700b8302085ebdc4396755cb3fb15793.ppt
- Количество слайдов: 42
 Quality Initiatives SCAI Quality Symposium 24 th Great Wall International Congress of Cardiology Asia Pacific Heart Congress 2013 October 12, 2013 Beijing, China Charles E. Chambers, MD, FSCAI President Elect, Society for Cardiovascular Angiography & Interventions Chairman, SCAI Laboratory Survey Committee Professor of Medicine and Radiology Pennsylvania State University College of Medicine Director Cardiac Catheterization Laboratories, Hershey Medical Center, PA
Quality Initiatives SCAI Quality Symposium 24 th Great Wall International Congress of Cardiology Asia Pacific Heart Congress 2013 October 12, 2013 Beijing, China Charles E. Chambers, MD, FSCAI President Elect, Society for Cardiovascular Angiography & Interventions Chairman, SCAI Laboratory Survey Committee Professor of Medicine and Radiology Pennsylvania State University College of Medicine Director Cardiac Catheterization Laboratories, Hershey Medical Center, PA
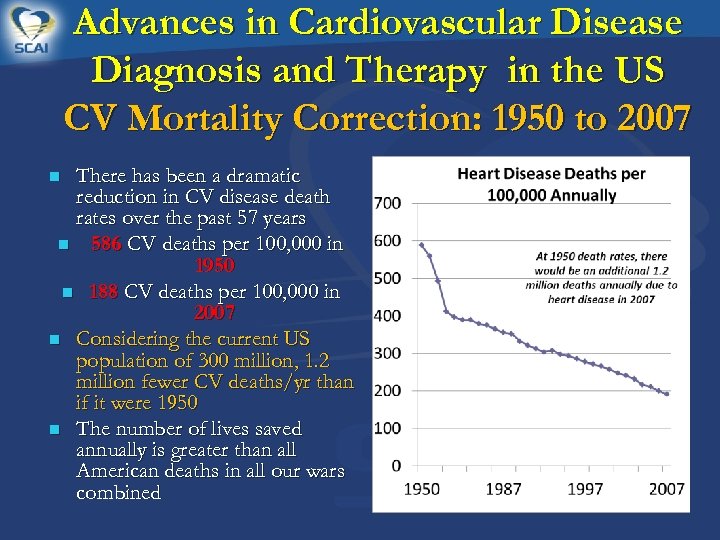 Advances in Cardiovascular Disease Diagnosis and Therapy in the US CV Mortality Correction: 1950 to 2007 There has been a dramatic reduction in CV disease death rates over the past 57 years n 586 CV deaths per 100, 000 in 1950 n 188 CV deaths per 100, 000 in 2007 n Considering the current US population of 300 million, 1. 2 million fewer CV deaths/yr than if it were 1950 n The number of lives saved annually is greater than all American deaths in all our wars combined n
Advances in Cardiovascular Disease Diagnosis and Therapy in the US CV Mortality Correction: 1950 to 2007 There has been a dramatic reduction in CV disease death rates over the past 57 years n 586 CV deaths per 100, 000 in 1950 n 188 CV deaths per 100, 000 in 2007 n Considering the current US population of 300 million, 1. 2 million fewer CV deaths/yr than if it were 1950 n The number of lives saved annually is greater than all American deaths in all our wars combined n
 Society for Cardiovascular Angiography and Intervention Mission Statement SCAI promotes excellence in invasive and interventional cardiovascular medicine through physician education and representation, and the advancement of quality standards to enhance patient care.
Society for Cardiovascular Angiography and Intervention Mission Statement SCAI promotes excellence in invasive and interventional cardiovascular medicine through physician education and representation, and the advancement of quality standards to enhance patient care.
 SCAI Publications 1978 -1998
SCAI Publications 1978 -1998
 …and the work continues
…and the work continues
 A Quality Improvement Program for all Cath Laboratories Recommendation COR LOE I C Every PCI program must have a quality improvement program that routinely: a) reviews ACC/SCAI 2011 PCI Guidelines Participation by every PCI program in a regional or national Update C I quality and outcomes of the entire program; b) reviews results of individual operators; c) includes risk adjustment; d) provides peer review of difficult or complicated cases, and; e) performs random case reviews PCI registry for the purpose of benchmarking its outcomes against current national norms Participation by all physicians that perform PCI in the American Board of Internal Medicine interventional cardiology board certification and maintenance of certification program IIa C
A Quality Improvement Program for all Cath Laboratories Recommendation COR LOE I C Every PCI program must have a quality improvement program that routinely: a) reviews ACC/SCAI 2011 PCI Guidelines Participation by every PCI program in a regional or national Update C I quality and outcomes of the entire program; b) reviews results of individual operators; c) includes risk adjustment; d) provides peer review of difficult or complicated cases, and; e) performs random case reviews PCI registry for the purpose of benchmarking its outcomes against current national norms Participation by all physicians that perform PCI in the American Board of Internal Medicine interventional cardiology board certification and maintenance of certification program IIa C
 How to Implement a Cath Lab Quality Program STEP 1: SCAI position paper on cath lab quality cited below STEP 2: Assemble Cath Lab QI Committee STEP 3: Determine Which Quality Measures to Follow STEP 4: Identify a database or method to capture data STEP 5: Develop Plan to Capture Data STEP 6: Analyze Data STEP 7: Using Benchmark Comparisons, Identify Quality Concerns STEP 8: Implement Plan. Do-Check-Act Cycle STEP 1: SCAI position paper on cath lab quality (www. scai. org, under header bar “guidelines and quality” select “guidelines”, access link to article under “ 2011, May”)
How to Implement a Cath Lab Quality Program STEP 1: SCAI position paper on cath lab quality cited below STEP 2: Assemble Cath Lab QI Committee STEP 3: Determine Which Quality Measures to Follow STEP 4: Identify a database or method to capture data STEP 5: Develop Plan to Capture Data STEP 6: Analyze Data STEP 7: Using Benchmark Comparisons, Identify Quality Concerns STEP 8: Implement Plan. Do-Check-Act Cycle STEP 1: SCAI position paper on cath lab quality (www. scai. org, under header bar “guidelines and quality” select “guidelines”, access link to article under “ 2011, May”)
 STEP 2: Assemble Cath Lab QI Committee a. Chairperson: a physician trusted by all e. g. , Director of Cath Lab or Interventional the Physician Champion b. Lab Staff Champion (Staff QA Coordinator) c. Physician Support (Invasive cardiologists) d. Physician Extenders e. Laboratory Support Staff Cath Lab technical director or chief technologist f. Cath Lab/Recovery Area g. Cath Lab Administrator/Hospital Administrator h. Consider cardiac surgeons, other cardiologists, internists, ER physicians & other representative from hospital QA department, and IT support. Team work is essential
STEP 2: Assemble Cath Lab QI Committee a. Chairperson: a physician trusted by all e. g. , Director of Cath Lab or Interventional the Physician Champion b. Lab Staff Champion (Staff QA Coordinator) c. Physician Support (Invasive cardiologists) d. Physician Extenders e. Laboratory Support Staff Cath Lab technical director or chief technologist f. Cath Lab/Recovery Area g. Cath Lab Administrator/Hospital Administrator h. Consider cardiac surgeons, other cardiologists, internists, ER physicians & other representative from hospital QA department, and IT support. Team work is essential
 Responsibilities of the Cath Lab Quality Committee 1. Regular Meetings (monthly) 2. Identify metrics of care to be monitored (from NCDR reports) 3. Review all serious adverse events (e. g. , death, emergency CABG) 4. Perform random film audits (e. g. , 1 case per MD for appropriateness, adequate imaging, outcome) 5. Review data on process and outcome metrics. 6. Identify quality issues. (e. g. , any complication with frequency > 90 th ile of peer hospitals). (e. g. , any physician with outlier incidence of complications) 7. Develop remediation plans, oversee implementation, check results. (i. e. , plan/do/check/act cycle) 8. Refer larger issues for appropriate intervention. (e. g. , disruptive physician behavior referred to department director) Bashore TM, Balter S, Barac A, Byrne JG, Cavendish JJ, Chambers CE, et al. : American College of Cardiology Foundation/Society for Cardiovascular Angiography and Interventions Clinical Expert Consensus Document on Cardiac Catheterization Laboratory Standards. Update 2012. J. Am. Coll. Cardiol. May 8, 2012 as doi: 10. 1016/j. jacc. 2012. 010.
Responsibilities of the Cath Lab Quality Committee 1. Regular Meetings (monthly) 2. Identify metrics of care to be monitored (from NCDR reports) 3. Review all serious adverse events (e. g. , death, emergency CABG) 4. Perform random film audits (e. g. , 1 case per MD for appropriateness, adequate imaging, outcome) 5. Review data on process and outcome metrics. 6. Identify quality issues. (e. g. , any complication with frequency > 90 th ile of peer hospitals). (e. g. , any physician with outlier incidence of complications) 7. Develop remediation plans, oversee implementation, check results. (i. e. , plan/do/check/act cycle) 8. Refer larger issues for appropriate intervention. (e. g. , disruptive physician behavior referred to department director) Bashore TM, Balter S, Barac A, Byrne JG, Cavendish JJ, Chambers CE, et al. : American College of Cardiology Foundation/Society for Cardiovascular Angiography and Interventions Clinical Expert Consensus Document on Cardiac Catheterization Laboratory Standards. Update 2012. J. Am. Coll. Cardiol. May 8, 2012 as doi: 10. 1016/j. jacc. 2012. 010.
 STEP 3: Determine Which Quality Measures to Follow n Structural Domain n Hospital and cath lab structure, Credentialing , Educational Efforts n Process Domain n Monitoring patient , System related, Guidelines related, Cost/Utilization n Outcomes Domain n Monitoring of outcomes on a regular basis including risk adjusted mortality, procedure related LOS, fluoro time, etc. , complications (30 days) with data sharing and reporting.
STEP 3: Determine Which Quality Measures to Follow n Structural Domain n Hospital and cath lab structure, Credentialing , Educational Efforts n Process Domain n Monitoring patient , System related, Guidelines related, Cost/Utilization n Outcomes Domain n Monitoring of outcomes on a regular basis including risk adjusted mortality, procedure related LOS, fluoro time, etc. , complications (30 days) with data sharing and reporting.
 STEP 4: Identify a Database or Method to Capture Data In the US, NCDR’s Cath/PCI Data Registry is easiest solution: 1. 85% of cath labs use cath PCI Registry 2. Quarterly reports summarize institution’s data 3. Results are compared to all hospitals 4. Trends are evident over time 5. Problem areas are easily identified (e. g. , >90 th %’tile complication rate) 6. Mortality data is risk adjusted
STEP 4: Identify a Database or Method to Capture Data In the US, NCDR’s Cath/PCI Data Registry is easiest solution: 1. 85% of cath labs use cath PCI Registry 2. Quarterly reports summarize institution’s data 3. Results are compared to all hospitals 4. Trends are evident over time 5. Problem areas are easily identified (e. g. , >90 th %’tile complication rate) 6. Mortality data is risk adjusted
 STEP 5: Develop Plan to Capture Data 1. Requires dedicated personnel (e. g. , nurses) a. Data from untrained personnel is unreliable (e. g. , floor nurses) 2. Docs must provide some technical data (e. g. , coronary anatomy details) 3. Docs can’t be relied on for all data (they won’t do it all) 4. Auto-population of database from in-lab system is ideal 5. Identifying post-PCI complications is most difficult part a. Confirm complications with interventionist before entry into database. 6. Work with other hospital departments to obtain data a. Health Physics/Rad Safety can provide data on radiation exposure b. Hospital QI committee can provide data on readmissions w/in 30 days
STEP 5: Develop Plan to Capture Data 1. Requires dedicated personnel (e. g. , nurses) a. Data from untrained personnel is unreliable (e. g. , floor nurses) 2. Docs must provide some technical data (e. g. , coronary anatomy details) 3. Docs can’t be relied on for all data (they won’t do it all) 4. Auto-population of database from in-lab system is ideal 5. Identifying post-PCI complications is most difficult part a. Confirm complications with interventionist before entry into database. 6. Work with other hospital departments to obtain data a. Health Physics/Rad Safety can provide data on radiation exposure b. Hospital QI committee can provide data on readmissions w/in 30 days
 STEP 6: Analyze Data For Individual Adverse Events 1. Review data, records, cine images, etc. a. Benchmark—“something that serves as a standard by which others may be measured or judged 2. QI Committee evaluates quality of care surrounding the adverse event a. relationship of event to procedure: direct, indirect, not related b. relationship of event to sub-optimal care: direct, indirect, not related c. quality of care: suboptimal, any opportunity for improvement Klein LW et al. Quality assessment and improvement in interventional cardiology: a position statement of the Society for Cardiac Angiography and Interventions. Cathet Cardiovasc Intervent 2011; 77: 927 -935
STEP 6: Analyze Data For Individual Adverse Events 1. Review data, records, cine images, etc. a. Benchmark—“something that serves as a standard by which others may be measured or judged 2. QI Committee evaluates quality of care surrounding the adverse event a. relationship of event to procedure: direct, indirect, not related b. relationship of event to sub-optimal care: direct, indirect, not related c. quality of care: suboptimal, any opportunity for improvement Klein LW et al. Quality assessment and improvement in interventional cardiology: a position statement of the Society for Cardiac Angiography and Interventions. Cathet Cardiovasc Intervent 2011; 77: 927 -935
 STEP 7: Using Benchmark Comparisons, Identify Quality Concerns
STEP 7: Using Benchmark Comparisons, Identify Quality Concerns
 STEP 8: Implement Plan-Do. Check-Act Cycle: Close the Loop 1. 2. 3. 4. Develop strategy to address problem Implement strategy Measure results If not satisfactory, repeat cycle Example for US: D 2 B is > 90 minutes routinely --- Plan: Evaluate opportunities for improvement --- Do: Implement pre-hospital EKG-based cath lab activation --- Check: Measure D 2 B after new system implemented --- Act: If D 2 B still > 90 minutes, repeat the cycle. Klein LW et al. Quality assessment and improvement in interventional cardiology: a position statement of the Society for Cardiac Angiography and Interventions. Cathet Cardiovasc Intervent 2011; 77: 927 -935
STEP 8: Implement Plan-Do. Check-Act Cycle: Close the Loop 1. 2. 3. 4. Develop strategy to address problem Implement strategy Measure results If not satisfactory, repeat cycle Example for US: D 2 B is > 90 minutes routinely --- Plan: Evaluate opportunities for improvement --- Do: Implement pre-hospital EKG-based cath lab activation --- Check: Measure D 2 B after new system implemented --- Act: If D 2 B still > 90 minutes, repeat the cycle. Klein LW et al. Quality assessment and improvement in interventional cardiology: a position statement of the Society for Cardiac Angiography and Interventions. Cathet Cardiovasc Intervent 2011; 77: 927 -935
 Now, How Can This Functioning Quality Assurance/ Quality Improvement Program Be Effective? n QA/QI Components n n n Clinical Proficiency Review Peer Review Equipment Performance ■ ■ QA/CQI Committee Data collection process ■ ■ ■ Direct patient-care related indicators System-specific indicators Guidelines-driven indicators Cost-related indicators Outcome-related indicators ■ Physical/Service ■ SCAI QI Toolkit
Now, How Can This Functioning Quality Assurance/ Quality Improvement Program Be Effective? n QA/QI Components n n n Clinical Proficiency Review Peer Review Equipment Performance ■ ■ QA/CQI Committee Data collection process ■ ■ ■ Direct patient-care related indicators System-specific indicators Guidelines-driven indicators Cost-related indicators Outcome-related indicators ■ Physical/Service ■ SCAI QI Toolkit
 The Society for Cardiovascular Angiography and Intervention Quality Improvement Toolkit (SCAI-QIT) There is Help!
The Society for Cardiovascular Angiography and Intervention Quality Improvement Toolkit (SCAI-QIT) There is Help!
 SCAI QIT Outline n Defining Quality in the Cath Lab n Operator Requirements n Staff Requirements n Procedural Quality n Benchmarking n Key conferences n Cath Lab Best Practices n Facility and Environmental Issues
SCAI QIT Outline n Defining Quality in the Cath Lab n Operator Requirements n Staff Requirements n Procedural Quality n Benchmarking n Key conferences n Cath Lab Best Practices n Facility and Environmental Issues
 Defining Quality in the Cath Lab n Structural Domain n Hospital/Cath lab structure, Credentialing , Education Efforts n Process Domain n Monitoring pt. , System/Guidelines related, Cost/Utilization n Outcomes Domain n Monitoring of outcomes on a regular basis n Risk adjusted mortality, procedure related LOS, fluoro time, etc. , complications (30 days) with data sharing and reporting
Defining Quality in the Cath Lab n Structural Domain n Hospital/Cath lab structure, Credentialing , Education Efforts n Process Domain n Monitoring pt. , System/Guidelines related, Cost/Utilization n Outcomes Domain n Monitoring of outcomes on a regular basis n Risk adjusted mortality, procedure related LOS, fluoro time, etc. , complications (30 days) with data sharing and reporting
 Operator and Staff Requirements n n ACLS certification should be completed yearly. All staff should have one of the following: n n n Nursing RN license. Radiation Technologist certification. Cardiovascular technologist professional training certificate.
Operator and Staff Requirements n n ACLS certification should be completed yearly. All staff should have one of the following: n n n Nursing RN license. Radiation Technologist certification. Cardiovascular technologist professional training certificate.
 Procedural Quality Benchmark—“something that serves as a standard by which others may be measured or judged” n Using external benchmarks allows you to see how your cath lab performs relative to: n Absolute standards, for example, n Joint Commission Sentinel Events: n Wrong patient; wrong body part n Fluoroscopy dose >1, 500 rads to a single field n Other cath labs in your region, nation, and worldwide n
Procedural Quality Benchmark—“something that serves as a standard by which others may be measured or judged” n Using external benchmarks allows you to see how your cath lab performs relative to: n Absolute standards, for example, n Joint Commission Sentinel Events: n Wrong patient; wrong body part n Fluoroscopy dose >1, 500 rads to a single field n Other cath labs in your region, nation, and worldwide n
 n Pre-procedure n n Informed Consent Sedation, Anesthesia and Analgesia Evaluation Procedure n Patient Preparation in Procedure Room n Sedation, Anesthesia Administration and Documentation n Optimal Catheterization Laboratory Team Post Procedure n n n Physician to Patient Communication Access Site Management Monitoring and Length of Stay Cath Lab Best Practices
n Pre-procedure n n Informed Consent Sedation, Anesthesia and Analgesia Evaluation Procedure n Patient Preparation in Procedure Room n Sedation, Anesthesia Administration and Documentation n Optimal Catheterization Laboratory Team Post Procedure n n n Physician to Patient Communication Access Site Management Monitoring and Length of Stay Cath Lab Best Practices
 QA and Cath Lab Conferences n n n Invasive Cardiology Morbidity and Mortality (Cath Lab M&M) n Separate from clinical cardiology M&M n Open review and assessment of cath lab complications and inhospital events following invasive cardiovascular procedures Invasive Case Review Conference (Angio Review) n Open review of random sample of cases n Diagnostic and interventional cases Catheterization Laboratory Educational Conference (Cath Conf) n Regular, frequent, formal educational events n Focus on cath lab practice and issues 1 http: //www. jointcommission. org/standards_information/jcfaqdetails. aspx? Standards. Faq. Id=311&Program. Id=1; accessed February 28, 2011 2 http: //www. acgme. org/outcome/implement/complete_PBLIBooklet. pdf; accessed March 1, 2011
QA and Cath Lab Conferences n n n Invasive Cardiology Morbidity and Mortality (Cath Lab M&M) n Separate from clinical cardiology M&M n Open review and assessment of cath lab complications and inhospital events following invasive cardiovascular procedures Invasive Case Review Conference (Angio Review) n Open review of random sample of cases n Diagnostic and interventional cases Catheterization Laboratory Educational Conference (Cath Conf) n Regular, frequent, formal educational events n Focus on cath lab practice and issues 1 http: //www. jointcommission. org/standards_information/jcfaqdetails. aspx? Standards. Faq. Id=311&Program. Id=1; accessed February 28, 2011 2 http: //www. acgme. org/outcome/implement/complete_PBLIBooklet. pdf; accessed March 1, 2011
 Facility and Environmental Issues n n n Infection control Radiation safety Operator and staff health – ergonomics (back pain, neck pain, etc. ) Information Storage and Inventory Equipment maintenance Best Worst
Facility and Environmental Issues n n n Infection control Radiation safety Operator and staff health – ergonomics (back pain, neck pain, etc. ) Information Storage and Inventory Equipment maintenance Best Worst
 n Core Measure Information n Accountability Measures Evolution of Performance Measurement at The Joint Commission Key Historical Activities Future Goals and Objectives Ongoing Activities n Specifications Manual for National Hospital Inpatient Quality Measures Specifications Manual for Joint Commission National Quality Core Measures However, at the end of the day… n n Someone is always watching. n n n Performance Measurement Initiatives n n n Development Initiatives Core Measure Sets Disease-Specific Care Certification Measures Staffing Certification Measures Library of Other Measures n Performance Measurement Systems n n n Performance Measurement System Information Core Systems List Core e. Measure Pilot Project Systems List Non-Core Systems List Candidate Systems List Facts About ORYX Performance Measurement Systems
n Core Measure Information n Accountability Measures Evolution of Performance Measurement at The Joint Commission Key Historical Activities Future Goals and Objectives Ongoing Activities n Specifications Manual for National Hospital Inpatient Quality Measures Specifications Manual for Joint Commission National Quality Core Measures However, at the end of the day… n n Someone is always watching. n n n Performance Measurement Initiatives n n n Development Initiatives Core Measure Sets Disease-Specific Care Certification Measures Staffing Certification Measures Library of Other Measures n Performance Measurement Systems n n n Performance Measurement System Information Core Systems List Core e. Measure Pilot Project Systems List Non-Core Systems List Candidate Systems List Facts About ORYX Performance Measurement Systems
 doing the Haka War Dance
doing the Haka War Dance
 Public Reporting
Public Reporting
 Peer Review n Internal Peer Review n n Large enough MD Pool Rotate Membership Unbiased/No secondary agendas External Peer Review n Considered the best assurance for an unbiased and accurate review n n No established data for this The specifics of peer review should be individualized to the lab, health system, and/or state
Peer Review n Internal Peer Review n n Large enough MD Pool Rotate Membership Unbiased/No secondary agendas External Peer Review n Considered the best assurance for an unbiased and accurate review n n No established data for this The specifics of peer review should be individualized to the lab, health system, and/or state
 Cath Lab Accreditation
Cath Lab Accreditation
 Cath Lab Accreditation for Cardiovascular Excellence is an independent, not for profit organization initially established by SCAI in 2009 with subsequent partnership from ACC. The mission of ACE is to ensure highquality patient care and promote patient safety in facilities performing invasive cardiac and endovascular procedures. ACE achieves this mission by setting standards for quality care, establishing requirements for accreditation, and providing peer review. ACE, the only cath lab accrediting organization, also provides tools and resources to support self-evaluation and quality improvement. Visit http: //www. cvexcel. org. What is ACE ? 30
Cath Lab Accreditation for Cardiovascular Excellence is an independent, not for profit organization initially established by SCAI in 2009 with subsequent partnership from ACC. The mission of ACE is to ensure highquality patient care and promote patient safety in facilities performing invasive cardiac and endovascular procedures. ACE achieves this mission by setting standards for quality care, establishing requirements for accreditation, and providing peer review. ACE, the only cath lab accrediting organization, also provides tools and resources to support self-evaluation and quality improvement. Visit http: //www. cvexcel. org. What is ACE ? 30
 Background n Standards based on guidelines, current literature n Including this ECD n Revised yearly , or more frequently if science demands n n Current Accreditation Programs n Carotid Artery Stenting n Cath/PCI Other Review Programs n Data Integrity n “Low Volume” Operator n External Peer Review n Appropriate Use Reviews n Customized Programs Pathway to Accreditation • • • Initial Application – Review by Nurse and Physician Reviewers • Policies and Procedures • Demographics, Appropriate Use, Outcome Measures, Standard Quality Metrics • Internal Peer Review Process Nurse Site Visit – Validation of NCDR reported data – Process and Facility Review Physician Data and Angiographic Review – Report • Deficiencies and Corrective Action Plans • Recommendation for Accreditation, Provisional Accreditation or Denial – Physician Site Visit for cause ACE Board Approval Ongoing support to implement corrective action plans • Shared experiences • Best Practices
Background n Standards based on guidelines, current literature n Including this ECD n Revised yearly , or more frequently if science demands n n Current Accreditation Programs n Carotid Artery Stenting n Cath/PCI Other Review Programs n Data Integrity n “Low Volume” Operator n External Peer Review n Appropriate Use Reviews n Customized Programs Pathway to Accreditation • • • Initial Application – Review by Nurse and Physician Reviewers • Policies and Procedures • Demographics, Appropriate Use, Outcome Measures, Standard Quality Metrics • Internal Peer Review Process Nurse Site Visit – Validation of NCDR reported data – Process and Facility Review Physician Data and Angiographic Review – Report • Deficiencies and Corrective Action Plans • Recommendation for Accreditation, Provisional Accreditation or Denial – Physician Site Visit for cause ACE Board Approval Ongoing support to implement corrective action plans • Shared experiences • Best Practices
 Continuing the Work… SCAI Think Tank on Quality, 2013 Initiative #1: SCAI-QIT Impact n Christopher J. White, MD, FSCAI, Past President SCAI Initiative #2: Personal Commitment to Quality n Theodore A. Bass, MD, FSCAI, SCAI President Initiative #3: Cath Lab Director Boot Camp n Charles E. Chambers, MD, FSCAI, SCAI President Elect
Continuing the Work… SCAI Think Tank on Quality, 2013 Initiative #1: SCAI-QIT Impact n Christopher J. White, MD, FSCAI, Past President SCAI Initiative #2: Personal Commitment to Quality n Theodore A. Bass, MD, FSCAI, SCAI President Initiative #3: Cath Lab Director Boot Camp n Charles E. Chambers, MD, FSCAI, SCAI President Elect
 SCAI-QIT Impact n Since its launch in May 2011, SCAI’s Quality Improvement Toolkit (SCAI-QIT) has provided the Society and the field of interventional cardiology with a proactive message about QUALITY. n SCAI-QIT has been funded by SCAI’s industry partners and has generated significant media coverage, portrayed as both a forward-looking effort to enhance quality and help the profession improve care: ‒ Incorporate Appropriate Use Criteria (AUC) into clinical decision making. ‒ A response to queries related to allegations of inappropriate/unnecessary procedures. SCAI-QIT CHAMPIONS N = 390 US States Represented 42 Total US 316 (81%) Countries Represented 31 Total OUS 74 (19%) Year 3: Measuring SCAI-QIT’s IMPACT: • Process improvement: ie. Peer-review conferences • Outcome improvement: ie. Improved bleeding rate
SCAI-QIT Impact n Since its launch in May 2011, SCAI’s Quality Improvement Toolkit (SCAI-QIT) has provided the Society and the field of interventional cardiology with a proactive message about QUALITY. n SCAI-QIT has been funded by SCAI’s industry partners and has generated significant media coverage, portrayed as both a forward-looking effort to enhance quality and help the profession improve care: ‒ Incorporate Appropriate Use Criteria (AUC) into clinical decision making. ‒ A response to queries related to allegations of inappropriate/unnecessary procedures. SCAI-QIT CHAMPIONS N = 390 US States Represented 42 Total US 316 (81%) Countries Represented 31 Total OUS 74 (19%) Year 3: Measuring SCAI-QIT’s IMPACT: • Process improvement: ie. Peer-review conferences • Outcome improvement: ie. Improved bleeding rate
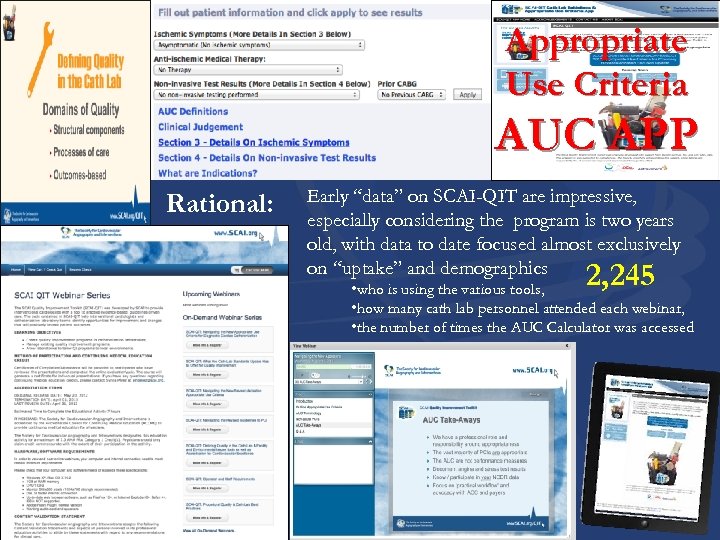 Appropriate Use Criteria AUC APP Rational: Early “data” on SCAI-QIT are impressive, especially considering the program is two years old, with data to date focused almost exclusively on “uptake” and demographics 2, 245 • who is using the various tools, • how many cath lab personnel attended each webinar, • the number of times the AUC Calculator was accessed
Appropriate Use Criteria AUC APP Rational: Early “data” on SCAI-QIT are impressive, especially considering the program is two years old, with data to date focused almost exclusively on “uptake” and demographics 2, 245 • who is using the various tools, • how many cath lab personnel attended each webinar, • the number of times the AUC Calculator was accessed
 Implementation n An efficient and cost-effective approach to measuring the impact of SCAI-QIT is to establish a “Research Steering Committee” – to select metrics, develop one or more hypotheses on the impact of SCAI-QIT tools on either cath lab processes (e. g. , benchmarking, check lists, or best practices) or patient outcomes (e. g. , reduction of complications). n Once these core components of the initial research project are established, the study group will expanded to include other active Quality Improvement Committee members and selected research “sites”.
Implementation n An efficient and cost-effective approach to measuring the impact of SCAI-QIT is to establish a “Research Steering Committee” – to select metrics, develop one or more hypotheses on the impact of SCAI-QIT tools on either cath lab processes (e. g. , benchmarking, check lists, or best practices) or patient outcomes (e. g. , reduction of complications). n Once these core components of the initial research project are established, the study group will expanded to include other active Quality Improvement Committee members and selected research “sites”.
 Personal Commitment to Quality: A Plan to Support Interventional Cardiology by Supporting Individual Interventionalists Theodore A. Bass, MD, SCAI President SCAI-QIT: our commitment to supporting SCAI-QIT quality one cath lab at a time. Now … SCAI commits to supporting quality one Fellow at a time.
Personal Commitment to Quality: A Plan to Support Interventional Cardiology by Supporting Individual Interventionalists Theodore A. Bass, MD, SCAI President SCAI-QIT: our commitment to supporting SCAI-QIT quality one cath lab at a time. Now … SCAI commits to supporting quality one Fellow at a time.
 Overview: Long-Term Goal: • To make “FSCAI” synonymous with quality • To grow awareness of the value of FSCAI to external audiences (public, referring providers, etc. ), so they will learn to seek FSCAI doctors • To demonstrate the profession’s (and the Society’s) commitment to Quality Paradigm Shift Requires Phased-in Approach: • Natural progression from guidelines & AUC to SCAI-QIT to expectations for individual members • Trickle down, starting with leaders & volunteers; then Quality Champions and committee members; new FSCAI applicants; eventually to all Fellows
Overview: Long-Term Goal: • To make “FSCAI” synonymous with quality • To grow awareness of the value of FSCAI to external audiences (public, referring providers, etc. ), so they will learn to seek FSCAI doctors • To demonstrate the profession’s (and the Society’s) commitment to Quality Paradigm Shift Requires Phased-in Approach: • Natural progression from guidelines & AUC to SCAI-QIT to expectations for individual members • Trickle down, starting with leaders & volunteers; then Quality Champions and committee members; new FSCAI applicants; eventually to all Fellows
 Personal Commitment to Quality Phase 1 Assemble a cross-disciplinary task force of SCAI leaders to develop a Menu of “tasks” with a pathway to completion and a plan for incentivizing participation. Test to see if meaningful, reasonable, measurable, valuable? Phase 2 Create a pilot program work in defined geographic area then evaluate and adjust. Phase 3 Roll-out to all Members with an invitation to participate and explanations of pathway, process, tools, resources, etc. Also make an announcement to External Audiences that SCAI takes commitment to Quality to a new level. This will provide a reassurance that interventional cardiologists are leading the way in quality care and why to look for FSCAI when choosing your cardiologist.
Personal Commitment to Quality Phase 1 Assemble a cross-disciplinary task force of SCAI leaders to develop a Menu of “tasks” with a pathway to completion and a plan for incentivizing participation. Test to see if meaningful, reasonable, measurable, valuable? Phase 2 Create a pilot program work in defined geographic area then evaluate and adjust. Phase 3 Roll-out to all Members with an invitation to participate and explanations of pathway, process, tools, resources, etc. Also make an announcement to External Audiences that SCAI takes commitment to Quality to a new level. This will provide a reassurance that interventional cardiologists are leading the way in quality care and why to look for FSCAI when choosing your cardiologist.
 Cath Lab Director’s Boot Camp Rationale Cath Lab Directors serve as influential decision makers and leaders with no job description or special training provided. SCAI can provide structured guidance and tools for improved, quality-based decision making. Assess/Understand Need Define Cath Lab Director Population Conduct Focus Groups during SCAI 2013 Conduct Online Survey Interview Thought Leaders
Cath Lab Director’s Boot Camp Rationale Cath Lab Directors serve as influential decision makers and leaders with no job description or special training provided. SCAI can provide structured guidance and tools for improved, quality-based decision making. Assess/Understand Need Define Cath Lab Director Population Conduct Focus Groups during SCAI 2013 Conduct Online Survey Interview Thought Leaders
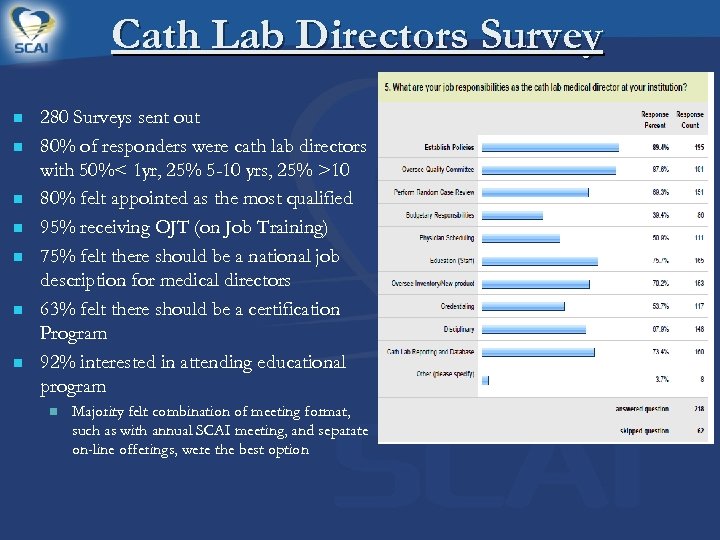 Cath Lab Directors Survey n n n n 280 Surveys sent out 80% of responders were cath lab directors with 50%< 1 yr, 25% 5 -10 yrs, 25% >10 80% felt appointed as the most qualified 95% receiving OJT (on Job Training) 75% felt there should be a national job description for medical directors 63% felt there should be a certification Program 92% interested in attending educational program n Majority felt combination of meeting format, such as with annual SCAI meeting, and separate on-line offerings, were the best option
Cath Lab Directors Survey n n n n 280 Surveys sent out 80% of responders were cath lab directors with 50%< 1 yr, 25% 5 -10 yrs, 25% >10 80% felt appointed as the most qualified 95% receiving OJT (on Job Training) 75% felt there should be a national job description for medical directors 63% felt there should be a certification Program 92% interested in attending educational program n Majority felt combination of meeting format, such as with annual SCAI meeting, and separate on-line offerings, were the best option
 Plan – Realize Synergies, Create Dynamic Opportunities Leverage Existing Resources n n n SCAI Quality Improvement Toolkit (QIT) SCAI live programming/Annual Meeting My. SCAI (e. SCAI) Community Determine Optimal Formats n n Define “Boot Camp” Concept – credential process vs. road map? Develop Online Resource n n n Static & dynamic presentations and videos Inclusion of QIT components Develop Live Programming n n Stand alone program for Cath Lab Directors? Sessions incorporated into existing programs? Cath Lab Director’s Boot Camp?
Plan – Realize Synergies, Create Dynamic Opportunities Leverage Existing Resources n n n SCAI Quality Improvement Toolkit (QIT) SCAI live programming/Annual Meeting My. SCAI (e. SCAI) Community Determine Optimal Formats n n Define “Boot Camp” Concept – credential process vs. road map? Develop Online Resource n n n Static & dynamic presentations and videos Inclusion of QIT components Develop Live Programming n n Stand alone program for Cath Lab Directors? Sessions incorporated into existing programs? Cath Lab Director’s Boot Camp?
 Final Thoughts and Questions SCAI is thankful for the opportunity to attend and present our quality initiatives. We as a society are dedicated to this effort and enthusiastic in partnering with all societies, countries, etc. , to promote universal application of quality standards for the best possible patient care.
Final Thoughts and Questions SCAI is thankful for the opportunity to attend and present our quality initiatives. We as a society are dedicated to this effort and enthusiastic in partnering with all societies, countries, etc. , to promote universal application of quality standards for the best possible patient care.


