851306ee2bc2260e310de34d530cb750.ppt
- Количество слайдов: 23

RECEPTA biopharma Partnering for cancer drug development: Why in Brazil? Jose Fernando Perez CEO 1

Vision To bring innovation in cancer drug development to Brazil in several stages of the value chain: – – – 2 m. Ab generation m. Ab development m. Ab production Pre-clinical Trials Phase I and Phase II Cancer Clinical Trials

Philosophy u Provide quality-of-life enhancing therapeutics to cancer patients in Brazil and worldwide, while offering a rewarding value proposition to our shareholders. u Build the necessary skills and resources to develop, produce and profitably market oncology products generated from our research and development activities, both in-house and through external collaborative strategic and commercial alliances. 3

Why in Brazil? : Leveraging of local advantages u u Excellent hospital facilities and qualified physicians with documented experience in the conduction of clinical trials u Lower costs of operation in Brazil than in traditional research regions for required personnel at all levels. u Excellent infrastructure of services: CROs & Logistics u 4 Highly qualified human resources: scientists and technicians: Brazil’s scientific output represents 3% of total world production Easier access to a diverse selection of informed patients and tumor samples for Phase I and II clinical trials as compared with the more traditional testing regions of the US, EU and Japan.

Why in Brazil? : Leveraging of local advantages u u The New Culture towards innovation: Priority in funding opportunities from several governmental sources (such as FINEP, BNDES, FAPESP) through R&D grants or low interest loans. Drug development is one the four stated priorities of the Brazilian industrial policy (PITCE) and biotechnology is also a priority as one the areas treated as carriers of the future. u 5 Engagement of required R&D talent through research fellowships and scholarships in public educational institutions at low cost. The Brazilian market needs new cancer drugs manufactured in the country to reduce imports

Partnership with the Ludwig Institute u u 6 The Ludwig Institute for Cancer Research (LICR) has licensed four antibodies to Recepta and is a major shareholder of the company. The agreement with the LICR creates conditions for an effective transfer of scientific and technological knowledge from the global network of scientists associated with the LICR to Recepta. The close collaboration with LICR’s scientists provides an extra validation of scientific and technological procedures. The presence of LICR as a shareholder offers Recepta an immediate international insertion.

Portfolio of Products – Antibody hu 3 S 193 – Anti-Lewis Y Successful IHC, Pre-Clinical and Phase I trials carried out by LICR as an Ovarian Cancer, with potential for ALL epithelial tumors. – Antibody MX-35 Successful IHC trials carried out for early Ovarian cancer by LICR and potential for other cancer remedies – Antibody hu 58 -1066 – Anti-Lewis B (Leb) Successful IHC trials carried out for Breast and Colorectal cancers by LICR and potential for other cancer remedies. – Antibody A-34 Successful IHC trials carried out for Gastric and Kidney cancers by LICR and potential for other cancer remedies. – Four new Antibodies in development Resulting from Recepta’s own R&D in Brazil 7

Virtue out of Necessity: An Unique Approach through Strategic Partnerships u u In contrast to other efforts, where entrepreneurs identify and sell existing research, Recepta starts from market needs and identifies academic resources inside universities and research centers for specific projects. u 8 Recepta’s novel R&D partnership model derives from thoroughly understanding academia potential and market needs, combined with the capacity to establish mutually beneficial partnerships. In other words, academia’s technological innovation is oriented to respond to market demand.
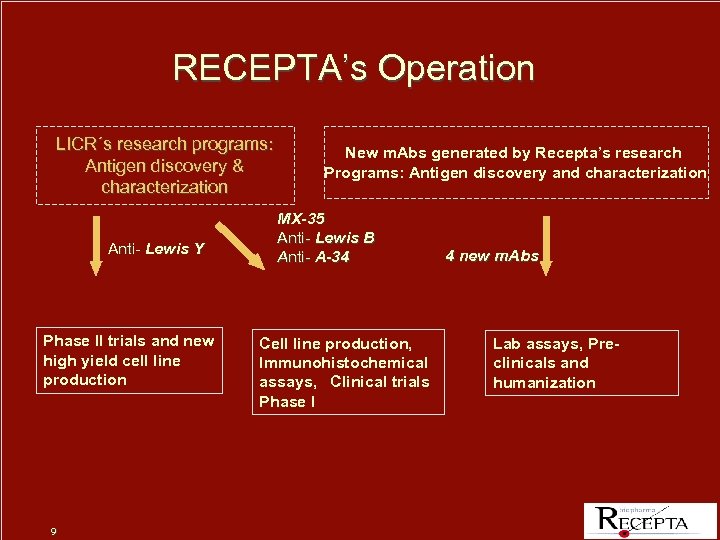
RECEPTA’s Operation LICR´s research programs: Antigen discovery & characterization Anti- Lewis Y Anti Phase II trials and new high yield cell line production 9 New m. Abs generated by Recepta’s research Programs: Antigen discovery and characterization MX-35 Anti- Lewis B Anti- A-34 Cell line production, Immunohistochemical assays, Clinical trials Phase I 4 new m. Abs Lab assays, Preclinicals and humanization

RECEPTA´s Operational Structure MAb R&D division Oncology division • • • Clinical Trials Management Immunohistochemistry Identification of new targets • • Generation of new antibodies High yield cell line construction • Preclinical Trials • Cell banks construction • Clinical trials Phase I (CRO, patients, hospital) • • GMP clinical grade large scale production (outsource) • Clinical trials Phase II CRO, patients, hospital) 10

Recepta´s Researchers and Project Support u Due to its aggressive partnership strategy, Recepta has already assembled an impressive group of qualified researchers: – – – 11 18 Ph. D 17 MSc 2 MD 6 graduates Total: 43
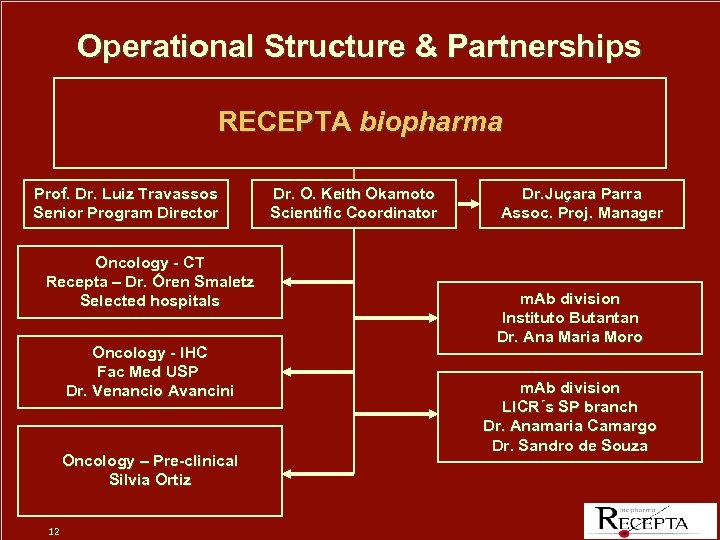
Operational Structure & Partnerships RECEPTA biopharma Prof. Dr. Luiz Travassos Senior Program Director Oncology - CT Recepta – Dr. Óren Smaletz Selected hospitals Oncology - IHC Fac Med USP Dr. Venancio Avancini Oncology – Pre-clinical Silvia Ortiz 12 Dr. O. Keith Okamoto Scientific Coordinator Dr. Juçara Parra Assoc. Proj. Manager m. Ab division Instituto Butantan Dr. Ana Maria Moro m. Ab division LICR´s SP branch Dr. Anamaria Camargo Dr. Sandro de Souza

Recepta´s Partner Institutions u Ludwig Institute for Cancer Research Global Local Branch – SP (new targets and m. Abs) u Instituto Butantan (Cell Biology) u Medical School of the University of São Paulo (Immunohistochemistry) u Hospitals (Clinical Trials): Sírio-Libanês, Alemão Oswaldo Cruz, Albert Einstein, National Institute of Cancer (INCA), Brazilian Institute for Cancer Control (IBCC), Baleia (BH) u MIT’s G-lab – Sloan School of Administration (valuation model) 13

RECEPTA´S m. Ab R&D Division: Partnership with Instituto Butantan u Instituto Butantan is a center of excellence for biomedical research maintained by the State of São Paulo Secretary of Health. Butantan conducts, both, basic and applied research in areas related to public health and is responsible for the production of more than 80% of the total amount of sera and vaccines used in Brazil u Recepta’s R&D project for financial support has been approved by FINEP to partially fund the installation and operation of a lab facility for the generation of high yield cell lines and cell banks. The researcher and project manager will be Dr. Ana Maria Moro, Ph. D, who works for Butantan and is one of Brazil’s most knowledgeable m. Ab experts. Dr. Ana Moro has worked as a consultant for Recepta from April to December 2005 during her license from Butantan. This circumstance places Dr. Moro in a privileged position to match the technological needs of the project with the challenges of the company. 14

RECEPTA´S m. Ab R&D Division: Partnership with LICR’s SP branch u u u 15 The São Paulo Branch of the Ludwig Institute for Cancer Research was established in 1983 at the Hospital do Cancer in São Paulo, Brazil. The Branch's research centers around furthering our understanding of human cancer genetics and the epidemiology of certain human cancers. The Branch has been integrally involved in several state-wide research initiatives, most notably the internationally acclaimed Human Cancer Genome Project, which linked many small facilities together to yield sequence data on genes expressed in cancer. Recepta has established a research and development partnership with the branch which includes: – identification of new targets, and; – generation of new monoclonal antibodies Two highly respected scientists, Dr. Anamaria Camargo, specialist in molecular genetics and Dr. Sandro de Souza, expert in bioinformatics will be leading the project.

RECEPTA´S Oncology Division u Immunopathology: Immunohistochemical assays u. A partnership agreement with the Medical School of the University of São Paulo was signed. u Recepta has provided the funding and the IHC research is currently being conducted by a team lead by Dr. Venancio Avancini Alves, Ph. D, MD and professor for pathology, with more than 30 yrs of experience in IHC. u Dr. Avancini’s team and facilities at the FMUSP has been site visited and approved by Dr. Achim Jungblüth, a renowned pathologist from MSKCC and LICR 16

RECEPTA´S Oncology Division Clinical Trials u A protocol design for an ovarian tumor Phase II Clinical Trial with Lewis Y has been prepared by Dr. Oren Smaletz in close collaboration with Dr. Alberto Wainstein from Biocancer, Dr. Eric Hoffman from the LICR New York Office and Dr. Andrew Scott from the LICR Melbourne Center. u Dr. Óren Smaletz, MD - oncology specialist with extensive experience in clinical trials at MSKCC currently working at the Einstein Hospital - has been hired to act as clinical coordinator for Recepta in clinical trials. u Recepta has identified several CROs and selected Euro. Trials Scientific Consultants for the first Lewis Y clinical trial. u New Lewis Y Phase II Clinical Trials are already being developed 17
18

RECEPTA´S Oncology Division Clinical Trials Hospitals u Recepta has established partnerships with hospitals known for their excellence in conducting clinical trials for its first Phase II clinical trial with anti-Lewis Y m. Ab. – Instituto de Ensino e Pesquisa do Hospital Sírio Libanês (SP) – – – u 19 Instituto de Ciências do Hospital Osvaldo Cruz (SP) Instituto de Pesquisa Albert Einstein (SP) National Institute of Cancer (INCA-RJ) Hospital da Baleia (BH) Brazilian Institute for Control of Cancer (IBCC-SP) This model will be replicated in the oncoming clinical trials

RECEPTA´S Funding Recepta has been awarded several grants under different programs funded by the federal and Sao Paulo state government. - Federal programs: FINEP: Program to fund partnership of Companies & Research Institutions FINEP: Subvention program - State Program: FAPESP: Program to fund partnership of Companies & Research Institutions 20

Funding Agencies for R&D in companies in Brazil Federal agencies: FINEP – the agency for innovation CNPq – National Research Council BNDES – National Bank for Economic and Social Development State Agency - São Paulo: FAPESP – The State of Sao Paulo Research Foundation 21

RECEPTA´S Funding u By using these federal and state programs to fund technological innovation Recepta’s investors account for an average of only 20% of the direct costs of R&D. u Recepta has been awarded a total of R$ 17. 2 million (US$ 10 million) grants from FINEP and FAPESP´s programs to fund parnerships between companies and research institutions. 22

Thank you! www. receptabiopharma. com 23
851306ee2bc2260e310de34d530cb750.ppt