0bc722e0d732f3ad90510b65904f5a54.ppt
- Количество слайдов: 20

Region I Advisory Board Meeting Wells Beach, ME June 9, 2008 Use and Verification of STD Nucleic Acid Amplification Tests for non-FDA Cleared Clinical Specimens

Presenter: Sally Liska, Dr. PH Director, San Francisco PH Lab sally_liska@sfdph. org “From Tommy Bahama [Levi Strauss] to LL Bean”

Background: San Francisco Public Health Laboratory • Local PH Lab – support Communicable Disease programs (STD, HIV, TB, etc. ) • Methods – microbiology, virology, serology, molecular diagnostics • Process > 100, 000 specimens/yr from DPH clinics, specialty clinics, jails, NGO clinics, “events”, etc.

Hx of CT/GC Lab Testing at the SFDPH Lab A. Culture for N. gonorrhoeae B. Nucleic Acid Amplification (NAAT) • Ligase Chain Reaction {LCR} – Abbott; 1995 • Strand Displacement Amplification {SDA} – Becton Dickinson; 2001 • Transcription Mediated Amplification {TMA} – Gen-Probe; 2004 • Polymerase Chain Reaction {PCR}
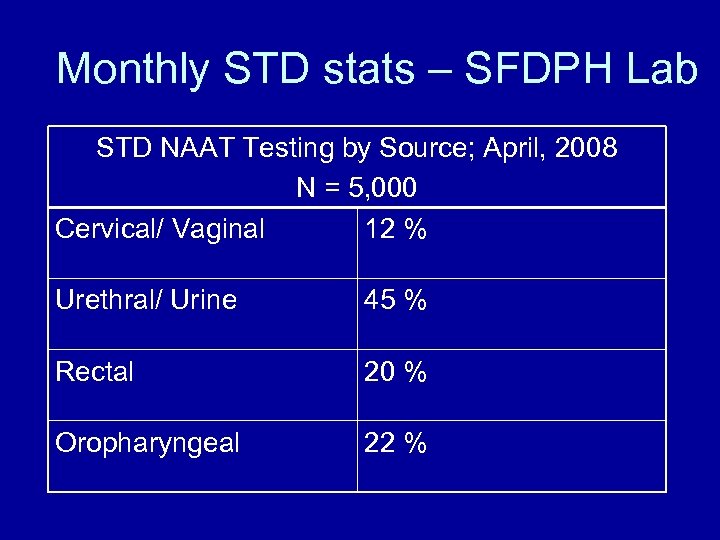
Monthly STD stats – SFDPH Lab STD NAAT Testing by Source; April, 2008 N = 5, 000 Cervical/ Vaginal 12 % Urethral/ Urine 45 % Rectal 20 % Oropharyngeal 22 %

Why Test Rectal & Oropharyngeal Specimens • Reduce acquisition and transmission of STD agents (Chlamydia, Gonorrhea, HIV) • Rectal infections can cause proctitis and increase risk of HIV infection • CDC recommendations for MSM – GC: annual screening of anatomical sites w/ possible exposure CT: annual screening for urethral and rectal infection
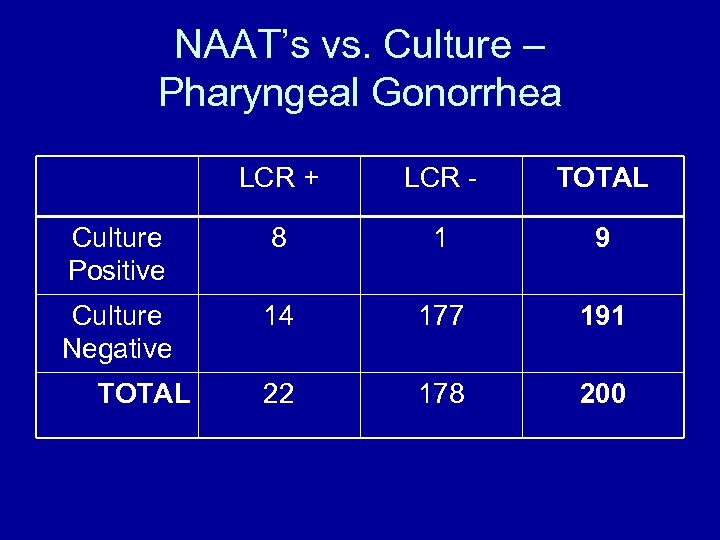
NAAT’s vs. Culture – Pharyngeal Gonorrhea LCR + LCR - TOTAL Culture Positive 8 1 9 Culture Negative 14 177 191 22 178 200 TOTAL

Performance Characteristics Pharyngeal Gonorrhea CULTURE LCR Sensitivity Specificity 47. 4 % 100 % 94. 7 % 97. 8 % Page-Shafer et al in CID, 2002; 34: 173 -6
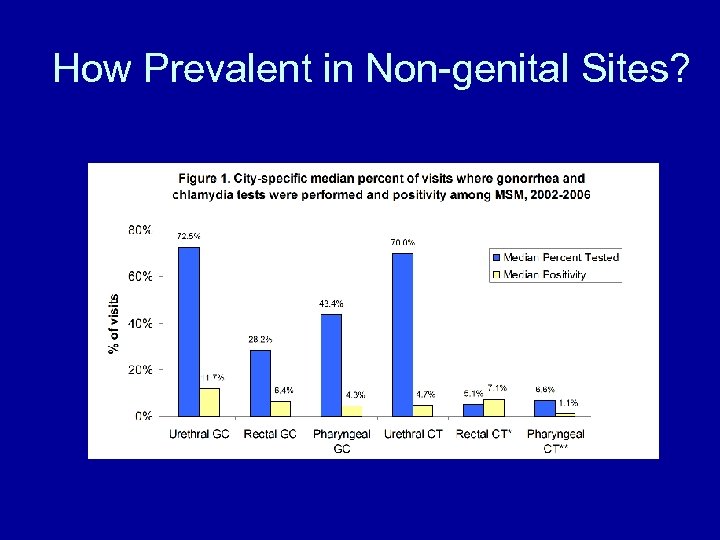
How Prevalent in Non-genital Sites?
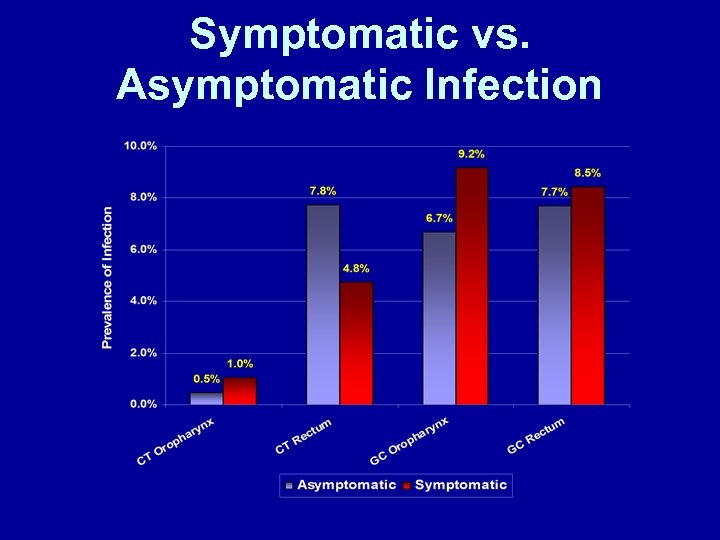
Symptomatic vs. Asymptomatic Infection
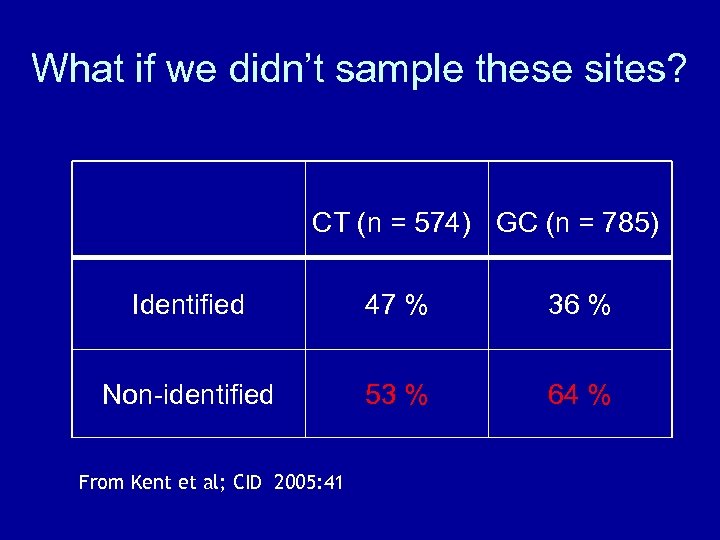
What if we didn’t sample these sites? CT (n = 574) GC (n = 785) Identified 47 % 36 % Non-identified 53 % 64 % From Kent et al; CID 2005: 41

What About Women? When to do rectal & pharyngeal screening in women, what are the indicators? 1. HRSA recommends screening HIV + women (by culture) 2. Medical indications – risk of reinfection if not treated for (rectal) infection? 3. PH indications – prevalence for these sites in women largely unknown

Use of NAAT’s for R & Ph specimens • • PRO’s More sensitive than culture More universal than CT culture Faster turn around time Can be automated CON’s • May be more expensive • NAAT’s not approved for these sites by FDA
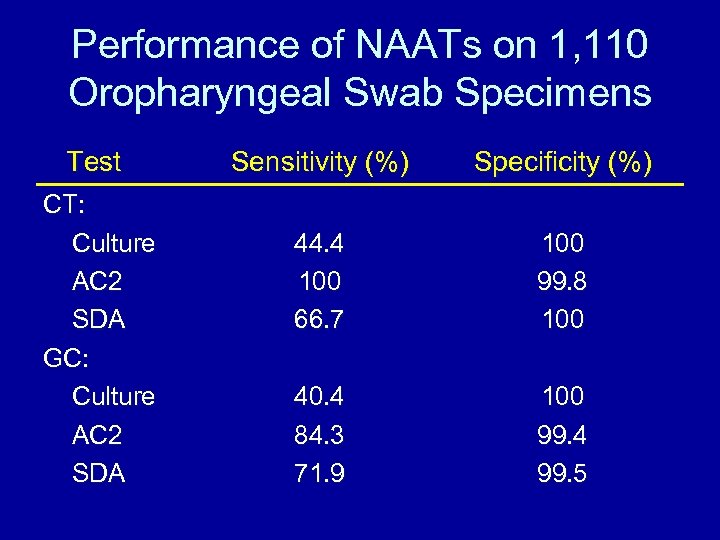
Performance of NAATs on 1, 110 Oropharyngeal Swab Specimens Test CT: Culture AC 2 SDA GC: Culture AC 2 SDA Sensitivity (%) Specificity (%) 44. 4 100 66. 7 100 99. 8 100 40. 4 84. 3 71. 9 100 99. 4 99. 5
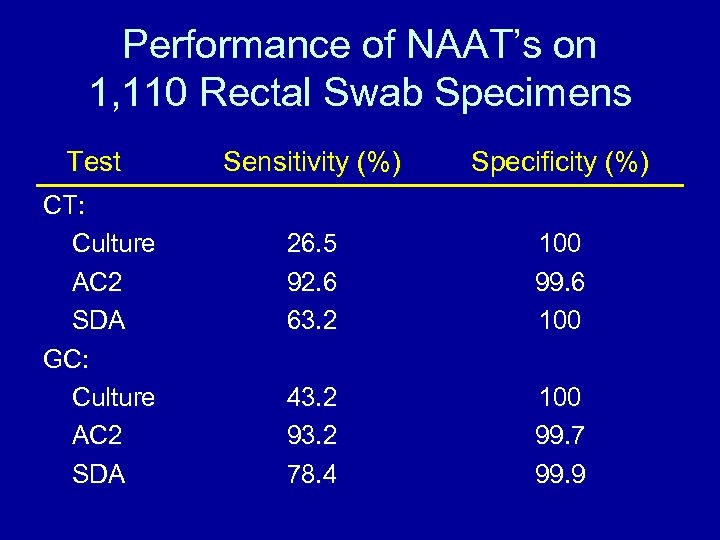
Performance of NAAT’s on 1, 110 Rectal Swab Specimens Test CT: Culture AC 2 SDA GC: Culture AC 2 SDA Sensitivity (%) Specificity (%) 26. 5 92. 6 63. 2 100 99. 6 100 43. 2 93. 2 78. 4 100 99. 7 99. 9

CLIA ’ 88 • Federal regulation w/ oversight on clinical labs – Dx, Rx & prevention of disease • Section 493. 1213 pertains to requirement for Verification of Method Performance for: – In-house developed method – Modification of a manufacturer’s procedure – Method not cleared by the FDA

Verification • A 1 -time process completed before the test is used for patient testing. • Requires determination of the test performance characteristics – ▪ Sensitivity ▪ Specificity ▪ Precision ▪ Accuracy ▪ Reportable range ▪ Reference range ▪ Other characteristics required for test performance From Cumitech 31; ASM press 1997

Verification Protocol • Purpose of test – e. g. screening for R GC • Write an SOP – pre to post-analytical testing • Procure specimens – – 20 Positive – 50 Negative • Set acceptability criteria • Statement of findings w/ regard to implementation – document!

Options • • Do a VERIFICATION of your NAAT Regionalize testing service Send specimens out to commercial lab Reimbursement – Medi. Cal will reimburse for testing multiple sites

Acknowledgements • SFDPH Diane Campbell, Leah Rauch & lab staff Charlotte Kent, Dr. Jeff Klausner & City Clinic clinicians • UCSF Julius Schachter & Jeanne Moncada • CA STD Control Drs. Gail Bolan & Chris Hall • CDC – Dr. John Papp • Manufacturers
0bc722e0d732f3ad90510b65904f5a54.ppt