1b864615646da8ad529b501d56119472.ppt
- Количество слайдов: 22

Prospective, Randomized Comparison of Heparin Plus IIb/IIIa Inhibition and Bivalirudin With or Without IIb/IIIa Inhibition in Patients with Acute Coronary Syndromes Gregg W. Stone MD for the ACUITY Investigators

Background: Current Management of ACS § Early invasive strategy if moderate-high risk 1, 2 § Median time to cath 21 hours 3 § Revascularization with PCI or CABG 1, 2 § 55% PCI, 12% CABG, 33% medical mgt 3 § Triple anti-platelet therapy 1, 2 § Aspirin § Clopidogrel (initiated pre or post angiography) § GP IIb/IIIa inhibitors - started upstream in all pts or in the CCL for PCI § Unfractionated or LMW heparin 1, 2, 4 1 Braunwald et al JACC 2002; 2 Bertrand et al. EHJ 2002; 3 www. crusade. org; 4 SYNERGY. JAMA 2004; 292: 45 -54

Bivalirudin as an Alternative to UFH/LMWH § Advantages of the direct thrombin inhibitor bivalirudin § No requirement for anti-thrombin III § Effective on clot-bound thrombin § Inhibits thrombin-mediated platelet activation § No interactions with PF-4 § Plasma half-life 25 minutes § No requirement for anticoagulant monitoring § Clinical results with bivalirudin in PCI § Similar protection from ischemic events as UFH + GP IIb/IIIa inhibitors, with markedly reduced bleeding 1 § Not previously tested in contemporary ACS patients REPLACE 2. Lincoff AM et al. JAMA 2003; 289: 853 -863

Bivalirudin in ACS: Hypotheses § In moderate-high risk patients with ACS undergoing an invasive strategy, compared to UFH or LMWH + GP IIb/IIIa inhibitors: • Bivalirudin + GP IIb/IIIa inhibitors will result in less adverse ischemic events and less bleeding • Bivalirudin alone will result in similar rates of ischemic events and markedly reduced bleeding
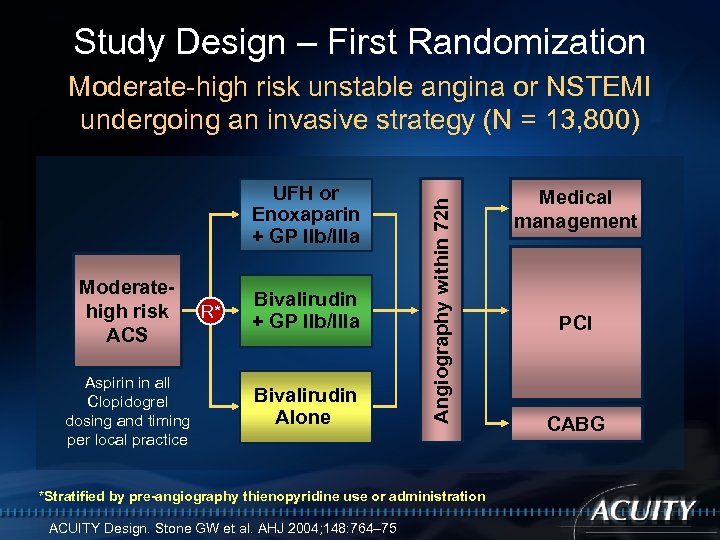
Study Design – First Randomization UFH or Enoxaparin + GP IIb/IIIa Moderatehigh risk ACS Aspirin in all Clopidogrel dosing and timing per local practice R* Bivalirudin + GP IIb/IIIa Bivalirudin Alone Angiography within 72 h Moderate-high risk unstable angina or NSTEMI undergoing an invasive strategy (N = 13, 800) *Stratified by pre-angiography thienopyridine use or administration ACUITY Design. Stone GW et al. AHJ 2004; 148: 764– 75 Medical management PCI CABG
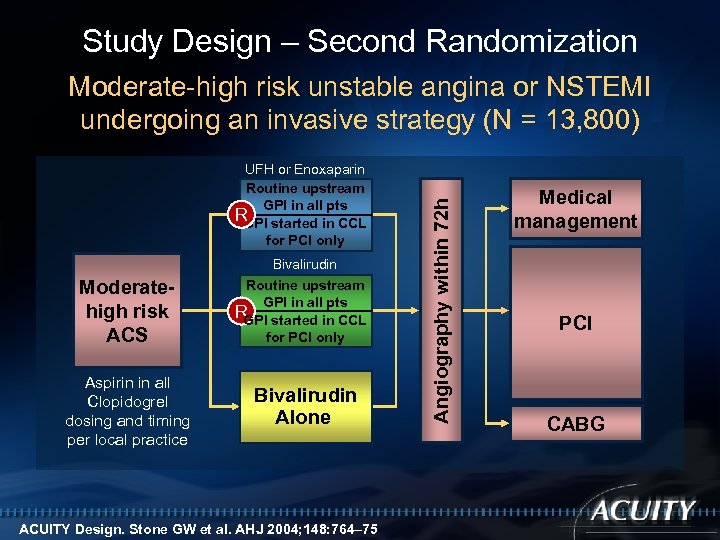
Study Design – Second Randomization UFH or Enoxaparin Routine upstream GPI in all pts R started in CCL GPI for PCI only Moderatehigh risk ACS Aspirin in all Clopidogrel dosing and timing per local practice Bivalirudin Routine upstream GPI in all pts R started in CCL GPI for PCI only Bivalirudin Alone ACUITY Design. Stone GW et al. AHJ 2004; 148: 764– 75 Angiography within 72 h Moderate-high risk unstable angina or NSTEMI undergoing an invasive strategy (N = 13, 800) Medical management PCI CABG

Major Entry Criteria Moderate-high risk unstable angina or NSTEMI Inclusion Criteria § Age ≥ 18 years § Chest pain ≥ 10’ within 24 h § At least one of: § New ST depression or transient ST elevation ≥ 1 mm § Troponin I, T, or CKMB § Documented CAD § All other 4 TIMI risk criteria - Age ≥ 65 years - Aspirin within 7 days - ≥ 2 angina episodes w/i 24 h - ≥ 3 cardiac risk factors § Written informed consent Exclusion Criteria § No angiography within 72 h § Acute STEMI or shock § Bleeding diathesis or major bleed within 2 weeks § Platelet count ≤ 100, 000/mm 3 § INR >1. 5 control § Cr. Cl ≤ 30 ml/min § Abcx or ≥ 2 prior LMWH doses § Prior UFH, LMWH (1 dose), eptifibatide and tirofiban were allowed § Allergy to drugs, contrast ACUITY Design. Stone GW et al. AHJ 2004; 148: 764– 75

Study Medications § Anti-thrombin agents (started pre angiography) UF Heparin U/Kg Enoxaparin mg/Kg Bivalirudin mg/kg Bolus 60 1. 0 sc bid 0. 1 iv Infusion/h 121 PCI CABG Medical mgt 0. 25 iv ACT 200 -250 s 0. 30 iv bolus 2 0. 75 iv bolus 3 0. 50 bolus iv 1. 75/h infusion iv 4 Per institution 5 None 6 Target a. PTT 50 -75 seconds If last enoxaparin dose ≥ 8 h - <16 h before PCI; 3 If maintenance dose discontinued or ≥ 16 h from last dose 4 Discontinued at end of PCI with option to continue at 0. 25 mg/kg for 4 -12 h if GPIIb/IIIa inhibitor not used 5 Bivalirudin option for off-pump same as PCI dose. For on-pump bivalirudin discontinued 2 hours before 6 Option to continue with pre-PCI anti-thrombotic regimen at physician discretion 1 2

3 Primary Endpoints (at 30 Days) 1. Composite net clinical benefit = 2. Ischemic composite or 3. Major bleeding § Death from any cause § Myocardial infarction - During medical Rx: Any biomarker elevation >ULN - Post PCI: CKMB >ULN with new Q waves or >3 x ULN w/o Q waves - Post CABG: CKMB >5 x ULN with new Q waves, >10 x ULN w/o Q waves § Unplanned revascularization for ischemia

3 Primary Endpoints (at 30 Days) 1. Composite net clinical benefit = 2. Ischemic composite or 3. Major bleeding § Non CABG related bleeding - Intracranial bleeding or intraocular bleeding - Retroperitoneal bleeding -Access site bleed requiring intervention/surgery - Hematoma ≥ 5 cm - Hgb ≥ 3 g/d. L with an overt source or ≥ 4 g/d. L w/o overt source - Blood product transfusion § Reoperation for bleeding

Statistical Plan § Anticipated event rates on UFH/Enox + GP IIb/IIIa § Ischemic composite: 6. 5% § Major bleeding: 9. 0% § Net clinical outcome: 12. 4% § Significance testing § Sequential non-inferiority and superiority tests § Non-inferiority tests: One-sided a=0. 025; =25% § Superiority tests: Two sided a=0. 05 § Hochberg procedure 1 used to control type I error § Sample size estimate = 13, 800 patients 1 Benjamini & Hochberg. J R Stat Soc 1995

Event Rates and Power Calculations UFH/Enox + GP IIb/IIIa Predicted Rate Endpoint Net clinical outcome 12. 4% Ischemic events 6. 5% Major bleeding 9. 0% NI = non-inferiority; Sup = superiority

Event Rates and Power Calculations UFH/Enox + GP IIb/IIIa Predicted Rate Bivalirudin + GP IIb/IIIa Rate Endpoint Power NI Sup Net clinical outcome 12. 4% 10. 3% 99% 88% Ischemic events 6. 5% 5. 3% 99% 67% Major bleeding 9. 0% 7. 5% 99% 73% NI = non-inferiority; Sup = superiority
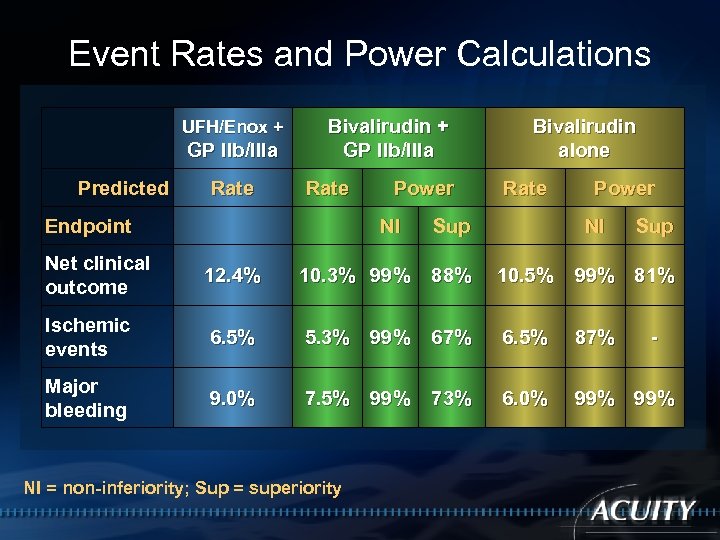
Event Rates and Power Calculations UFH/Enox + GP IIb/IIIa Predicted Rate Bivalirudin + GP IIb/IIIa Rate Endpoint Power NI Bivalirudin alone Rate Sup Power NI Sup Net clinical outcome 12. 4% 10. 3% 99% 88% 10. 5% 99% 81% Ischemic events 6. 5% 5. 3% 99% 67% 6. 5% 87% Major bleeding 9. 0% 7. 5% 99% 73% 6. 0% 99% NI = non-inferiority; Sup = superiority -

Study Organization Principal Investigator Gregg W. Stone Columbia University, NYC, NY Executive Committee Michel Bertrand Hosp. Cardiologique, Lambersart, France A. Michael Lincoff Cleveland Clinic, Cleveland, Ohio Jeffrey W. Moses Columbia University, NYC, NY Magnus Ohman Duke University, Durham, NC Harvey D. White Green Lane Hosp. , Auckland, NZ

Study Organization Steering Committee Frederick Feit (Chair) New York University NYC, NY Antonio Columbo (EU Chair) Ospedael San Raphael Milano, Italy Ramin Ebrahimi VA Medical Ctr West Los Angeles Los Angelas, CA Lars Hvilsted Rasmussen Alborg Sygehus, Afdeling Syd Alborg, Denmark Hans-Jürgen Rupprecht Gpr Klinikum Rüsselsheim, Germany Phil Aylward Flinders Medical Centre Bedford Park, Australia Angel Cequier Ciutat Sanitària Belvitge Barcelona, Spain Walter Desmet University Hospital, Gasthuisberg Leuven, Belgium Harold Darius Krankenhaus Neukölln Berlin, Germany Martial Hamon University Hospital Caen Cedex, France James Hoekstra Wake Forest University Lewisville, NC Charles V. Pollack Pennsylvania Hospital Philadelphia, PA

Study Organization Statistical Committee Stuart Pocock London School of Hygiene and Tropical Medicine, London, UK Jim Ware Harvard University, Boston, Mass. Data Monitoring The Medicines Company/Nycomed Data Management e. Trials/The Medicines Company Clinical Events Committee Roxana Mehran, Director Cardiovascular Research Foundation, NY Angio. Core Laboratory Alexandra Lansky, Director Health Economics and Cost-Effectiveness David J. Cohen (Chair) Cardiovascular Research Foundation, NY Beth Israel Deaconess, Boston, Mass.

Study Organization Biomarker Substudy George Dangas (Chair) Columbia University, NYC, NY W. Craig Hooper CDC, Atlanta, GA Steven R. Steinhubl University of Kentucky, Lexington, KY Data Safety and Monitoring Committee Bernard J. Gersh (Chair) Mayo Clinic, Rochester, Minn David Faxon Brigham & Women’s Hosp. , Boston, Mass. Spencer King Fuqua Heart Center, Atlanta, GA Stuart Pocock London, UK Hartzell Schaff Mayo Clinic, Rochester, Minn David O. Williams Rhode Island Hosp. , Providence, RI
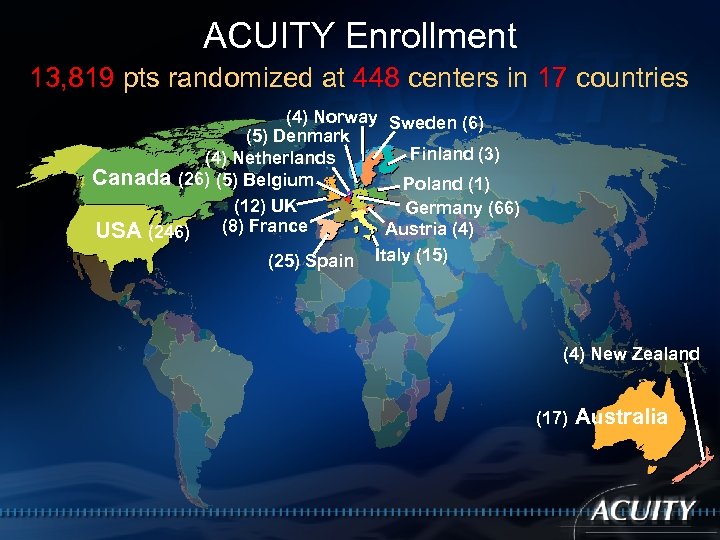
ACUITY Enrollment 13, 819 pts randomized at 448 centers in 17 countries (4) Norway Sweden (6) (5) Denmark Finland (3) (4) Netherlands Canada (26) (5) Belgium Poland (1) (12) UK Germany (66) Austria (4) USA (246) (8) France (25) Spain Italy (15) (4) New Zealand (17) Australia
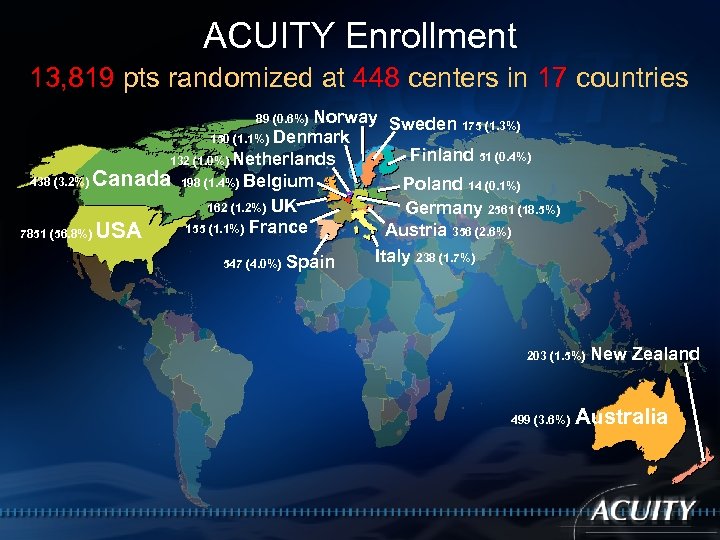
ACUITY Enrollment 13, 819 pts randomized at 448 centers in 17 countries Norway Sweden 175 (1. 3%) 150 (1. 1%) Denmark Finland 51 (0. 4%) 132 (1. 0%) Netherlands 438 (3. 2%) Canada 198 (1. 4%) Belgium Poland 14 (0. 1%) 162 (1. 2%) UK Germany 2561 (18. 5%) 155 (1. 1%) France Austria 356 (2. 6%) 7851 (56. 8%) USA Italy 238 (1. 7%) 547 (4. 0%) Spain 89 (0. 6%) 203 (1. 5%) 499 (3. 6%) New Zealand Australia

Top 20 Enrollers B. Mc. Laurin, MD R. H. Strasser, MD D. Cox, MD T. Stuckey, MD Anderson Area Medical Center, Anderson, SC Mid Carolina Cardiology, Charlotte, NC Technische Universitt Dresden, Germany Lebauer CV Research Foundation, Greensboro, NC M. Zubair Jafar, MD I. H. Lieber, MD H. Chandna, MD R. Zelman, MD F. Harmann, MD J. Neuzner, MD Hudson Valley Heart Center, Poughkeepsie, NY Victoria Heart and Vascular Center, Victoria, TX Universittsklilnik Schleswig. Holstein Campus Lbeck, Germany F. Leisch, MD Allgemeines ffentl. Krankenhaus der Landershauptstadt Linz, Austria M. Desaga, MD Klinikum Dachu der Landeshaupstadt Linz, Germany North Houston Heart Center, Kingwood, TX Cape Cod Research Institute, Hyannis, MA Klinikum Kassel Gmb. H, Kassel, GA K. Huber, MD Welhelminespital der Stadt Wien, Vienna, Austria H. White, MD Auckland City Hospital, Auckland, New Zealand M. Buerke, MD Klinikum der Martin-Luther. Universitt Halle-Wittenber, Halle, Germany S. Konstantinides, MD George-August-Universitt Gttingen Goettingen, Germany K. E. Hauptmann, MD Krankenhaus der Barmherzigen Bruder Medizinische Klinkill, Trier, Germany E. Mostel, MD Palm Beach Gardens Medical Center/Palm Beach Cardiology, Palm Beach Gardens, FL A. Cequier, MD Ciutat Santaria Bellvitge, Barcelona, Spain M. Mockel, MD Charite Universitatsmedizin Berlin, Germany
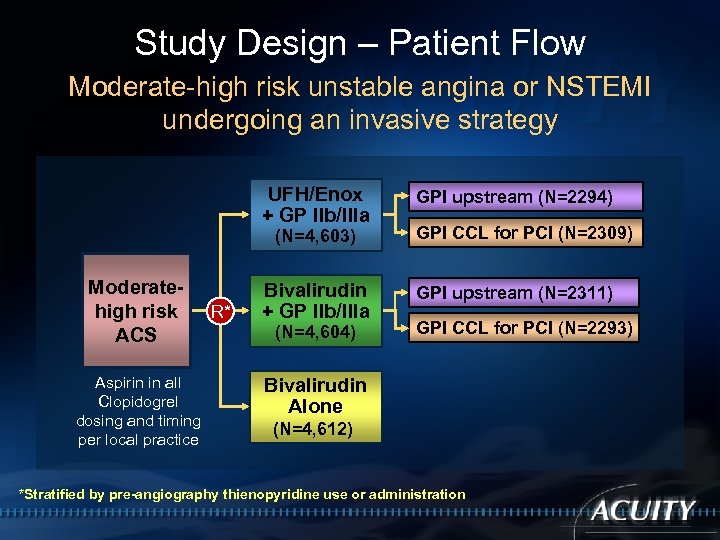
Study Design – Patient Flow Moderate-high risk unstable angina or NSTEMI undergoing an invasive strategy UFH/Enox + GP IIb/IIIa GPI upstream (N=2294) Bivalirudin + GP IIb/IIIa GPI upstream (N=2311) (N=4, 603) Moderatehigh risk ACS Aspirin in all Clopidogrel dosing and timing per local practice R* (N=4, 604) GPI CCL for PCI (N=2309) GPI CCL for PCI (N=2293) Bivalirudin Alone (N=4, 612) *Stratified by pre-angiography thienopyridine use or administration
1b864615646da8ad529b501d56119472.ppt