 Распространенность химических элементов Илья Векслер лекция в ПГНИУ, 7 октября 2013
Распространенность химических элементов Илья Векслер лекция в ПГНИУ, 7 октября 2013
 7. 10. 2013, 9: 45 ауд. 623 н (8 корпус) «Распространенность химических элементов» 8. 10. 2013, 11: 30 ауд. 603 н (8 корпус) «Условия и продукты мантийного плавления» 9. 10. 2013, 11: 30 ауд. 632 н (8 корпус) «Расслоенные интрузии»
7. 10. 2013, 9: 45 ауд. 623 н (8 корпус) «Распространенность химических элементов» 8. 10. 2013, 11: 30 ауд. 603 н (8 корпус) «Условия и продукты мантийного плавления» 9. 10. 2013, 11: 30 ауд. 632 н (8 корпус) «Расслоенные интрузии»
 Michail Vasil‘evic LOMONOSOV 1711 - 1765 Student der Marburger Universität 1736 – 1739 «Везде исследуйте всечасно Что есть велико и прекрасно Чего еще не видел свет»
Michail Vasil‘evic LOMONOSOV 1711 - 1765 Student der Marburger Universität 1736 – 1739 «Везде исследуйте всечасно Что есть велико и прекрасно Чего еще не видел свет»
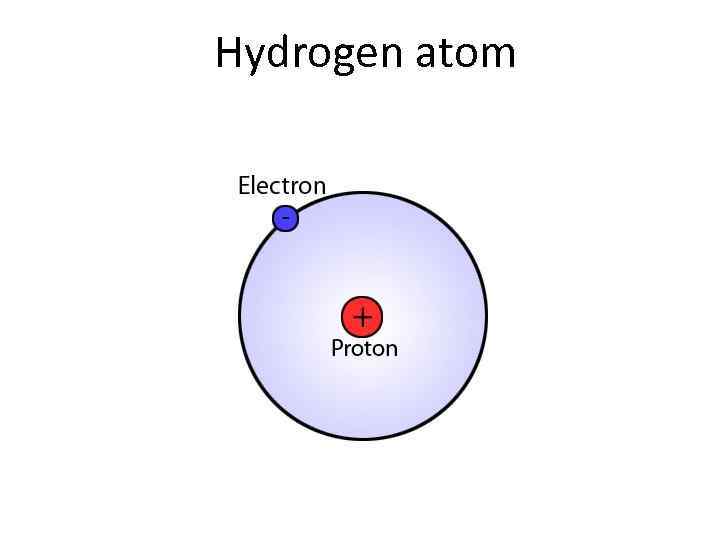 Hydrogen atom
Hydrogen atom
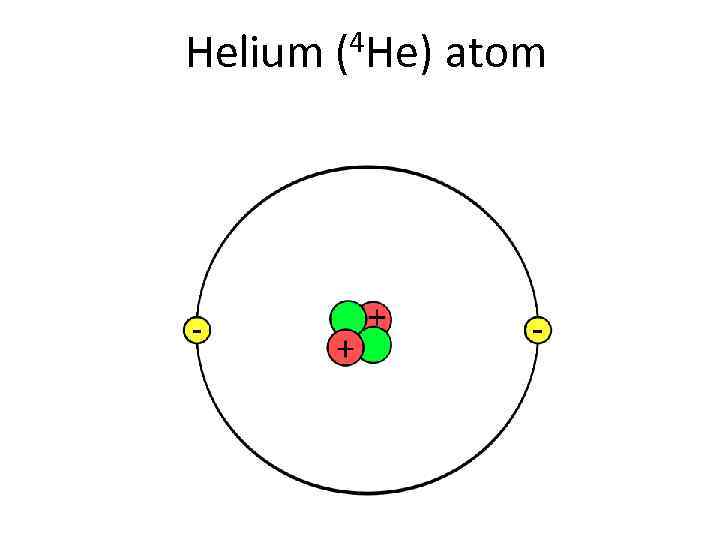 Helium (4 He) atom
Helium (4 He) atom
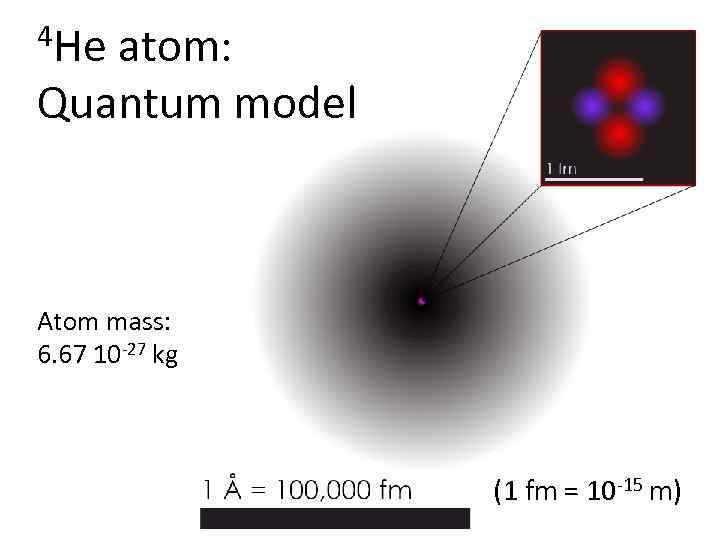 4 He atom: Quantum model Atom mass: 6. 67 10 -27 kg (1 fm = 10 -15 m)
4 He atom: Quantum model Atom mass: 6. 67 10 -27 kg (1 fm = 10 -15 m)
 How did H and He atoms (= 99 % of all the atoms in the Universe) form ?
How did H and He atoms (= 99 % of all the atoms in the Universe) form ?
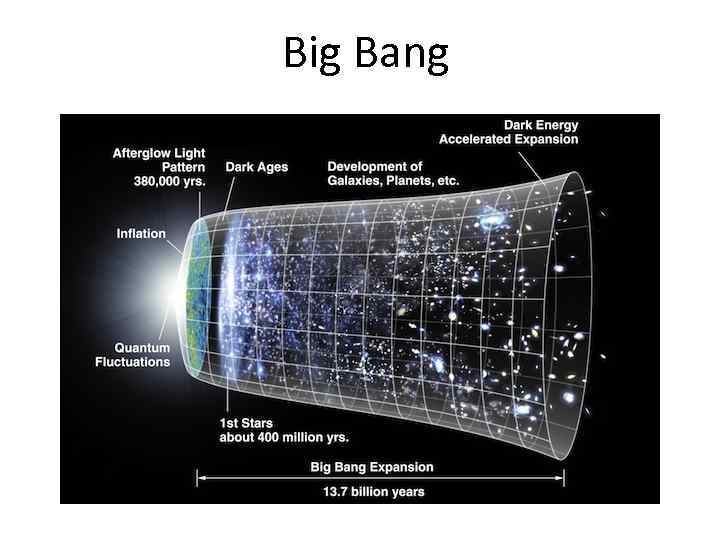 Big Bang
Big Bang
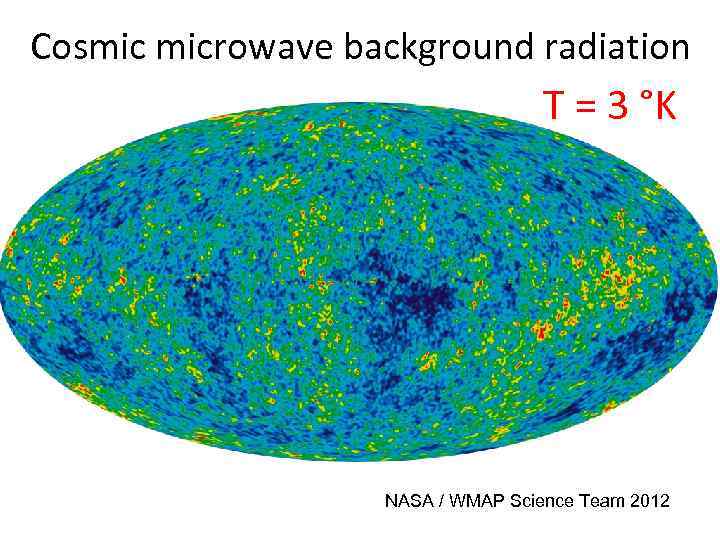 Cosmic microwave background radiation T = 3 °K NASA / WMAP Science Team 2012
Cosmic microwave background radiation T = 3 °K NASA / WMAP Science Team 2012
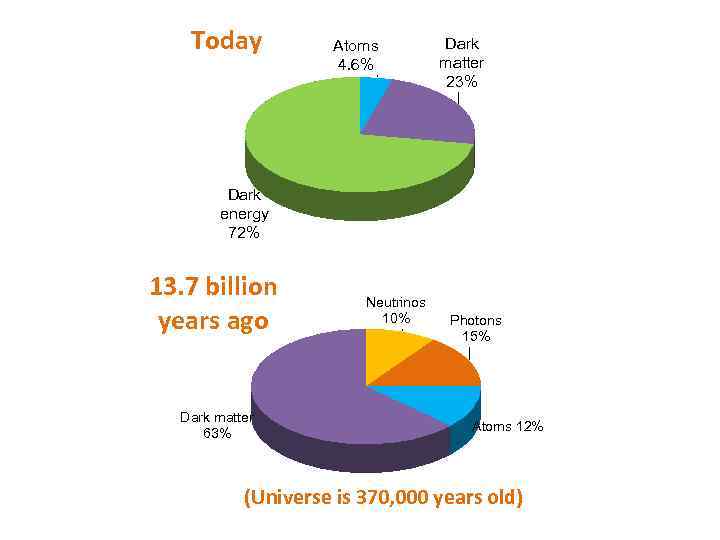 Today Atoms 4. 6% Dark matter 23% Dark energy 72% 13. 7 billion years ago Dark matter 63% Neutrinos 10% Photons 15% Atoms 12% (Universe is 370, 000 years old)
Today Atoms 4. 6% Dark matter 23% Dark energy 72% 13. 7 billion years ago Dark matter 63% Neutrinos 10% Photons 15% Atoms 12% (Universe is 370, 000 years old)
 “α-β-γ paper” Ralph Alpher 1921 - 2007 George Gamow 1904 - 1968
“α-β-γ paper” Ralph Alpher 1921 - 2007 George Gamow 1904 - 1968
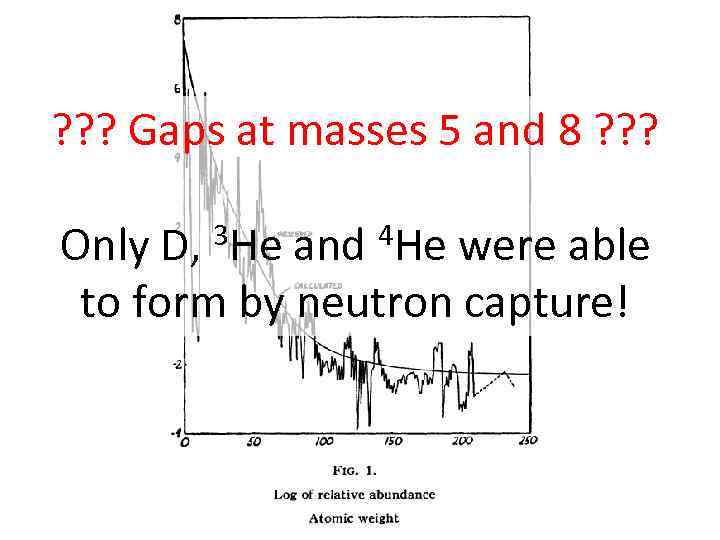 ? ? ? Gaps at masses 5 and 8 ? ? ? Only D, 3 He and 4 He were able to form by neutron capture!
? ? ? Gaps at masses 5 and 8 ? ? ? Only D, 3 He and 4 He were able to form by neutron capture!
 Where do the heavy elements form ?
Where do the heavy elements form ?
 Fred Hoyle 1915 - 2001
Fred Hoyle 1915 - 2001
 Our star - the Sun
Our star - the Sun
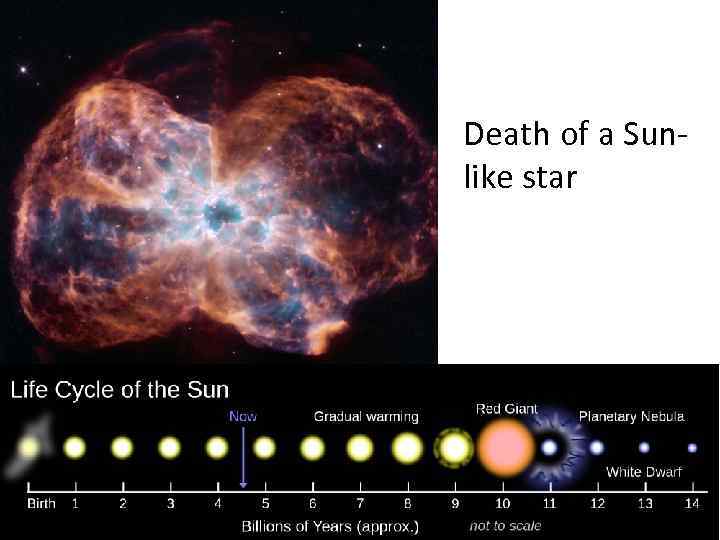 Death of a Sunlike star
Death of a Sunlike star
 Supernova 1987 A
Supernova 1987 A
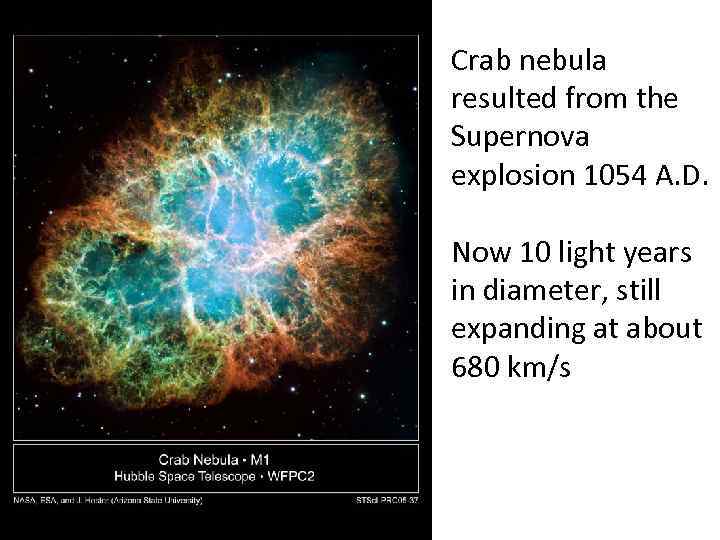 Crab nebula resulted from the Supernova explosion 1054 A. D. Now 10 light years in diameter, still expanding at about 680 km/s
Crab nebula resulted from the Supernova explosion 1054 A. D. Now 10 light years in diameter, still expanding at about 680 km/s
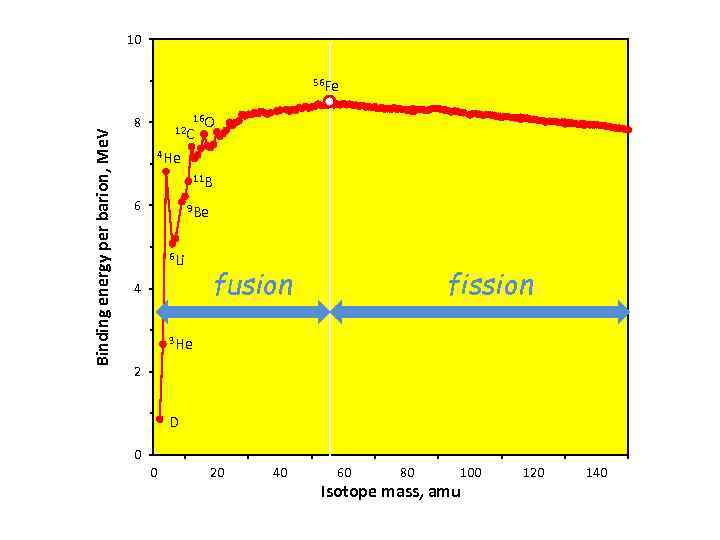 10 Binding energy per barion, Me. V 56 Fe 16 O 12 C 8 4 He 11 B 6 9 Be 6 Li 4 fusion fission 3 He 2 D 0 0 20 40 60 80 100 Isotope mass, amu 120 140
10 Binding energy per barion, Me. V 56 Fe 16 O 12 C 8 4 He 11 B 6 9 Be 6 Li 4 fusion fission 3 He 2 D 0 0 20 40 60 80 100 Isotope mass, amu 120 140
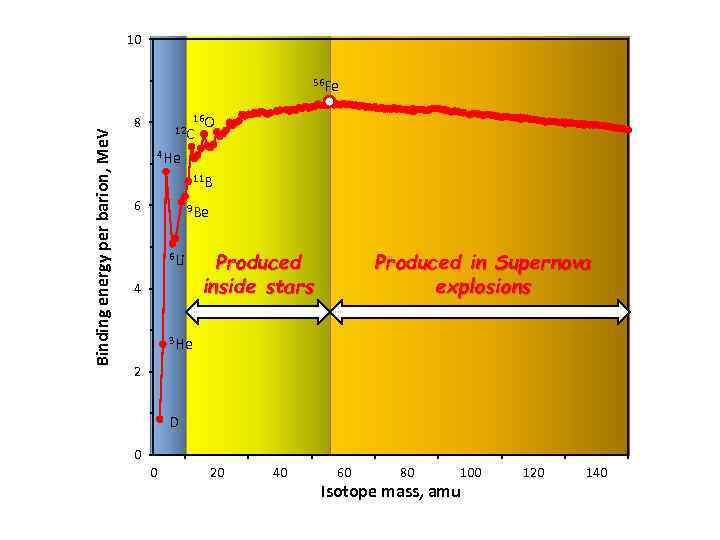 10 Binding energy per barion, Me. V 56 Fe 16 O 12 C 8 4 He 11 B 6 9 Be 6 Li 4 Produced inside stars Produced in Supernova explosions 3 He 2 D 0 0 20 40 60 80 100 Isotope mass, amu 120 140
10 Binding energy per barion, Me. V 56 Fe 16 O 12 C 8 4 He 11 B 6 9 Be 6 Li 4 Produced inside stars Produced in Supernova explosions 3 He 2 D 0 0 20 40 60 80 100 Isotope mass, amu 120 140
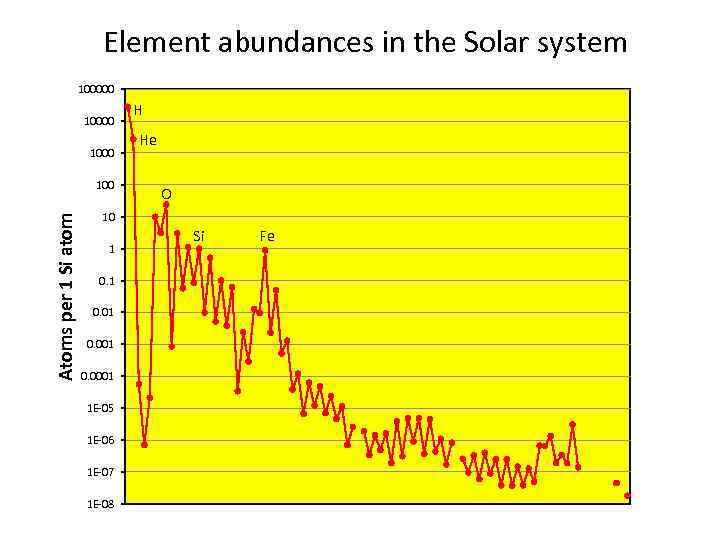 Element abundances in the Solar system 100000 1000 Atoms per 1 Si atom 100 H He O 10 1 0. 01 0. 0001 1 E-05 1 E-06 1 E-07 1 E-08 Si Fe
Element abundances in the Solar system 100000 1000 Atoms per 1 Si atom 100 H He O 10 1 0. 01 0. 0001 1 E-05 1 E-06 1 E-07 1 E-08 Si Fe
 Condensation: from gas to dust
Condensation: from gas to dust
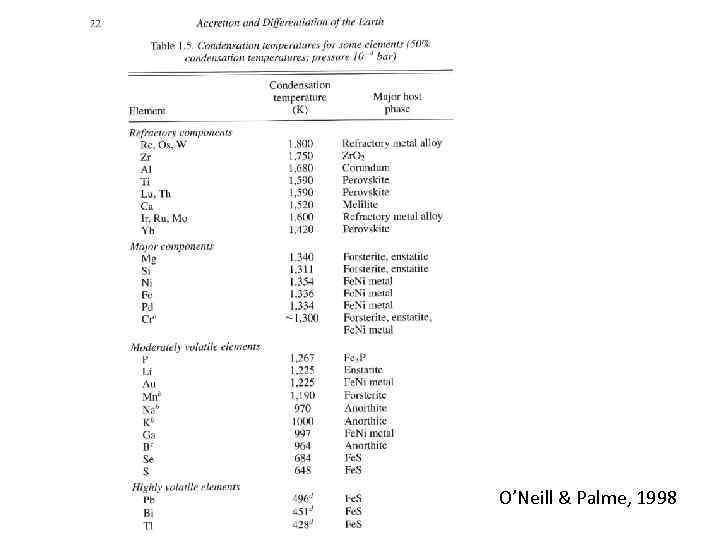 O’Neill & Palme, 1998
O’Neill & Palme, 1998
 Allende meteorite (2 Feb 1969) 1 cm 3
Allende meteorite (2 Feb 1969) 1 cm 3
 Allende meteorite (2 Feb 1969) CAI 1 cm 3
Allende meteorite (2 Feb 1969) CAI 1 cm 3
 Accretion: from dust and gas to stars and planets
Accretion: from dust and gas to stars and planets
 The Solar system
The Solar system
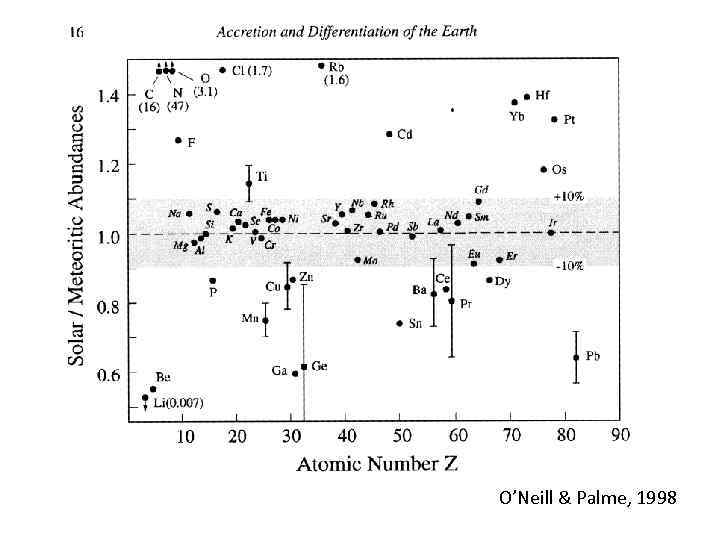 O’Neill & Palme, 1998
O’Neill & Palme, 1998
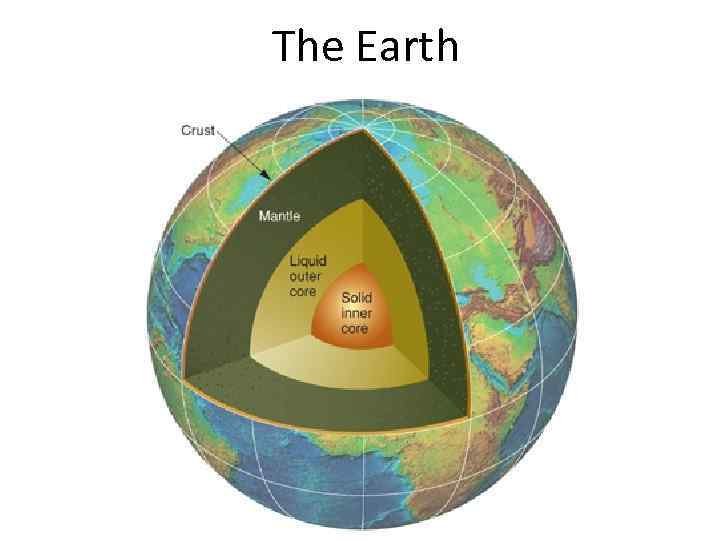 The Earth
The Earth
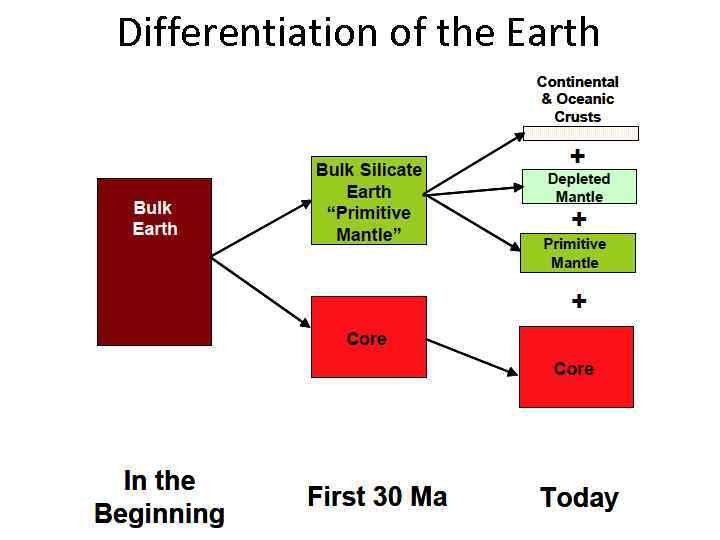 Differentiation of the Earth
Differentiation of the Earth
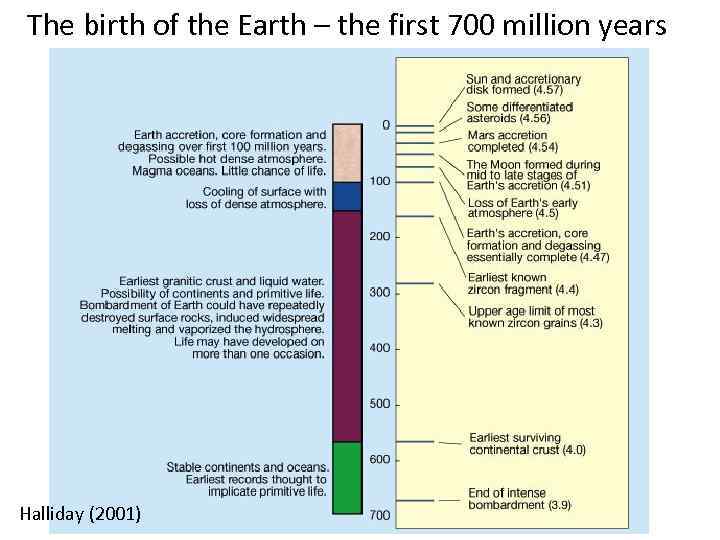 The birth of the Earth – the first 700 million years Halliday (2001)
The birth of the Earth – the first 700 million years Halliday (2001)
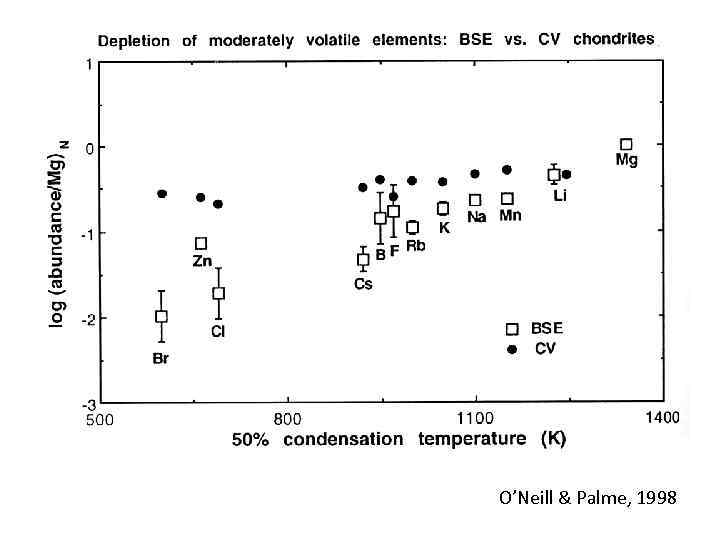 O’Neill & Palme, 1998
O’Neill & Palme, 1998
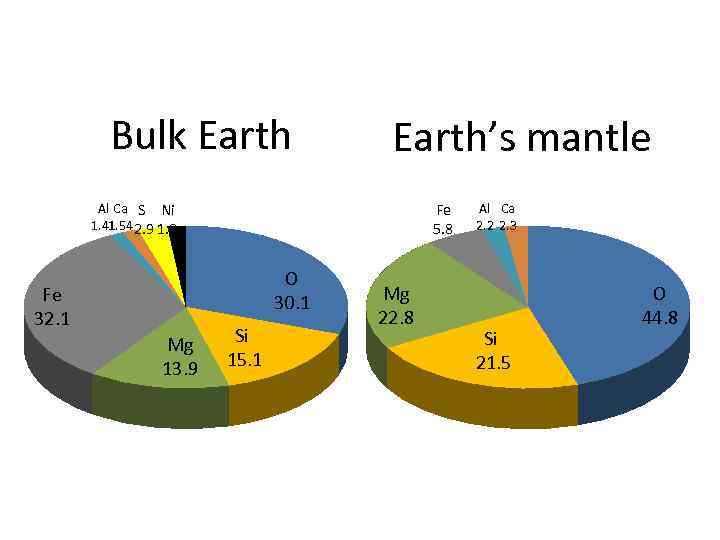 Bulk Earth’s mantle Al Ca S Ni 1. 41 2. 9 1. 8 1. 54 Fe 5. 8 O 30. 1 Fe 32. 1 Mg 13. 9 Si 15. 1 Mg 22. 8 Al Ca 2. 2 2. 3 Si 21. 5 O 44. 8
Bulk Earth’s mantle Al Ca S Ni 1. 41 2. 9 1. 8 1. 54 Fe 5. 8 O 30. 1 Fe 32. 1 Mg 13. 9 Si 15. 1 Mg 22. 8 Al Ca 2. 2 2. 3 Si 21. 5 O 44. 8
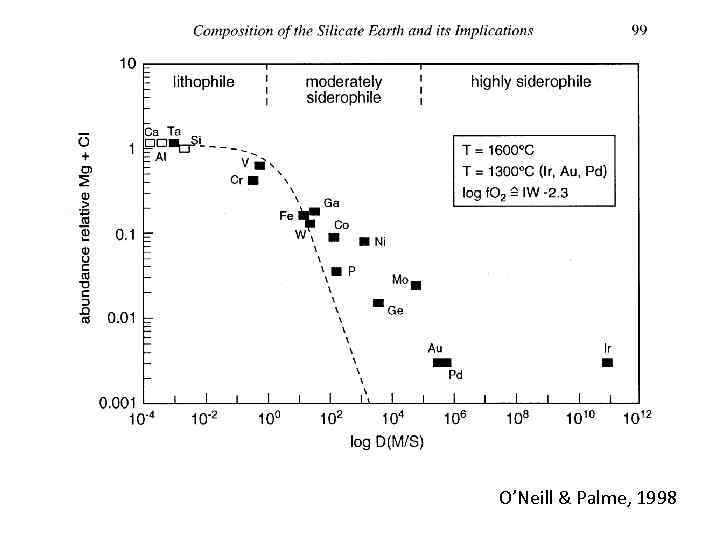 O’Neill & Palme, 1998
O’Neill & Palme, 1998
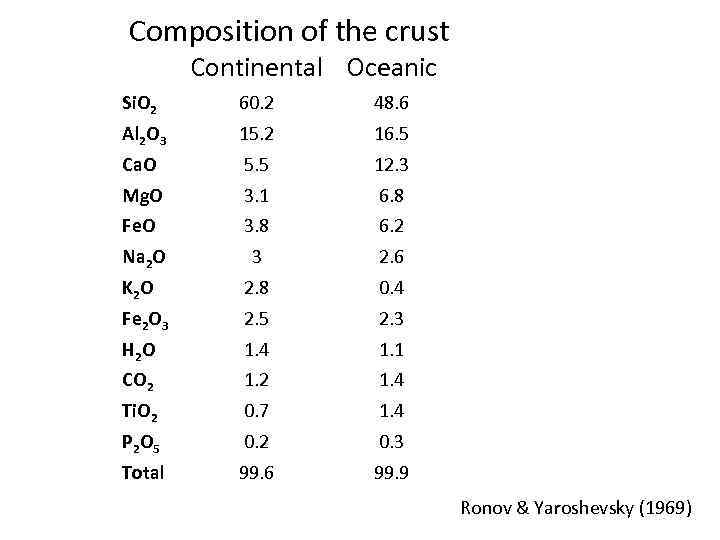 Composition of the crust Continental Oceanic Si. O 2 60. 2 48. 6 Al 2 O 3 15. 2 16. 5 Ca. O 5. 5 12. 3 Mg. O 3. 1 6. 8 Fe. O 3. 8 6. 2 Na 2 O 3 2. 6 K 2 O 2. 8 0. 4 Fe 2 O 3 2. 5 2. 3 H 2 O 1. 4 1. 1 CO 2 1. 4 Ti. O 2 0. 7 1. 4 P 2 O 5 0. 2 0. 3 Total 99. 6 99. 9 Ronov & Yaroshevsky (1969)
Composition of the crust Continental Oceanic Si. O 2 60. 2 48. 6 Al 2 O 3 15. 2 16. 5 Ca. O 5. 5 12. 3 Mg. O 3. 1 6. 8 Fe. O 3. 8 6. 2 Na 2 O 3 2. 6 K 2 O 2. 8 0. 4 Fe 2 O 3 2. 5 2. 3 H 2 O 1. 4 1. 1 CO 2 1. 4 Ti. O 2 0. 7 1. 4 P 2 O 5 0. 2 0. 3 Total 99. 6 99. 9 Ronov & Yaroshevsky (1969)
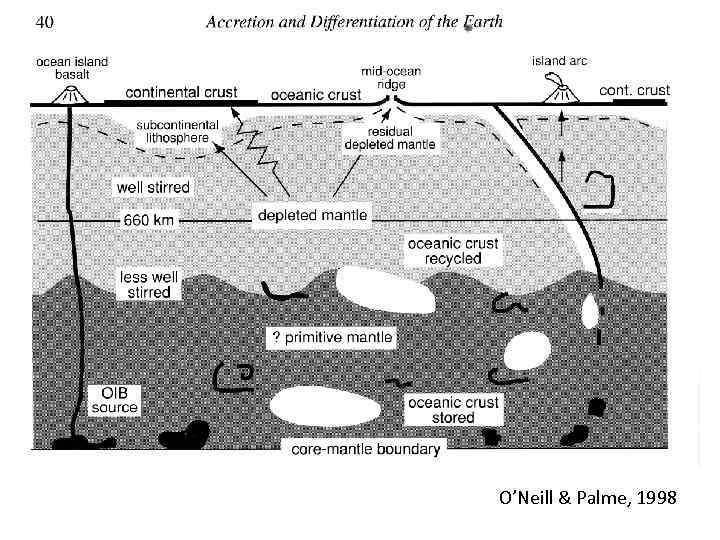 O’Neill & Palme, 1998
O’Neill & Palme, 1998