033ff1848757ce083b08ab94092e5e3b.ppt
- Количество слайдов: 45
 Protein Structure, classification, Prediction and Proteomics
Protein Structure, classification, Prediction and Proteomics
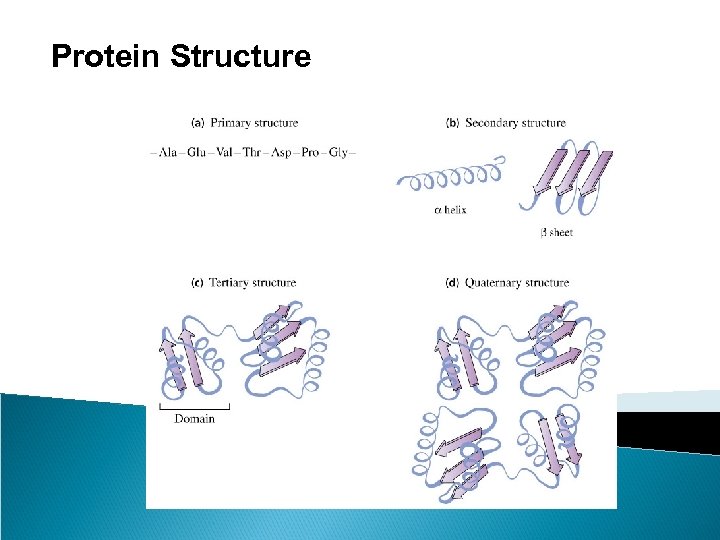 Protein Structure
Protein Structure
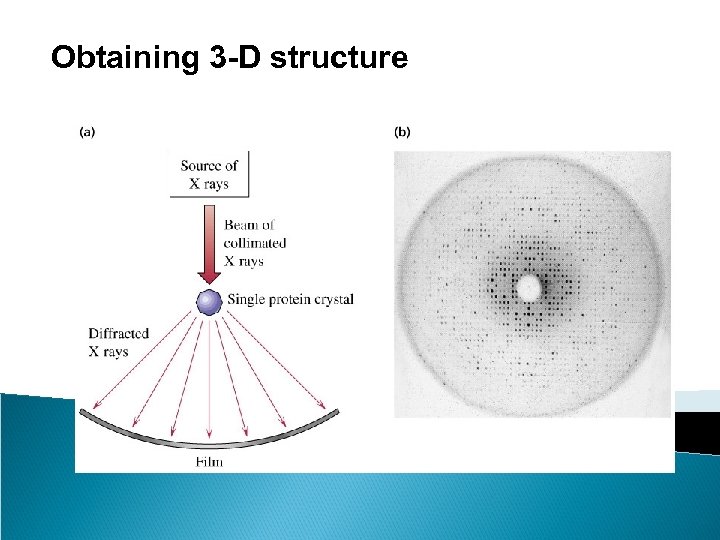 Obtaining 3 -D structure
Obtaining 3 -D structure
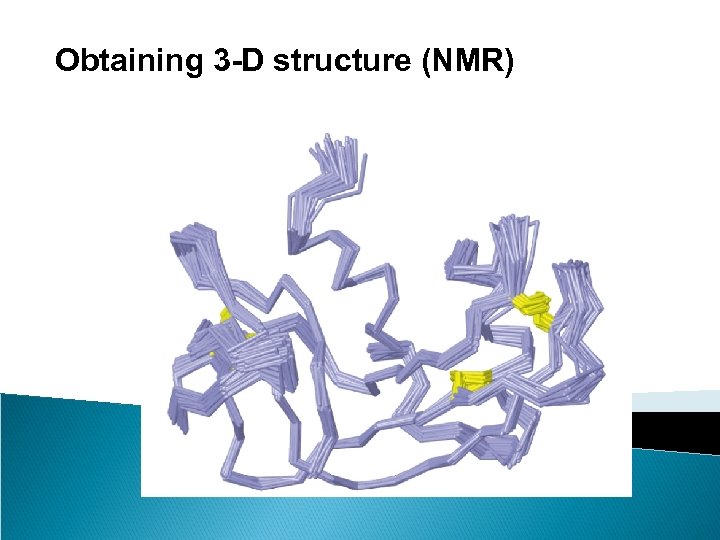 Obtaining 3 -D structure (NMR)
Obtaining 3 -D structure (NMR)
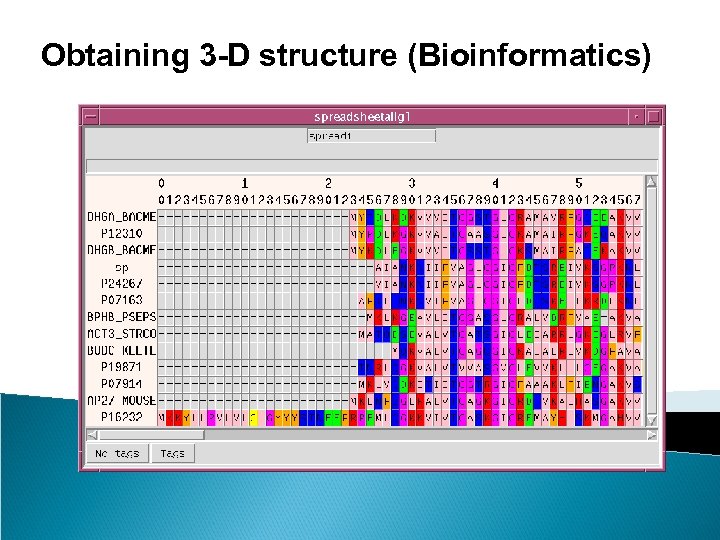 Obtaining 3 -D structure (Bioinformatics)
Obtaining 3 -D structure (Bioinformatics)
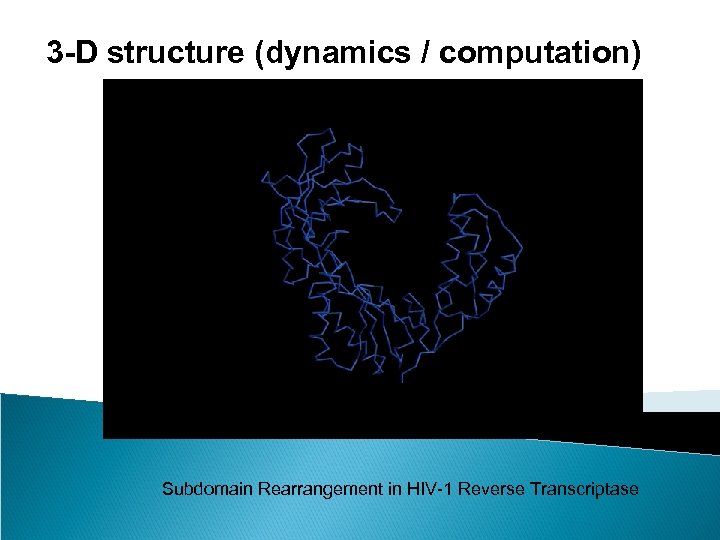 3 -D structure (dynamics / computation) Subdomain Rearrangement in HIV-1 Reverse Transcriptase
3 -D structure (dynamics / computation) Subdomain Rearrangement in HIV-1 Reverse Transcriptase
 Protein Databases Uni. Prot is the universal protein database, a central repository of protein data created by combining Swiss-Prot, Tr. EMBL and PIR. This makes it the world's most comprehensive resource on protein information. The Protein Information Resource (PIR), located at Georgetown University Medical Center (GUMC), is an integrated public bioinformatics resource to support genomic and proteomic research, and scientific studies. Swiss-Prot is a curated biological database of protein sequences from different species created in 1986 by Amos Bairoch during his Ph. D and developed by the Swiss Institute of Bioinformatics and the European Bioinformatics Institute. Pfam is a large collection of multiple sequence alignments and hidden Markov models covering many common protein domains and families. PDB NCBI http: //proteome. nih. gov/links. html
Protein Databases Uni. Prot is the universal protein database, a central repository of protein data created by combining Swiss-Prot, Tr. EMBL and PIR. This makes it the world's most comprehensive resource on protein information. The Protein Information Resource (PIR), located at Georgetown University Medical Center (GUMC), is an integrated public bioinformatics resource to support genomic and proteomic research, and scientific studies. Swiss-Prot is a curated biological database of protein sequences from different species created in 1986 by Amos Bairoch during his Ph. D and developed by the Swiss Institute of Bioinformatics and the European Bioinformatics Institute. Pfam is a large collection of multiple sequence alignments and hidden Markov models covering many common protein domains and families. PDB NCBI http: //proteome. nih. gov/links. html
 Pub. Med – Protein Databases The Protein database contains sequence data from the translated coding regions from DNA sequences in Gen. Bank, EMBL, and DDBJ as well as protein sequences submitted to Protein Information Resource (PIR), SWISS-PROT, Protein Research Foundation (PRF), and Protein Data Bank (PDB) (sequences from solved structures). The Structure database or Molecular Modeling Database (MMDB) contains experimental data from crystallographic and NMR structure determinations. The data for MMDB are obtained from the Protein Data Bank (PDB). The NCBI has cross-linked structural data to bibliographic information, to the sequence databases, and to the NCBI taxonomy. Use Cn 3 D, the NCBI 3 D structure viewer, for easy interactive visualization of molecular structures from Entrez. Tutorial: http: //www. pdb. org/pdbstatic/tutorials/tutorial. html
Pub. Med – Protein Databases The Protein database contains sequence data from the translated coding regions from DNA sequences in Gen. Bank, EMBL, and DDBJ as well as protein sequences submitted to Protein Information Resource (PIR), SWISS-PROT, Protein Research Foundation (PRF), and Protein Data Bank (PDB) (sequences from solved structures). The Structure database or Molecular Modeling Database (MMDB) contains experimental data from crystallographic and NMR structure determinations. The data for MMDB are obtained from the Protein Data Bank (PDB). The NCBI has cross-linked structural data to bibliographic information, to the sequence databases, and to the NCBI taxonomy. Use Cn 3 D, the NCBI 3 D structure viewer, for easy interactive visualization of molecular structures from Entrez. Tutorial: http: //www. pdb. org/pdbstatic/tutorials/tutorial. html
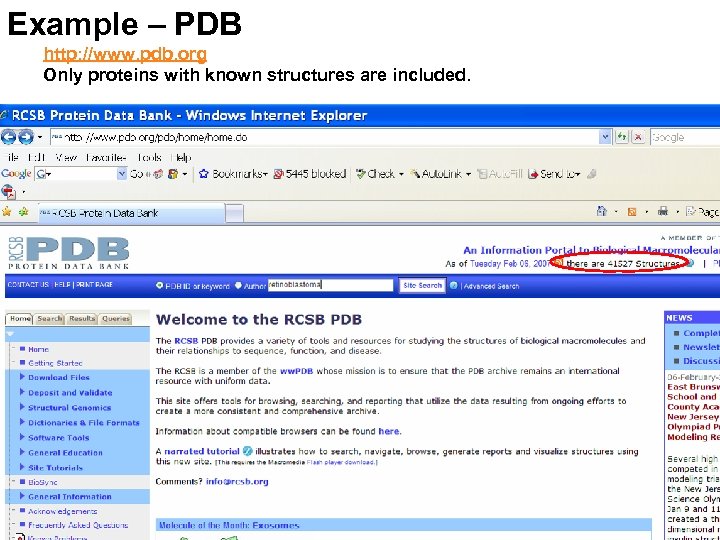 Example – PDB http: //www. pdb. org Only proteins with known structures are included.
Example – PDB http: //www. pdb. org Only proteins with known structures are included.
 Protein Visualization Softwares • • Cn 3 d Ras. Mol TOPS Chime • • • DSSP Molscript Ribbons MSMS Surfnet …
Protein Visualization Softwares • • Cn 3 d Ras. Mol TOPS Chime • • • DSSP Molscript Ribbons MSMS Surfnet …
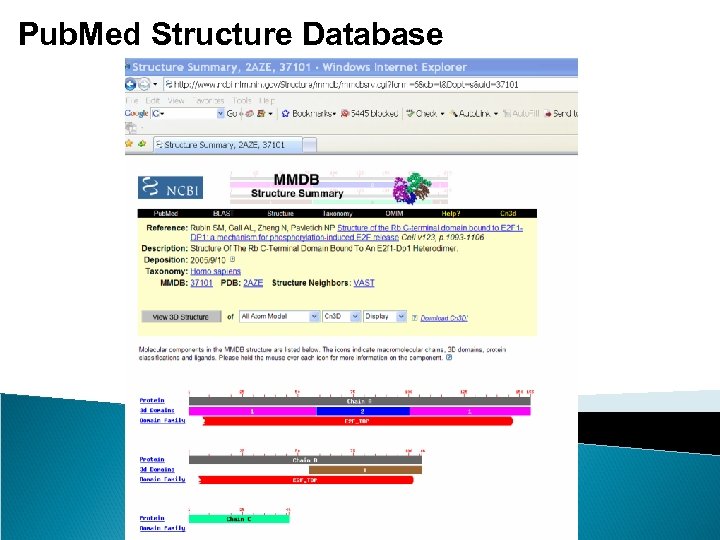 Pub. Med Structure Database
Pub. Med Structure Database
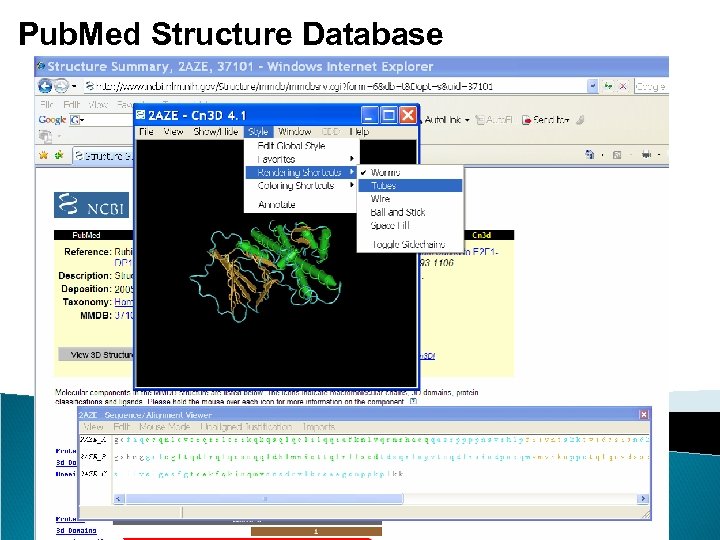 Pub. Med Structure Database
Pub. Med Structure Database
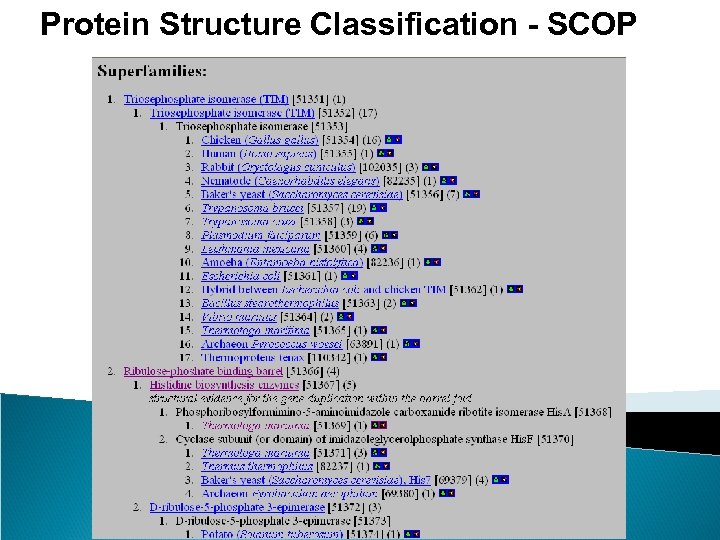
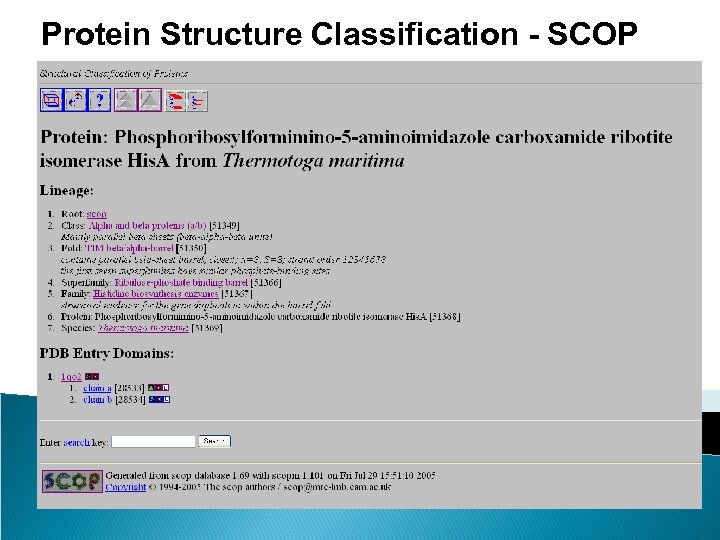
 Protein Structure Classification - SCOP • Structure Classification Of Proteins database • http: //scop. mrc-lmb. cam. ac. uk/scop/ • Hierarchical Clustering • Family – clear evolutionarily relationship • Superfamily – probable common evolutionary origin • Fold – major structural similarity • Boundaries between levels are more or less subjective • Conservative evolutionary classification leads to many new divisions at the family and superfamily levels, therefore it is recommended to first focus on higher levels in the classification tree.
Protein Structure Classification - SCOP • Structure Classification Of Proteins database • http: //scop. mrc-lmb. cam. ac. uk/scop/ • Hierarchical Clustering • Family – clear evolutionarily relationship • Superfamily – probable common evolutionary origin • Fold – major structural similarity • Boundaries between levels are more or less subjective • Conservative evolutionary classification leads to many new divisions at the family and superfamily levels, therefore it is recommended to first focus on higher levels in the classification tree.
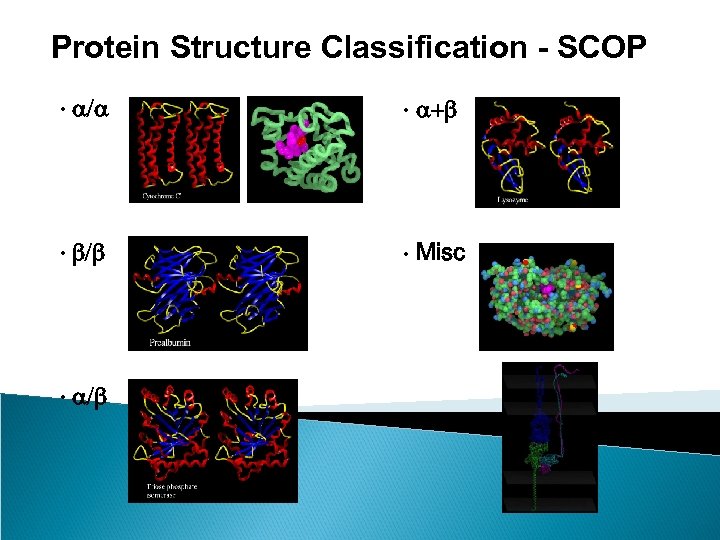 Protein Structure Classification - SCOP • a/a • a+b • b/b • Misc • a/b
Protein Structure Classification - SCOP • a/a • a+b • b/b • Misc • a/b
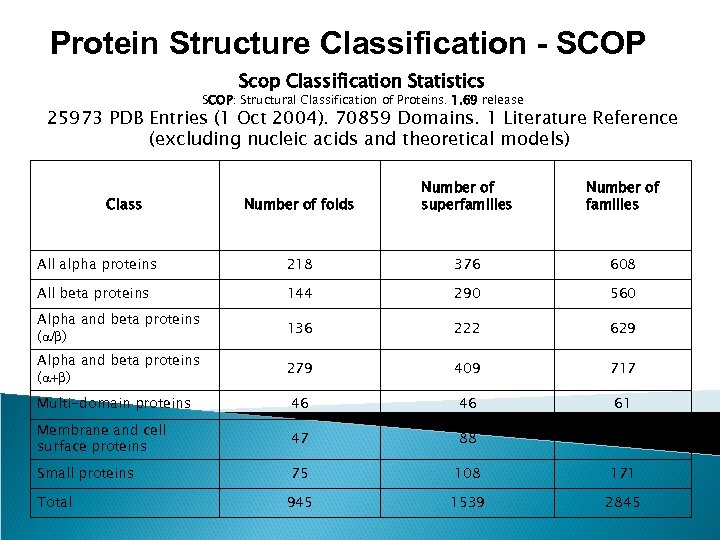 Protein Structure Classification - SCOP Scop Classification Statistics SCOP: Structural Classification of Proteins. 1. 69 release 25973 PDB Entries (1 Oct 2004). 70859 Domains. 1 Literature Reference (excluding nucleic acids and theoretical models) Number of folds Number of superfamilies Number of families All alpha proteins 218 376 608 All beta proteins 144 290 560 Alpha and beta proteins (a/b) 136 222 629 Alpha and beta proteins (a+b) 279 409 717 Multi-domain proteins 46 46 61 Membrane and cell surface proteins 47 88 99 Small proteins 75 108 171 945 1539 2845 Class Total
Protein Structure Classification - SCOP Scop Classification Statistics SCOP: Structural Classification of Proteins. 1. 69 release 25973 PDB Entries (1 Oct 2004). 70859 Domains. 1 Literature Reference (excluding nucleic acids and theoretical models) Number of folds Number of superfamilies Number of families All alpha proteins 218 376 608 All beta proteins 144 290 560 Alpha and beta proteins (a/b) 136 222 629 Alpha and beta proteins (a+b) 279 409 717 Multi-domain proteins 46 46 61 Membrane and cell surface proteins 47 88 99 Small proteins 75 108 171 945 1539 2845 Class Total
 Protein Structure Classification - SCOP
Protein Structure Classification - SCOP
 Protein Structure Classification - SCOP
Protein Structure Classification - SCOP
 Protein Structure Classification - SCOP
Protein Structure Classification - SCOP
 Protein Structure Classification - SCOP
Protein Structure Classification - SCOP
 Protein Structure Classification - SCOP
Protein Structure Classification - SCOP
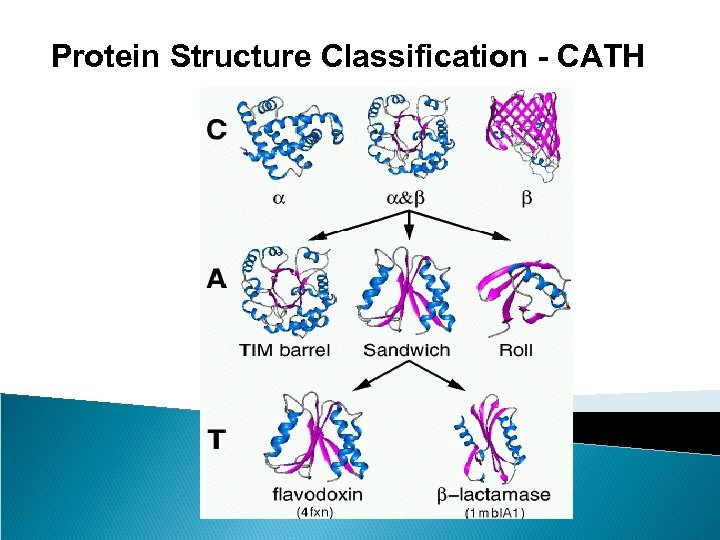
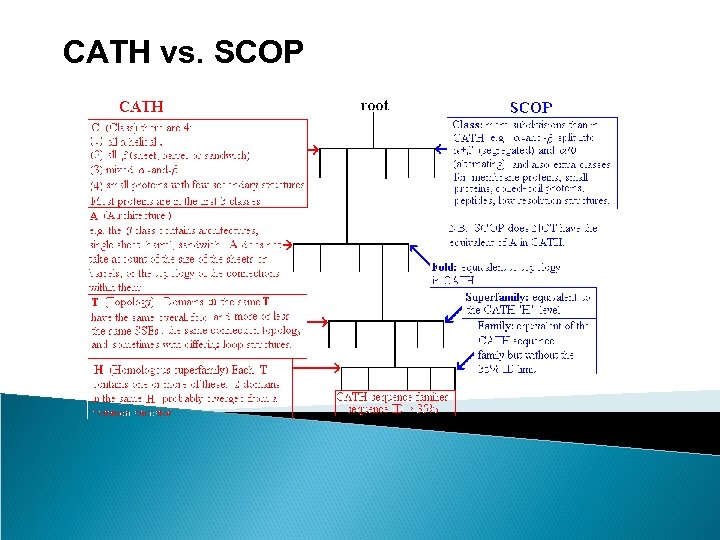
 Protein Structure Classification - CATH • CATH Protein Structure Classification • http: //www. cathdb. info/latest/index. html • CATH is a hierarchical classification of protein domain structures, which clusters proteins at four major levels, Class(C), Architecture(A), Topology(T) and Homologous superfamily (H). • Class, derived from secondary structure content, is assigned for more than 90% of protein structures automatically. • Architecture, which describes the gross orientation of secondary structures, independent of connectivities, is currently assigned manually. • The topology level clusters structures into fold groups according to their topological connections and numbers of secondary structures. • The homologous superfamilies cluster proteins with highly similar structures and functions. The assignments of structures to fold groups and homologous superfamilies are made by sequence and structure comparisons.
Protein Structure Classification - CATH • CATH Protein Structure Classification • http: //www. cathdb. info/latest/index. html • CATH is a hierarchical classification of protein domain structures, which clusters proteins at four major levels, Class(C), Architecture(A), Topology(T) and Homologous superfamily (H). • Class, derived from secondary structure content, is assigned for more than 90% of protein structures automatically. • Architecture, which describes the gross orientation of secondary structures, independent of connectivities, is currently assigned manually. • The topology level clusters structures into fold groups according to their topological connections and numbers of secondary structures. • The homologous superfamilies cluster proteins with highly similar structures and functions. The assignments of structures to fold groups and homologous superfamilies are made by sequence and structure comparisons.
 Protein Structure Classification - CATH
Protein Structure Classification - CATH
 CATH vs. SCOP
CATH vs. SCOP
 Secondary Structure Prediction AGADIR - An algorithm to predict the helical content of peptides APSSP - Advanced Protein Secondary Structure Prediction Server GOR - Garnier et al, 1996 HNN - Hierarchical Neural Network method (Guermeur, 1997) Jpred - A consensus method for protein secondary structure prediction at University of Dundee JUFO - Protein secondary structure prediction from sequence (neural network) nn. Predict - University of California at San Francisco (UCSF) Porter - University College Dublin Predict. Protein - PHDsec, PHDacc, PHDhtm, PHDtopology, PHDthreader, Max. Hom, Eval. Sec from Columbia University Prof - Cascaded Multiple Classifiers for Secondary Structure Prediction PSA - Bio. Molecular Engineering Research Center (BMERC) / Boston PSIpred - Various protein structure prediction methods at Brunel University SOPMA - Geourjon and Deléage, 1995 SSpro - Secondary structure prediction using bidirectional recurrent neural networks at University of California DLP - Domain linker prediction at RIKEN http: //us. expasy. org/tools/#secondary
Secondary Structure Prediction AGADIR - An algorithm to predict the helical content of peptides APSSP - Advanced Protein Secondary Structure Prediction Server GOR - Garnier et al, 1996 HNN - Hierarchical Neural Network method (Guermeur, 1997) Jpred - A consensus method for protein secondary structure prediction at University of Dundee JUFO - Protein secondary structure prediction from sequence (neural network) nn. Predict - University of California at San Francisco (UCSF) Porter - University College Dublin Predict. Protein - PHDsec, PHDacc, PHDhtm, PHDtopology, PHDthreader, Max. Hom, Eval. Sec from Columbia University Prof - Cascaded Multiple Classifiers for Secondary Structure Prediction PSA - Bio. Molecular Engineering Research Center (BMERC) / Boston PSIpred - Various protein structure prediction methods at Brunel University SOPMA - Geourjon and Deléage, 1995 SSpro - Secondary structure prediction using bidirectional recurrent neural networks at University of California DLP - Domain linker prediction at RIKEN http: //us. expasy. org/tools/#secondary
 Secondary Structure Prediction - HNN • http: //npsa-pbil. ibcp. fr/cgi-bin/secpred_hnn. pl • >gi|78099986|sp|P 0 ABK 2|CYDB_ECOLI Cytochrome d ubiquinol oxidase subunit 2 (Cytochrome d ubiquinol oxidase subunit II) (Cytochrome bd-I oxidase subunit II) MIDYEVLRFIWWLLVGVLLIGFAVTDGFDMGVGMLTRFLGRNDTERRIMINSIAPHWDGNQVWLITAGGA LFAAWPMVYAAAFSGFYVAMILVLASLFFRPVGFDYRSKIEETRWRNMWDWGIFIGSFVPPLVIGVAFGN LLQGVPFNVDEYLRLYYTGNFFQLLNPFGLLAGVVSVGMIITQGATYLQMRTVGELHLRTRATAQVAALV TLVCFALAGVWVMYGIDGYVVKSTMDHYAASNPLNKEVVREAGAWLVNFNNTPILWAIPALGVVLPLLTI LTARMDKAAWAFVFSSLTLACIILTAGIAMFPFVMPSSTMMNASLTMWDATSSQLTLNVMTWVAVVLVPIILLY TAWCYWKMFGRITKEDIERNTHSLY
Secondary Structure Prediction - HNN • http: //npsa-pbil. ibcp. fr/cgi-bin/secpred_hnn. pl • >gi|78099986|sp|P 0 ABK 2|CYDB_ECOLI Cytochrome d ubiquinol oxidase subunit 2 (Cytochrome d ubiquinol oxidase subunit II) (Cytochrome bd-I oxidase subunit II) MIDYEVLRFIWWLLVGVLLIGFAVTDGFDMGVGMLTRFLGRNDTERRIMINSIAPHWDGNQVWLITAGGA LFAAWPMVYAAAFSGFYVAMILVLASLFFRPVGFDYRSKIEETRWRNMWDWGIFIGSFVPPLVIGVAFGN LLQGVPFNVDEYLRLYYTGNFFQLLNPFGLLAGVVSVGMIITQGATYLQMRTVGELHLRTRATAQVAALV TLVCFALAGVWVMYGIDGYVVKSTMDHYAASNPLNKEVVREAGAWLVNFNNTPILWAIPALGVVLPLLTI LTARMDKAAWAFVFSSLTLACIILTAGIAMFPFVMPSSTMMNASLTMWDATSSQLTLNVMTWVAVVLVPIILLY TAWCYWKMFGRITKEDIERNTHSLY
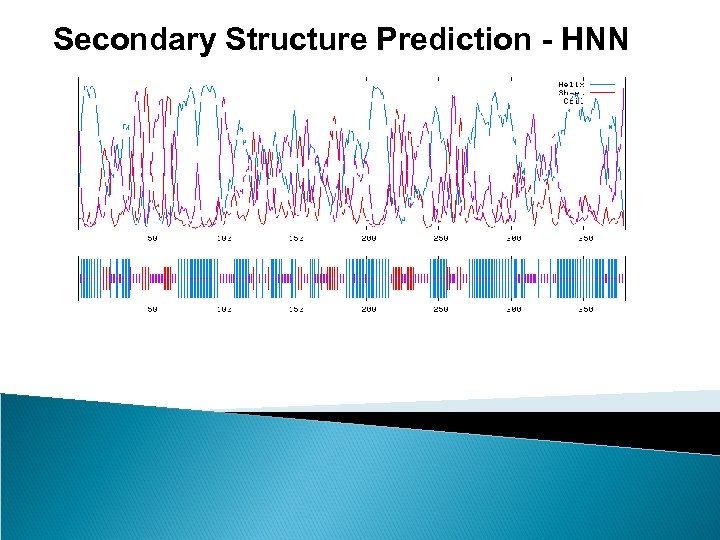
 Secondary Structure Prediction - HNN Sequence length : 379 HNN : Alpha helix (Hh) : 209 is 55. 15% 310 helix (Gg) : 0 is 0. 00% Pi helix (Ii) : 0 is 0. 00% Beta bridge (Bb) : 0 is 0. 00% Extended strand (Ee) : 55 is 14. 51% Beta turn (Tt) : 0 is 0. 00% Bend region (Ss) : 0 is 0. 00% Random coil (Cc) : 115 is 30. 34% Ambigous states (? ) : 0 is 0. 00% Other states : 0 is 0. 00% 10 20 30 40 50 60 70 | | | | MIDYEVLRFIWWLLVGVLLIGFAVTDGFDMGVGMLTRFLGRNDTERRIMINSIAPHWDGNQVWLITAGGA ccchhhhhhhheeeeehccchhhhhheecccccceeeeeeccch LFAAWPMVYAAAFSGFYVAMILVLASLFFRPVGFDYRSKIEETRWRNMWDWGIFIGSFVPPLVIGVAFGN hhhhhhhhhhhhhhhccccchhhhhcceeehccchccheehhhhhc LLQGVPFNVDEYLRLYYTGNFFQLLNPFGLLAGVVSVGMIITQGATYLQMRTVGELHLRTRATAQVAALV hhcccccchhhhheeeeccchhhhhcchceccceeeeeccchhhhhhhchhhhh TLVCFALAGVWVMYGIDGYVVKSTMDHYAASNPLNKEVVREAGAWLVNFNNTPILWAIPALGVVLPLLTI hhhhhhccceeeeeeccccccchhhhhheeccccceeeeccchhhhh LTARMDKAAWAFVFSSLTLACIILTAGIAMFPFVMPSSTMMNASLTMWDATSSQLTLNVMTWVAVVLVPI hhhhhhhhhhhhhcchhhcccchhhccccchhhhhhhhhh ILLYTAWCYWKMFGRITKEDIERNTHSLY hhhhhhhhcchhhhhhhccccc
Secondary Structure Prediction - HNN Sequence length : 379 HNN : Alpha helix (Hh) : 209 is 55. 15% 310 helix (Gg) : 0 is 0. 00% Pi helix (Ii) : 0 is 0. 00% Beta bridge (Bb) : 0 is 0. 00% Extended strand (Ee) : 55 is 14. 51% Beta turn (Tt) : 0 is 0. 00% Bend region (Ss) : 0 is 0. 00% Random coil (Cc) : 115 is 30. 34% Ambigous states (? ) : 0 is 0. 00% Other states : 0 is 0. 00% 10 20 30 40 50 60 70 | | | | MIDYEVLRFIWWLLVGVLLIGFAVTDGFDMGVGMLTRFLGRNDTERRIMINSIAPHWDGNQVWLITAGGA ccchhhhhhhheeeeehccchhhhhheecccccceeeeeeccch LFAAWPMVYAAAFSGFYVAMILVLASLFFRPVGFDYRSKIEETRWRNMWDWGIFIGSFVPPLVIGVAFGN hhhhhhhhhhhhhhhccccchhhhhcceeehccchccheehhhhhc LLQGVPFNVDEYLRLYYTGNFFQLLNPFGLLAGVVSVGMIITQGATYLQMRTVGELHLRTRATAQVAALV hhcccccchhhhheeeeccchhhhhcchceccceeeeeccchhhhhhhchhhhh TLVCFALAGVWVMYGIDGYVVKSTMDHYAASNPLNKEVVREAGAWLVNFNNTPILWAIPALGVVLPLLTI hhhhhhccceeeeeeccccccchhhhhheeccccceeeeccchhhhh LTARMDKAAWAFVFSSLTLACIILTAGIAMFPFVMPSSTMMNASLTMWDATSSQLTLNVMTWVAVVLVPI hhhhhhhhhhhhhcchhhcccchhhccccchhhhhhhhhh ILLYTAWCYWKMFGRITKEDIERNTHSLY hhhhhhhhcchhhhhhhccccc
 Secondary Structure Prediction - HNN
Secondary Structure Prediction - HNN
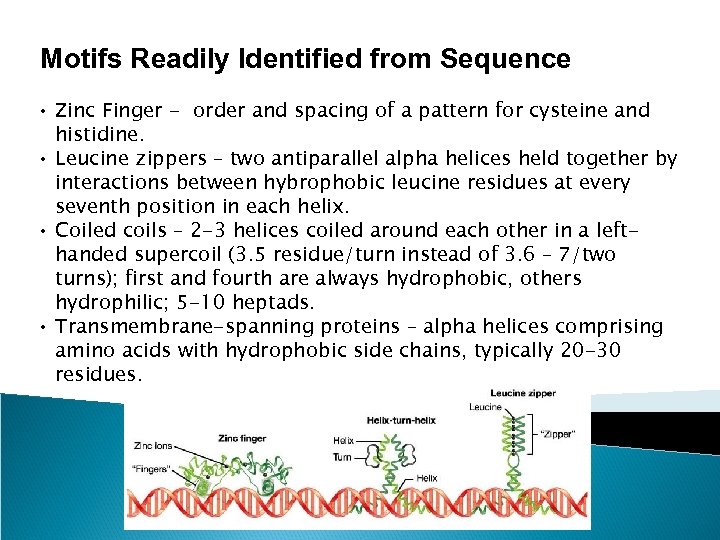 Motifs Readily Identified from Sequence • Zinc Finger - order and spacing of a pattern for cysteine and histidine. • Leucine zippers – two antiparallel alpha helices held together by interactions between hybrophobic leucine residues at every seventh position in each helix. • Coiled coils – 2 -3 helices coiled around each other in a lefthanded supercoil (3. 5 residue/turn instead of 3. 6 – 7/two turns); first and fourth are always hydrophobic, others hydrophilic; 5 -10 heptads. • Transmembrane-spanning proteins – alpha helices comprising amino acids with hydrophobic side chains, typically 20 -30 residues.
Motifs Readily Identified from Sequence • Zinc Finger - order and spacing of a pattern for cysteine and histidine. • Leucine zippers – two antiparallel alpha helices held together by interactions between hybrophobic leucine residues at every seventh position in each helix. • Coiled coils – 2 -3 helices coiled around each other in a lefthanded supercoil (3. 5 residue/turn instead of 3. 6 – 7/two turns); first and fourth are always hydrophobic, others hydrophilic; 5 -10 heptads. • Transmembrane-spanning proteins – alpha helices comprising amino acids with hydrophobic side chains, typically 20 -30 residues.
 Topology Prediction PSORT - Prediction of protein subcellular localization Target. P - Prediction of subcellular location DAS - Prediction of transmembrane regions in prokaryotes using the Dense Alignment Surface method (Stockholm University) HMMTOP - Prediction of transmembrane helices and topology of proteins (Hungarian Academy of Sciences) Predict. Protein - Prediction of transmembrane helix location and topology (Columbia University) SOSUI - Prediction of transmembrane regions (Nagoya University, Japan) TMAP - Transmembrane detection based on multiple sequence alignment (Karolinska Institut; Sweden) TMHMM - Prediction of transmembrane helices in proteins (CBS; Denmark) TMpred - Prediction of transmembrane regions and protein orientation (EMBnet. CH) Top. Pred - Topology prediction of membrane proteins (France) http: //us. expasy. org/tools
Topology Prediction PSORT - Prediction of protein subcellular localization Target. P - Prediction of subcellular location DAS - Prediction of transmembrane regions in prokaryotes using the Dense Alignment Surface method (Stockholm University) HMMTOP - Prediction of transmembrane helices and topology of proteins (Hungarian Academy of Sciences) Predict. Protein - Prediction of transmembrane helix location and topology (Columbia University) SOSUI - Prediction of transmembrane regions (Nagoya University, Japan) TMAP - Transmembrane detection based on multiple sequence alignment (Karolinska Institut; Sweden) TMHMM - Prediction of transmembrane helices in proteins (CBS; Denmark) TMpred - Prediction of transmembrane regions and protein orientation (EMBnet. CH) Top. Pred - Topology prediction of membrane proteins (France) http: //us. expasy. org/tools
 Tertiary Structure Prediction Comparative modeling SWISS-MODEL - An automated knowledge-based protein modelling server 3 Djigsaw - Three-dimensional models for proteins based on homologues of known structure CPHmodels - Automated neural-network based protein modelling server ESy. Pred 3 D - Automated homology modeling program using neural networks Geno 3 d - Automatic modeling of protein three-dimensional structure SDSC 1 - Protein Structure Homology Modeling Server Threading 3 D-PSSM - Protein fold recognition using 1 D and 3 D sequence profiles coupled with secondary structure information (Foldfit) Fugue - Sequence-structure homology recognition HHpred - Protein homology detection and structure prediction by HMM-HMM comparison Libellula - Neural network approach to evaluate fold recognition results LOOPP - Sequence to sequence, sequence to structure, and structure to structure alignment SAM-T 02 - HMM-based Protein Structure Prediction Threader - Protein fold recognition Pro. Sup - Protein structure superimposition SWEET - Constructing 3 D models of saccharides from their sequences Ab initio HMMSTR/Rosetta - Prediction of protein structure from sequence http: //us. expasy. org/tools
Tertiary Structure Prediction Comparative modeling SWISS-MODEL - An automated knowledge-based protein modelling server 3 Djigsaw - Three-dimensional models for proteins based on homologues of known structure CPHmodels - Automated neural-network based protein modelling server ESy. Pred 3 D - Automated homology modeling program using neural networks Geno 3 d - Automatic modeling of protein three-dimensional structure SDSC 1 - Protein Structure Homology Modeling Server Threading 3 D-PSSM - Protein fold recognition using 1 D and 3 D sequence profiles coupled with secondary structure information (Foldfit) Fugue - Sequence-structure homology recognition HHpred - Protein homology detection and structure prediction by HMM-HMM comparison Libellula - Neural network approach to evaluate fold recognition results LOOPP - Sequence to sequence, sequence to structure, and structure to structure alignment SAM-T 02 - HMM-based Protein Structure Prediction Threader - Protein fold recognition Pro. Sup - Protein structure superimposition SWEET - Constructing 3 D models of saccharides from their sequences Ab initio HMMSTR/Rosetta - Prediction of protein structure from sequence http: //us. expasy. org/tools
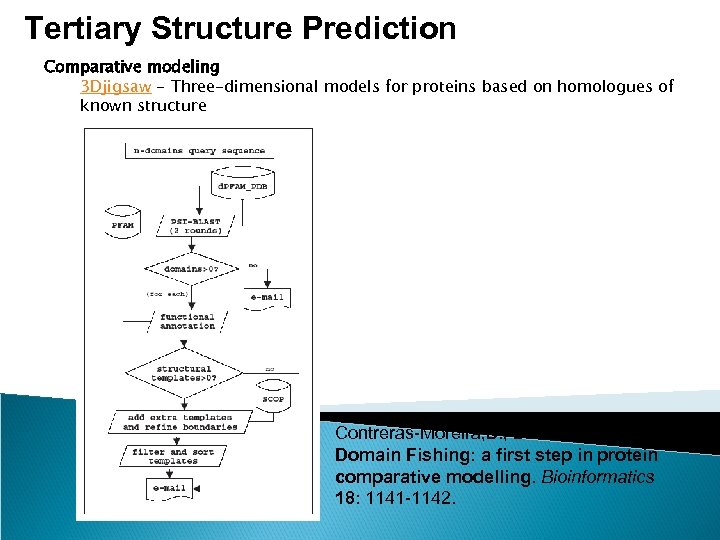 Tertiary Structure Prediction Comparative modeling 3 Djigsaw - Three-dimensional models for proteins based on homologues of known structure Contreras-Moreira, B. , Bates, P. A. (2002) Domain Fishing: a first step in protein comparative modelling. Bioinformatics 18: 1141 -1142.
Tertiary Structure Prediction Comparative modeling 3 Djigsaw - Three-dimensional models for proteins based on homologues of known structure Contreras-Moreira, B. , Bates, P. A. (2002) Domain Fishing: a first step in protein comparative modelling. Bioinformatics 18: 1141 -1142.
 Tertiary Structure Prediction Threading 3 D-PSSM - Protein fold recognition using 1 D and 3 D sequence profiles coupled with secondary structure information (Foldfit) Fugue - Sequence-structure homology recognition HHpred - Protein homology detection and structure prediction by HMM-HMM comparison Libellula - Neural network approach to evaluate fold recognition results LOOPP - Sequence to sequence, sequence to structure, and structure to structure alignment SAM-T 02 - HMM-based Protein Structure Prediction Threader - Protein fold recognition Pro. Sup - Protein structure superimposition SWEET - Constructing 3 D models of saccharides from their sequences
Tertiary Structure Prediction Threading 3 D-PSSM - Protein fold recognition using 1 D and 3 D sequence profiles coupled with secondary structure information (Foldfit) Fugue - Sequence-structure homology recognition HHpred - Protein homology detection and structure prediction by HMM-HMM comparison Libellula - Neural network approach to evaluate fold recognition results LOOPP - Sequence to sequence, sequence to structure, and structure to structure alignment SAM-T 02 - HMM-based Protein Structure Prediction Threader - Protein fold recognition Pro. Sup - Protein structure superimposition SWEET - Constructing 3 D models of saccharides from their sequences
 Tertiary Structure Prediction Threading The term threading was first coined by Jones, Taylor and Thornton in 1992, and originally referred specifically to the use of a full 3 -D structure atomic representation of the protein template in fold recognition. Today, the terms threading and fold recognition are frequently (though somewhat incorrectly) used interchangeably. The basic idea is that the target sequence (the protein sequence for which the structure is being predicted) is threaded through the backbone structures of a collection of template proteins (known as the fold library) and a “goodness of fit” score calculated for each sequence-structure alignment. This goodness of fit is often derived in terms of an empirical energy function, based on statistics derived from known protein structures, but many other scoring functions have been proposed and tried over the years. Threading methods share some of the characteristics of both comparative modelling methods (the sequence alignment aspect) and ab initio prediction methods (predicting structure based on identifying low-energy conformations of the target protein). http: //en. wikipedia. org/wiki/Threading_%28 protein_sequence%29
Tertiary Structure Prediction Threading The term threading was first coined by Jones, Taylor and Thornton in 1992, and originally referred specifically to the use of a full 3 -D structure atomic representation of the protein template in fold recognition. Today, the terms threading and fold recognition are frequently (though somewhat incorrectly) used interchangeably. The basic idea is that the target sequence (the protein sequence for which the structure is being predicted) is threaded through the backbone structures of a collection of template proteins (known as the fold library) and a “goodness of fit” score calculated for each sequence-structure alignment. This goodness of fit is often derived in terms of an empirical energy function, based on statistics derived from known protein structures, but many other scoring functions have been proposed and tried over the years. Threading methods share some of the characteristics of both comparative modelling methods (the sequence alignment aspect) and ab initio prediction methods (predicting structure based on identifying low-energy conformations of the target protein). http: //en. wikipedia. org/wiki/Threading_%28 protein_sequence%29
 Tertiary Structure Prediction Ab initio (de novo) • From scratch – using physical property instead of known structures • Mimic folding process – minimize certain energy function, stochastic modeling (e. g. , simulated annealing) • Computationally expensive – requires large clusters, large machines (e. g. , IBM Blue. Gene) or distributed computing, currently only work for small peptides • Big potential in the future – understand the dynamics, accuracy, and applications in drug development
Tertiary Structure Prediction Ab initio (de novo) • From scratch – using physical property instead of known structures • Mimic folding process – minimize certain energy function, stochastic modeling (e. g. , simulated annealing) • Computationally expensive – requires large clusters, large machines (e. g. , IBM Blue. Gene) or distributed computing, currently only work for small peptides • Big potential in the future – understand the dynamics, accuracy, and applications in drug development
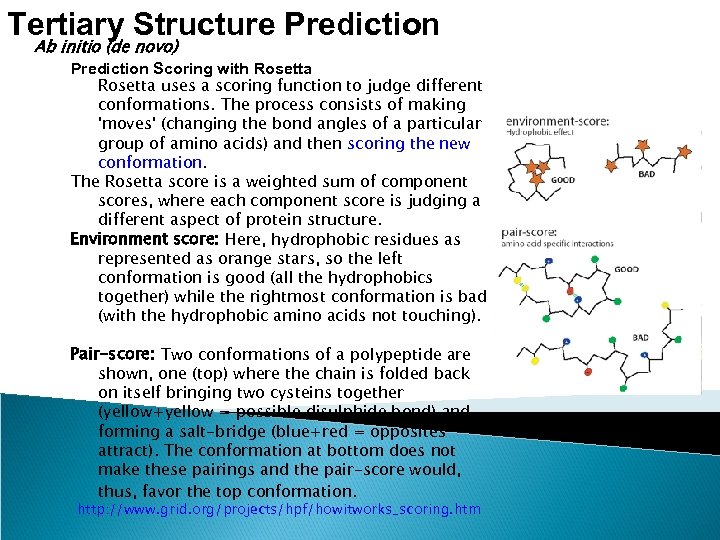 Tertiary Structure Prediction Ab initio (de novo) Prediction Scoring with Rosetta uses a scoring function to judge different conformations. The process consists of making 'moves' (changing the bond angles of a particular group of amino acids) and then scoring the new conformation. The Rosetta score is a weighted sum of component scores, where each component score is judging a different aspect of protein structure. Environment score: Here, hydrophobic residues as represented as orange stars, so the left conformation is good (all the hydrophobics together) while the rightmost conformation is bad (with the hydrophobic amino acids not touching). Pair-score: Two conformations of a polypeptide are shown, one (top) where the chain is folded back on itself bringing two cysteins together (yellow+yellow = possible disulphide bond) and forming a salt-bridge (blue+red = opposites attract). The conformation at bottom does not make these pairings and the pair-score would, thus, favor the top conformation. http: //www. grid. org/projects/hpf/howitworks_scoring. htm
Tertiary Structure Prediction Ab initio (de novo) Prediction Scoring with Rosetta uses a scoring function to judge different conformations. The process consists of making 'moves' (changing the bond angles of a particular group of amino acids) and then scoring the new conformation. The Rosetta score is a weighted sum of component scores, where each component score is judging a different aspect of protein structure. Environment score: Here, hydrophobic residues as represented as orange stars, so the left conformation is good (all the hydrophobics together) while the rightmost conformation is bad (with the hydrophobic amino acids not touching). Pair-score: Two conformations of a polypeptide are shown, one (top) where the chain is folded back on itself bringing two cysteins together (yellow+yellow = possible disulphide bond) and forming a salt-bridge (blue+red = opposites attract). The conformation at bottom does not make these pairings and the pair-score would, thus, favor the top conformation. http: //www. grid. org/projects/hpf/howitworks_scoring. htm
 Evaluation - CASP - Critical Assessment of Techniques for Protein Structure Prediction, is a community-wide experiment (though it is commonly referred to as a competition) for protein structure prediction taking place every two years since 1994. (http: //predictioncenter. org/) The main goal of CASP is to obtain an in-depth and objective assessment of our current abilities and inabilities in the area of protein structure prediction. To this end, participants will predict as much as possible about a set of soon to be known structures. These will be true predictions, not ‘post-dictions’ made on already known structures. CASP 7 will particularly address the following questions: 1. Are the models produced similar to the corresponding experimental structure? 2. Is the mapping of the target sequence onto the proposed structure (i. e. the alignment) correct? 3. Have similar structures that a model can be based on been identified? 4. Are comparative models more accurate than can be obtained by simply copying the best template? 5. Has there been progress from the earlier CASPs? 6. What methods are most effective? 7. Where can future effort be most productively focused?
Evaluation - CASP - Critical Assessment of Techniques for Protein Structure Prediction, is a community-wide experiment (though it is commonly referred to as a competition) for protein structure prediction taking place every two years since 1994. (http: //predictioncenter. org/) The main goal of CASP is to obtain an in-depth and objective assessment of our current abilities and inabilities in the area of protein structure prediction. To this end, participants will predict as much as possible about a set of soon to be known structures. These will be true predictions, not ‘post-dictions’ made on already known structures. CASP 7 will particularly address the following questions: 1. Are the models produced similar to the corresponding experimental structure? 2. Is the mapping of the target sequence onto the proposed structure (i. e. the alignment) correct? 3. Have similar structures that a model can be based on been identified? 4. Are comparative models more accurate than can be obtained by simply copying the best template? 5. Has there been progress from the earlier CASPs? 6. What methods are most effective? 7. Where can future effort be most productively focused?
 Evaluation - CASP Evaluation of the results is carried out in the following prediction categories: • tertiary structure prediction (all CASPs) • secondary structure prediction (dropped after CASP 5) • prediction of structure complexes (CASP 2 only; a separate experiment CAPRI - carries on this subject) • residue-residue contact prediction (starting CASP 4) • disordered regions prediction (starting CASP 5) • domain boundary prediction (starting CASP 6) • function prediction (starting CASP 6) • model quality assessment (starting CASP 7) • model refinement (starting CASP 7) Tertiary structure prediction category was further subdivided into • homology modelling • fold recognition (also called protein threading; Note, this is incorrect as threading is a method) • de novo structure prediction Now referred to as 'New Fold' as many methods apply evaluation, or scoring, functions that are biased by knowledge of native protein structures, such an example would be an artificial neural network.
Evaluation - CASP Evaluation of the results is carried out in the following prediction categories: • tertiary structure prediction (all CASPs) • secondary structure prediction (dropped after CASP 5) • prediction of structure complexes (CASP 2 only; a separate experiment CAPRI - carries on this subject) • residue-residue contact prediction (starting CASP 4) • disordered regions prediction (starting CASP 5) • domain boundary prediction (starting CASP 6) • function prediction (starting CASP 6) • model quality assessment (starting CASP 7) • model refinement (starting CASP 7) Tertiary structure prediction category was further subdivided into • homology modelling • fold recognition (also called protein threading; Note, this is incorrect as threading is a method) • de novo structure prediction Now referred to as 'New Fold' as many methods apply evaluation, or scoring, functions that are biased by knowledge of native protein structures, such an example would be an artificial neural network.
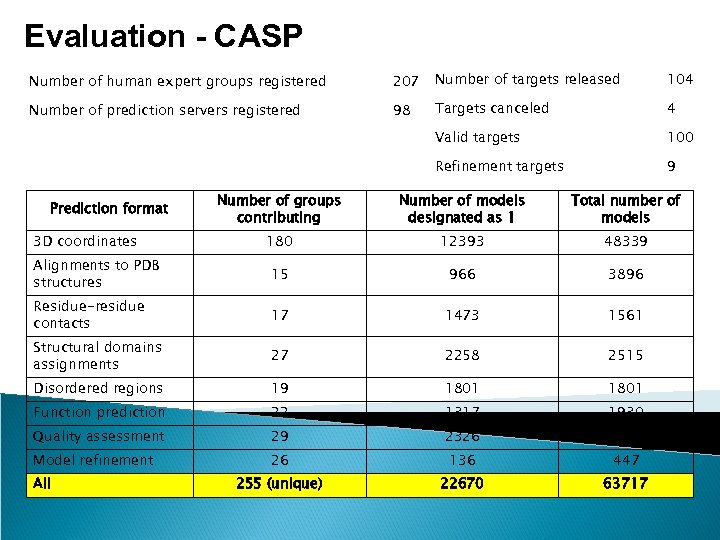 Evaluation - CASP Number of human expert groups registered 207 Number of targets released 104 Number of prediction servers registered 98 Targets canceled 4 Valid targets 100 Refinement targets 9 Number of groups contributing Number of models designated as 1 Total number of models 180 12393 48339 Alignments to PDB structures 15 966 3896 Residue-residue contacts 17 1473 1561 Structural domains assignments 27 2258 2515 Disordered regions 19 1801 Function prediction 22 1317 1930 Quality assessment 29 2326 3228 Model refinement 26 136 447 255 (unique) 22670 63717 Prediction format 3 D coordinates All
Evaluation - CASP Number of human expert groups registered 207 Number of targets released 104 Number of prediction servers registered 98 Targets canceled 4 Valid targets 100 Refinement targets 9 Number of groups contributing Number of models designated as 1 Total number of models 180 12393 48339 Alignments to PDB structures 15 966 3896 Residue-residue contacts 17 1473 1561 Structural domains assignments 27 2258 2515 Disordered regions 19 1801 Function prediction 22 1317 1930 Quality assessment 29 2326 3228 Model refinement 26 136 447 255 (unique) 22670 63717 Prediction format 3 D coordinates All
 Proteomics The term proteome was coined by Mark Wilkins in 1995 and is used to describe the entire complement of proteins in a given biological organism or system at a given time, i. e. the protein products of the genome. The term has been applied to several different types of biological systems. A cellular proteome is the collection of proteins found in a particular cell type under a particular set of environmental conditions such as exposure to hormone stimulation.
Proteomics The term proteome was coined by Mark Wilkins in 1995 and is used to describe the entire complement of proteins in a given biological organism or system at a given time, i. e. the protein products of the genome. The term has been applied to several different types of biological systems. A cellular proteome is the collection of proteins found in a particular cell type under a particular set of environmental conditions such as exposure to hormone stimulation.
 Proteomics vs. Genomics The proteome is larger than the genome, especially in eukaryotes, in the sense that there are more proteins than genes. This is due to alternative Splicing_(genetics) splicing of genes and post-translational modifications like glycosylation or phosphorylation. The proteome has at least two levels of complexity lacking in the genome. When the genome is defined by the sequence of nucleotides, the proteome cannot be limited to the sum of the sequences of the proteins present. Knowledge of the proteome requires knowledge of (1) the structure of the proteins in the proteome and (2) the functional interaction between the proteins.
Proteomics vs. Genomics The proteome is larger than the genome, especially in eukaryotes, in the sense that there are more proteins than genes. This is due to alternative Splicing_(genetics) splicing of genes and post-translational modifications like glycosylation or phosphorylation. The proteome has at least two levels of complexity lacking in the genome. When the genome is defined by the sequence of nucleotides, the proteome cannot be limited to the sum of the sequences of the proteins present. Knowledge of the proteome requires knowledge of (1) the structure of the proteins in the proteome and (2) the functional interaction between the proteins.
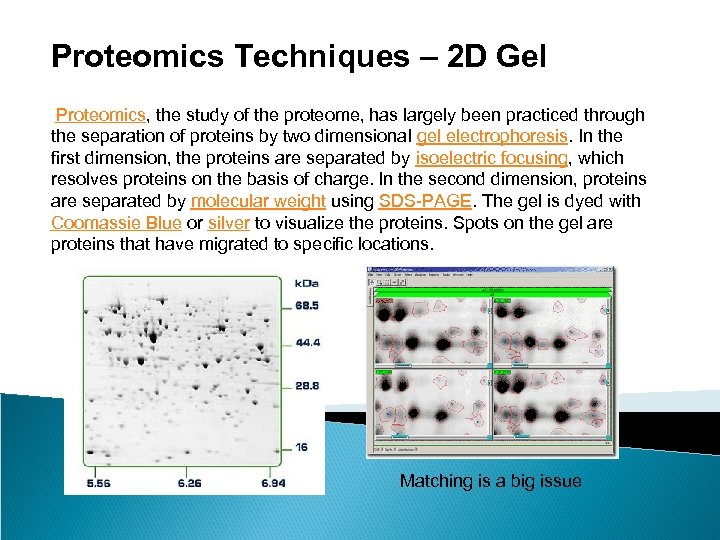 Proteomics Techniques – 2 D Gel Proteomics, the study of the proteome, has largely been practiced through the separation of proteins by two dimensional gel electrophoresis. In the first dimension, the proteins are separated by isoelectric focusing, which resolves proteins on the basis of charge. In the second dimension, proteins are separated by molecular weight using SDS-PAGE. The gel is dyed with Coomassie Blue or silver to visualize the proteins. Spots on the gel are proteins that have migrated to specific locations. Matching is a big issue
Proteomics Techniques – 2 D Gel Proteomics, the study of the proteome, has largely been practiced through the separation of proteins by two dimensional gel electrophoresis. In the first dimension, the proteins are separated by isoelectric focusing, which resolves proteins on the basis of charge. In the second dimension, proteins are separated by molecular weight using SDS-PAGE. The gel is dyed with Coomassie Blue or silver to visualize the proteins. Spots on the gel are proteins that have migrated to specific locations. Matching is a big issue
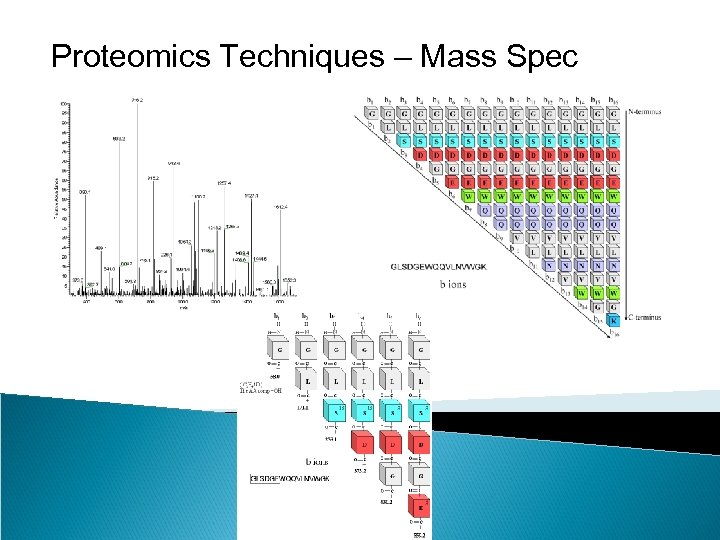
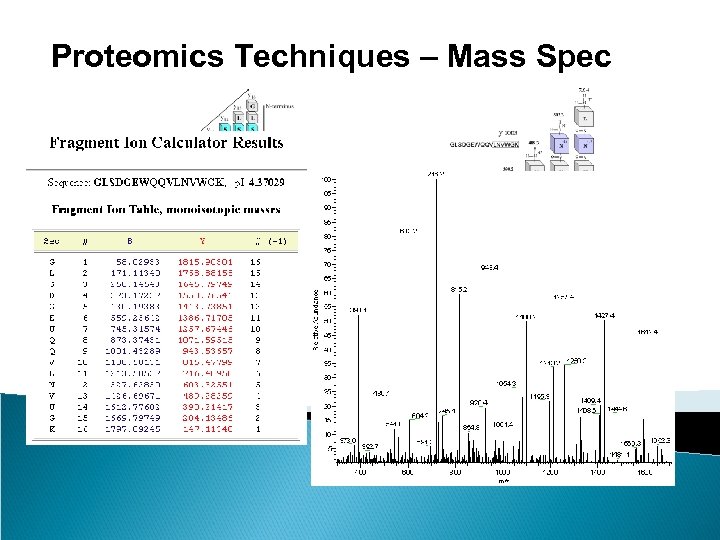
 Proteomics Techniques – Mass Spec Peptide mass fingerprinting identifies a protein by cleaving it into short peptides and then deduces the protein's identity by matching the observed peptide masses against a sequence database. Tandem mass spectrometry, on the other hand, can get sequence information from individual peptides by isolating them, colliding them with a nonreactive gas, and then cataloging the fragment ions produced.
Proteomics Techniques – Mass Spec Peptide mass fingerprinting identifies a protein by cleaving it into short peptides and then deduces the protein's identity by matching the observed peptide masses against a sequence database. Tandem mass spectrometry, on the other hand, can get sequence information from individual peptides by isolating them, colliding them with a nonreactive gas, and then cataloging the fragment ions produced.
 Proteomics Techniques – Mass Spec
Proteomics Techniques – Mass Spec
 Proteomics Techniques – Mass Spec
Proteomics Techniques – Mass Spec
 Proteomics Techniques – Microarray Measures m. RNA level, no change in m. RNA does not necessarily mean no change in protein expression and function due to effects of posttranslational modulation.
Proteomics Techniques – Microarray Measures m. RNA level, no change in m. RNA does not necessarily mean no change in protein expression and function due to effects of posttranslational modulation.


