6b22f51d4a30d0d4943e3965c855f9a0.ppt
- Количество слайдов: 43
 Prion Diseases CMED 526/EPI 526 - May 6 th, 2009 Robert Harrington, DVM, Ph. D USDA – Agricultural Research Service UW – Dept. of Comparative Medicine rdhdvm@u. washington. edu
Prion Diseases CMED 526/EPI 526 - May 6 th, 2009 Robert Harrington, DVM, Ph. D USDA – Agricultural Research Service UW – Dept. of Comparative Medicine rdhdvm@u. washington. edu
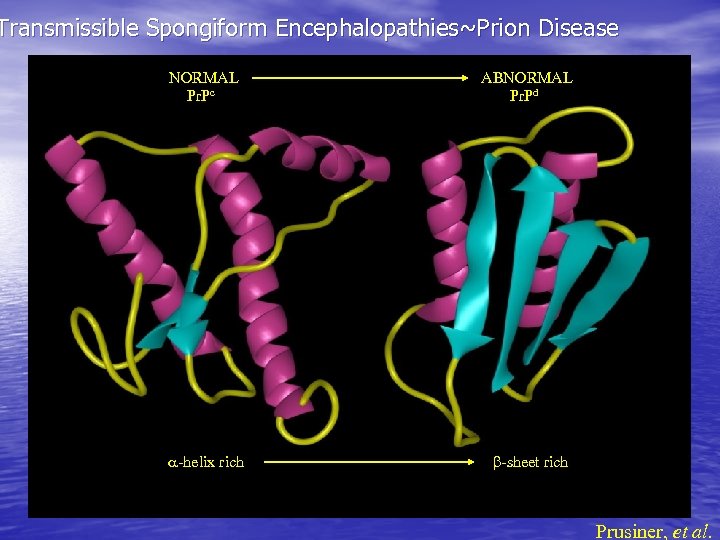 Transmissible Spongiform Encephalopathies~Prion Disease NORMAL Pr. Pc ABNORMAL Pr. Pd -helix rich -sheet rich Prusiner, et al.
Transmissible Spongiform Encephalopathies~Prion Disease NORMAL Pr. Pc ABNORMAL Pr. Pd -helix rich -sheet rich Prusiner, et al.
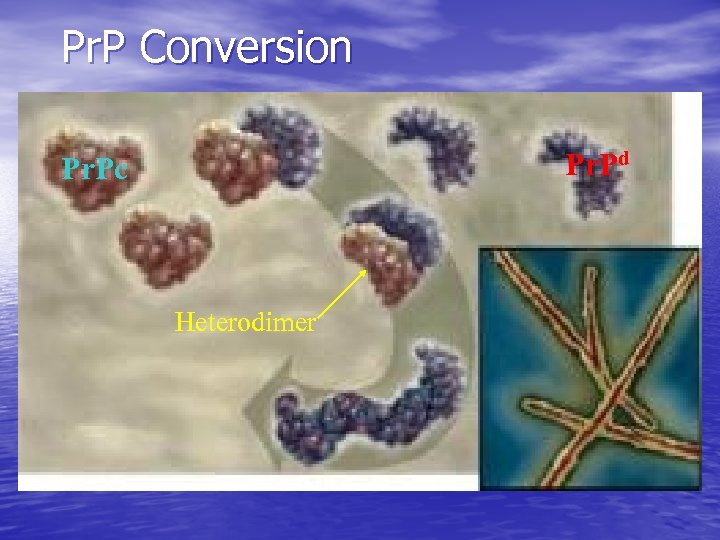 Pr. P Conversion Pr. Pd Pr. Pc Heterodimer
Pr. P Conversion Pr. Pd Pr. Pc Heterodimer
 TSE Pathogenesis • Transmission occurs by oral route • Pr. Pd localizes to regional lymphoid tissue – Transient in some species • Migration to central nervous system – Retrograde along nerves – Blood-borne transport • Accumulation in brain with subsequent neurodegeneration
TSE Pathogenesis • Transmission occurs by oral route • Pr. Pd localizes to regional lymphoid tissue – Transient in some species • Migration to central nervous system – Retrograde along nerves – Blood-borne transport • Accumulation in brain with subsequent neurodegeneration
 Diagnosis • Postmortem – – Microscopic pathology Immunohistochemistry ELISA, Immunoblotting Bioassay – – Same techniques as above applied to: Brain biopsy Tonsil, lymph node, third eyelid biopsy, or rectal ? Blood test ? • Antemortem
Diagnosis • Postmortem – – Microscopic pathology Immunohistochemistry ELISA, Immunoblotting Bioassay – – Same techniques as above applied to: Brain biopsy Tonsil, lymph node, third eyelid biopsy, or rectal ? Blood test ? • Antemortem
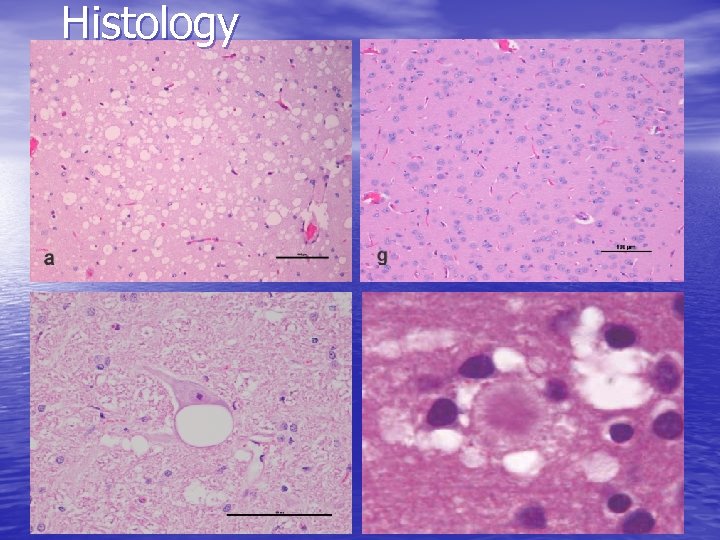 Histology
Histology
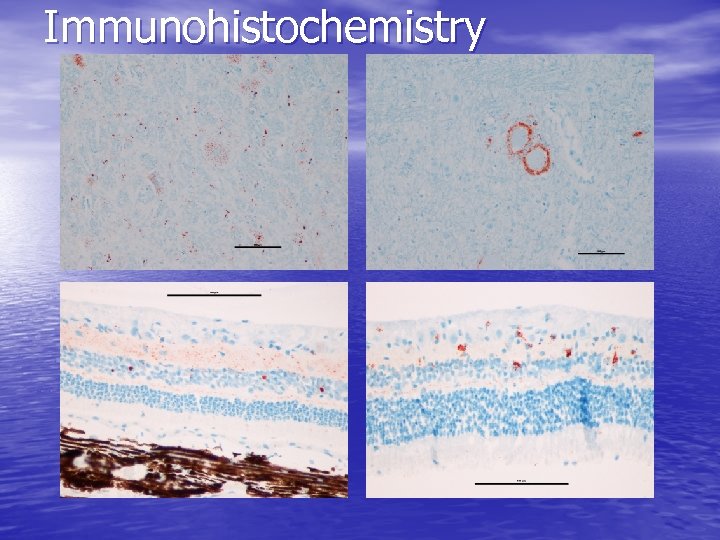 Immunohistochemistry
Immunohistochemistry
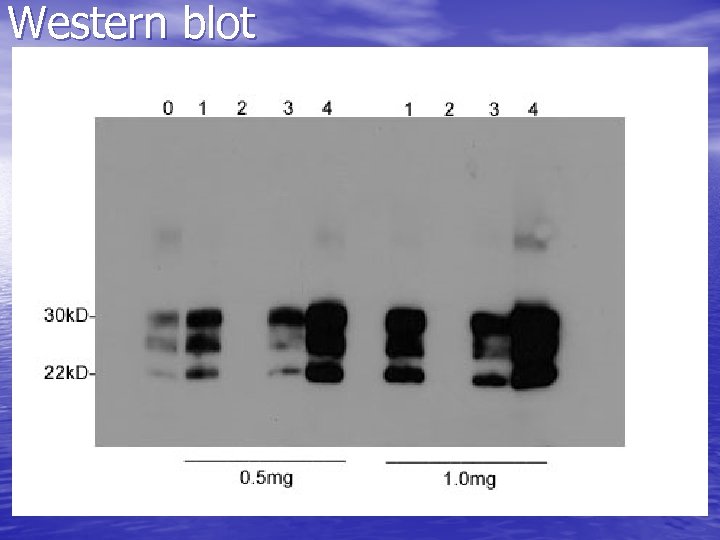 Western blot
Western blot
 Animal Prion Disease • Scrapie - sheep, goats • Chronic Wasting Disease (CWD) - deer, elk, moose • Bovine Spongiform Encephalopathy (BSE) - cattle • Transmissible mink encephalopathy (TME) - mink • Feline spongiform encephalopathy - large & • domestic cats Spongiform encephalopathy of captive ungulates exotic hoof-stock in zoological parks
Animal Prion Disease • Scrapie - sheep, goats • Chronic Wasting Disease (CWD) - deer, elk, moose • Bovine Spongiform Encephalopathy (BSE) - cattle • Transmissible mink encephalopathy (TME) - mink • Feline spongiform encephalopathy - large & • domestic cats Spongiform encephalopathy of captive ungulates exotic hoof-stock in zoological parks
 Human Prion Disease • Sporadic – Creutzfeldt-Jakob disease (CJD) • Familial (genetic) – Familial CJD – Gerstman-Straussler-Scheinker Syndrome (GSS) – Fatal Familial Insomnia (FFI) • Acquired by transmission – Kuru – Iatrogenic CJD (neurosurgical instruments, dura mater grafts) – Variant CJD (v. CJD)
Human Prion Disease • Sporadic – Creutzfeldt-Jakob disease (CJD) • Familial (genetic) – Familial CJD – Gerstman-Straussler-Scheinker Syndrome (GSS) – Fatal Familial Insomnia (FFI) • Acquired by transmission – Kuru – Iatrogenic CJD (neurosurgical instruments, dura mater grafts) – Variant CJD (v. CJD)
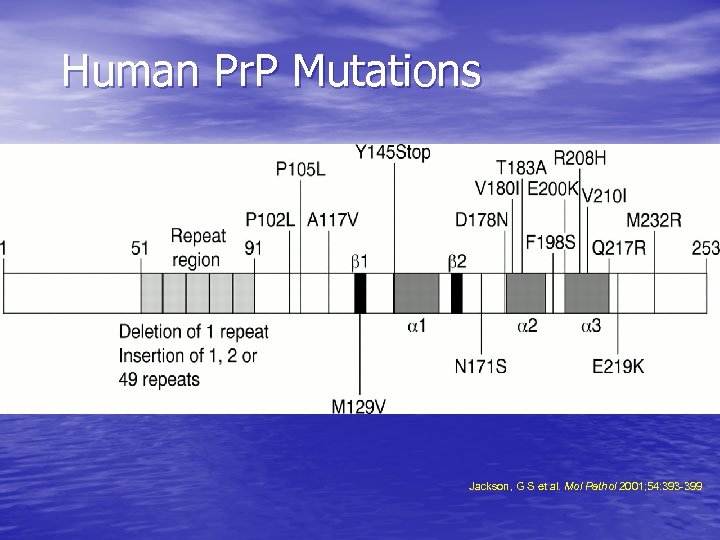 Human Pr. P Mutations Jackson, G S et al. Mol Pathol 2001; 54: 393 -399
Human Pr. P Mutations Jackson, G S et al. Mol Pathol 2001; 54: 393 -399
 Transmission Within Species vertical and horizontal COMMON in utero, fetal fluids, fetal membranes horizontal Oral (urine, feces, or blood? ) UNCOMMON Foodborne Direct only through bite wounds Foodborne (MBM) No direct transmission from cow to cow Foodborne, blood, tissue transplant, HGH, instruments
Transmission Within Species vertical and horizontal COMMON in utero, fetal fluids, fetal membranes horizontal Oral (urine, feces, or blood? ) UNCOMMON Foodborne Direct only through bite wounds Foodborne (MBM) No direct transmission from cow to cow Foodborne, blood, tissue transplant, HGH, instruments
 Species Barrier Concept • Transmission within a species may occur readily • Barrier between species limits transmission – Inefficient transmission – Extended incubation times – Low or non-existent rate of disease • Serial passage – Required to overcome species barrier – Progressive reduction in incubation time – Increased rate of disease
Species Barrier Concept • Transmission within a species may occur readily • Barrier between species limits transmission – Inefficient transmission – Extended incubation times – Low or non-existent rate of disease • Serial passage – Required to overcome species barrier – Progressive reduction in incubation time – Increased rate of disease
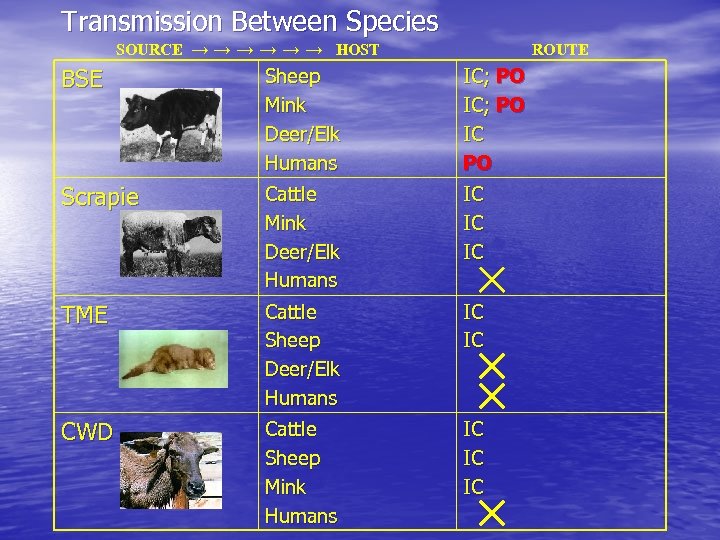 Transmission Between Species SOURCE → → → HOST ROUTE BSE Sheep Mink Deer/Elk Humans IC; PO IC PO Scrapie Cattle Mink Deer/Elk Humans IC IC IC TME Cattle Sheep Deer/Elk Humans IC IC CWD Cattle Sheep Mink Humans IC IC IC
Transmission Between Species SOURCE → → → HOST ROUTE BSE Sheep Mink Deer/Elk Humans IC; PO IC PO Scrapie Cattle Mink Deer/Elk Humans IC IC IC TME Cattle Sheep Deer/Elk Humans IC IC CWD Cattle Sheep Mink Humans IC IC IC
 Recognition of BSE • Late 1985: Unusual neurologic disease in UK cattle • Insidious onset – – – Irritabilty, agression Motor system impairment (ataxia) Difficulty in rising (e. g. “downer cow”) Decreased milk production Wasting Death • Predominantly dairy cattle – Feeding practices – Relative herd age • Neuropathology similar to Scrapie – Vacuolation, Pr. Pd, astrocytosis, Scrapie associated fibrils
Recognition of BSE • Late 1985: Unusual neurologic disease in UK cattle • Insidious onset – – – Irritabilty, agression Motor system impairment (ataxia) Difficulty in rising (e. g. “downer cow”) Decreased milk production Wasting Death • Predominantly dairy cattle – Feeding practices – Relative herd age • Neuropathology similar to Scrapie – Vacuolation, Pr. Pd, astrocytosis, Scrapie associated fibrils
 Cause of BSE • Ruminant tissue in food chain – – – Meat and bone meal (MBM) Scrapie Sporadic BSE in cattle – – Human tissue? Toxin? Environmental? Other? • Alternative theories
Cause of BSE • Ruminant tissue in food chain – – – Meat and bone meal (MBM) Scrapie Sporadic BSE in cattle – – Human tissue? Toxin? Environmental? Other? • Alternative theories
 BSE Epidemic • ~180, 000 cumulative cases in UK • Peaked at 37, 000 cases per annum in 1992 • Recycling of ruminant tissue in food chain implicated • Progressive decline with introduction of feed bans
BSE Epidemic • ~180, 000 cumulative cases in UK • Peaked at 37, 000 cases per annum in 1992 • Recycling of ruminant tissue in food chain implicated • Progressive decline with introduction of feed bans
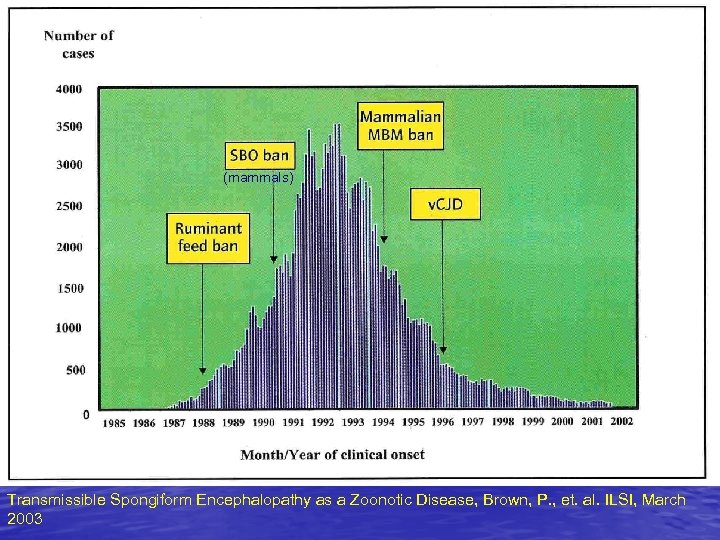 (mammals) Transmissible Spongiform Encephalopathy as a Zoonotic Disease, Brown, P. , et. al. ILSI, March 2003
(mammals) Transmissible Spongiform Encephalopathy as a Zoonotic Disease, Brown, P. , et. al. ILSI, March 2003
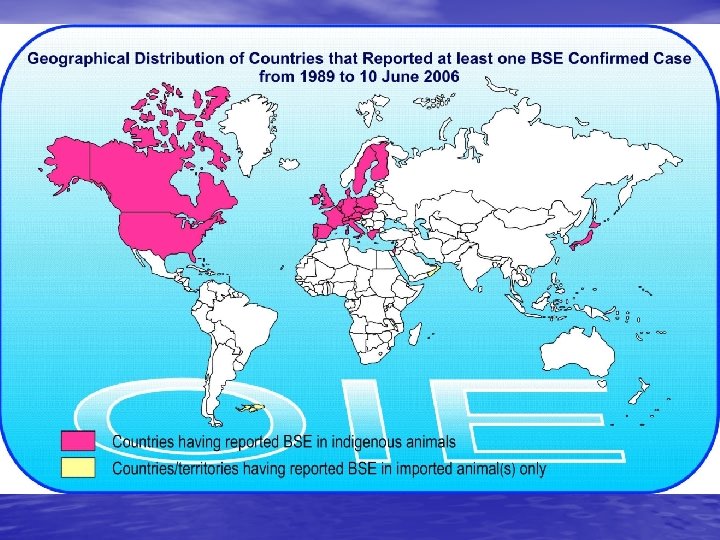
 Spread of BSE Epidemic • 1990: Domestic BSE detected in Switzerland, • • • imported cases in Portugal 1999: 7 other EU countries with domestic BSE Jan 2000 to Oct 2002: 11 additional EU countries 2001: BSE detected in Japan 2002: BSE detected in Israel 2003: BSE in Canadian cow
Spread of BSE Epidemic • 1990: Domestic BSE detected in Switzerland, • • • imported cases in Portugal 1999: 7 other EU countries with domestic BSE Jan 2000 to Oct 2002: 11 additional EU countries 2001: BSE detected in Japan 2002: BSE detected in Israel 2003: BSE in Canadian cow
 BSE in the United States • 30 -40 million cattle slaughtered/year • 1997: ban on feeding US cattle meat-andbone meal • 3 cases to date - RARE! – 2003: 6. 5 yo dairy cow imported from Canada – 2004: 12 yo beef cow born and raised in Texas – 2006: 10 yo beef cow in Alabama uncertain origin – Were there previously unrecognized cases?
BSE in the United States • 30 -40 million cattle slaughtered/year • 1997: ban on feeding US cattle meat-andbone meal • 3 cases to date - RARE! – 2003: 6. 5 yo dairy cow imported from Canada – 2004: 12 yo beef cow born and raised in Texas – 2006: 10 yo beef cow in Alabama uncertain origin – Were there previously unrecognized cases?
 BSE in the United States • 2003: Additional measures post WA BSE case – “downer” cattle excluded from human consumption – Ban on SRM from animals >30 months of age from human consumption – Ban on mechanically-separated meat
BSE in the United States • 2003: Additional measures post WA BSE case – “downer” cattle excluded from human consumption – Ban on SRM from animals >30 months of age from human consumption – Ban on mechanically-separated meat
 New variant CJD • Unusual form of neurologic disease in teenagers and young adults • Spongiform encephalopathy • Neuropathology not consistent with sporadic forms of CJD • Stimulus for US National Prion Disease Center
New variant CJD • Unusual form of neurologic disease in teenagers and young adults • Spongiform encephalopathy • Neuropathology not consistent with sporadic forms of CJD • Stimulus for US National Prion Disease Center
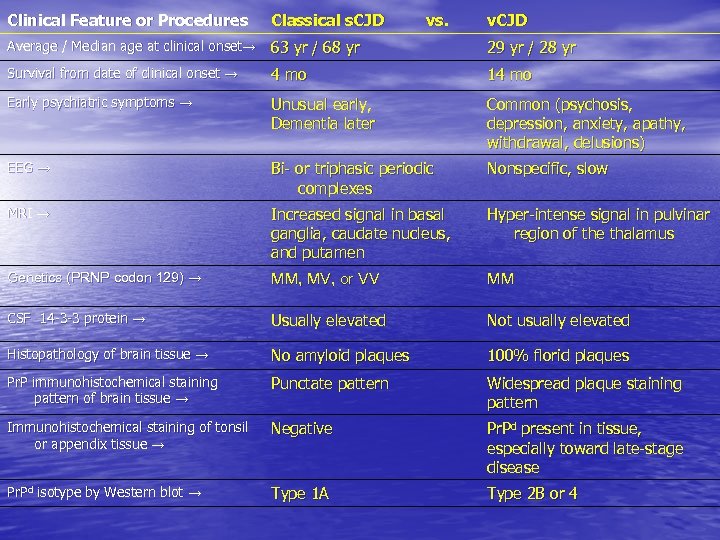 Clinical Feature or Procedures Classical s. CJD vs. v. CJD Average / Median age at clinical onset→ 63 yr / 68 yr 29 yr / 28 yr Survival from date of clinical onset → 4 mo 14 mo Early psychiatric symptoms → Unusual early, Dementia later Common (psychosis, depression, anxiety, apathy, withdrawal, delusions) EEG → Bi- or triphasic periodic complexes Nonspecific, slow MRI → Increased signal in basal ganglia, caudate nucleus, and putamen Hyper-intense signal in pulvinar region of the thalamus Genetics (PRNP codon 129) → MM, MV, or VV MM CSF 14 -3 -3 protein → Usually elevated Not usually elevated Histopathology of brain tissue → No amyloid plaques 100% florid plaques Pr. P immunohistochemical staining pattern of brain tissue → Punctate pattern Widespread plaque staining pattern Immunohistochemical staining of tonsil or appendix tissue → Negative Pr. Pd present in tissue, especially toward late-stage disease Pr. Pd isotype by Western blot → Type 1 A Type 2 B or 4
Clinical Feature or Procedures Classical s. CJD vs. v. CJD Average / Median age at clinical onset→ 63 yr / 68 yr 29 yr / 28 yr Survival from date of clinical onset → 4 mo 14 mo Early psychiatric symptoms → Unusual early, Dementia later Common (psychosis, depression, anxiety, apathy, withdrawal, delusions) EEG → Bi- or triphasic periodic complexes Nonspecific, slow MRI → Increased signal in basal ganglia, caudate nucleus, and putamen Hyper-intense signal in pulvinar region of the thalamus Genetics (PRNP codon 129) → MM, MV, or VV MM CSF 14 -3 -3 protein → Usually elevated Not usually elevated Histopathology of brain tissue → No amyloid plaques 100% florid plaques Pr. P immunohistochemical staining pattern of brain tissue → Punctate pattern Widespread plaque staining pattern Immunohistochemical staining of tonsil or appendix tissue → Negative Pr. Pd present in tissue, especially toward late-stage disease Pr. Pd isotype by Western blot → Type 1 A Type 2 B or 4
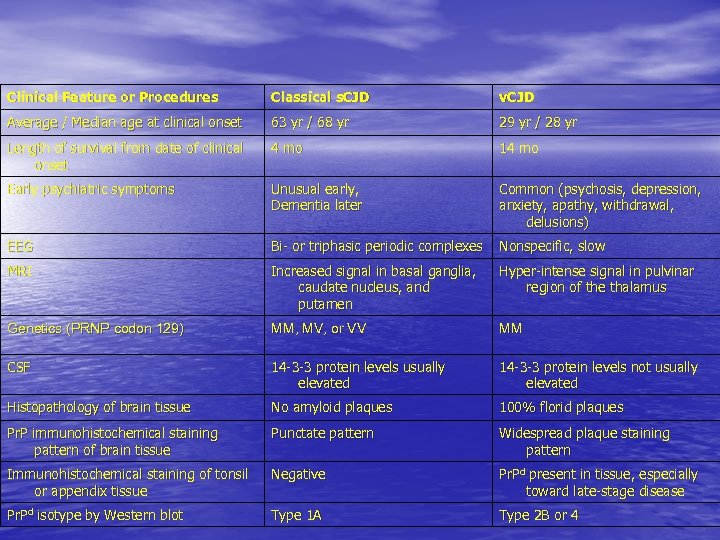 Clinical Feature or Procedures Classical s. CJD v. CJD Average / Median age at clinical onset 63 yr / 68 yr 29 yr / 28 yr Length of survival from date of clinical onset 4 mo 14 mo Early psychiatric symptoms Unusual early, Dementia later Common (psychosis, depression, anxiety, apathy, withdrawal, delusions) EEG Bi- or triphasic periodic complexes Nonspecific, slow MRI Increased signal in basal ganglia, caudate nucleus, and putamen Hyper-intense signal in pulvinar region of the thalamus Genetics (PRNP codon 129) MM, MV, or VV MM CSF 14 -3 -3 protein levels usually elevated 14 -3 -3 protein levels not usually elevated Histopathology of brain tissue No amyloid plaques 100% florid plaques Pr. P immunohistochemical staining pattern of brain tissue Punctate pattern Widespread plaque staining pattern Immunohistochemical staining of tonsil or appendix tissue Negative Pr. Pd present in tissue, especially toward late-stage disease Pr. Pd isotype by Western blot Type 1 A Type 2 B or 4
Clinical Feature or Procedures Classical s. CJD v. CJD Average / Median age at clinical onset 63 yr / 68 yr 29 yr / 28 yr Length of survival from date of clinical onset 4 mo 14 mo Early psychiatric symptoms Unusual early, Dementia later Common (psychosis, depression, anxiety, apathy, withdrawal, delusions) EEG Bi- or triphasic periodic complexes Nonspecific, slow MRI Increased signal in basal ganglia, caudate nucleus, and putamen Hyper-intense signal in pulvinar region of the thalamus Genetics (PRNP codon 129) MM, MV, or VV MM CSF 14 -3 -3 protein levels usually elevated 14 -3 -3 protein levels not usually elevated Histopathology of brain tissue No amyloid plaques 100% florid plaques Pr. P immunohistochemical staining pattern of brain tissue Punctate pattern Widespread plaque staining pattern Immunohistochemical staining of tonsil or appendix tissue Negative Pr. Pd present in tissue, especially toward late-stage disease Pr. Pd isotype by Western blot Type 1 A Type 2 B or 4
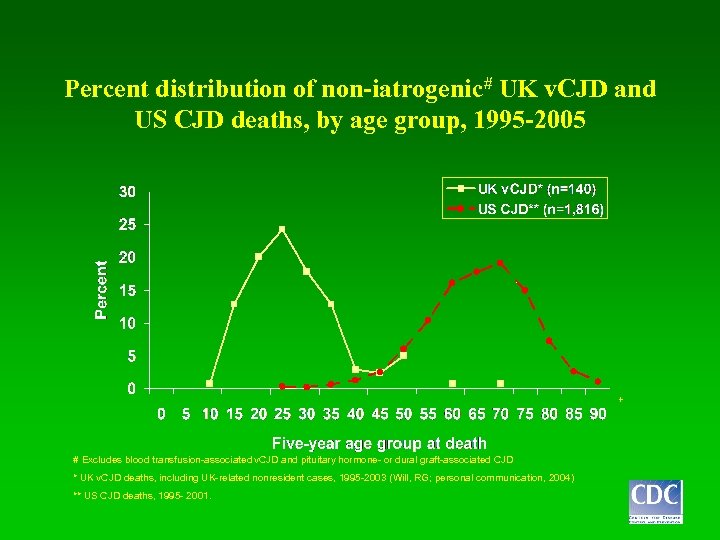 Percent distribution of non-iatrogenic# UK v. CJD and US CJD deaths, by age group, 1995 -2005 + # Excludes blood transfusion-associated v. CJD and pituitary hormone- or dural graft-associated CJD * UK v. CJD deaths, including UK-related nonresident cases, 1995 -2003 (Will, RG; personal communication, 2004) ** US CJD deaths, 1995 - 2001.
Percent distribution of non-iatrogenic# UK v. CJD and US CJD deaths, by age group, 1995 -2005 + # Excludes blood transfusion-associated v. CJD and pituitary hormone- or dural graft-associated CJD * UK v. CJD deaths, including UK-related nonresident cases, 1995 -2003 (Will, RG; personal communication, 2004) ** US CJD deaths, 1995 - 2001.
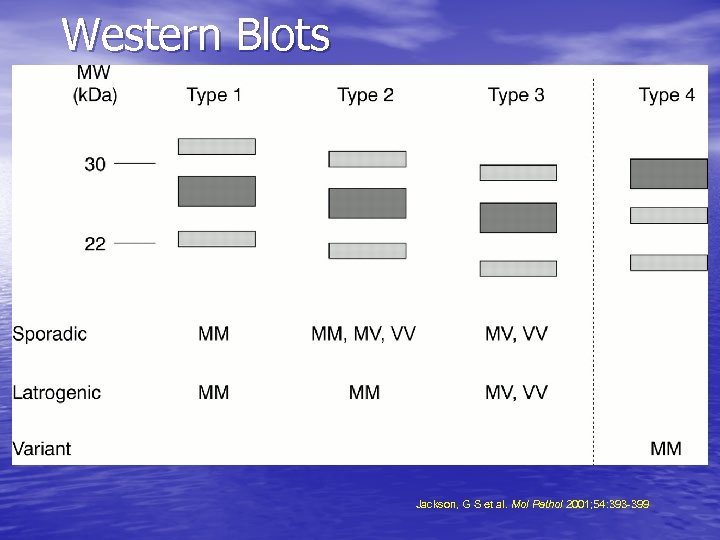 Western Blots Jackson, G S et al. Mol Pathol 2001; 54: 393 -399
Western Blots Jackson, G S et al. Mol Pathol 2001; 54: 393 -399
 BSE-v. CJD Link • New variant disease that differs from classical CJD – Similarities to BSE – Geographically related to areas of BSE – Hypothesis: consumption of contaminated beef products • Epidemiologic curve • Animal challenge studies • Molecular biology
BSE-v. CJD Link • New variant disease that differs from classical CJD – Similarities to BSE – Geographically related to areas of BSE – Hypothesis: consumption of contaminated beef products • Epidemiologic curve • Animal challenge studies • Molecular biology
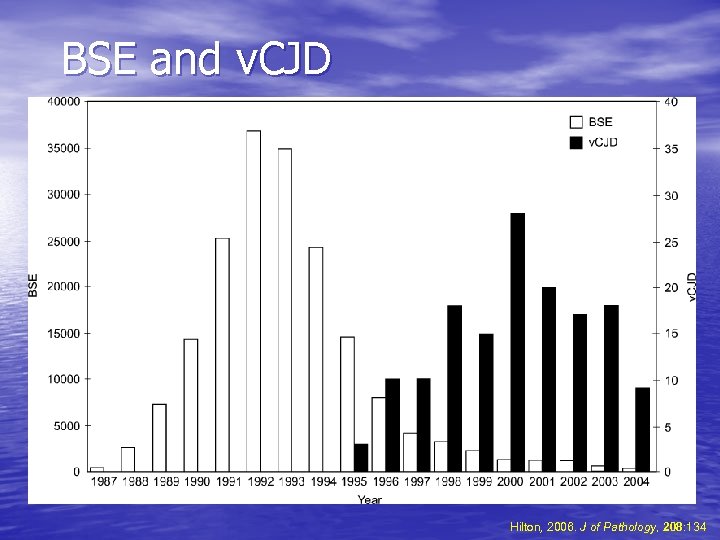 BSE and v. CJD Hilton, 2006. J of Pathology, 208: 134
BSE and v. CJD Hilton, 2006. J of Pathology, 208: 134
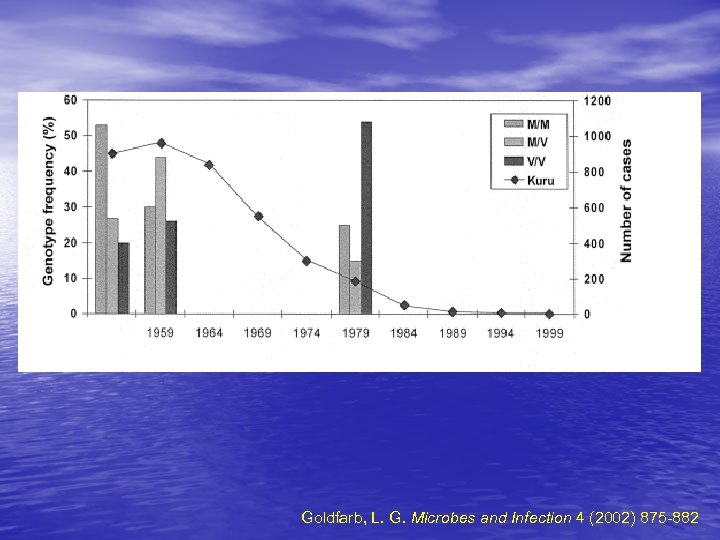 Goldfarb, L. G. Microbes and Infection 4 (2002) 875 -882
Goldfarb, L. G. Microbes and Infection 4 (2002) 875 -882
 Animal Challenge Studies • BSE → primates ≈ v. CJD → primates – Similar lesions and biochemistry • Transgenic mice – BSE → humanized mice – v. CJD → bovinized mice – Lesions, biochemistry of BSE ≈ v. CJD regardless of mouse type – Both differ from s. CJD
Animal Challenge Studies • BSE → primates ≈ v. CJD → primates – Similar lesions and biochemistry • Transgenic mice – BSE → humanized mice – v. CJD → bovinized mice – Lesions, biochemistry of BSE ≈ v. CJD regardless of mouse type – Both differ from s. CJD
 Western Blots Jackson, G S et al. Mol Pathol 2001; 54: 393 -399
Western Blots Jackson, G S et al. Mol Pathol 2001; 54: 393 -399
 Continuing US Cattle Surveillance • USDA National Veterinary Services Laboratory – AAVLD certified labs, refer positives to NVSL • If 1 case per 1 million slaughter then 95% CI requires: – All slaughters = 2, 995, 731 – Suspect cattle = 40, 000 (neuro signs, fallen, “downers”) – 45, 803 samples in 2007 – Meets OIE “controlled risk” classification • National animal ID system
Continuing US Cattle Surveillance • USDA National Veterinary Services Laboratory – AAVLD certified labs, refer positives to NVSL • If 1 case per 1 million slaughter then 95% CI requires: – All slaughters = 2, 995, 731 – Suspect cattle = 40, 000 (neuro signs, fallen, “downers”) – 45, 803 samples in 2007 – Meets OIE “controlled risk” classification • National animal ID system
 Continuing US Human Surveillance • National Prion Disease Center – Established 1996 • Monitoring for unusual trends in mortality data • ↑ # of autopsies in US, ↑ # of referrals • CJD monitoring in CWD endemic areas
Continuing US Human Surveillance • National Prion Disease Center – Established 1996 • Monitoring for unusual trends in mortality data • ↑ # of autopsies in US, ↑ # of referrals • CJD monitoring in CWD endemic areas
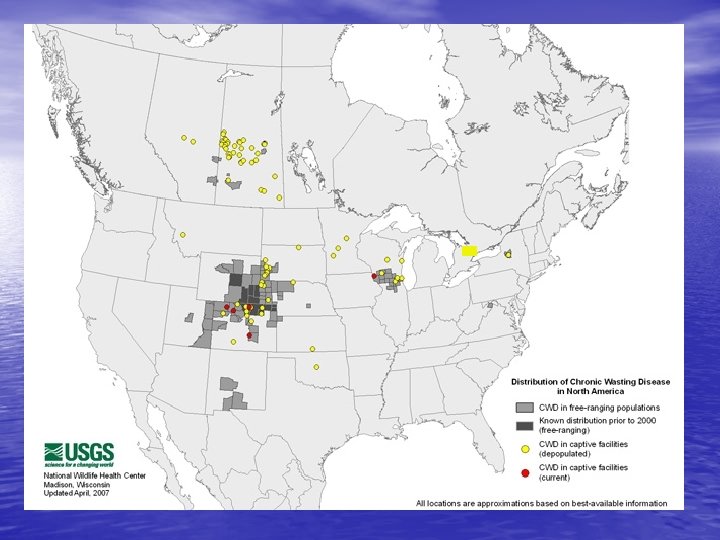
 CWD to Humans? • CJD flatline in endemic areas • Challenge studies indicate natural transmission is unlikely – Cattle – Mink – Humanized Tg mice
CWD to Humans? • CJD flatline in endemic areas • Challenge studies indicate natural transmission is unlikely – Cattle – Mink – Humanized Tg mice
 CWD risk reduction? • Hunting – Avoid endemic areas – Test animals in effected areas • Consumption – Don’t eat brain, nerves, spleen, lymph nodes, or eyes – Avoid composite foods (sausage, head cheese)
CWD risk reduction? • Hunting – Avoid endemic areas – Test animals in effected areas • Consumption – Don’t eat brain, nerves, spleen, lymph nodes, or eyes – Avoid composite foods (sausage, head cheese)
 Conclusion • Prion diseases vary by species, distinct differences • TSE transmission usually limited to within a species • Only Scrapie and CWD are readily transmissible • Species barrier limits transmission between species
Conclusion • Prion diseases vary by species, distinct differences • TSE transmission usually limited to within a species • Only Scrapie and CWD are readily transmissible • Species barrier limits transmission between species
 Questions?
Questions?
 Do you think this study supports the association of beef consumption as a primary risk factor for development of v. CJD?
Do you think this study supports the association of beef consumption as a primary risk factor for development of v. CJD?
 Did the study authors take adequate measures to address the limitations to the study design? What other measures could they have considered?
Did the study authors take adequate measures to address the limitations to the study design? What other measures could they have considered?
 Are there other ways that the question of risk factors for v. CJD may be addressed?
Are there other ways that the question of risk factors for v. CJD may be addressed?


