71e0b536bce63384bd32aa05d6ee060c.ppt
- Количество слайдов: 39
 REGULATORY GUIDELINES FOR PRECLINICAL AND CLINICAL VALIDATION OF TRADITIONAL MEDICINES BY Dr. Basavaraj K. Nanjwade Associate Professor of Pharmaceutics Department of Pharmacology JN Medical College Belgaum-10 e-mail: bknanjwade@yahoo. co. in 19/08/2007 KLE University, Belgaum, India 1
REGULATORY GUIDELINES FOR PRECLINICAL AND CLINICAL VALIDATION OF TRADITIONAL MEDICINES BY Dr. Basavaraj K. Nanjwade Associate Professor of Pharmaceutics Department of Pharmacology JN Medical College Belgaum-10 e-mail: bknanjwade@yahoo. co. in 19/08/2007 KLE University, Belgaum, India 1
 Traditional Medicine “Traditional medicine refers to health practices, approaches, knowledge and beliefs incorporating plant, animal and mineral based medicines, spiritual therapies, manual techniques and exercises, applied singularly or in combination to treat, diagnose and prevent illnesses or maintain well-being” 19/08/2007 KLE University, Belgaum, India 2
Traditional Medicine “Traditional medicine refers to health practices, approaches, knowledge and beliefs incorporating plant, animal and mineral based medicines, spiritual therapies, manual techniques and exercises, applied singularly or in combination to treat, diagnose and prevent illnesses or maintain well-being” 19/08/2007 KLE University, Belgaum, India 2
 Traditional Medicine/CAM n Non-Industrialized countries population uses for primary health care needs. E. g. Africa up to 80% n In industrialized countries, adaptations of traditional medicine are “Complementary” or “Alternative” medicine E. g. USA, EU, Australia etc. 19/08/2007 KLE University, Belgaum, India 3
Traditional Medicine/CAM n Non-Industrialized countries population uses for primary health care needs. E. g. Africa up to 80% n In industrialized countries, adaptations of traditional medicine are “Complementary” or “Alternative” medicine E. g. USA, EU, Australia etc. 19/08/2007 KLE University, Belgaum, India 3
 Use and Popularity of TM/CAM n 30%-50% of the total medicinal consumption - China. n 60% first line treatment - Ghana, Nigeria and Zambia n Over 50% of the population have used CAM at least once – Europe & North America n 75% of people living with HIV/AIDS use TM/CAM - San Francisco, London & South Africa. 19/08/2007 KLE University, Belgaum, India 4
Use and Popularity of TM/CAM n 30%-50% of the total medicinal consumption - China. n 60% first line treatment - Ghana, Nigeria and Zambia n Over 50% of the population have used CAM at least once – Europe & North America n 75% of people living with HIV/AIDS use TM/CAM - San Francisco, London & South Africa. 19/08/2007 KLE University, Belgaum, India 4
 Use and Popularity of TM/CAM n 158 million of the adult population use complementary medicines – US n US $ 17 billion was spent on traditional remedies in 2000 n In UK annual expenditure on alternation medicine is US $ 230 million n The global market for herbal medicines currently stands at over US $ 60 billion annual and is growing steadily 19/08/2007 KLE University, Belgaum, India 5
Use and Popularity of TM/CAM n 158 million of the adult population use complementary medicines – US n US $ 17 billion was spent on traditional remedies in 2000 n In UK annual expenditure on alternation medicine is US $ 230 million n The global market for herbal medicines currently stands at over US $ 60 billion annual and is growing steadily 19/08/2007 KLE University, Belgaum, India 5
 TCM Products n Traditional Chinese Medicine products are taken to mean “finished TCM products and proprietary products” n A finished product is one that has undergone all stages of production, including packaging in its final containers and labeling. n A single TCM formula or product may contain between six to twenty different ingredients 19/08/2007 KLE University, Belgaum, India 6
TCM Products n Traditional Chinese Medicine products are taken to mean “finished TCM products and proprietary products” n A finished product is one that has undergone all stages of production, including packaging in its final containers and labeling. n A single TCM formula or product may contain between six to twenty different ingredients 19/08/2007 KLE University, Belgaum, India 6
 TCM Products n Chines medicinal materials showed that 86. 8%, 12. 5% and 0. 7% of the products examined could be classified as herb, animal and mineral, respectively. 19/08/2007 KLE University, Belgaum, India 7
TCM Products n Chines medicinal materials showed that 86. 8%, 12. 5% and 0. 7% of the products examined could be classified as herb, animal and mineral, respectively. 19/08/2007 KLE University, Belgaum, India 7
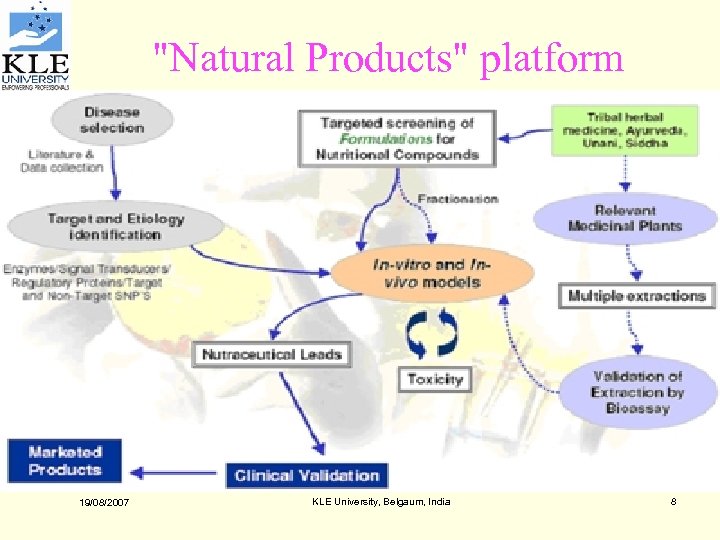 "Natural Products" platform 19/08/2007 KLE University, Belgaum, India 8
"Natural Products" platform 19/08/2007 KLE University, Belgaum, India 8
 Pre-clinical studies n Pre-clinical studies serve a vital role in the drug discovery and development processes. n These studies can be used to identify lead compounds likely to possess favourable biopharmaceutic and pharmacokinetic properties in humans. n In addition, they can facilitate transition through the discovery - development interface and decrease the need for expensive and time-consuming clinical studies. 19/08/2007 KLE University, Belgaum, India 9
Pre-clinical studies n Pre-clinical studies serve a vital role in the drug discovery and development processes. n These studies can be used to identify lead compounds likely to possess favourable biopharmaceutic and pharmacokinetic properties in humans. n In addition, they can facilitate transition through the discovery - development interface and decrease the need for expensive and time-consuming clinical studies. 19/08/2007 KLE University, Belgaum, India 9
 Pre-clinical studies include - in vivo animal models - isolated perfused liver, kidney, intestine, hind limb and heart - animal and human liver microsomes - bioavailability studies - pharmacokinetic studies - prediction of oral absorption in humans - determination of mechanisms of intestinal absorption 19/08/2007 KLE University, Belgaum, India 10
Pre-clinical studies include - in vivo animal models - isolated perfused liver, kidney, intestine, hind limb and heart - animal and human liver microsomes - bioavailability studies - pharmacokinetic studies - prediction of oral absorption in humans - determination of mechanisms of intestinal absorption 19/08/2007 KLE University, Belgaum, India 10
 Pre-clinical studies include - assessment of transport, distribution and elimination of compounds - validated models for cytochrome P 450 enzymes - metabolism studies in human liver microsomes - assessment of potential for metabolic drug – drug interactions - analysis of drugs and metabolites in biological matrices 19/08/2007 KLE University, Belgaum, India 11
Pre-clinical studies include - assessment of transport, distribution and elimination of compounds - validated models for cytochrome P 450 enzymes - metabolism studies in human liver microsomes - assessment of potential for metabolic drug – drug interactions - analysis of drugs and metabolites in biological matrices 19/08/2007 KLE University, Belgaum, India 11
 Limitations of preclinical studies 1. Suitable pharmacological models have not yet been developed for many common diseases 2. Toxicity testing is time-consuming & expensive. 3. Large numbers of animals must be used. 4. Extrapolation of toxicity data from animals to humans is not completely reliable. 5. Rare adverse effects are unlikely to be detected 19/08/2007 KLE University, Belgaum, India 12
Limitations of preclinical studies 1. Suitable pharmacological models have not yet been developed for many common diseases 2. Toxicity testing is time-consuming & expensive. 3. Large numbers of animals must be used. 4. Extrapolation of toxicity data from animals to humans is not completely reliable. 5. Rare adverse effects are unlikely to be detected 19/08/2007 KLE University, Belgaum, India 12
 Clinical studies include Two types of clinical research 1. Methodology/study design 2. Clinical trials a. Phase one study b. Phase two study c. Phase three study d. Phase four study/Late Phase three-Post Marketing 19/08/2007 KLE University, Belgaum, India 13
Clinical studies include Two types of clinical research 1. Methodology/study design 2. Clinical trials a. Phase one study b. Phase two study c. Phase three study d. Phase four study/Late Phase three-Post Marketing 19/08/2007 KLE University, Belgaum, India 13
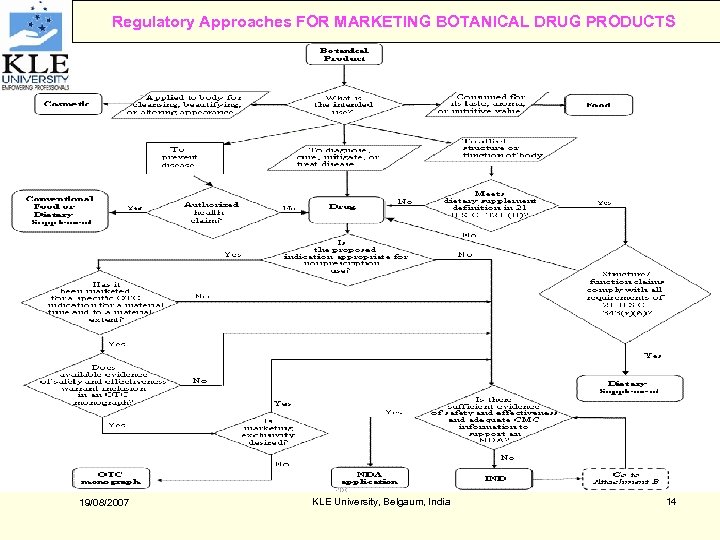 Regulatory Approaches FOR MARKETING BOTANICAL DRUG PRODUCTS 19/08/2007 KLE University, Belgaum, India 14
Regulatory Approaches FOR MARKETING BOTANICAL DRUG PRODUCTS 19/08/2007 KLE University, Belgaum, India 14
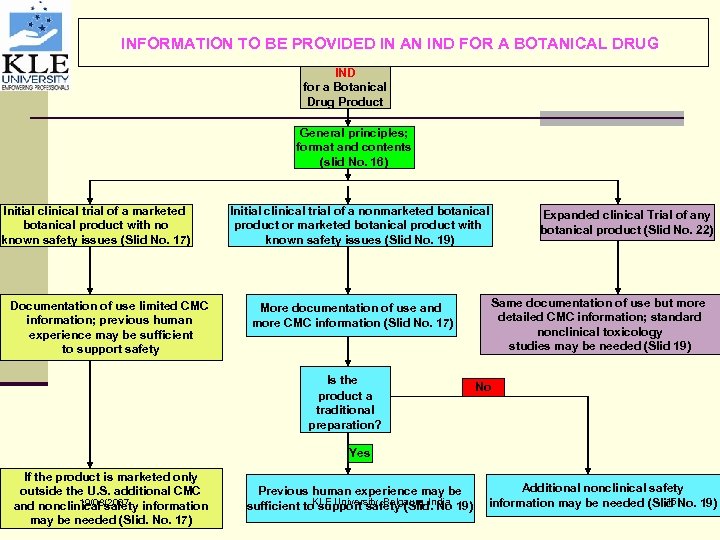 INFORMATION TO BE PROVIDED IN AN IND FOR A BOTANICAL DRUG IND for a Botanical Drug Product General principles; format and contents (slid No. 16) Initial clinical trial of a marketed botanical product with no known safety issues (Slid No. 17) Documentation of use limited CMC information; previous human experience may be sufficient to support safety Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues (Slid No. 19) More documentation of use and more CMC information (Slid No. 17) Is the product a traditional preparation? Expanded clinical Trial of any botanical product (Slid No. 22) Same documentation of use but more detailed CMC information; standard nonclinical toxicology studies may be needed (Slid 19) No Yes If the product is marketed only outside the U. S. additional CMC 19/08/2007 and nonclinical safety information may be needed (Slid. No. 17) Previous human experience may be sufficient to. KLE University, Belgaum, India 19) support safety (Slid. No Additional nonclinical safety 15 information may be needed (Slid No. 19)
INFORMATION TO BE PROVIDED IN AN IND FOR A BOTANICAL DRUG IND for a Botanical Drug Product General principles; format and contents (slid No. 16) Initial clinical trial of a marketed botanical product with no known safety issues (Slid No. 17) Documentation of use limited CMC information; previous human experience may be sufficient to support safety Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues (Slid No. 19) More documentation of use and more CMC information (Slid No. 17) Is the product a traditional preparation? Expanded clinical Trial of any botanical product (Slid No. 22) Same documentation of use but more detailed CMC information; standard nonclinical toxicology studies may be needed (Slid 19) No Yes If the product is marketed only outside the U. S. additional CMC 19/08/2007 and nonclinical safety information may be needed (Slid. No. 17) Previous human experience may be sufficient to. KLE University, Belgaum, India 19) support safety (Slid. No Additional nonclinical safety 15 information may be needed (Slid No. 19)
 General principal & format and contents A. IND information for different categories of botanicals B. Basic format for INDs 1. Cover sheet 2. Table of contents 3. Introductory statement and general investigational plan 4. Investigators Brochure 5. Protocol 6. Chemistry, Manufacturing and Controls 7. Pharmacology and Toxicological Information 8. Previous Human Experience with the Product 19/08/2007 KLE University, Belgaum, India 16
General principal & format and contents A. IND information for different categories of botanicals B. Basic format for INDs 1. Cover sheet 2. Table of contents 3. Introductory statement and general investigational plan 4. Investigators Brochure 5. Protocol 6. Chemistry, Manufacturing and Controls 7. Pharmacology and Toxicological Information 8. Previous Human Experience with the Product 19/08/2007 KLE University, Belgaum, India 16
 Initial clinical trial of a marketed botanical product with no known safety issues A. Description of Product and Documentation of Human Use 1. Description of Botanicals Used 2. Currently Marketed Use B. Chemistry, Manufacturing, and Controls 1. Botanical Raw Material 2. Botanical Drug Substance 3. Botanical Drug Product a. Quantitative description b. The composition or quantitative description manufacturer's certificate of analysis 4. Placebo 5. Labeling 6. Environmental Assessment or Claim of Categorical Exclusion 19/08/2007 KLE University, Belgaum, India 17
Initial clinical trial of a marketed botanical product with no known safety issues A. Description of Product and Documentation of Human Use 1. Description of Botanicals Used 2. Currently Marketed Use B. Chemistry, Manufacturing, and Controls 1. Botanical Raw Material 2. Botanical Drug Substance 3. Botanical Drug Product a. Quantitative description b. The composition or quantitative description manufacturer's certificate of analysis 4. Placebo 5. Labeling 6. Environmental Assessment or Claim of Categorical Exclusion 19/08/2007 KLE University, Belgaum, India 17
 Initial clinical trial of a marketed botanical product with no known safety issues C. Pharmacology 1. All Marketed Botanical Products 2. Foreign- Marketed Botanical Products D. Bioavailability E. Clinical Considerations 19/08/2007 KLE University, Belgaum, India 18
Initial clinical trial of a marketed botanical product with no known safety issues C. Pharmacology 1. All Marketed Botanical Products 2. Foreign- Marketed Botanical Products D. Bioavailability E. Clinical Considerations 19/08/2007 KLE University, Belgaum, India 18
 Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues A. Description of Product & Documentation of Human Use 1. Description of Botanicals Used 2. History of Use 3. Current Investigational Use B. Chemistry, Manufacturing, & Controls 1. Botanical Raw Material 2. Botanical Drug Substance a. A Quantitative b. The quantitative description (strength) c. Manufacturer (Processor) d. Manufacturing process e. Quality control tests 19/08/2007 KLE University, Belgaum, India 19
Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues A. Description of Product & Documentation of Human Use 1. Description of Botanicals Used 2. History of Use 3. Current Investigational Use B. Chemistry, Manufacturing, & Controls 1. Botanical Raw Material 2. Botanical Drug Substance a. A Quantitative b. The quantitative description (strength) c. Manufacturer (Processor) d. Manufacturing process e. Quality control tests 19/08/2007 KLE University, Belgaum, India 19
 Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues f. Container g. Container label 3. Botanical Drug Product a. Qualitative description b. Composition, or quantitative description c. Manufacturer d. Manufacturing process e. Quality control test f. Container/Closure g. Stability data on the drug product 4. Placebo 5. Labeling 19/08/2007 KLE University, Belgaum, India 20
Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues f. Container g. Container label 3. Botanical Drug Product a. Qualitative description b. Composition, or quantitative description c. Manufacturer d. Manufacturing process e. Quality control test f. Container/Closure g. Stability data on the drug product 4. Placebo 5. Labeling 19/08/2007 KLE University, Belgaum, India 20
 Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues 6. Environmental Assessment or Claim of Categorical Exclusion C. Preclinical Safety Assessment 1. Traditional Preparations 2. Others D. Bioavailability E. Clinical Considerations 19/08/2007 KLE University, Belgaum, India 21
Initial clinical trial of a nonmarketed botanical product or marketed botanical product with known safety issues 6. Environmental Assessment or Claim of Categorical Exclusion C. Preclinical Safety Assessment 1. Traditional Preparations 2. Others D. Bioavailability E. Clinical Considerations 19/08/2007 KLE University, Belgaum, India 21
 Expanded clinical Trial of any botanical product A. B. a. b. - Description of Product & Documentation of Human Experience Chemistry, Manufacturing, & Controls Botanical raw material Botanical drug substance Qualitative identification Chemical identification Specifications Manufacturing process Quality control tests Test methods Reference standards Containers & closure 19/08/2007 KLE University, Belgaum, India 22
Expanded clinical Trial of any botanical product A. B. a. b. - Description of Product & Documentation of Human Experience Chemistry, Manufacturing, & Controls Botanical raw material Botanical drug substance Qualitative identification Chemical identification Specifications Manufacturing process Quality control tests Test methods Reference standards Containers & closure 19/08/2007 KLE University, Belgaum, India 22
 Expanded clinical Trial of any botanical product Stability data Container label C. Botanical drug product - Qualitative description & the composition - Acceptance specifications - Manufacturing process - Quality control tests - Test methods - Containers & closure - Stability data d. Placebo e. Labeling f. Claim of categorical exclusion - 19/08/2007 KLE University, Belgaum, India 23
Expanded clinical Trial of any botanical product Stability data Container label C. Botanical drug product - Qualitative description & the composition - Acceptance specifications - Manufacturing process - Quality control tests - Test methods - Containers & closure - Stability data d. Placebo e. Labeling f. Claim of categorical exclusion - 19/08/2007 KLE University, Belgaum, India 23
 Expanded clinical Trial of any botanical product 2. End-of-Phase 3 Clinical Studies & Pre-NDA Considerations a. Botanical raw materials b. Manufacturing process c. Batch-to-batch consistency d. Specifications e. Analytical methods & test procedures f. Reference standard g. Stability-indicating analytical h. Comparison of the similarities and/or differences in CMC among preclinical and clinical i. Manufacturing and testing facilities j. Claim for categorical exclusion 19/08/2007 KLE University, Belgaum, India 24
Expanded clinical Trial of any botanical product 2. End-of-Phase 3 Clinical Studies & Pre-NDA Considerations a. Botanical raw materials b. Manufacturing process c. Batch-to-batch consistency d. Specifications e. Analytical methods & test procedures f. Reference standard g. Stability-indicating analytical h. Comparison of the similarities and/or differences in CMC among preclinical and clinical i. Manufacturing and testing facilities j. Claim for categorical exclusion 19/08/2007 KLE University, Belgaum, India 24
 Expanded clinical Trial of any botanical product C. Preclinical Safety Assessment (including Pre-NDA) 1. Repeat-Dose General Toxicity Studies 2. Non-clinical Pharmacokinetic/Toxic-kinetic Studies 3. Reproductive Toxicology 4. Genotoxicity Studies 5. Carcinogenicity Studies 6. Special Pharmacology/Toxicology Studies 7. Regulatory Considerations D. Bio-availability & Drug-Drug Interactions E. Clinical Considerations 19/08/2007 KLE University, Belgaum, India 25
Expanded clinical Trial of any botanical product C. Preclinical Safety Assessment (including Pre-NDA) 1. Repeat-Dose General Toxicity Studies 2. Non-clinical Pharmacokinetic/Toxic-kinetic Studies 3. Reproductive Toxicology 4. Genotoxicity Studies 5. Carcinogenicity Studies 6. Special Pharmacology/Toxicology Studies 7. Regulatory Considerations D. Bio-availability & Drug-Drug Interactions E. Clinical Considerations 19/08/2007 KLE University, Belgaum, India 25
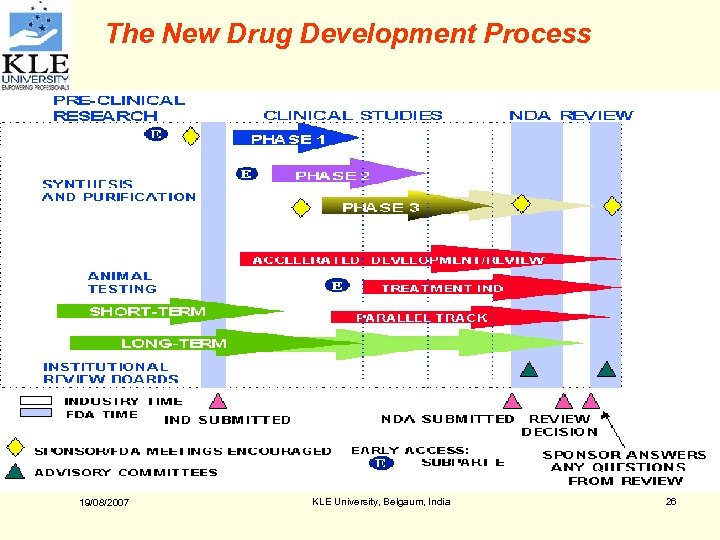 The New Drug Development Process 19/08/2007 KLE University, Belgaum, India 26
The New Drug Development Process 19/08/2007 KLE University, Belgaum, India 26
 19/08/2007 KLE University, Belgaum, India 27
19/08/2007 KLE University, Belgaum, India 27
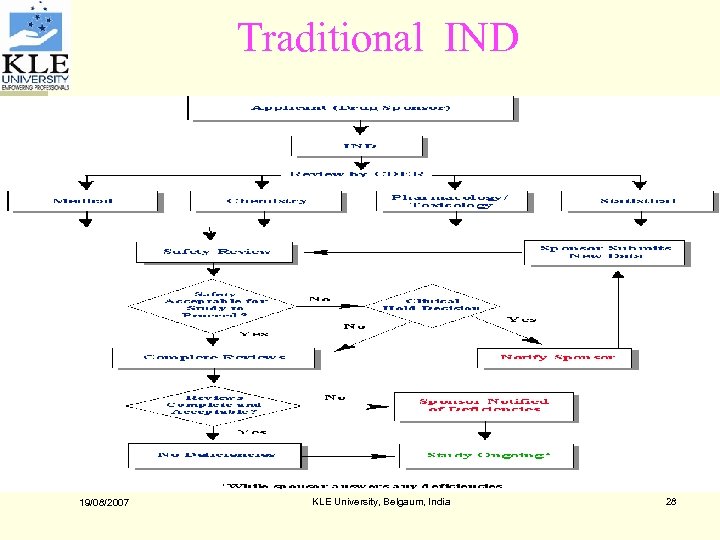 Traditional IND 19/08/2007 KLE University, Belgaum, India 28
Traditional IND 19/08/2007 KLE University, Belgaum, India 28
 19/08/2007 KLE University, Belgaum, India 29
19/08/2007 KLE University, Belgaum, India 29
 19/08/2007 KLE University, Belgaum, India 30
19/08/2007 KLE University, Belgaum, India 30
 19/08/2007 KLE University, Belgaum, India 31
19/08/2007 KLE University, Belgaum, India 31
 19/08/2007 KLE University, Belgaum, India 32
19/08/2007 KLE University, Belgaum, India 32
 19/08/2007 KLE University, Belgaum, India 33
19/08/2007 KLE University, Belgaum, India 33
 Conclusions of Exploratory IND 19/08/2007 KLE University, Belgaum, India 34
Conclusions of Exploratory IND 19/08/2007 KLE University, Belgaum, India 34
 Conclusions of Exploratory IND 19/08/2007 KLE University, Belgaum, India 35
Conclusions of Exploratory IND 19/08/2007 KLE University, Belgaum, India 35
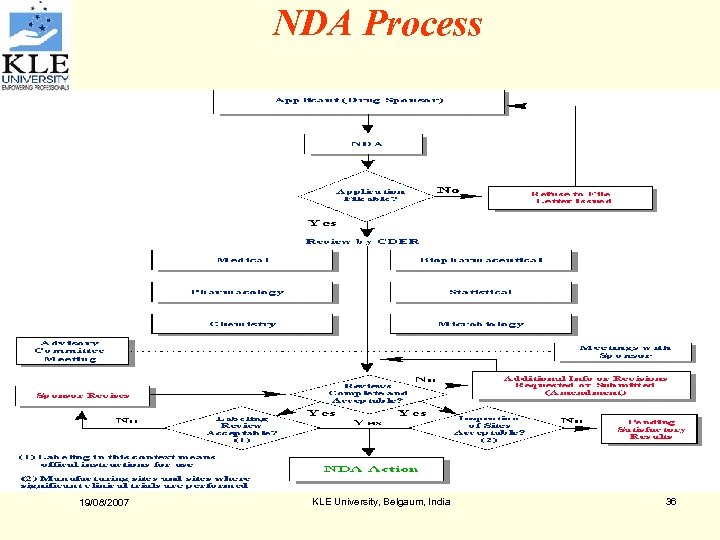 NDA Process 19/08/2007 KLE University, Belgaum, India 36
NDA Process 19/08/2007 KLE University, Belgaum, India 36
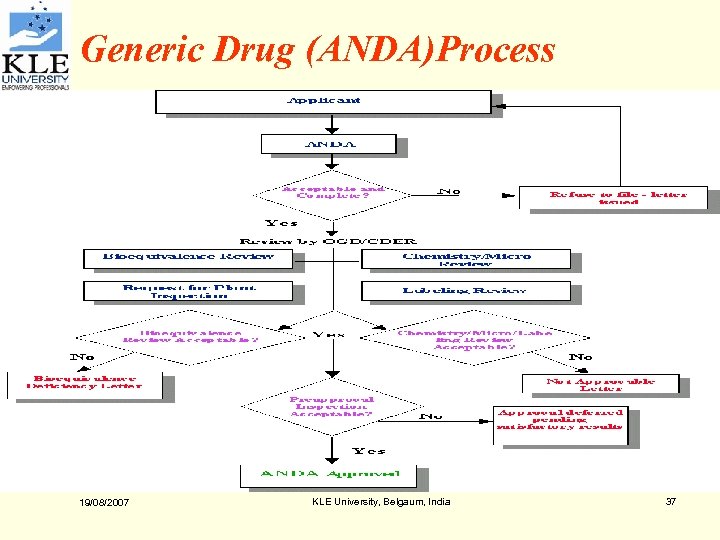 Generic Drug (ANDA)Process 19/08/2007 KLE University, Belgaum, India 37
Generic Drug (ANDA)Process 19/08/2007 KLE University, Belgaum, India 37
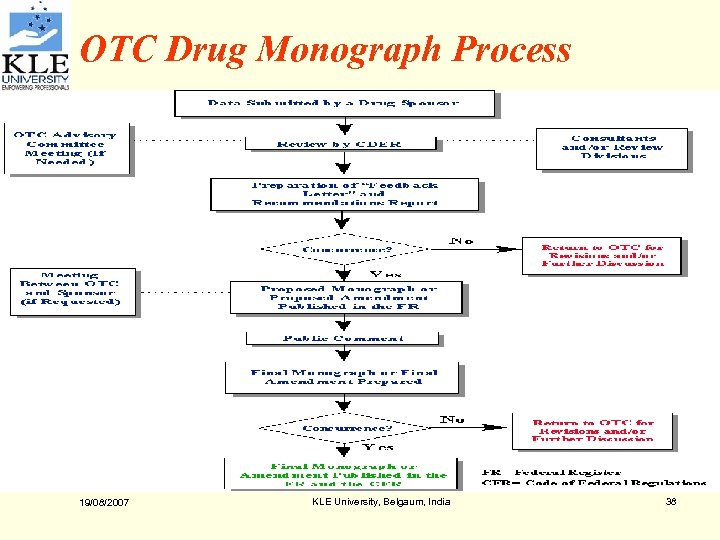 OTC Drug Monograph Process 19/08/2007 KLE University, Belgaum, India 38
OTC Drug Monograph Process 19/08/2007 KLE University, Belgaum, India 38
 e-mail: bknanjwade@yahoo. co. in 19/08/2007 KLE University, Belgaum, India 39
e-mail: bknanjwade@yahoo. co. in 19/08/2007 KLE University, Belgaum, India 39


