b703f3e8493740b45d8ff6f36d9cdf61.ppt
- Количество слайдов: 49
 Prior Art Senior Design Course City College, Fall 2007
Prior Art Senior Design Course City College, Fall 2007
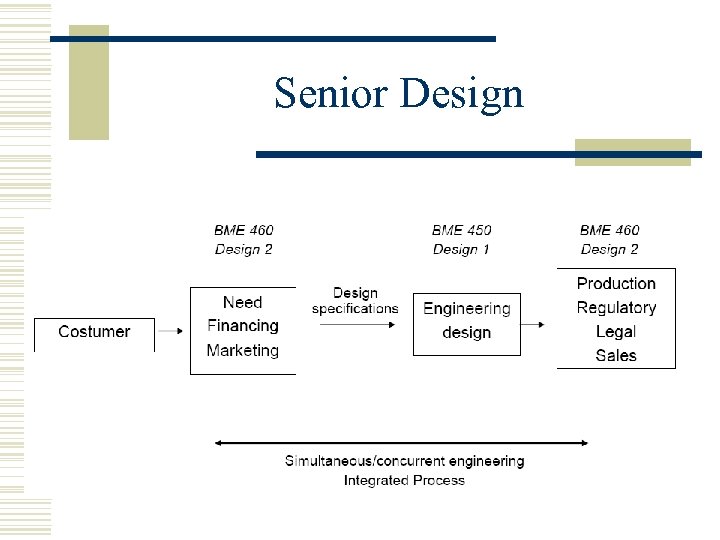 Senior Design
Senior Design
 Main steps in Design Process w 1. Determine design specifications / requirements, understand the problem. w 2. Prepare a project proposal. w 3. Prepare an initial timeline for the project. w 4. Consider existing/alternative solutions, prior art search. w 5. Generate multiple (alternative) solutions
Main steps in Design Process w 1. Determine design specifications / requirements, understand the problem. w 2. Prepare a project proposal. w 3. Prepare an initial timeline for the project. w 4. Consider existing/alternative solutions, prior art search. w 5. Generate multiple (alternative) solutions
 Main steps in Design Process w 6. Evaluate alternatives by comparing them to design requirements and to each other. Steps 5 -6 are iterative and can be conducted using: n n n a) Brain-storming, block-diagram, paper design b) Engineering/quantitative analysis (e. g. computer modeling/simulation) c) Prototyping, components and different levels of abstraction w 7. Decide on applicable solution w 8. Communicate results w The entire design process must be carefully documented.
Main steps in Design Process w 6. Evaluate alternatives by comparing them to design requirements and to each other. Steps 5 -6 are iterative and can be conducted using: n n n a) Brain-storming, block-diagram, paper design b) Engineering/quantitative analysis (e. g. computer modeling/simulation) c) Prototyping, components and different levels of abstraction w 7. Decide on applicable solution w 8. Communicate results w The entire design process must be carefully documented.
 Prior Art w Generally, an invention is something that is found by reaching out into the unknown. w Since an invention cannot be defined by describing something that is still unknown, the only alternative is to state what is not an invention. w This is done by defining what is in the prior art.
Prior Art w Generally, an invention is something that is found by reaching out into the unknown. w Since an invention cannot be defined by describing something that is still unknown, the only alternative is to state what is not an invention. w This is done by defining what is in the prior art.
 Prior Art w Prior art may be defined very broadly as the entire body of knowledge from the beginning of time to the present. w In most patent laws, prior art or state of the art is all information that has been disclosed to the public in any form before a given date.
Prior Art w Prior art may be defined very broadly as the entire body of knowledge from the beginning of time to the present. w In most patent laws, prior art or state of the art is all information that has been disclosed to the public in any form before a given date.
 Prior Art w However, under United States patent law, secret prior art, such as secret sales, qualifies as prior art in certain circumstances. w In Europe, prior art does not include information kept secret, whether from trade secrecy or just a simple lack of interest in publication.
Prior Art w However, under United States patent law, secret prior art, such as secret sales, qualifies as prior art in certain circumstances. w In Europe, prior art does not include information kept secret, whether from trade secrecy or just a simple lack of interest in publication.
 Prior Art w In most patent laws, prior art is expected to provide a description sufficient to inform the average worker in the field (or the man skilled in the art), published in fixed form and made available in public libraries.
Prior Art w In most patent laws, prior art is expected to provide a description sufficient to inform the average worker in the field (or the man skilled in the art), published in fixed form and made available in public libraries.
 Prior Art w In most patent laws, prior art does not include unpublished work or mere conversations (though according to the Europen Patent Convention, oral disclosures also form prior art – see Art. 54(2) EPC). It is disputed whether traditional knowledge (e. g. of medical properties of a certain plant) constitutes prior art.
Prior Art w In most patent laws, prior art does not include unpublished work or mere conversations (though according to the Europen Patent Convention, oral disclosures also form prior art – see Art. 54(2) EPC). It is disputed whether traditional knowledge (e. g. of medical properties of a certain plant) constitutes prior art.
 Prior Art w The term prior art is mainly used in the patent field. Patents disclose to society how an invention is practiced, in return for the right (during a limited term) to exclude others from manufacturing, selling, offering for sale or using the patented invention without the patentee's permission.
Prior Art w The term prior art is mainly used in the patent field. Patents disclose to society how an invention is practiced, in return for the right (during a limited term) to exclude others from manufacturing, selling, offering for sale or using the patented invention without the patentee's permission.
 Prior Art w Patent offices deal with prior art searches in the context of the patent granting procedure. To assess the validity of a patent application, patent offices explore the prior art that was disclosed before the invention occurred (in the United States and all first-to-invent patent systems) or before the filing date (in Europe and all first-to-file patent systems).
Prior Art w Patent offices deal with prior art searches in the context of the patent granting procedure. To assess the validity of a patent application, patent offices explore the prior art that was disclosed before the invention occurred (in the United States and all first-to-invent patent systems) or before the filing date (in Europe and all first-to-file patent systems).
 The jet engine w Hero, an Egyptian scientist from Alexandria, developed the first "jet engine" about the year 100 BC. Known as the "aeolipile"
The jet engine w Hero, an Egyptian scientist from Alexandria, developed the first "jet engine" about the year 100 BC. Known as the "aeolipile"
 The jet engine w Leonardo Da Vinci designed a device called the "chimney jack" around the year 1500 AD. The chimney jack was used to turn a roasting skewer. This reaction-type turbine worked on the principle of heat raising gases from the roasting tire. As the hot air rose, it passed through fanlike blades that turned the roast through a series of gears
The jet engine w Leonardo Da Vinci designed a device called the "chimney jack" around the year 1500 AD. The chimney jack was used to turn a roasting skewer. This reaction-type turbine worked on the principle of heat raising gases from the roasting tire. As the hot air rose, it passed through fanlike blades that turned the roast through a series of gears
 The jet engine w In 1629 an Italian engineer, Giovanni Branca, was probably the first to invent an actual impulse turbine. This device, a stamping mill, was generated by a steam-powered turbine. A jet nozzle directed steam onto a horizontally mounted turbine wheel, which then turned an arrangement of gears that operated his mill
The jet engine w In 1629 an Italian engineer, Giovanni Branca, was probably the first to invent an actual impulse turbine. This device, a stamping mill, was generated by a steam-powered turbine. A jet nozzle directed steam onto a horizontally mounted turbine wheel, which then turned an arrangement of gears that operated his mill
 w In 1687 Isaac Newton attempted to put his newly formulated laws of motion to the test with his "steam wagon". He tried to propel the wagon by directing steam through a nozzle pointed rearward. Steam was produced by a boiler mounted on the wagon. Due to lack of power from the steam, this vehicle didn't operate
w In 1687 Isaac Newton attempted to put his newly formulated laws of motion to the test with his "steam wagon". He tried to propel the wagon by directing steam through a nozzle pointed rearward. Steam was produced by a boiler mounted on the wagon. Due to lack of power from the steam, this vehicle didn't operate
 The jet engine w In 1791 John Barber, was the first to patent a design that used thermodynamic cycle of the modem gas turbine. His design contained the basics of the modem gas turbine it had a compressor, a combustion chamber, and a turbine. The main difference in his design was that the turbine was equipped with a chain-driven reciprocating type of compressor. He intended its use for jet propulsion
The jet engine w In 1791 John Barber, was the first to patent a design that used thermodynamic cycle of the modem gas turbine. His design contained the basics of the modem gas turbine it had a compressor, a combustion chamber, and a turbine. The main difference in his design was that the turbine was equipped with a chain-driven reciprocating type of compressor. He intended its use for jet propulsion
 w In January 1930 an Englishman, Frank Whittle, submitted a patent application for a gas turbine for jet propulsion. It wasn't until the summer of 1939 that the Air Ministry awarded Power Jets Ltd a contract to design a flight engine. In May 1941 the Whittle W 1 engine made its first flight mounted in the Gloster Model E 28/39 aircraft. This airplane would later achieve a speed of 370 MPH in level flight with 1000 pounds of thrust.
w In January 1930 an Englishman, Frank Whittle, submitted a patent application for a gas turbine for jet propulsion. It wasn't until the summer of 1939 that the Air Ministry awarded Power Jets Ltd a contract to design a flight engine. In May 1941 the Whittle W 1 engine made its first flight mounted in the Gloster Model E 28/39 aircraft. This airplane would later achieve a speed of 370 MPH in level flight with 1000 pounds of thrust.
 Where? w Scientific Journals w Design Magazines w Patent Offices w Internet w Company web sites w Design books, texts, software, databases, etc
Where? w Scientific Journals w Design Magazines w Patent Offices w Internet w Company web sites w Design books, texts, software, databases, etc
 Scientific Journals w BME design related journals as Anti-cancer drug design, Artificial intelligence for engineering design, analysis and manufacturing, Computer aided design, Design automation for embedded systems, Design engineering, Design management review, Design news, Design Patents, Design studies, Drug design and discovery, Electronic design, Finite elements in analysis and design, Formal methods in system design, Global design news, Hospitality design, ID : magazine of international design (New York, N. Y. 1984), …
Scientific Journals w BME design related journals as Anti-cancer drug design, Artificial intelligence for engineering design, analysis and manufacturing, Computer aided design, Design automation for embedded systems, Design engineering, Design management review, Design news, Design Patents, Design studies, Drug design and discovery, Electronic design, Finite elements in analysis and design, Formal methods in system design, Global design news, Hospitality design, ID : magazine of international design (New York, N. Y. 1984), …
 Scientific Journals w International Journal of Design Computing, International journal of mechanics and materials in design, Internet electronic journal of molecular design, Journal of computer-aided materials design, Journal of engineering design, Journal of sustainable product design, Materials & design, Mechanics based design of structures and machines, Printed circuit design & manufacture, Research in engineering design, SBS digital design)
Scientific Journals w International Journal of Design Computing, International journal of mechanics and materials in design, Internet electronic journal of molecular design, Journal of computer-aided materials design, Journal of engineering design, Journal of sustainable product design, Materials & design, Mechanics based design of structures and machines, Printed circuit design & manufacture, Research in engineering design, SBS digital design)
 Design Magazines w BME design related magazines as http: //www. medicaldesign. com/, Medical Device & Diagnostic Industry, Medical Product Manufacturing News, IVD Technology, MX, European Medical Device Manufacturer, Medical Electronics Manufacturing, Pharmaceutical & Medical Packaging News, Medical design technology, Medical Device & Diagnostic Industry magazine, etc)
Design Magazines w BME design related magazines as http: //www. medicaldesign. com/, Medical Device & Diagnostic Industry, Medical Product Manufacturing News, IVD Technology, MX, European Medical Device Manufacturer, Medical Electronics Manufacturing, Pharmaceutical & Medical Packaging News, Medical design technology, Medical Device & Diagnostic Industry magazine, etc)
 Medicaldesign. com
Medicaldesign. com


 Patent and regulation Offices w http: //devicelink. com/links/regulatory. html w Intellectual Property Sites: n n www. patentcafe. com - a good site for general inventor info, www. uspto. gov This is the official Web site of the United States Patent and Trademark Office, an Agency of the US Department of Commerce.
Patent and regulation Offices w http: //devicelink. com/links/regulatory. html w Intellectual Property Sites: n n www. patentcafe. com - a good site for general inventor info, www. uspto. gov This is the official Web site of the United States Patent and Trademark Office, an Agency of the US Department of Commerce.
 Patent & Standards Organizations w www. fda. gov/cdrh/ Food and drug administration w Association for the Advancement of Medical Instrumentation (AAMI) http: //www. aami. org/ w American National Standards Institute (ANSI) http: //www. ansi. org/ w International Electrotechnical Commission (IEC) http: //www. iec. ch/ w CDRH Office and Division Associated with Recognized Standards w Processes: 510(K), PMA, PDP, IDE, HDE, GMP, DESIGN CONTROLS
Patent & Standards Organizations w www. fda. gov/cdrh/ Food and drug administration w Association for the Advancement of Medical Instrumentation (AAMI) http: //www. aami. org/ w American National Standards Institute (ANSI) http: //www. ansi. org/ w International Electrotechnical Commission (IEC) http: //www. iec. ch/ w CDRH Office and Division Associated with Recognized Standards w Processes: 510(K), PMA, PDP, IDE, HDE, GMP, DESIGN CONTROLS
 Patent and regulation Offices w European: http: //www. atlanticbridge. co. uk/page 7. htm CE Marking of Medical devices. w National Institute of Industrial Property (France): http: //www. inpi. fr/inpi/accueil. htm w For information about legislative requirements from the European Union, the European Directives, and their relation to European Standards in particular industrial sectors, visit: http: //www. newapproach. org/
Patent and regulation Offices w European: http: //www. atlanticbridge. co. uk/page 7. htm CE Marking of Medical devices. w National Institute of Industrial Property (France): http: //www. inpi. fr/inpi/accueil. htm w For information about legislative requirements from the European Union, the European Directives, and their relation to European Standards in particular industrial sectors, visit: http: //www. newapproach. org/
 Internet w General search engines (Google, yahoo, etc); w Scientific/medical search engines (Pub Med, ISI, IEEE, Bio. One Journals Online, JSTOR (General Science, Mathematics and Statistics Collections), Kluwer Online, LINK - (from Springer Publishing Group), Oxford University Press, Proquest Direct, Science. Direct - (from Elsevier), Synergy - (from Blackwell Science and Munksgaard), Wiley Interscience
Internet w General search engines (Google, yahoo, etc); w Scientific/medical search engines (Pub Med, ISI, IEEE, Bio. One Journals Online, JSTOR (General Science, Mathematics and Statistics Collections), Kluwer Online, LINK - (from Springer Publishing Group), Oxford University Press, Proquest Direct, Science. Direct - (from Elsevier), Synergy - (from Blackwell Science and Munksgaard), Wiley Interscience
 Internet w Relevant web-based resources. Medical device news links http: //www. abledata. com; www. bmesource. org; http: //enablingdevices. com, http: //www. devicelink. com/ , http: //www. mdtmag. com/scripts/default. asp, www. medicaldevicesonline. com/
Internet w Relevant web-based resources. Medical device news links http: //www. abledata. com; www. bmesource. org; http: //enablingdevices. com, http: //www. devicelink. com/ , http: //www. mdtmag. com/scripts/default. asp, www. medicaldevicesonline. com/
 Relevant company web sites n n n n ATL Acuson Agilent Colin, Corometrics Critikon Criticare Datascope IVAC Littman Med-Scan Medison Physio. Control Omron Sensormedics n n n n Perimed Schiller Ash Medical Pressurometer American Medical Systems Puritan-Bennett Dinamap Omni. Trak Infinium Nicolet SPO Medical Diasonics, Fuji GE, n n n n Hitachi, HP, Marquette, Medinotes, NEC-Mitsubishi, Mennen Motorola, Nonin, Philips, Nihon Kohden Siemens, Space Labs, Tektronix, Toshiba, Welch Allyn, etc.
Relevant company web sites n n n n ATL Acuson Agilent Colin, Corometrics Critikon Criticare Datascope IVAC Littman Med-Scan Medison Physio. Control Omron Sensormedics n n n n Perimed Schiller Ash Medical Pressurometer American Medical Systems Puritan-Bennett Dinamap Omni. Trak Infinium Nicolet SPO Medical Diasonics, Fuji GE, n n n n Hitachi, HP, Marquette, Medinotes, NEC-Mitsubishi, Mennen Motorola, Nonin, Philips, Nihon Kohden Siemens, Space Labs, Tektronix, Toshiba, Welch Allyn, etc.
 BME design books w King, Paul H, Fries, Richard C, Design of Biomedical Devices and Systems, Marcel Dekker, 2002. w Fries, Richard C. Reliable Design of Medical Devices, New York, Marcel Dekker, 1997. w Witkin, Karen B. Clinical Evaluation of Medical Devices: Principles and Case Studies, Humana Press, 1997. (excellent on evaluation) w Geddes, Leslie, Medical Device Accidents With Illustrative Cases, New York, CRC Press, 1998. ( an interesting collection of mishaps) w Casey, S. , Set Phasers on Stun and other True Tales of Design, Technology, and Human Error, Santa Barbara, CA, Aegean Publishing Company, 1993. (Excellent reading on many mishaps. . . )
BME design books w King, Paul H, Fries, Richard C, Design of Biomedical Devices and Systems, Marcel Dekker, 2002. w Fries, Richard C. Reliable Design of Medical Devices, New York, Marcel Dekker, 1997. w Witkin, Karen B. Clinical Evaluation of Medical Devices: Principles and Case Studies, Humana Press, 1997. (excellent on evaluation) w Geddes, Leslie, Medical Device Accidents With Illustrative Cases, New York, CRC Press, 1998. ( an interesting collection of mishaps) w Casey, S. , Set Phasers on Stun and other True Tales of Design, Technology, and Human Error, Santa Barbara, CA, Aegean Publishing Company, 1993. (Excellent reading on many mishaps. . . )
 ME/EE/Comp. Eng related design texts w Bock, Peter, getting it right, R&D Methods for Science and Engineering. New York, 2001, Academic Press. (Generalist text on both R & D methodology, good source. ) w Ulrich, K. T. , Eppinger, S. D. Product Design & Development, New York, 2000, Mc. Graw Hill (Excellent general purpose text, useable from ME to Management. ) w Design in the New Millennium: Advanced Engineering Environments: Phase 2, National Academy Press. See http: //www. nap. edu/books/0309071259/html/ for an electronic copy and print order forms. w Pahl, G. , Beitz, W. , Engineering Design, A Systematic Approach, London, Springer-Verlag, 1988. (Classic, good, but hard to read)
ME/EE/Comp. Eng related design texts w Bock, Peter, getting it right, R&D Methods for Science and Engineering. New York, 2001, Academic Press. (Generalist text on both R & D methodology, good source. ) w Ulrich, K. T. , Eppinger, S. D. Product Design & Development, New York, 2000, Mc. Graw Hill (Excellent general purpose text, useable from ME to Management. ) w Design in the New Millennium: Advanced Engineering Environments: Phase 2, National Academy Press. See http: //www. nap. edu/books/0309071259/html/ for an electronic copy and print order forms. w Pahl, G. , Beitz, W. , Engineering Design, A Systematic Approach, London, Springer-Verlag, 1988. (Classic, good, but hard to read)
 ME/EE/Comp. Eng related design texts w Dieter, G. E. , Engineering Design, New York, Mc. Graw Hill, 2000. (Good general purpose ME text) w Suh, N. The Principles of Design New York, Oxford University Press, 1990. (an axiomatic approach to design using information principles) w Wilcox, A. Engineering Design For Electrical Engineers, Englewood Cliffs N. J. , Prentice Hall, 1990. (Easy) w Ingle, Kathryn A. Reverse Engineering, New York, Mc. Graw-Hill Inc. , 1994. (excellent ME/EE reverse engineering examples) w Middendorf, William H. Design of Devices and Systems, New York, Marcel Dekker Inc. , 1990. (Good overview, good coverage of Human Factors. . . )
ME/EE/Comp. Eng related design texts w Dieter, G. E. , Engineering Design, New York, Mc. Graw Hill, 2000. (Good general purpose ME text) w Suh, N. The Principles of Design New York, Oxford University Press, 1990. (an axiomatic approach to design using information principles) w Wilcox, A. Engineering Design For Electrical Engineers, Englewood Cliffs N. J. , Prentice Hall, 1990. (Easy) w Ingle, Kathryn A. Reverse Engineering, New York, Mc. Graw-Hill Inc. , 1994. (excellent ME/EE reverse engineering examples) w Middendorf, William H. Design of Devices and Systems, New York, Marcel Dekker Inc. , 1990. (Good overview, good coverage of Human Factors. . . )
 ME/EE/Comp. Eng related design texts w Carper, Kenneth L. Forensic Engineering, New York, Elsevier, 1989. . (interesting case studies. . . ) w Burgess, J. Designing for Humans: The Human Factor in Engineering, Princeton, Petrocelli Books, 1986. (Any of you needing ergonomics. . . ) w Schach, Steven R. , Classical and Object-oriented Software Engineering, Irwin Press, Chicago, 1996 (Excellent source text for software design) w Dym, Clive L. Engineering Design, a Synthesis of Views, Cambridge University Press, 1994 (An interesting non-denominational overview of the design process, taxonomy, and trends)
ME/EE/Comp. Eng related design texts w Carper, Kenneth L. Forensic Engineering, New York, Elsevier, 1989. . (interesting case studies. . . ) w Burgess, J. Designing for Humans: The Human Factor in Engineering, Princeton, Petrocelli Books, 1986. (Any of you needing ergonomics. . . ) w Schach, Steven R. , Classical and Object-oriented Software Engineering, Irwin Press, Chicago, 1996 (Excellent source text for software design) w Dym, Clive L. Engineering Design, a Synthesis of Views, Cambridge University Press, 1994 (An interesting non-denominational overview of the design process, taxonomy, and trends)
 ME/EE/Comp. Eng related design texts w Dieter, George E. Engineering Design, A Materials and Processing Approach, Mc. Graw Hill, 2000. (A Mechanical Engineering text, excellent in coverage of the overall process of design. ) w "Curriculum Design, from an art towards a science" by S Waks (Technion U, Israel), ISBN # 3 -9804190 -0 -2 is recommended for planning purposes, if you are outlining a new design or related course. . w Chaffin, DB, Andersson, GBJ, "Occupational Biomechanics", Wiley, 1991. (some human factors use. ) w Eggert, Rudolph. "Engineering Design", Prentice Hall, 2005. (ME & aerospace oriented, several useful chapters)
ME/EE/Comp. Eng related design texts w Dieter, George E. Engineering Design, A Materials and Processing Approach, Mc. Graw Hill, 2000. (A Mechanical Engineering text, excellent in coverage of the overall process of design. ) w "Curriculum Design, from an art towards a science" by S Waks (Technion U, Israel), ISBN # 3 -9804190 -0 -2 is recommended for planning purposes, if you are outlining a new design or related course. . w Chaffin, DB, Andersson, GBJ, "Occupational Biomechanics", Wiley, 1991. (some human factors use. ) w Eggert, Rudolph. "Engineering Design", Prentice Hall, 2005. (ME & aerospace oriented, several useful chapters)
 Intellectual Property/ Miscellaneous w Sunar, D. G. , The Expert Witness Handbook-a Guide for Engineers, Belmont CA, Professional Publications, 1989. (overview of expert witness responsibilities. . . ) w Poynter, D. The Expert Witness Handbook, Tips and Techniques for the Litigation Consultant, Para Publishing, 1987. w Foltz, R. , Penn, T. Protecting Engineering Ideas & Inventions, Cleveland Ohio, Penn Institute, 1989. (In-depth coverage of the titled topic, of value in your professional activities. . . ) w LT Kahn, JM Corrigan, MS Donaldson, To Err is Human, Building a Safer Health System. National Academy Press, Washington, 2000. (Super statistics, proof that BMEs are needed!) w Nahum, Alan m. , Melvin, John W. “Accidental Injury, Biomechanics and Prevention”, Springer, New York, 2002.
Intellectual Property/ Miscellaneous w Sunar, D. G. , The Expert Witness Handbook-a Guide for Engineers, Belmont CA, Professional Publications, 1989. (overview of expert witness responsibilities. . . ) w Poynter, D. The Expert Witness Handbook, Tips and Techniques for the Litigation Consultant, Para Publishing, 1987. w Foltz, R. , Penn, T. Protecting Engineering Ideas & Inventions, Cleveland Ohio, Penn Institute, 1989. (In-depth coverage of the titled topic, of value in your professional activities. . . ) w LT Kahn, JM Corrigan, MS Donaldson, To Err is Human, Building a Safer Health System. National Academy Press, Washington, 2000. (Super statistics, proof that BMEs are needed!) w Nahum, Alan m. , Melvin, John W. “Accidental Injury, Biomechanics and Prevention”, Springer, New York, 2002.
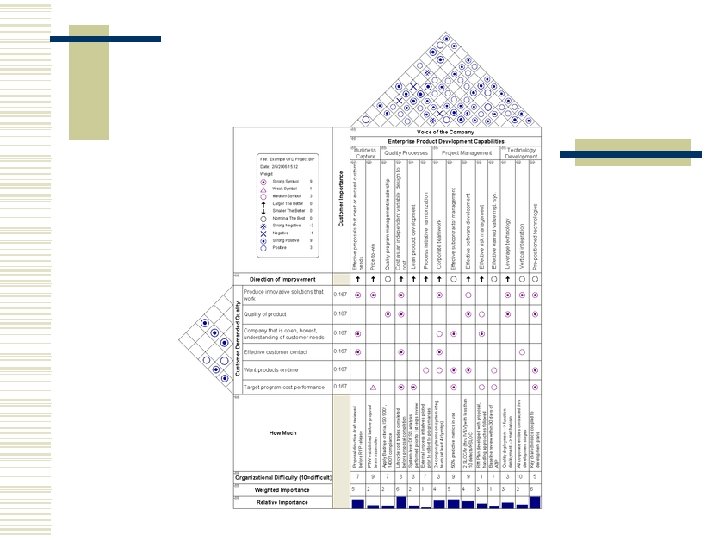
 Quality Function Deployment w Quality function deployment or "QFD" is a flexible and comprehensive group decision making technique used in product or service development, brand marketing, and product management. w QFD can strongly help an organization focus on the critical characteristics of a new or existing product or service from the separate viewpoints of the customer market segments, company, or technology-development needs. The results of the technique yield transparent and visible graphs and matrices that can be reused for future product/service developments
Quality Function Deployment w Quality function deployment or "QFD" is a flexible and comprehensive group decision making technique used in product or service development, brand marketing, and product management. w QFD can strongly help an organization focus on the critical characteristics of a new or existing product or service from the separate viewpoints of the customer market segments, company, or technology-development needs. The results of the technique yield transparent and visible graphs and matrices that can be reused for future product/service developments
 QFD/TRIZ Materials w William Eureka, Nancy Ryan, The Customer-Driven Company: Managerial Perspectives on Quality Function Deployment, ASI Press, Dearborn MI 1994 (Good QFD overview/tutorial). w John Terninko, Step-by-Step QFD, Customer-Driven Product Design, 2 nd Edition, St Lucie Press, Boca Raton, FL, 1997 (ditto) w Cohen, L. "Quality Function Deployment: How to Make QFD Work for You" Addison Wesley, 1995 (good, generic coverage) w http: //www. qualisoft. com/ - for QFD information, download the demo… w http: //www. totalqualitysoftware. co. uk for qfd 2000 information w Altshuller, G. S. "Creativity as an Exact Science" Gordon & Breach Science, 1984. (translation of the original TRIZ material) w http: //www. ideationtriz. com for TRIZ materials, several publications available. w http: //www. techoptimizer. com for another TRIZ type of site, software for invention.
QFD/TRIZ Materials w William Eureka, Nancy Ryan, The Customer-Driven Company: Managerial Perspectives on Quality Function Deployment, ASI Press, Dearborn MI 1994 (Good QFD overview/tutorial). w John Terninko, Step-by-Step QFD, Customer-Driven Product Design, 2 nd Edition, St Lucie Press, Boca Raton, FL, 1997 (ditto) w Cohen, L. "Quality Function Deployment: How to Make QFD Work for You" Addison Wesley, 1995 (good, generic coverage) w http: //www. qualisoft. com/ - for QFD information, download the demo… w http: //www. totalqualitysoftware. co. uk for qfd 2000 information w Altshuller, G. S. "Creativity as an Exact Science" Gordon & Breach Science, 1984. (translation of the original TRIZ material) w http: //www. ideationtriz. com for TRIZ materials, several publications available. w http: //www. techoptimizer. com for another TRIZ type of site, software for invention.
 Ethics w http: //www. cnn. com/HEALTH/bioethics/archive. index. html for some of our ethical questions w http: //www. cwru. edu/med/bioethics. html. CWRU ethics site, comprehensive! w http: //www. nspe. org/ethics/eh 1 -code. asp NSPE ethics web site, links to others
Ethics w http: //www. cnn. com/HEALTH/bioethics/archive. index. html for some of our ethical questions w http: //www. cwru. edu/med/bioethics. html. CWRU ethics site, comprehensive! w http: //www. nspe. org/ethics/eh 1 -code. asp NSPE ethics web site, links to others
 Biomaterials/Biomimetics w Hill, D. , "Design Engineering of Biomaterials for Medical Devices", Wiley, 1998. ( A BME specific biomaterials oriented text. 2/3 Biomaterials, 1/3 design, all very good. ) w Wise, D. L. Biomaterials and Bioengineering Handbook, Dekker, 2000 (Very comprehensive) w Sarikaya, M, Aksay, I. Biomimetics, Design and Processing of Materials, AIP, 1995. w Ratner, B. , Hoffman, A. , Schoen, F. , Lemons, J. Biomaterials Science: An Introduction to Materials in Medicine, Academic Press, 1996. (Excellent coverage of a wide variety of topics. )
Biomaterials/Biomimetics w Hill, D. , "Design Engineering of Biomaterials for Medical Devices", Wiley, 1998. ( A BME specific biomaterials oriented text. 2/3 Biomaterials, 1/3 design, all very good. ) w Wise, D. L. Biomaterials and Bioengineering Handbook, Dekker, 2000 (Very comprehensive) w Sarikaya, M, Aksay, I. Biomimetics, Design and Processing of Materials, AIP, 1995. w Ratner, B. , Hoffman, A. , Schoen, F. , Lemons, J. Biomaterials Science: An Introduction to Materials in Medicine, Academic Press, 1996. (Excellent coverage of a wide variety of topics. )
 Design Software w Design, Simulation commercial products and services, etc. Specialized packages, such as designsafe[i] – for safety analyses of devices and processes, FMEA analysis – for failure mode analyses, and QFD designer - for quality/function/deployment applications are recommended as courses become more specialized. Recommendations for senior or graduate design aids include software such as Working Model 2 -D and 3 -D modeler[ii] for motion analysis and trajectory modeling. [i] Design Safety Engineering, Ann Arbor MI [ii] Knowledge Revolution, San Mateo CA
Design Software w Design, Simulation commercial products and services, etc. Specialized packages, such as designsafe[i] – for safety analyses of devices and processes, FMEA analysis – for failure mode analyses, and QFD designer - for quality/function/deployment applications are recommended as courses become more specialized. Recommendations for senior or graduate design aids include software such as Working Model 2 -D and 3 -D modeler[ii] for motion analysis and trajectory modeling. [i] Design Safety Engineering, Ann Arbor MI [ii] Knowledge Revolution, San Mateo CA
![Design Software w CAD software such as Solid. Works[iii] or Think 3[iv] is recommended Design Software w CAD software such as Solid. Works[iii] or Think 3[iv] is recommended](https://present5.com/presentation/b703f3e8493740b45d8ff6f36d9cdf61/image-48.jpg) Design Software w CAD software such as Solid. Works[iii] or Think 3[iv] is recommended for design layouts, if the students have had introductory courses in the use of such software, or have the time to learn it as needed. Given sufficient preparation, prototypes from these packages can be generated and tested. The program Tech. Optimizer[v] is strongly recommended for advanced level design courses, if one can afford the license cost. Tech. Optimizer will guide one through and assist in the design process, once sufficient time is spent learning how to use the software. [iii] Solid. Works Corp, Concord MA [iv] Think 3, Santa Clara CA [v] Invention Machine Corp. , Boston MA
Design Software w CAD software such as Solid. Works[iii] or Think 3[iv] is recommended for design layouts, if the students have had introductory courses in the use of such software, or have the time to learn it as needed. Given sufficient preparation, prototypes from these packages can be generated and tested. The program Tech. Optimizer[v] is strongly recommended for advanced level design courses, if one can afford the license cost. Tech. Optimizer will guide one through and assist in the design process, once sufficient time is spent learning how to use the software. [iii] Solid. Works Corp, Concord MA [iv] Think 3, Santa Clara CA [v] Invention Machine Corp. , Boston MA
 Design Software w Options in the use of the software routines that guide one through product or process analysis, evaluation of feature transfers, and generation of similar effect listings. At a bare minimum, this program can be used to do both a general web search using a logical Boolean search, and to do a similar patent search of either or both the US Patent Office listing or the Japanese Patent Office listings.
Design Software w Options in the use of the software routines that guide one through product or process analysis, evaluation of feature transfers, and generation of similar effect listings. At a bare minimum, this program can be used to do both a general web search using a logical Boolean search, and to do a similar patent search of either or both the US Patent Office listing or the Japanese Patent Office listings.


