e2e8377fe0aa515c2c545e89a8f8eda5.ppt
- Количество слайдов: 21
 PRIORITY MEDICINES FOR EUROPE AND THE WORLD for the A report prepared by WHO Netherlands Government by Warren Kaplan Richard Laing and Saloni Tanna Eduardo Sabaté Joyce Wilson Ann Wilberforce Marjolein Willemen Monique Renevier Lisa Greenough Kathy Hurst
PRIORITY MEDICINES FOR EUROPE AND THE WORLD for the A report prepared by WHO Netherlands Government by Warren Kaplan Richard Laing and Saloni Tanna Eduardo Sabaté Joyce Wilson Ann Wilberforce Marjolein Willemen Monique Renevier Lisa Greenough Kathy Hurst
 Objectives of Priority Medicines Project • Provide a methodology for identifying pharmaceutical “gaps” from a public health perspective, for Europe and the World. • Provide a public-health based pharmaceutical R&D agenda for use by the EU in the 7 th Framework Programme, “Good public policy should spend public funds on areas of greatest public needs”
Objectives of Priority Medicines Project • Provide a methodology for identifying pharmaceutical “gaps” from a public health perspective, for Europe and the World. • Provide a public-health based pharmaceutical R&D agenda for use by the EU in the 7 th Framework Programme, “Good public policy should spend public funds on areas of greatest public needs”
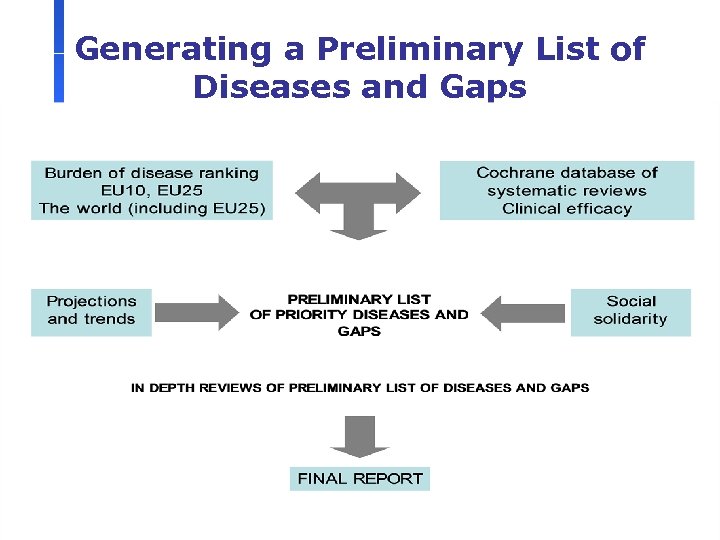 Generating a Preliminary List of Diseases and Gaps
Generating a Preliminary List of Diseases and Gaps
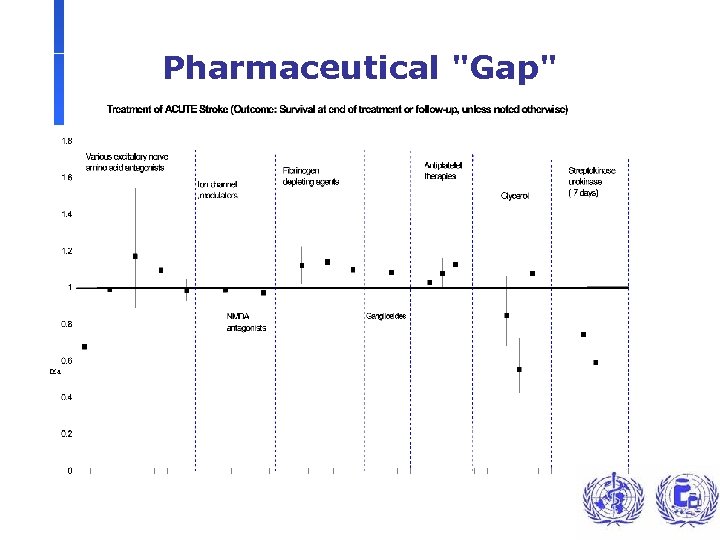 Pharmaceutical "Gap"
Pharmaceutical "Gap"
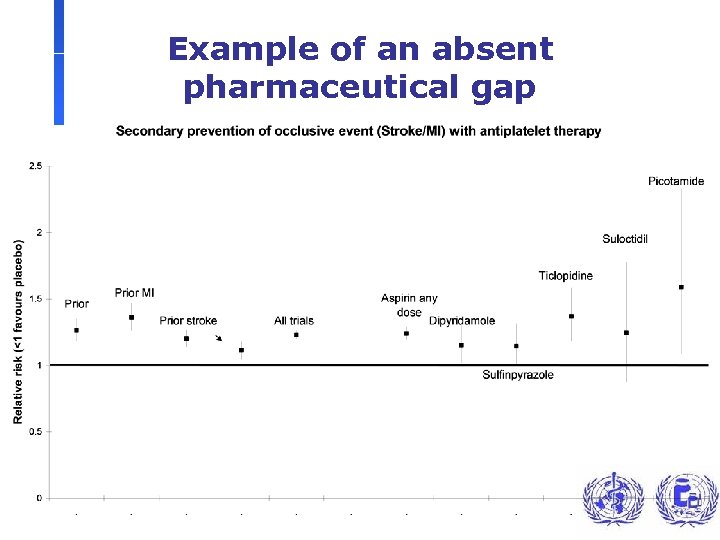 Example of an absent pharmaceutical gap
Example of an absent pharmaceutical gap
 "Commonality of interest"
"Commonality of interest"
 Special Needs for Women, Children, and the Elderly • All groups neglected in drug development • Complicated by different physiology & metabolism • Recent improvements in situation of women and children • Considerable gaps remain for the elderly who use the most medicines
Special Needs for Women, Children, and the Elderly • All groups neglected in drug development • Complicated by different physiology & metabolism • Recent improvements in situation of women and children • Considerable gaps remain for the elderly who use the most medicines
 Determining Value for Innovation and Setting Prices Octavia Quintana Trias, DG Research "How do you value innovation? Special thanks to David Henry, Sue Hill & Danielle Lang
Determining Value for Innovation and Setting Prices Octavia Quintana Trias, DG Research "How do you value innovation? Special thanks to David Henry, Sue Hill & Danielle Lang
 Mr Günter VERHEUGEN (Enterprise and Industry) September 2004 "As for the economic aspects of medicinal products, the Commission could, following deliberations on pricing and reimbursement methods, play a part in developing alternative approaches aimed at promoting innovation and making it financially rewarding. Similarly, questions concerning national procedures for determining therapeutic value of new medicinal products must also be tackled. "
Mr Günter VERHEUGEN (Enterprise and Industry) September 2004 "As for the economic aspects of medicinal products, the Commission could, following deliberations on pricing and reimbursement methods, play a part in developing alternative approaches aimed at promoting innovation and making it financially rewarding. Similarly, questions concerning national procedures for determining therapeutic value of new medicinal products must also be tackled. "
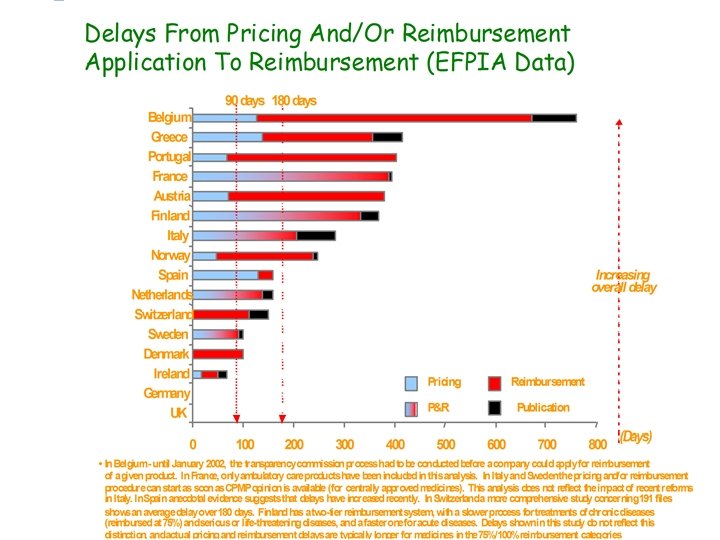
 The Problem • European government have a dual role as purchasers and in promoting innovation • All OECD countries except USA regulate pharmaceutical prices using up to five different methods (Jacobzone) • Price setting process is unpredictable for level and time to a decision (“Black box”) • Pharmaceuticals are among the most highly regulated products • Pharmaceuticals are only a small proportion of total health care costs in developed countries www. pmprb-cepmb. gc. ca/symposium 2002/ present/Jacobzone-Oct 7 -OECD/
The Problem • European government have a dual role as purchasers and in promoting innovation • All OECD countries except USA regulate pharmaceutical prices using up to five different methods (Jacobzone) • Price setting process is unpredictable for level and time to a decision (“Black box”) • Pharmaceuticals are among the most highly regulated products • Pharmaceuticals are only a small proportion of total health care costs in developed countries www. pmprb-cepmb. gc. ca/symposium 2002/ present/Jacobzone-Oct 7 -OECD/
 Differential Pricing and Parallel Trade • Differential pricing already occurs for many consumer products • For pharmaceuticals dramatic differences already exist between countries for vaccines, oral contraceptives, antimalarials and ARVs • Economic theory indicates that where the true marginal costs of a product are low, price discrimination will increase the total revenue of a company, so long as parallel trade can be minimized • A number of technical solutions have been proposed to limit pharmaceutical parallel trade, such as unique presentation and country-specific labelling and packaging measures
Differential Pricing and Parallel Trade • Differential pricing already occurs for many consumer products • For pharmaceuticals dramatic differences already exist between countries for vaccines, oral contraceptives, antimalarials and ARVs • Economic theory indicates that where the true marginal costs of a product are low, price discrimination will increase the total revenue of a company, so long as parallel trade can be minimized • A number of technical solutions have been proposed to limit pharmaceutical parallel trade, such as unique presentation and country-specific labelling and packaging measures
 Parallel Trade - The Evidence Panos Kanavos (LSE) 2003 • Examined 19 top-selling drugs in six Northern European "destination countries" of imports -- account for almost a quarter of all prescription drug sales in Europe. • Combined sales of reimported versions of these drugs saved 0. 3 percent to 3. 6 % of their annual drug budgets, or slightly more than $100 million. • Savings were not passed on to consumers • Drug prices in Northern and Southern Europe did not become more similar. • The biggest gains from selling imported drugs (46% of the sales of all products studied) went straight to the repackagers. http: //www. washingtonpost. com/wp-dyn/articles/A 60984 -2004 Sep 29. html
Parallel Trade - The Evidence Panos Kanavos (LSE) 2003 • Examined 19 top-selling drugs in six Northern European "destination countries" of imports -- account for almost a quarter of all prescription drug sales in Europe. • Combined sales of reimported versions of these drugs saved 0. 3 percent to 3. 6 % of their annual drug budgets, or slightly more than $100 million. • Savings were not passed on to consumers • Drug prices in Northern and Southern Europe did not become more similar. • The biggest gains from selling imported drugs (46% of the sales of all products studied) went straight to the repackagers. http: //www. washingtonpost. com/wp-dyn/articles/A 60984 -2004 Sep 29. html
 Spreading the Burden of Drug Development Costs and Valuing new medicines • Rewarding innovation is a key aspect of pricing i. e. real innovation should be rewarded! • Pricing should reflect ability to pay possibly measured as GNI per capita • Pharmaco-economics already provides the methods for reactive price setting and could be used proactively
Spreading the Burden of Drug Development Costs and Valuing new medicines • Rewarding innovation is a key aspect of pricing i. e. real innovation should be rewarded! • Pricing should reflect ability to pay possibly measured as GNI per capita • Pharmaco-economics already provides the methods for reactive price setting and could be used proactively
 Proposed Method (Henry 2002) • The World Bank has suggested that health care interventions may be considered cost -effective if they buy a year of healthy life for less than the national average per capita GNI • The method proposed for further evaluation is based on GNI per capita and efficacy measures
Proposed Method (Henry 2002) • The World Bank has suggested that health care interventions may be considered cost -effective if they buy a year of healthy life for less than the national average per capita GNI • The method proposed for further evaluation is based on GNI per capita and efficacy measures
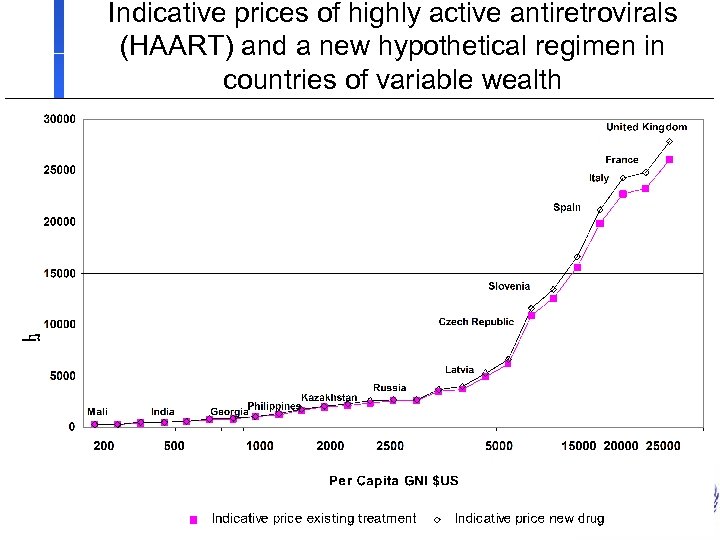 Indicative prices of highly active antiretrovirals (HAART) and a new hypothetical regimen in countries of variable wealth
Indicative prices of highly active antiretrovirals (HAART) and a new hypothetical regimen in countries of variable wealth
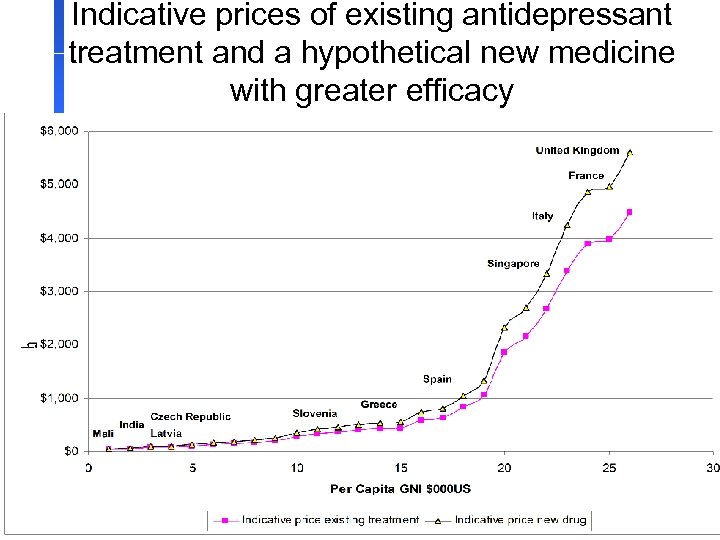 Indicative prices of existing antidepressant treatment and a hypothetical new medicine with greater efficacy
Indicative prices of existing antidepressant treatment and a hypothetical new medicine with greater efficacy
 Conclusions (1) • Possible to incorporate measures of clinical performance and national wealth in determining indicative prices for pharmaceuticals. • Does not mean that the indicative price is necessarily the ‘right’ price, the lowest possible price or an affordable price. • No guarantee that manufacturers will be interested in manufacturing products on this basis.
Conclusions (1) • Possible to incorporate measures of clinical performance and national wealth in determining indicative prices for pharmaceuticals. • Does not mean that the indicative price is necessarily the ‘right’ price, the lowest possible price or an affordable price. • No guarantee that manufacturers will be interested in manufacturing products on this basis.
 Conclusions (2) • A more predictable system that encourages medicines development and achieves affordability of access in countries of variable wealth requires reconsideration of current policies towards parallel trade. • Parallel imports tend to favour high-income countries, which can benefit from the prices paid in lower-income countries. May hurt lower income countries. Reference pricing encourages higher prices for poorer countries • Any inhibition of parallel trade conflicts with European law, which highlights the need for a wide debate & research on these issues.
Conclusions (2) • A more predictable system that encourages medicines development and achieves affordability of access in countries of variable wealth requires reconsideration of current policies towards parallel trade. • Parallel imports tend to favour high-income countries, which can benefit from the prices paid in lower-income countries. May hurt lower income countries. Reference pricing encourages higher prices for poorer countries • Any inhibition of parallel trade conflicts with European law, which highlights the need for a wide debate & research on these issues.
 Reviewer Comments • “The model is theoretically interesting but impractical. ” • “There is such a strong commitment to the ideal of a European Common Market that any restrictions on parallel trade would be unacceptable. ” • “Technical requirements for such an approach would be impossible to undertake proactively. ”
Reviewer Comments • “The model is theoretically interesting but impractical. ” • “There is such a strong commitment to the ideal of a European Common Market that any restrictions on parallel trade would be unacceptable. ” • “Technical requirements for such an approach would be impossible to undertake proactively. ”
 Final Summary • “If rewarding innovation is one of the intended effects of reimbursement policies, then setting prices related to the level of clinical efficacy and the national wealth as measured by GNI per capita would be a logical and transparent approach. ” • A researchable topic!
Final Summary • “If rewarding innovation is one of the intended effects of reimbursement policies, then setting prices related to the level of clinical efficacy and the national wealth as measured by GNI per capita would be a logical and transparent approach. ” • A researchable topic!


