergence, tennis games are also about regulatory completion and emulation in a race to set the global standard of protection. The regulation may start as excessively precautionary in one country, only to be taken over by another country for one reason or another, leading to a muddled picture.
Transatlantic tennis Games : what is it? More complex than convergence, tennis games are also about regulatory completion and emulation in a race to set the global standard of protection. The regulation may start as excessively precautionary in one country, only to be taken over by another country for one reason or another, leading to a muddled picture.
 Criteria (1)How levels of risk acceptability for one particular risk may vary over time and shape regulatory developments; (2) How international policy discussions and processes may affect these level of risk acceptance and impact on a shared or ‘harmonised’ outcome.
Criteria (1)How levels of risk acceptability for one particular risk may vary over time and shape regulatory developments; (2) How international policy discussions and processes may affect these level of risk acceptance and impact on a shared or ‘harmonised’ outcome.
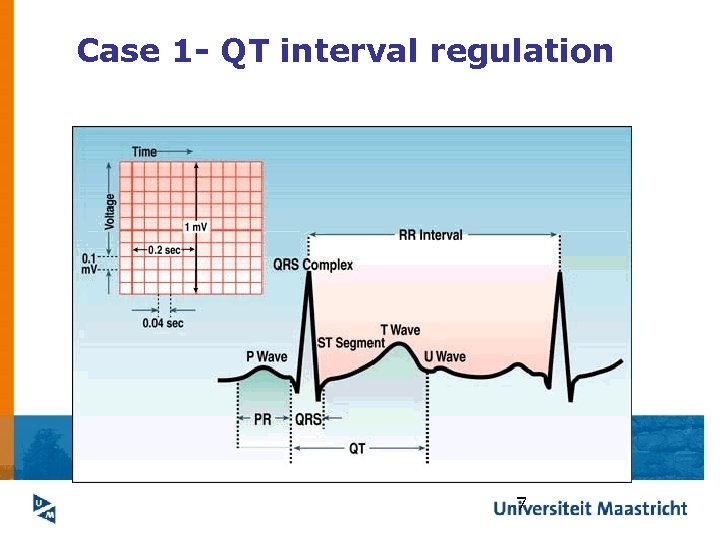 Case 1 - QT interval regulation 7
Case 1 - QT interval regulation 7
 Basic facts about QT prolongation ü QT prolongation can degenerate into a potentially fatal form of tachyarrhythmia called Torsades de Pointes (Td. P). Dessertenne 1966. ü QT interval varies naturally (e. g. age and gender variability) Meyerberg 1999. ü A large number of cardiac and noncardiac drugs prolong QT. About 90 noncardiac drugs reported. Shah 2002. 8
Basic facts about QT prolongation ü QT prolongation can degenerate into a potentially fatal form of tachyarrhythmia called Torsades de Pointes (Td. P). Dessertenne 1966. ü QT interval varies naturally (e. g. age and gender variability) Meyerberg 1999. ü A large number of cardiac and noncardiac drugs prolong QT. About 90 noncardiac drugs reported. Shah 2002. 8
 Mix scientific messages? “There is no clear correlation between QT prolongation and the risk of death”. “There is no scientific evidence indicating that a certain QT interval is related to x% increase of death”. NIH 2004. But… “ QT is the best and only surrogate marker for Td. P, especially so because the definition of this arrhythmia requires prolongation of the QT as a preceding event. Haverkamp and al. 2000; Shah 2002. 9
Mix scientific messages? “There is no clear correlation between QT prolongation and the risk of death”. “There is no scientific evidence indicating that a certain QT interval is related to x% increase of death”. NIH 2004. But… “ QT is the best and only surrogate marker for Td. P, especially so because the definition of this arrhythmia requires prolongation of the QT as a preceding event. Haverkamp and al. 2000; Shah 2002. 9
 Triggers for action 1985. Approval of Terfenadine by FDA, a drug prescribed for hay fever and itchy rash. No cardiovascular events. Botstein, 1993. 1990. Some cases of sudden death observed. First case report (Dec. ) Symptomatic Td. P occurring with the use of Terfenadine. 1991 Similar worries concerning Terodiline (used against incontinence) Connolly et al. 1991; Stewart et al. , 1992 FDA calling for more research on subpopulation at risk, announcing warning labels and sending “Dear Doctor” letters 10
Triggers for action 1985. Approval of Terfenadine by FDA, a drug prescribed for hay fever and itchy rash. No cardiovascular events. Botstein, 1993. 1990. Some cases of sudden death observed. First case report (Dec. ) Symptomatic Td. P occurring with the use of Terfenadine. 1991 Similar worries concerning Terodiline (used against incontinence) Connolly et al. 1991; Stewart et al. , 1992 FDA calling for more research on subpopulation at risk, announcing warning labels and sending “Dear Doctor” letters 10
 In 1996, similar cases of unexplained death among patients taking Cisapride, meant to cure dysfunctions of the oesophagi. Wysowski and al. 1996. Despite the lack of epidemiological studies (Darpö 2001), FDA decided the risk was unacceptable. Terfenadine was withdrawn from the US market in 1998. “Voluntary” withdrawal of Cisapride in 2000 (WHO annual index). 11
In 1996, similar cases of unexplained death among patients taking Cisapride, meant to cure dysfunctions of the oesophagi. Wysowski and al. 1996. Despite the lack of epidemiological studies (Darpö 2001), FDA decided the risk was unacceptable. Terfenadine was withdrawn from the US market in 1998. “Voluntary” withdrawal of Cisapride in 2000 (WHO annual index). 11
 The EU : assessment or precaution? The European regulator (EMEA) did withdraw Terfanadine nor Cisapride EMEA’s convened in 1997 an ad-hoc expert group to “provide reassurance concerning the safe clinical usage of such products” Outcome: “Points to consider” document acknowledging potential risks and suggesting general recommendations for future applicants. Human and non-human testing envisaged 12
The EU : assessment or precaution? The European regulator (EMEA) did withdraw Terfanadine nor Cisapride EMEA’s convened in 1997 an ad-hoc expert group to “provide reassurance concerning the safe clinical usage of such products” Outcome: “Points to consider” document acknowledging potential risks and suggesting general recommendations for future applicants. Human and non-human testing envisaged 12
 From globalisation of concerns to trilateral action Consultation Workshop, Washington DC, 01/03 Tripartite (Europe-US-Japan) ICH Expert Working Group (EWG) composed of 6 stakeholders (public + private) EWG meetings: Tokyo, Brussels, Osaka, Washington DC (7 -10/06/04) “Step 2” document signed on 10/06/04 and open for public comments 06 -10/04 Endorsement : “Step 4” final document May 2005 End of 2005: S 7 B (non clinical) and E 14 (clinical) guidance 13
From globalisation of concerns to trilateral action Consultation Workshop, Washington DC, 01/03 Tripartite (Europe-US-Japan) ICH Expert Working Group (EWG) composed of 6 stakeholders (public + private) EWG meetings: Tokyo, Brussels, Osaka, Washington DC (7 -10/06/04) “Step 2” document signed on 10/06/04 and open for public comments 06 -10/04 Endorsement : “Step 4” final document May 2005 End of 2005: S 7 B (non clinical) and E 14 (clinical) guidance 13
 Areas of disagreement with major consequences – Levels of thresholds – Calculation of thresholds – Need for clinical (human) studies E 14 does not rule out the most restrictive options 14
Areas of disagreement with major consequences – Levels of thresholds – Calculation of thresholds – Need for clinical (human) studies E 14 does not rule out the most restrictive options 14
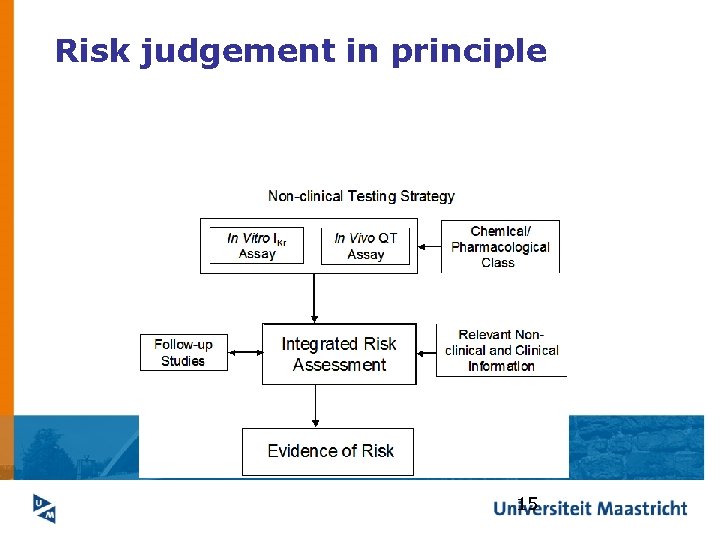 Risk judgement in principle 15
Risk judgement in principle 15
 What basis for risk judgement in practice? Weak consensus on science, no acceptability tools liked to risk quantification , Industry asked to perform signal (hazard) detection thorough QT/QTc studies. How much will QT regulation cost? Not clear Low concerns for stigmatisation effects of drugs that approach the upper limit 16
What basis for risk judgement in practice? Weak consensus on science, no acceptability tools liked to risk quantification , Industry asked to perform signal (hazard) detection thorough QT/QTc studies. How much will QT regulation cost? Not clear Low concerns for stigmatisation effects of drugs that approach the upper limit 16
 QT interval conclusions ‘Race to the top’ based on: - signal identification of possible hazard rather than probabilistic estimates on increased risk - Little attention paid to NRC-type risk management measures (Risk/benefit, risk perception studies etc. ) 17
QT interval conclusions ‘Race to the top’ based on: - signal identification of possible hazard rather than probabilistic estimates on increased risk - Little attention paid to NRC-type risk management measures (Risk/benefit, risk perception studies etc. ) 17
 Case 2 - Acomplia – Acomplia (rimonabant) is an anti-obesity drug manufactured by Sanofi-aventis. Approved for marketing in the European Union in June 2006. – Sanofi argued that Acomplia can also prevent the risk of cardiovascular disease. – Prior to its approval, Sanofi claimed the drug was also potentially capable of treating depression and helping people stop smoking.
Case 2 - Acomplia – Acomplia (rimonabant) is an anti-obesity drug manufactured by Sanofi-aventis. Approved for marketing in the European Union in June 2006. – Sanofi argued that Acomplia can also prevent the risk of cardiovascular disease. – Prior to its approval, Sanofi claimed the drug was also potentially capable of treating depression and helping people stop smoking.
 Sanofi’s new blockbuster? Rimonabant is a selective antagonist of cannabinoid type 1 (CB 1) receptor. The first member of a new class of compounds that target a novel physiological system, the endocannabinoid system (ECS). (EMEA Scientific discussion) Considerable media attention (both negative and positive)
Sanofi’s new blockbuster? Rimonabant is a selective antagonist of cannabinoid type 1 (CB 1) receptor. The first member of a new class of compounds that target a novel physiological system, the endocannabinoid system (ECS). (EMEA Scientific discussion) Considerable media attention (both negative and positive)
 Initial discussions on safety “Depressive disorders reported with rimonabant 20 mg were usually mild or moderate in severity. Most of the cases resolved with the corrective measures undertaken, either discontinuation or anti-depressant treatment. Overall, psychiatric adverse events or adverse symptoms from the CNS were more common among rimonabant treated patients than among placebo treated. These adverse events were dose dependent”
Initial discussions on safety “Depressive disorders reported with rimonabant 20 mg were usually mild or moderate in severity. Most of the cases resolved with the corrective measures undertaken, either discontinuation or anti-depressant treatment. Overall, psychiatric adverse events or adverse symptoms from the CNS were more common among rimonabant treated patients than among placebo treated. These adverse events were dose dependent”
 US/Europe On 13 June 2007, the FDA independent advisory committee unanimously voted against approval. Why? Suicidal thoughts were reported in patients under clinical trials “Regulators worried about cardiovascular risks ten years ago, psychiatric effects are today’s concern” (Patient)
US/Europe On 13 June 2007, the FDA independent advisory committee unanimously voted against approval. Why? Suicidal thoughts were reported in patients under clinical trials “Regulators worried about cardiovascular risks ten years ago, psychiatric effects are today’s concern” (Patient)
 Impact on EMA “EMEA did not want to seem weaker than its American counterpart” – Industry respondent Version contested by agency. Yet. . .
Impact on EMA “EMEA did not want to seem weaker than its American counterpart” – Industry respondent Version contested by agency. Yet. . .
 EMA’s changing position 14 June 2007 EMA Communication on US stop on prescribing 19 July 2007 Press release and Q&As Restriction of prescriptions to susceptible patients (taking antidepressants) EMEA/330598/2007 “Acomplia must not be used in patients on antidepressants or with major depression” 25 July 2007 EMEA/318931/2007
EMA’s changing position 14 June 2007 EMA Communication on US stop on prescribing 19 July 2007 Press release and Q&As Restriction of prescriptions to susceptible patients (taking antidepressants) EMEA/330598/2007 “Acomplia must not be used in patients on antidepressants or with major depression” 25 July 2007 EMEA/318931/2007
 Restriction In May 2008, the CHMP recommended updating the product information to “reflect the fact that depression may occur as a side effect of Acomplia in patients who have no obvious risk factors apart from obesity itself”, and “to advise prescribers to monitor patients for signs and symptoms of psychiatric disorders, particularly depression, after the start of treatment”. More studies asked, more discussion with FDA
Restriction In May 2008, the CHMP recommended updating the product information to “reflect the fact that depression may occur as a side effect of Acomplia in patients who have no obvious risk factors apart from obesity itself”, and “to advise prescribers to monitor patients for signs and symptoms of psychiatric disorders, particularly depression, after the start of treatment”. More studies asked, more discussion with FDA
 Withdrawal 23 October 2008. “The CHMP considered that the new data from postmarketing experience and ongoing clinical trials indicated that serious psychiatric disorders may be more common than in the clinical trials used in the initial assessment of the medicine. The CHMP was also of the opinion that these psychiatric side effects could not be adequately addressed by further risk minimisation measures”.
Withdrawal 23 October 2008. “The CHMP considered that the new data from postmarketing experience and ongoing clinical trials indicated that serious psychiatric disorders may be more common than in the clinical trials used in the initial assessment of the medicine. The CHMP was also of the opinion that these psychiatric side effects could not be adequately addressed by further risk minimisation measures”.
 Dilemma: clinical trials “CHMP suspended the marketing of Acomplia but not the clinical trials. This made no sense. One week after national authorities, which are the only competent authority in this matter decided to suspend clinical trials” (Regulator) Pro: “With strong safeguards on prescription we could have explored the potential of this product” (Industry) Against: “how is it possible to maintain double standards? ” (Regulator)
Dilemma: clinical trials “CHMP suspended the marketing of Acomplia but not the clinical trials. This made no sense. One week after national authorities, which are the only competent authority in this matter decided to suspend clinical trials” (Regulator) Pro: “With strong safeguards on prescription we could have explored the potential of this product” (Industry) Against: “how is it possible to maintain double standards? ” (Regulator)
 Acomplia conclusions EMA risk assessors feel they cannot afford to ignore FDA. Transatlantic discussion leads to more conservative interpretation of relatively stable clinical data and to more stringency. Major remaining difference: clinical trials. Unlike FDA some corners of EMA still in favour of more trials.
Acomplia conclusions EMA risk assessors feel they cannot afford to ignore FDA. Transatlantic discussion leads to more conservative interpretation of relatively stable clinical data and to more stringency. Major remaining difference: clinical trials. Unlike FDA some corners of EMA still in favour of more trials.
 Lessons from tennis games 1 - Emulation of concerns 2 - Sharing objectives enhanced by international processes 3 - Mutually-feeding race to the top 4 - Regulatory culture of each block remains critical for delivery 5 - Therefore convergence remains limited at delivery
Lessons from tennis games 1 - Emulation of concerns 2 - Sharing objectives enhanced by international processes 3 - Mutually-feeding race to the top 4 - Regulatory culture of each block remains critical for delivery 5 - Therefore convergence remains limited at delivery
 Other possible tennis games? Chemical policy and how Europe set the standards [donna le ton] after REACH being an influence on calls for reforming US regulation? Pharmaceuticals in the environment: Europeans influencing water pollution standards from pharmaceutical products. Terrorism including aspects of financing of terrorism. US leading standards
Other possible tennis games? Chemical policy and how Europe set the standards [donna le ton] after REACH being an influence on calls for reforming US regulation? Pharmaceuticals in the environment: Europeans influencing water pollution standards from pharmaceutical products. Terrorism including aspects of financing of terrorism. US leading standards


