5405c1ccd9a1ae08bde932f2d383e752.ppt
- Количество слайдов: 55

Proteomics GET THE BIOLOGICAL ANSWERS YOU WANT SAMPLE PREP AND SEPARATIONS Cindy L. James, Ph. D Protein Biochemist

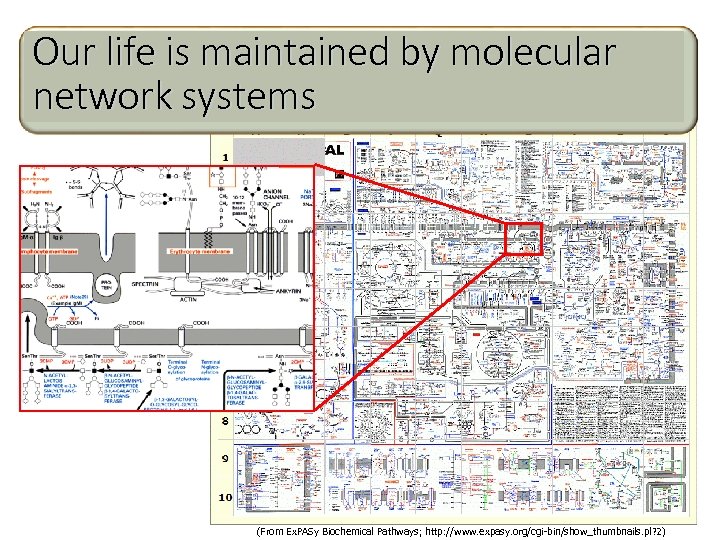
Our life is maintained by molecular network systems (From Ex. PASy Biochemical Pathways; http: //www. expasy. org/cgi-bin/show_thumbnails. pl? 2)

Protein interactions are at the core of the entire system of any living cell… …AN ABSOLUTE REQUIREMENT TO UNDERSTAND! A C P I G YT OL LYC T Y WA H

Same genome, different proteome
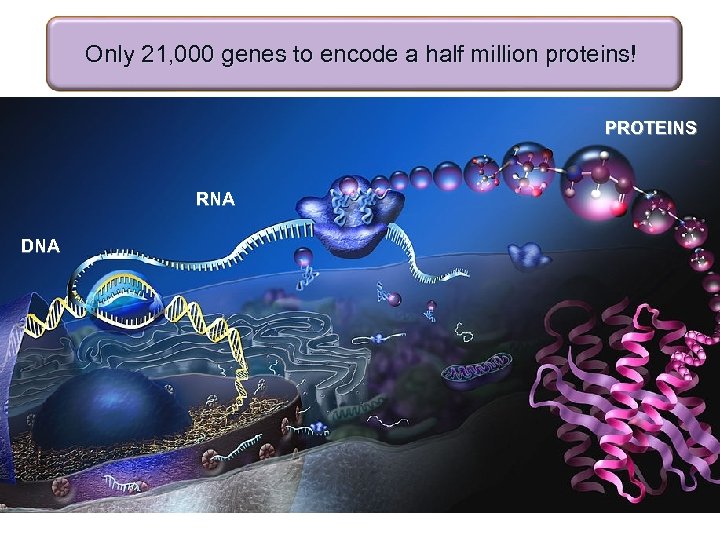
Only 21, 000 genes to encode a half million proteins! PROTEINS RNA DNA
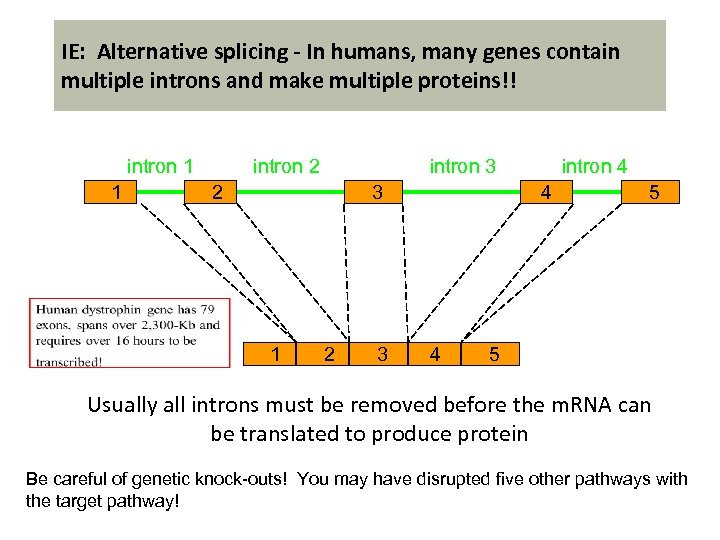
IE: Alternative splicing - In humans, many genes contain multiple introns and make multiple proteins!! intron 1 1 intron 3 intron 2 2 3 1 2 3 intron 4 4 4 5 5 Usually all introns must be removed before the m. RNA can be translated to produce protein Be careful of genetic knock-outs! You may have disrupted five other pathways with the target pathway!
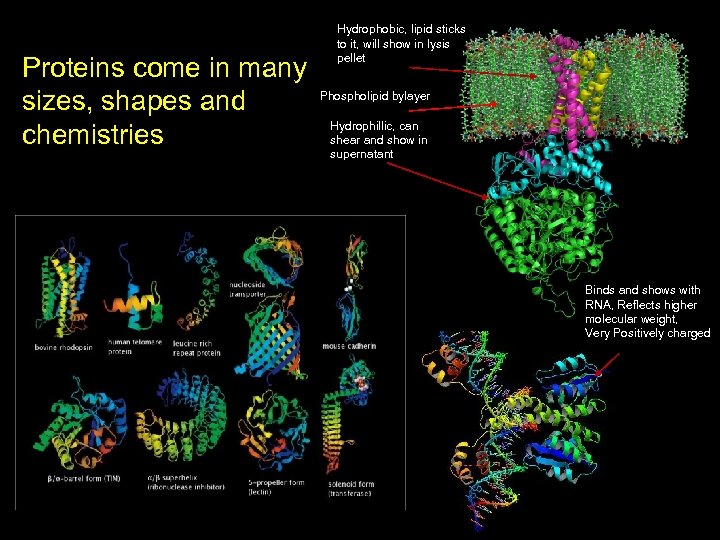
Hydrophobic, lipid sticks to it, will show in lysis pellet Proteins come in many Phospholipid bylayer sizes, shapes and Hydrophillic, can chemistries shear and show in supernatant Binds and shows with RNA, Reflects higher molecular weight, Very Positively charged

What do you want to know? ◦ Which proteins are present? In what isoforms? ◦ What post-translational modifications? ◦ In what concentrations (quantification)? ◦ What “signature” does protein have that will relate to other pathogenic or cancer related proteins ◦ What pathways and involved proteins will assist in determination of drug therapy ◦ WHO DOES IT ‘PLAY’ WITH? Today, we largely address these questions via mass spectrometry, but ◦ GOOD SAMPLE PREPARATION IS ESSENTIAL!!
Not Every Sample or Cell-line has the same proteome! Our samples come from: Bacteria Feces Food Hair Plants Roots Sediment Seeds Sludge Tree Tissue Urine Nasal secretion Yeast Blood Plasma Saliva Urine For Example: Tissue samples obtained from a biopsy, or Biopsy during surgical removal of a tumor can be used to classify the type of tumor found in the patient!

Not all Proteins are produced by the cell in equal amounts! Favored research drug targets (signal proteins) are actually low in abundance! ◦ kinases, ◦ proteases ◦ hydrolases of all sorts ◦ receptors (most likely) ◦ Researcher needs to aim for regulatory choke point and bottleneck proteins for targets

Some Experiment Challenges Statistics! Analyses = difficult to duplicate ◦Ie: Statistically better = grow many plates at once and harvest all at once, not many different growths. Proteome will be more equal between samples Difficult to prepare pure samples Cellular protein expression very sensitive to environmental conditions AND p. H’s Gel work may not run identically from time to time
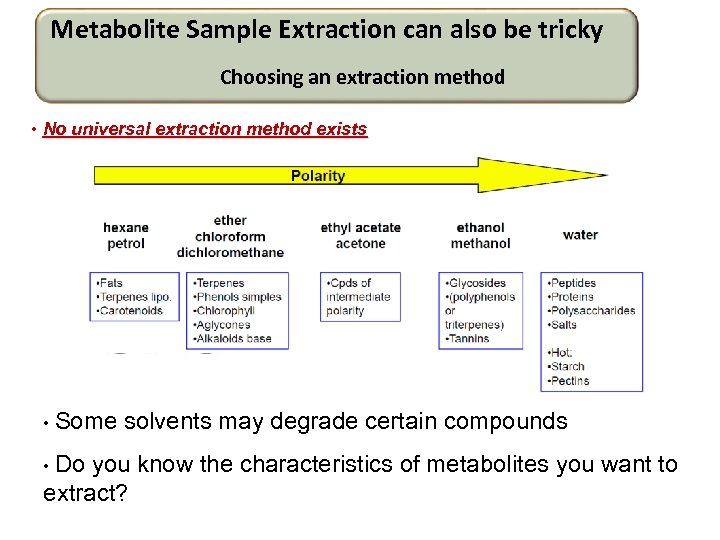
Metabolite Sample Extraction can also be tricky Choosing an extraction method • No universal extraction method exists • Some solvents may degrade certain compounds • Do you know the characteristics of metabolites you want to extract?

Notes on Sample Preparation Contaminants (nucleic acids, lipids, and carbohydrates) can cause problems There is no single protocol for cleaning up the protein sample! Researchers combine procedures to reduce unwanted components.

Things to ask before sample prep and why What are you looking for? ◦ Are they fishing for lots of global proteins, or looking for one precious gem? Where do you expect to find it? ◦ Do they think the protein is nuclear? Cytosolic? Expressed out of the cell? Does your protein have any characteristics that make it different from all others? ◦ Does it bind DNA? Ligands? ATP? Is your protein hydrophobic? Bind lipids? Sugars? What is the protein’s PI? (isoelectric point) Has anyone published on this or something similar?
OPEN YOUR CELLS AND TISSUES

Does your lysis buffer make a difference? YES!!! Cells and tissues need to be lysed to release the proteins of interest. Lysis buffers differ in ability to solubilize proteins. SDS and other ionic detergents are harshest but give the highest yield Are you doing IP’s and WB’s? ANTIBODIES: - recognize reduced and denatured protein…use these conditions first. - Some antibodies will only recognize a protein in native, non-denatured form - don’t use denaturing detergent (SDS, deoxycholate, and somewhat less denaturing, Triton X-100). IP FOR PROTEIN-PROTEIN STUDIES: - Use non-ionic IGEPAL CA-630, less denaturing and better for kinase activity and protein interactions Do not use RIPA if looking for protein: protein interactions. Are you looking for phosphorylation of proteins, protein-protein interactions, or membrane bound Use Sulfo-betanes, IGEPAL CA-630 Buffer – for cytoplasmic, membrane-bound, or whole cell extracts. If protein of interest isn’t completely extracted from insoluble material or aggregates, use harsher ionic detergents that assist proteins into solution. Are you interested in total protein levels of a protein Try RIPA buffer can give lower background in immunoprecipitation, but can denature some proteins.
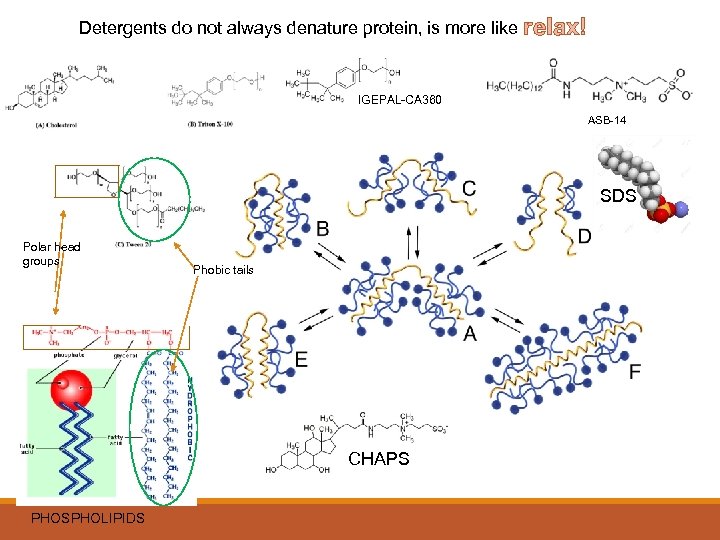
Detergents do not always denature protein, is more like relax! IGEPAL-CA 360 ASB-14 SDS Polar head groups Phobic tails CHAPS PHOSPHOLIPIDS

Protein location Whole Cell Cytoplasmic (soluble) Cytoplasmic (cytoskeletal bound) Membrane bound Nuclear Mitochondria Buffer recommended RIPA Tris-HCl Tris-Triton CJ’s Mem. Lysis or RIPA use nuclear fraction protocol and RIPA use mitochondrial fraction protocol, high salt & IGEPAL Enrich for your elusive target protein by fractionation. Can load more of the protein per gel lane. Will help removal of potentially crossreactive proteins from unused fractions. FOR MEMBRANE PROTEINS CJ’S Mem. Lysis Buffer – used for signal and membrane proteins. A chaotrope plus aminosulfobetaine OR similar to compete with lipid/hydrophobic amino acid that involve imbedded membrane proteins. Detergent fatty-acid chain should mimic the lipids it wants to dissolve. Since cell walls are different for membrane proteins, the detergent used should have similar number of carbons and polar-head ionics to match it!

Separation by subfraction GET YOUR PROTEINS AWAY FROM ALL THE OTHER KIDS IN THE PLAYGROUND!
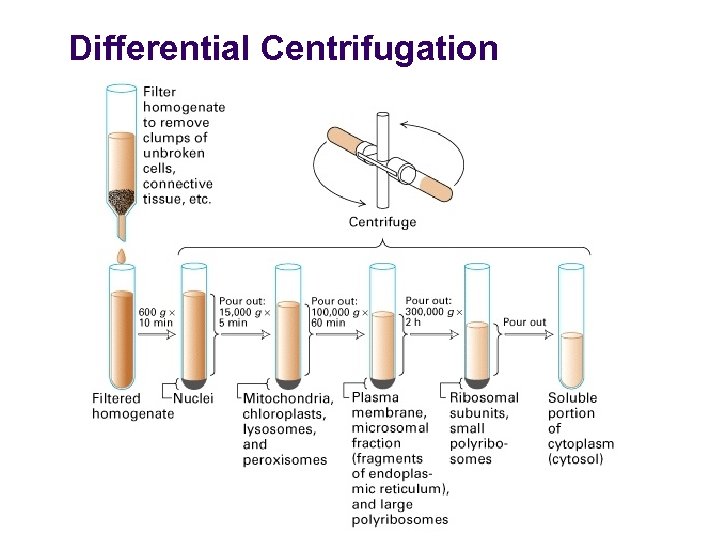
Differential Centrifugation
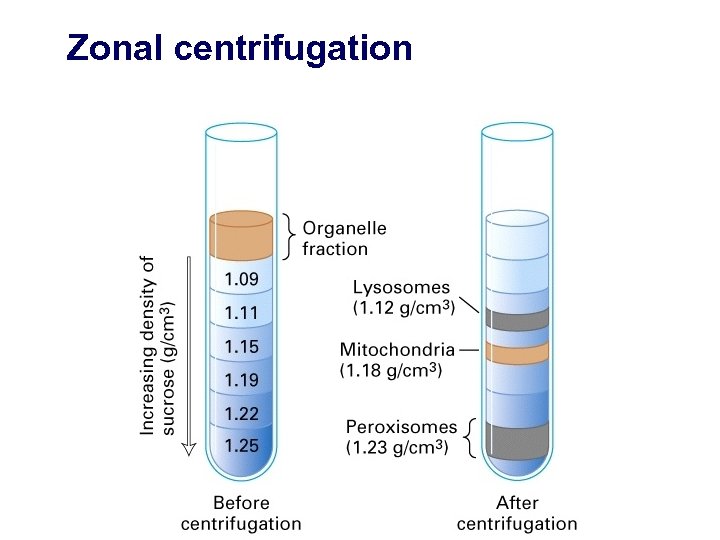
Zonal centrifugation

Common Separation techniques

Summary of initial steps of protein purification • • Choose source of proteins. Solubilize proteins. Stabilize proteins. Specific assay for protein of interest – Enzymatic activity, immunological activity, physical characteristics (e. g. molecular mass, spectroscopic properties, etc. ), biological activity • Assay should be: – – Specific Rapid Sensitive Quantitative

Know the charge of your protein!

Know the p. I of your proteins of interest! Every protein has a p. I (point where there is an OVERALL ZERO charge, not where there are no charges on your protein!) INDUSTRY RULE: If your buffer is at the p. I of the protein(s) you are after, you WILL lose your protein in precipitate!
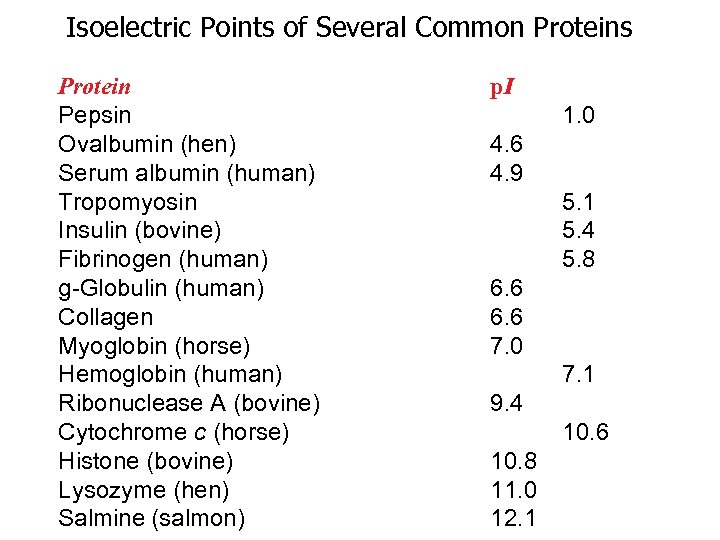
Isoelectric Points of Several Common Proteins Protein Pepsin Ovalbumin (hen) Serum albumin (human) Tropomyosin Insulin (bovine) Fibrinogen (human) g-Globulin (human) Collagen Myoglobin (horse) Hemoglobin (human) Ribonuclease A (bovine) Cytochrome c (horse) Histone (bovine) Lysozyme (hen) Salmine (salmon) p. I 1. 0 4. 6 4. 9 5. 1 5. 4 5. 8 6. 6 7. 0 7. 1 9. 4 10. 6 10. 8 11. 0 12. 1
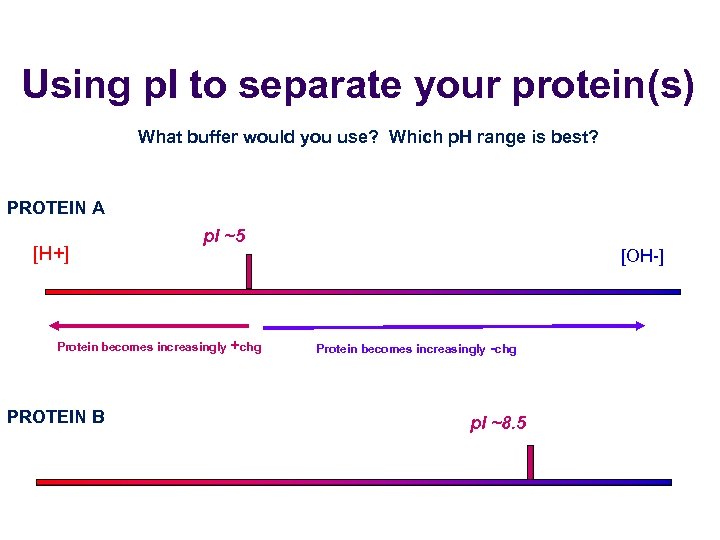
Using p. I to separate your protein(s) What buffer would you use? Which p. H range is best? PROTEIN A [H+] p. I ~5 Protein becomes increasingly +chg PROTEIN B [OH-] Protein becomes increasingly -chg p. I ~8. 5
Salting Out Your Protein • Solubilized proteins can be purified based on overall charge, ionic strength, polarity • Ammonium sulfate (NH 4 SO 4) commonly used to “salt out” • Takes away water making protein less soluble because hydrophobic interactions increase • Different aliquots taken as function of salt concentration to get closer to desired protein sample of interest (30, 40, 50, 75% increments) • One fraction has protein of interest
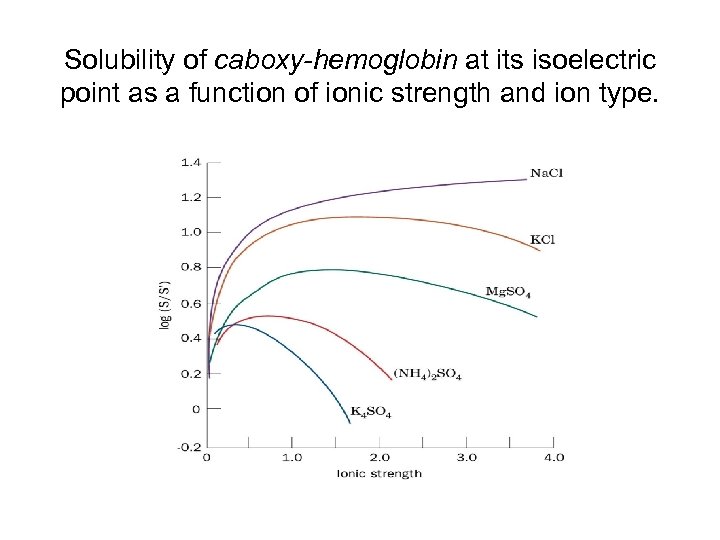
Solubility of caboxy-hemoglobin at its isoelectric point as a function of ionic strength and ion type.

Solubility of proteins • Water-miscible organic solvents also precipitate proteins. – Acetone, ethanol – Low dielectric constants lower the solvating power of their aqueous solutions for dissolved ions. • This technique is done at low temperatures (0 ºC) because at higher temperatures, the solvent evaporates. • Can magnify the differences in salting out procedures. • Some water-miscible organic solvents (DMF, DMSO) are good at s • Solubilizing proteins (high dielectric constants). Certain ions (I-, Cl. O 4 -, SCN-, Li+, Mg 2+, Ca 2+ ) increase the solubility of proteins rather than salting out!

Solubility of proteins • A protein in a p. H near its isolectric point is not subject to salting in. • As the p. H is moved away from the p. I of the protein, the protein’s net charge increases and it is easier to salt in. • Salts inhibit interactions between neighboring molecules in the protein that promote aggregation and precipitation. • p. I’s of proteins can be used to precipitate proteins.

Separating by Column Chromatography
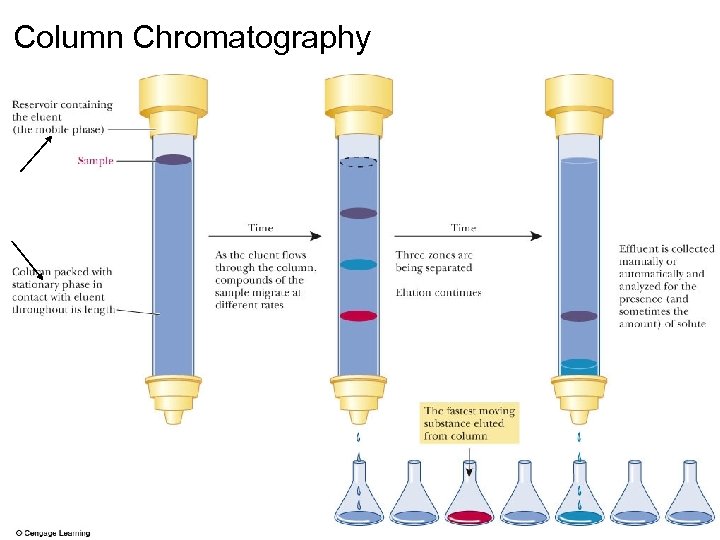
Column Chromatography
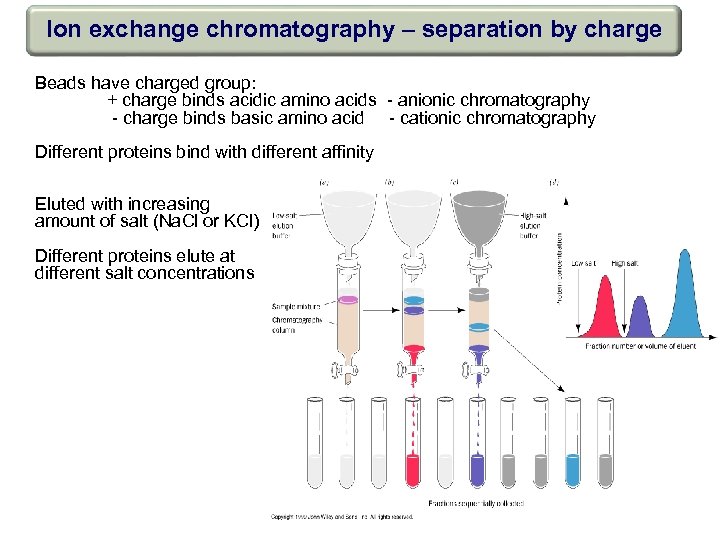
Ion exchange chromatography – separation by charge Beads have charged group: + charge binds acidic amino acids - anionic chromatography - charge binds basic amino acid - cationic chromatography Different proteins bind with different affinity Eluted with increasing amount of salt (Na. Cl or KCl) Different proteins elute at different salt concentrations
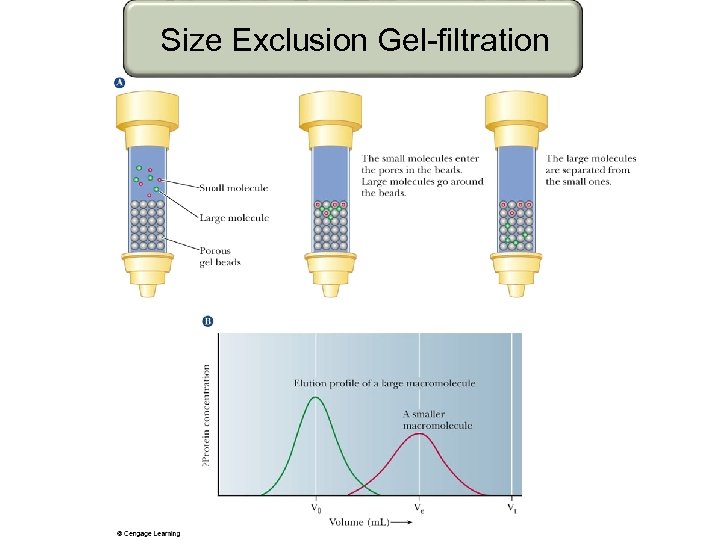
Size Exclusion Gel-filtration
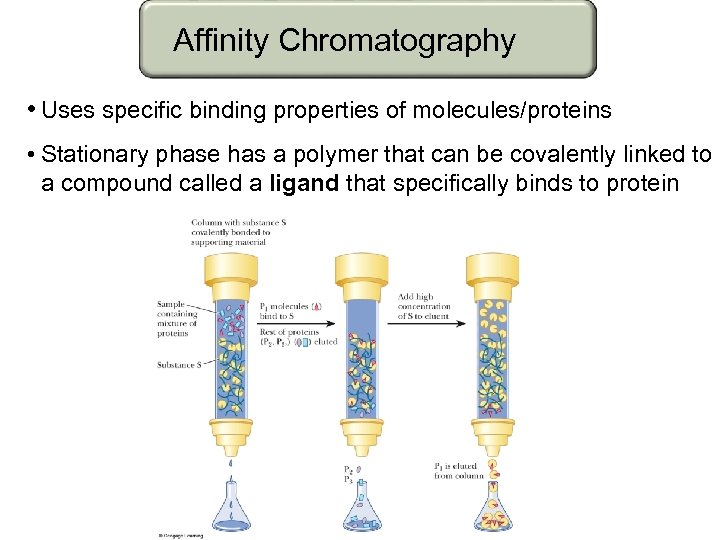
Affinity Chromatography • Uses specific binding properties of molecules/proteins • Stationary phase has a polymer that can be covalently linked to a compound called a ligand that specifically binds to protein
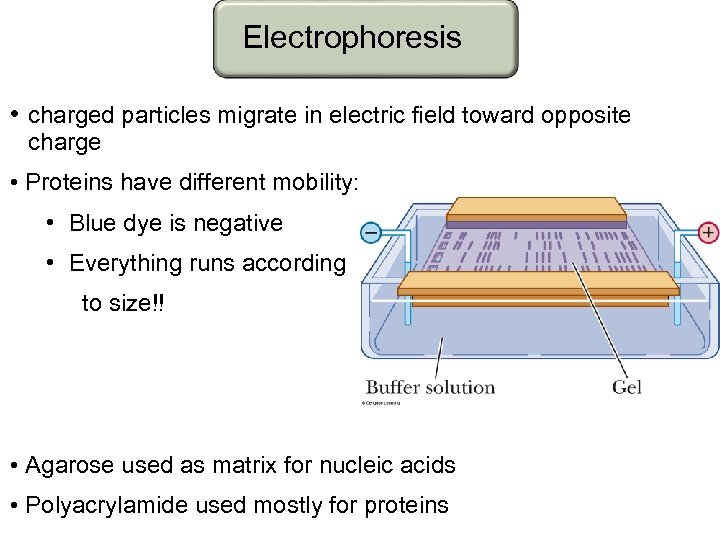
Electrophoresis • charged particles migrate in electric field toward opposite charge • Proteins have different mobility: • Blue dye is negative • Everything runs according to size!! • Agarose used as matrix for nucleic acids • Polyacrylamide used mostly for proteins

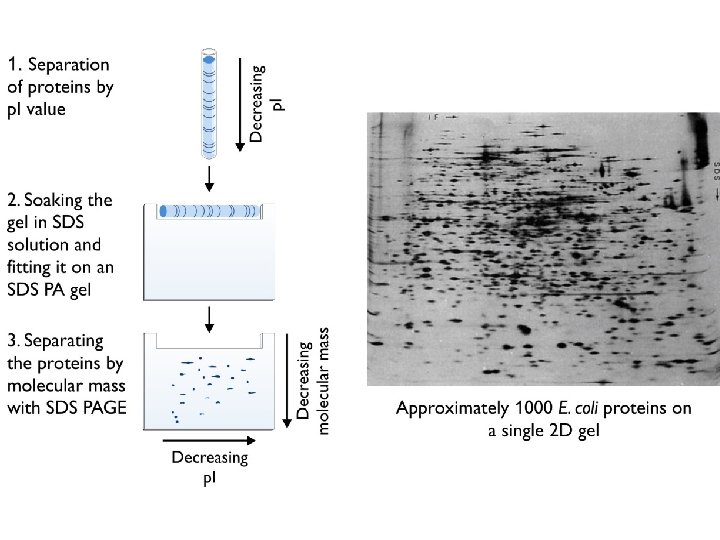
2 -D Gel Electrophoresis IEF (Isoelectric Focusing) • Powerful technology to separate proteins. • Separates thousands of proteins onto polymer gel. • Allows physical properties of proteins to be picked out separately for analysis and identification. • First dimension is IEF to separate by p. I by p. H
Di. GE Difference gel electrophoresis Jeremy Keirsey – in house expert Keirsey. 1@osu. edu
DIGE – a visual 2 D approach Clinical drug stimulated cell line
Technical Issues

Where do you think your protein is? “It’s exported to the plasma” or “it’s taken up by neuro-transmitters” or “it’s in the blood”. Examples: “Exported to plasma” – proteins go through the cell wall by – p themselves, or with carrier associations. The researcher looks for these by examining the cell in growth media and harvesting media. strategy: precipitate protein out of growth media (Am. SO 4 only, as TCA or Chloroform as the latter may cause their protein to inactivate). Be careful! Media has a LOT manufacture added proteins that can interfere! of

Examples Continued… “It’s a neurotransmitter” – very lipidy samples. May also be extremely glycosylated. Part of the protein may be hydrophobic and part hydrophilic! strategy: you will need to do some prep so that the lipids and/or sugars will not interfere with downstream experiments “It’s in the blood” – Ok! One of the most common proteins in blood is hemoglobin. It is so abundant that it may interfere as well as the iron! strategy: remove hemoglobin by affinity chromatography and albumen by CIBA-Blue chromatography

What are you looking for? “A signal protein” or “A nuclear protein” or “A structural protein”…answers give you a great place to start! Examples: Signal Proteins are post-translationally modified, in low abundance and most copies of the protein will NOT have the signal on it. strategy: look for phosphorylations or nitrations on 2 D, or use Pro. Q or nitro-tyrosine antibodies on 2 D or western blot Nuclear Proteins locate to the nucleus. A large percentage of them are positively charged and can be complexed with the nuclear membrane strategy: enrich for the nucleus only. Use cationic chromatography to separate out.

Protein Interactions When analyzing a new protein, ask – to what proteins does it bind? ◦ strategy: Use new protein as an affinity agent to isolate its binding partners ◦ Bind protein or TAG-protein to resin and run fresh lysate over it ◦ May detect low affinity, transient, or cellular environment specific interactions – protein can be crosslinked to its ligand! ◦ Maybe use an immunoprecipitation or CO-IP

Enrich for Modifications! Glycosylation? Total = concavalin (Con. A) Wheat Germ Agglutinin(WGA) Mannose = Concavalin, snowdrop lectin, lentil lectin Sialic Acid = Wheat Germ agglutinin, Elderberry lectin O-glycan = Jacalin, Peanut Agglutinin Enrich for Phosphate IMAC (immobilized metal) and strong cation exchange Enrich for Nitration Antibody IP or CO-IP Enrich for Acetylation Antibody IP for acetyl-lysine

Not seeing what you want? - Are the proteins expressing in the cell lines he is looking in? - Were all the cells grown all at the same time? Harvest at EXACTLY the same time? - The cell has its own growth phases…the proteome will change THE ENTIRE TIME! - Many larger protein-protein interactions will bind to the inner side of the membrane walls or to structural proteins - Make sure as they check their soluble fraction – they ALSO check their pellet for their proteins! Many just think “cell debris” and throw them away! - Is the p. H right for the protein in question? If the buffer is ANYWHERE near the p. I of the protein, the protein will precipitate out of the solution.

IS IT A TECHNICAL PROBLEM? …Affects proteome and metabolome! Structural Proteins AND glycolytic pathways will all upregulate! WASH WITH MEDIA, NOT PBS. EXCHANGE MORE OFTEN WITH ½ NEW MEDIA! Pathways affected: Glucose Inositol Betaine Taurine Choline Na+ K+ Proline JNK-p 38 ‘ERK-type. MAP kinases’ ‘Tyrosine Kinases’ + HUNDREDS MORE!
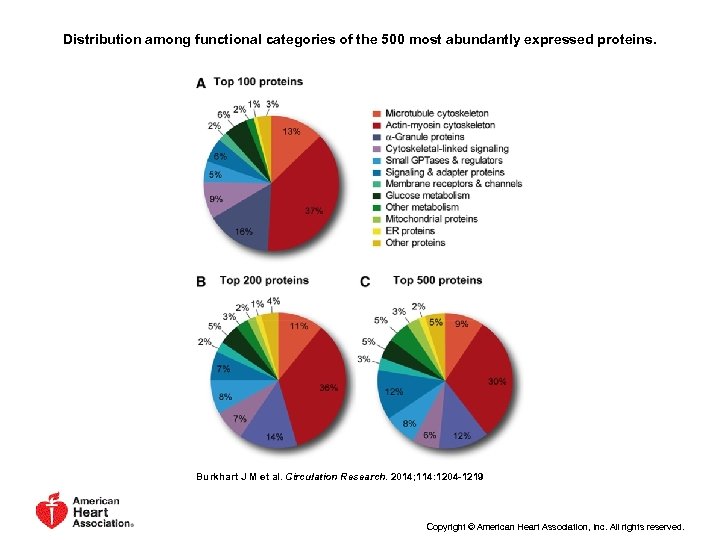
Distribution among functional categories of the 500 most abundantly expressed proteins. Burkhart J M et al. Circulation Research. 2014; 114: 1204 -1219 Copyright © American Heart Association, Inc. All rights reserved.

ARE YOU LOOKING FOR A NEEDLE IN A HAYSTACK? We can help you!

At OSU, we examine issues that scientists deal with concerning dependable and reproducible data from biological experiments! - isolate proteins - Study them alone or Study them in combination delicate - Cutting edge science requires and complicated customized approaches - Isolate proteins - Assure that biological activity is maintained, if desired We give advice on most experimental design to enhance the probability of success!

SERVICES at OSU Proteomics Just ask! • • • Protein Growth, Induction and Expression, Protein purification Subcloning into recombinant cell lines, Plasmid design DIGE Develop novel protein protocols, individualized for experiment Selective subfractionation, Salt fractionation, Enrichment, Solubility screening, Inclusion body isolation Western Blotting, Far Western Blotting, Immunoprecipitation and Coimmunoprecipitation, Protein-Protein interaction studies Classic chromatography: Affinity –Tag purification, ionic exchange, HIC reverse phase, SEC gel chromatography 100, 300, Immobilized metal affinity chromatography (IMAC), Heparin affinity: Protein A/G affinity column, ENDOTOXIN removal SDS-PAGE and DNA Electrophoresis, reduced and/or non-reduced Pro. Q, Lava. Purple, Sypro and other gel staining Fluorescent and Bradford Protein Quantitation Mass Spectrometry for protein identification

THANKS FOR LISTENING! You can find us at… Mass Spec and Proteomics and Protein Expression and Purification Facility Biomedical Research Tower Room 250 460 West 12 th Street Columbus, Ohio Lab: 614 -247 -8789 Arpad Somogyi, Ph. D – somogyi. 16@osu. edu Cindy L. James, Ph. D – james. 456@osu. edu
5405c1ccd9a1ae08bde932f2d383e752.ppt