b6d3b348b86300ab24e552ac4bb812eb.ppt
- Количество слайдов: 9
 PROFICIENCY TESTING OF IN-HOUSE NAT ASSAYS USED FOR BLOOD SCREENING XXI So. GAT International Working Group Meeting on the Standardization of NAT for the Safety Testing of Blood, Tissues and Organs for Blood-Borne Pathogens 28 - 29 May 2009 Brussels, Belgium Julia Kreß Michael Chudy Micha Nübling Paul-Ehrlich-Institut, Langen, Germany WHO Collaborating Centre for Quality Assurance of Blood Products and in vitro Diagnostic Devices
PROFICIENCY TESTING OF IN-HOUSE NAT ASSAYS USED FOR BLOOD SCREENING XXI So. GAT International Working Group Meeting on the Standardization of NAT for the Safety Testing of Blood, Tissues and Organs for Blood-Borne Pathogens 28 - 29 May 2009 Brussels, Belgium Julia Kreß Michael Chudy Micha Nübling Paul-Ehrlich-Institut, Langen, Germany WHO Collaborating Centre for Quality Assurance of Blood Products and in vitro Diagnostic Devices
 German Regulations for NAT Blood Donor Screening § 1999: first mandatory NAT was introduced for HCV (< 5. 000 IU/m. L ID) § 2004: NAT was implemented for HIV-1 (< 10. 000 IU/m. L ID) § HBV NAT is voluntarily performed by many blood donation services § in-house developed NAT assays, CE-marked diagnostic assays (off-label-use) and CE-marked NAT screening assays may be used § assays are validated for the individual pool size (10 to 96 donations) § validation studies are assessed by PEI 1 § NAT systems undergo regular external quality assessment programs organised by PEI
German Regulations for NAT Blood Donor Screening § 1999: first mandatory NAT was introduced for HCV (< 5. 000 IU/m. L ID) § 2004: NAT was implemented for HIV-1 (< 10. 000 IU/m. L ID) § HBV NAT is voluntarily performed by many blood donation services § in-house developed NAT assays, CE-marked diagnostic assays (off-label-use) and CE-marked NAT screening assays may be used § assays are validated for the individual pool size (10 to 96 donations) § validation studies are assessed by PEI 1 § NAT systems undergo regular external quality assessment programs organised by PEI
 In-house NAT Proficiency Study 2008: Objective § verification of the efficiency of in-house NAT assays for the detection of HCV, HIV-1 and HBV in blood donations regarding § § analytical sensitivity genotype / subtype sensitivity specificity reproducibility § the participation in the proficiency study is mandatory for HCV and HIV-1 NATs, voluntary for HBV NAT 2
In-house NAT Proficiency Study 2008: Objective § verification of the efficiency of in-house NAT assays for the detection of HCV, HIV-1 and HBV in blood donations regarding § § analytical sensitivity genotype / subtype sensitivity specificity reproducibility § the participation in the proficiency study is mandatory for HCV and HIV-1 NATs, voluntary for HBV NAT 2
 In-house NAT Proficiency Study 2008: Study Design 3 § detection limit of the HCV, HIV-1 and HBV NATs with respect to the pool size § testing panels: calibrated PEI reference preparation, two positive materials and negative plasma § HIV-1 -samples: one missed by CTM v 1, one with discrepant results § 0. 5 log dilution series starting with the required minimum sensitivity § characterization of panels by CE-certified NAT screening systems: cobas Taq. Screen MPX Test, Procleix Ultrio Assay § dilution of samples individually for each lab simulating the pool size § encoding of labs and samples § sample shipment on dry ice
In-house NAT Proficiency Study 2008: Study Design 3 § detection limit of the HCV, HIV-1 and HBV NATs with respect to the pool size § testing panels: calibrated PEI reference preparation, two positive materials and negative plasma § HIV-1 -samples: one missed by CTM v 1, one with discrepant results § 0. 5 log dilution series starting with the required minimum sensitivity § characterization of panels by CE-certified NAT screening systems: cobas Taq. Screen MPX Test, Procleix Ultrio Assay § dilution of samples individually for each lab simulating the pool size § encoding of labs and samples § sample shipment on dry ice
 In-house NAT Proficiency Study 2008: Participants § Invited laboratories: § in-house NAT screening assays (non-CE-marked / CEmarked) § CE-marked diagnostic assays used for screening (offlabel-use) § CE-marked screening system with large pool sizes (48, 96) § HCV: 30 labs 16 in-house NATs, 16 diagnostic assays, 3 cobas Taq. Screen MPX (5 labs: 2 different methods) § HIV-1: 30 labs 15 in-house NATs, 15 diagnostic assays, 3 cobas Taq. Screen MPX (3 labs: 2 different methods) 4 § HBV: 21 labs 14 in-house NATs, 6 diagnostic assays, 3 cobas
In-house NAT Proficiency Study 2008: Participants § Invited laboratories: § in-house NAT screening assays (non-CE-marked / CEmarked) § CE-marked diagnostic assays used for screening (offlabel-use) § CE-marked screening system with large pool sizes (48, 96) § HCV: 30 labs 16 in-house NATs, 16 diagnostic assays, 3 cobas Taq. Screen MPX (5 labs: 2 different methods) § HIV-1: 30 labs 15 in-house NATs, 15 diagnostic assays, 3 cobas Taq. Screen MPX (3 labs: 2 different methods) 4 § HBV: 21 labs 14 in-house NATs, 6 diagnostic assays, 3 cobas
 In-house NAT Proficiency Study 2008: HCV Results 5
In-house NAT Proficiency Study 2008: HCV Results 5
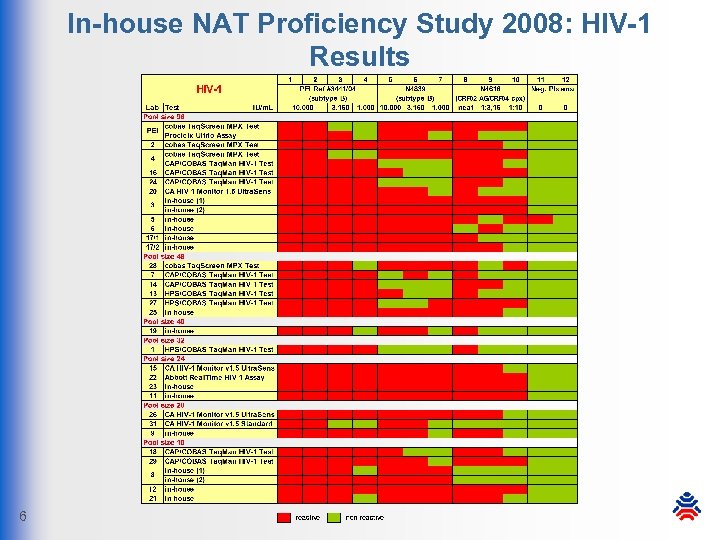 In-house NAT Proficiency Study 2008: HIV-1 Results 6
In-house NAT Proficiency Study 2008: HIV-1 Results 6
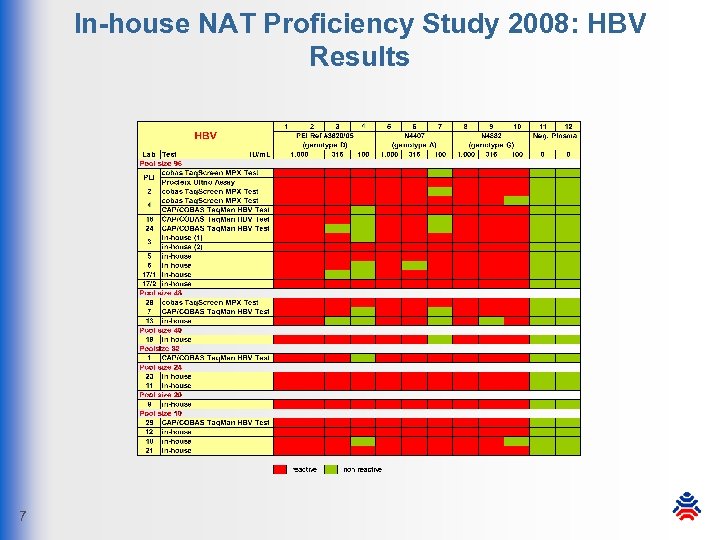 In-house NAT Proficiency Study 2008: HBV Results 7
In-house NAT Proficiency Study 2008: HBV Results 7
 In-house NAT Proficiency Study 2008: Conclusion § all participants meet the PEI sensitivity requirements § in most labs NAT assays show higher sensitivity than required § high specificity: only 1 false-positive result by 1 lab § HIV-1: CTM v 1 -missed sample was detected by other systems § due to high mutation rates there is a certain amount of risk that infectious donations are missed by HIV-1 NATs § voluntary HBV NAT assays: high sensitivity and specificity 8 § in-house NAT systems and off-label-use systems under proper conditions are still suitable for blood donor
In-house NAT Proficiency Study 2008: Conclusion § all participants meet the PEI sensitivity requirements § in most labs NAT assays show higher sensitivity than required § high specificity: only 1 false-positive result by 1 lab § HIV-1: CTM v 1 -missed sample was detected by other systems § due to high mutation rates there is a certain amount of risk that infectious donations are missed by HIV-1 NATs § voluntary HBV NAT assays: high sensitivity and specificity 8 § in-house NAT systems and off-label-use systems under proper conditions are still suitable for blood donor


