c6e6b712805de86b13ff03981a121b3d.ppt
- Количество слайдов: 77

Principle of Atomic Absorption Spectrophotometry Mr. Charnchai Suracheep

Introduction Atomic Absorption Spectrophotometry, which are standard instruments for the determination of metal elements, are widely applied of samples, such as agriculture chemical, clinical and biochemistry, minerals, food and drugs, environmental and other.

Principle of Atomic Absorption Spectrophotometer Principle of the Atomic Absorption Method Atomized elements each absorb energy of a wavelength that is peculiar to that element. The atomic absorption method uses as its light source a hollow cathode lamp which emits light of a wavelength that is peculiar to each element. Elements within a solution are heated in a flame or electrically (2000 K to 3000 K) and subsequently determined using the fact that the degree of absorption will vary with its concentration. Light absorption process of atoms
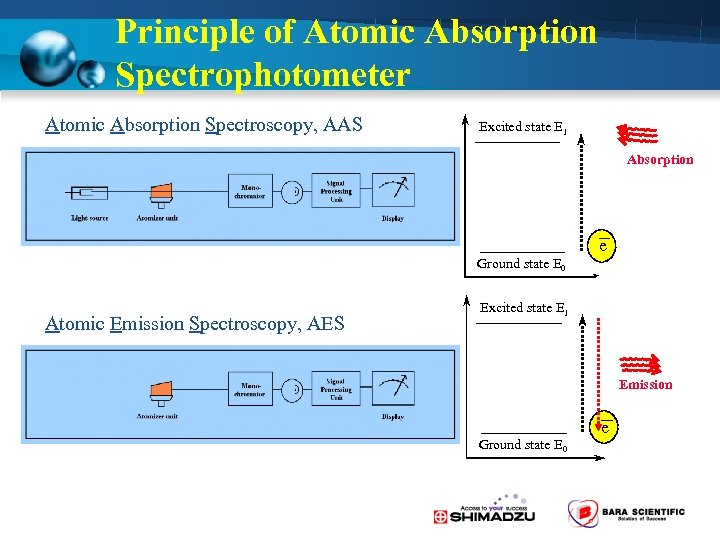
Principle of Atomic Absorption Spectrophotometer Atomic Absorption Spectroscopy, AAS Excited state E 1 Absorption Ground state E 0 Atomic Emission Spectroscopy, AES Excited state E 1 e e Emission Ground state E 0 e

Electronic Transition Sodium (Na) energy states Excited state (II) 3. 6 e. V Excited state (I) 2. 2 e. V 330. 3 nm 589. 0 nm Ground state 0. 0 e. V
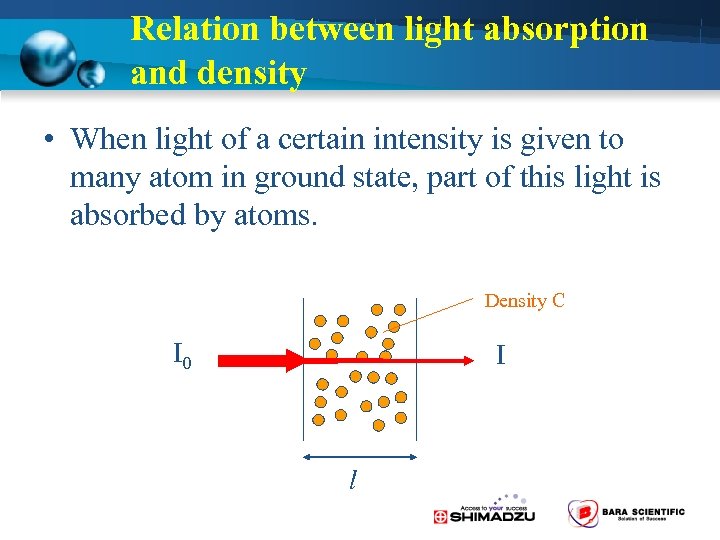
Relation between light absorption and density • When light of a certain intensity is given to many atom in ground state, part of this light is absorbed by atoms. Density C I 0 I l
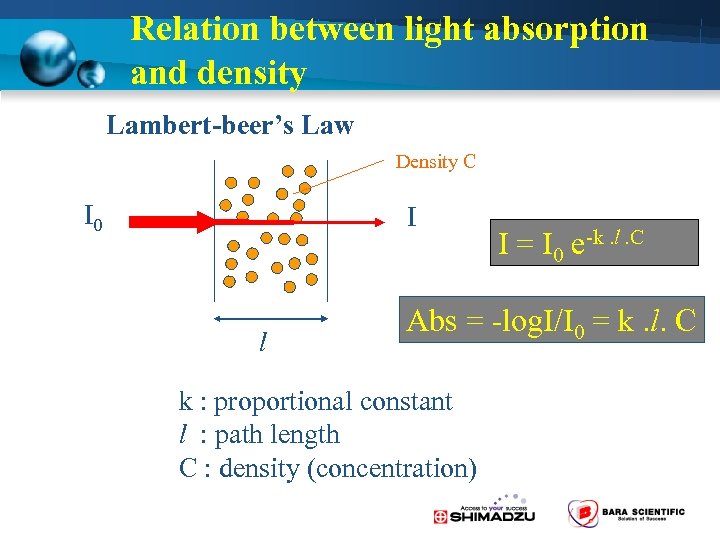
Relation between light absorption and density Lambert-beer’s Law Density C I 0 I l I = I 0 e-k. l. C Abs = -log. I/I 0 = k. l. C k : proportional constant l : path length C : density (concentration)
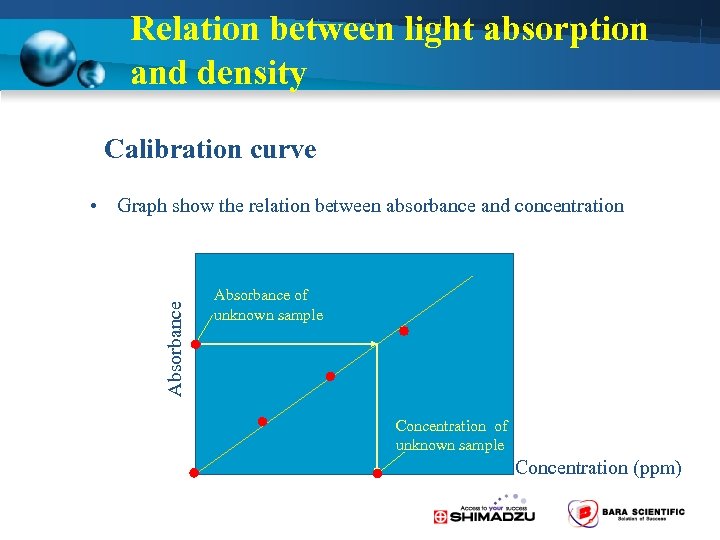
Relation between light absorption and density Calibration curve Absorbance • Graph show the relation between absorbance and concentration Absorbance of unknown sample Concentration (ppm)

Atomization method • Atomic absorption spectrometry measures absorption of free atom. • “Free atom” means an atom not combined with other atoms. • Elements in the sample to be analyzed are not in the free state, and are combined with other elements invariably to make a so-called molecule.

Atomization method • The combination must be cut off by some means to free the atoms. • This is called “atomization” • 2 types: - Flame method - Flameless method
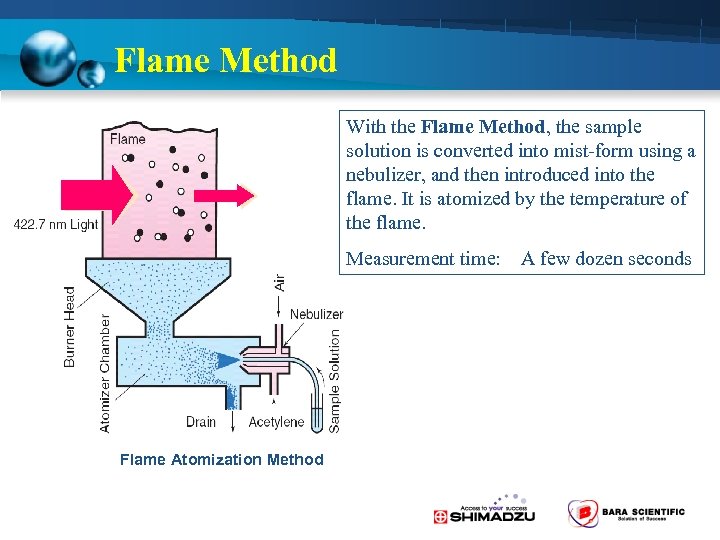
Flame Method With the Flame Method, the sample solution is converted into mist-form using a nebulizer, and then introduced into the flame. It is atomized by the temperature of the flame. Measurement time: A few dozen seconds Flame Atomization Method
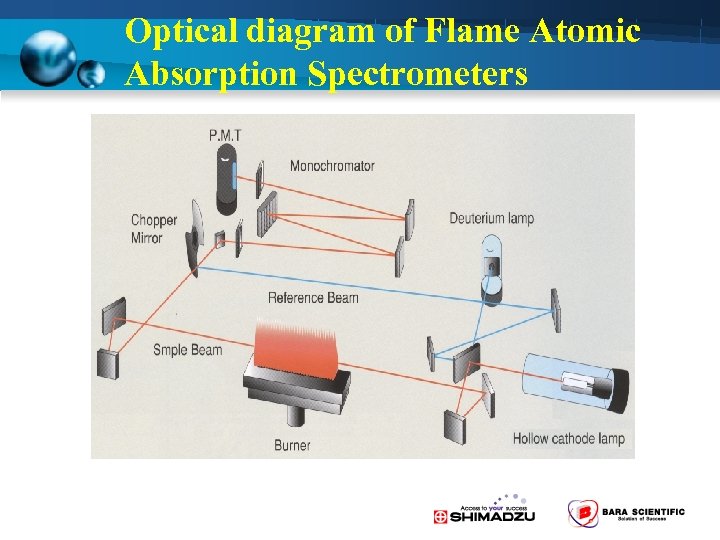
Optical diagram of Flame Atomic Absorption Spectrometers

Flame Method Flame selection • These flames vary in temperature, reducibility and transmission characteristics. • Selected according to the element being analyzed, and properties of the sample. • Argon-hydrogen : Max. temp. 1, 577 0 C • Air-hydrogen : Max. temp. 2, 045 0 C • Air-acetylene : Max. temp. 2, 300 0 C • Nitrous oxide-acetylene : Max. temp. 2, 955 0 C (For elements are hard to combine with oxygen (Al, Si, V, Ti, etc. ))

Flame Method Flame selection

Flameless Method (Graphite Furnace) Graphite holder Graphite cap Cooling block Sample inlet Aperture plate socket シール Seal Eject arm Graphite tube Spring Fixing knob Graphite tube

Flameless Method (Graphite Furnace) • Sample is injected in the formed graphite tube. • An electric current of 300 ampere (maximum) is applied to the tube.
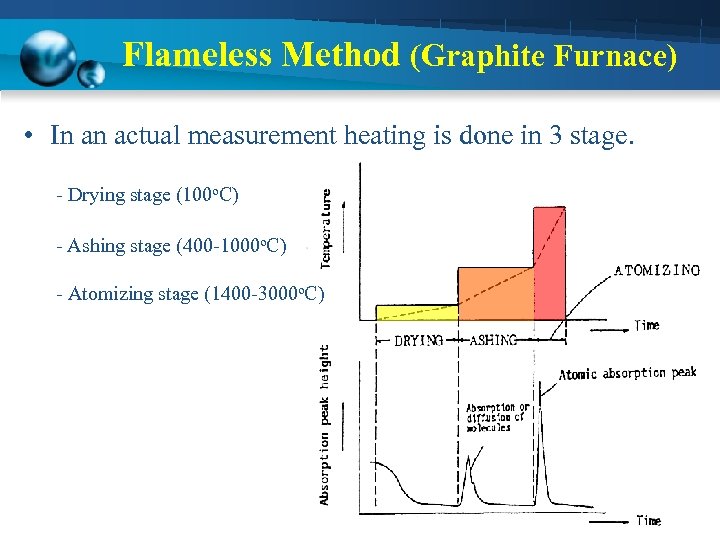
Flameless Method (Graphite Furnace) • In an actual measurement heating is done in 3 stage. - Drying stage (100 o. C) - Ashing stage (400 -1000 o. C) - Atomizing stage (1400 -3000 o. C)

Other atomic absorption methods • Methods having higher sensitivity than normal flame atomic absorption or electrothermal atomic absorption • Used for special elements including arsenic, selenium and mercury. • Use chemical reactions in the process of atomization to vaporize in the form of an atom or simple molecule.

Hydride Vapor Generation Technique • As, Se, Sb, Sn, Te, Bi, Hg and other metals produce a metal hydride by this method Elements Concentration (ppb) As 5~20 Sb 5~20 Te 5~20 Bi 5~20 Se 10~40 Hg 20~80 Sn 30~90 6 BH 4 - +As 3++ 3 H+ 3 B 2 H 6+3 H 2 +As. H 3 (gas) Peristal tic Manifold Pump Absorption Cell Burner Head of AAS Reactio n Coil Samp Na. BH 4 HCl le Carrier Gas Ar Structural Diagram of Hydride Vapor Generator Gas Liquid Separato r Drain
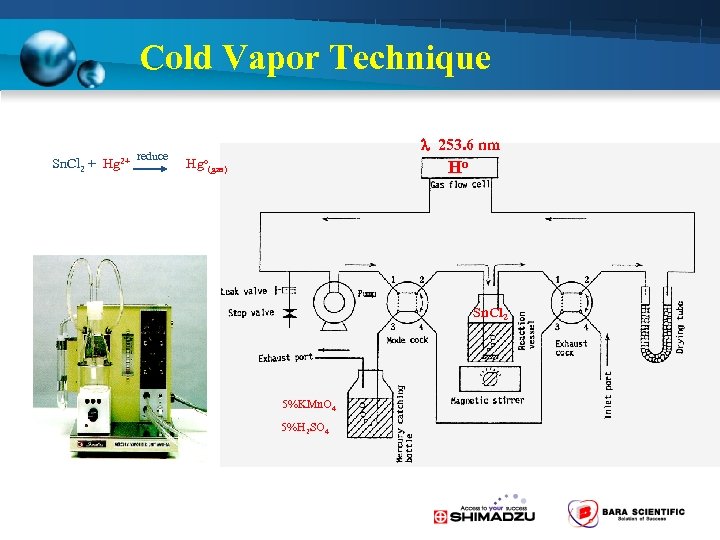
Cold Vapor Technique Sn. Cl 2 + Hg 253. 6 nm reduce 2+ Hgo(gas) Ho Sn. Cl 2 5%KMn. O 4 5%H 2 SO 4
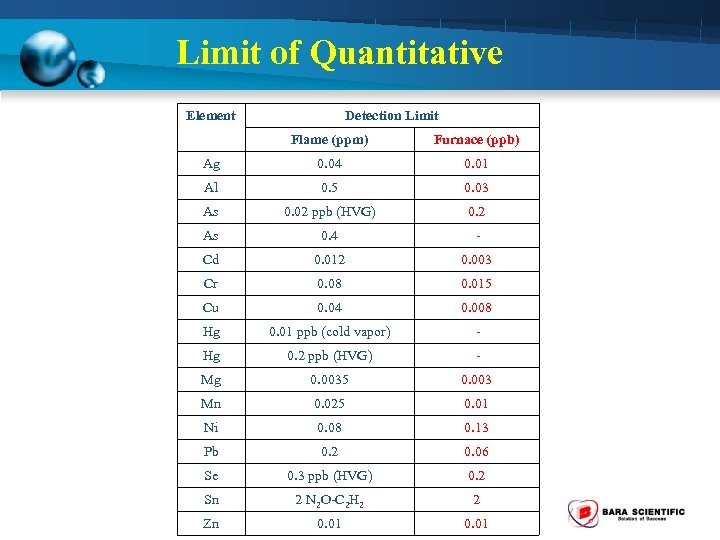
Limit of Quantitative Element Detection Limit Flame (ppm) Furnace (ppb) Ag 0. 04 0. 01 Al 0. 5 0. 03 As 0. 02 ppb (HVG) 0. 2 As 0. 4 - Cd 0. 012 0. 003 Cr 0. 08 0. 015 Cu 0. 04 0. 008 Hg 0. 01 ppb (cold vapor) - Hg 0. 2 ppb (HVG) - Mg 0. 0035 0. 003 Mn 0. 025 0. 01 Ni 0. 08 0. 13 Pb 0. 2 0. 06 Se 0. 3 ppb (HVG) 0. 2 Sn 2 N 2 O-C 2 H 2 2 Zn 0. 01

Interference effects • Physical interference • Spectral interference • Chemical interference

Physical interference • Flame – Spray efficiency fluctuations due to difference in viscosity and surface tension between the standard and sample. • Furnace – Sample dispersion ; Measurement value fluctuations due to tube temperature distribution – Viscosity within the graphite furnace ; Adherence to sample tip causing errors in collection quantity. • Example: samples, such as blood or juice, containing numerous organic components.

Spectral interference • Spectral absorption line overlapping with the absorption line of the target element. • Absorption and scattering by molecules
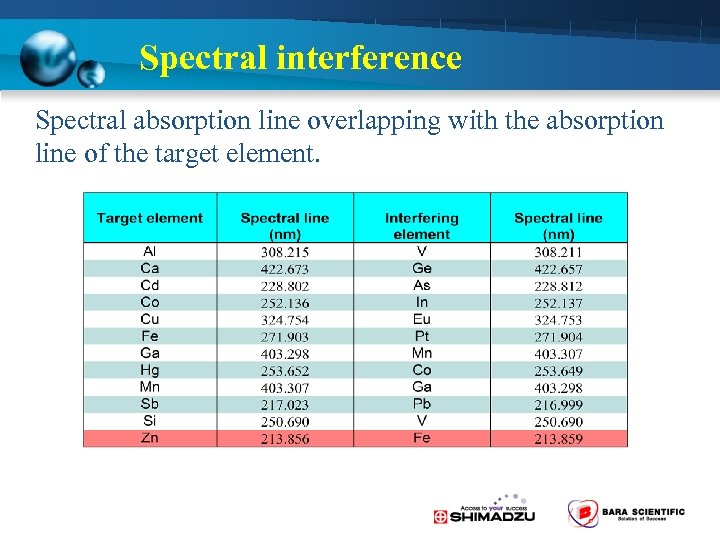
Spectral interference Spectral absorption line overlapping with the absorption line of the target element.
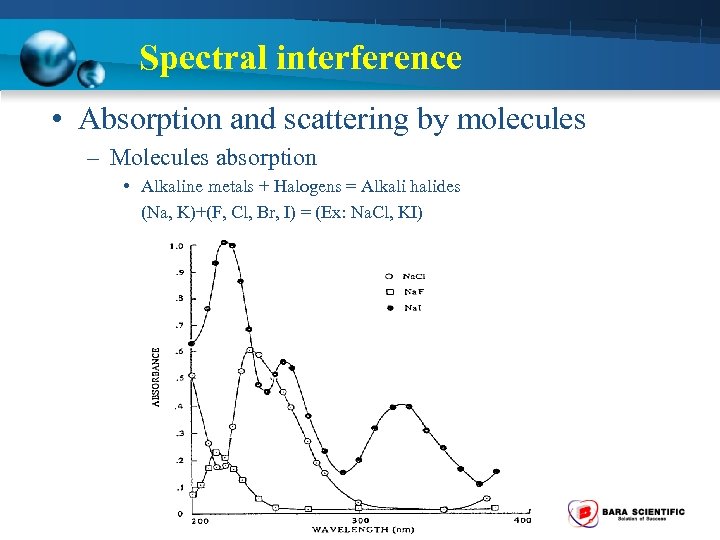
Spectral interference • Absorption and scattering by molecules – Molecules absorption • Alkaline metals + Halogens = Alkali halides (Na, K)+(F, Cl, Br, I) = (Ex: Na. Cl, KI)

Chemical interference • Generation of non-separable compounds by coexisting matrices –Example : influence of PO 4 -, Si. O 2 relative to Ca, Mg in flame analysis • (generation of Ca 2 PO 4) • Generation of low boiling point compounds by coexisting matrices –Example: influence of chloride ions relative to Cd in furnace analyses • (generation of Cd. Cl 2)

Matrix modifier effect • Masking of obstructing matrices • Influence of phosphate on Ca is masked by La • Conversion of obstructing matrices to compounds that easily undergo sublimation or evaporation – Sublimation agent • Example: removal of chloride ion by ammonium salt of nitric acid or phosphoric acid • Conversion of measured elements to stable oxides or metallic intermediary compounds – Stabilizing agent: • Example: creation of measured element alloy using white metals (Pd, Pt, Rh)

Application examples of the matrix modifier method

Standard Addition Method No. 1 No. 4 No. 2 No. 3 10 ml 20 ml 10 ml X X+0. 1 X+0. 2 X+0. 3 100 ml Mg concentration after filled up 10 ml Unknown sample 30 ml Solvent 1. 0 ppm X Standard solution (ppm : mg/1000 ml)

Standard Addition Method Calibration Curve of Standard Addition Method Concentration of unknown sample

Background Correction 2 -Way Background Correction is Standard • D 2 lamp method ( 190 -430 nm) – Molecular absorption • Self-Reversal (SR) method – Spectra interference
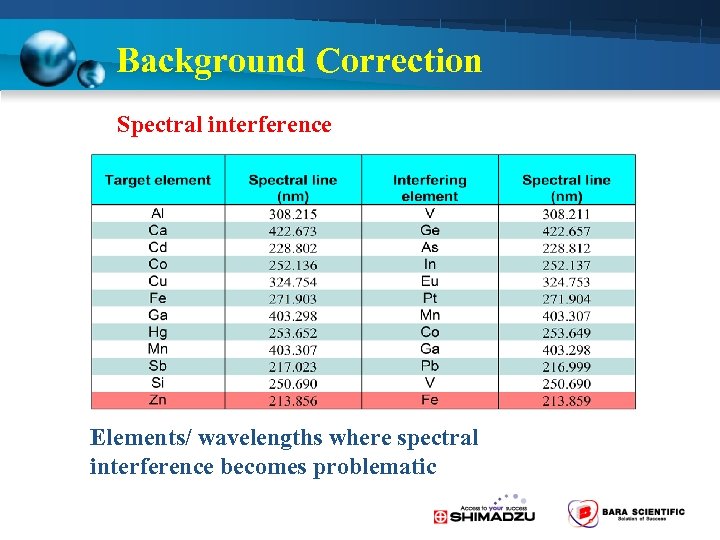
Background Correction Spectral interference Elements/ wavelengths where spectral interference becomes problematic
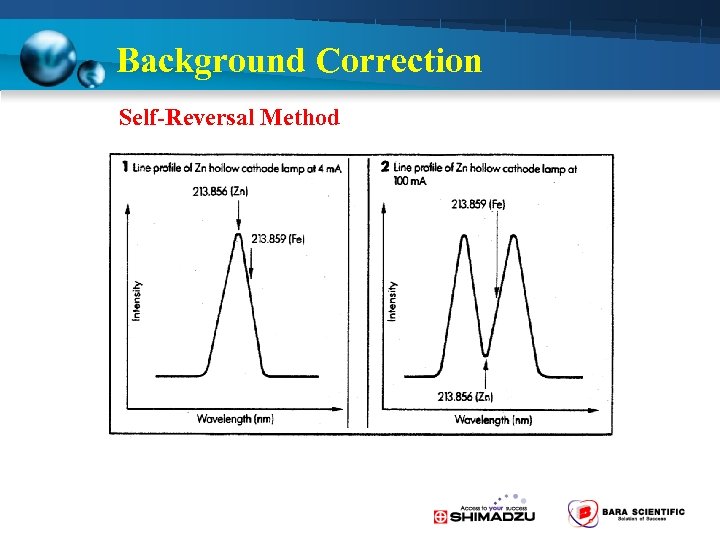
Background Correction Self-Reversal Method
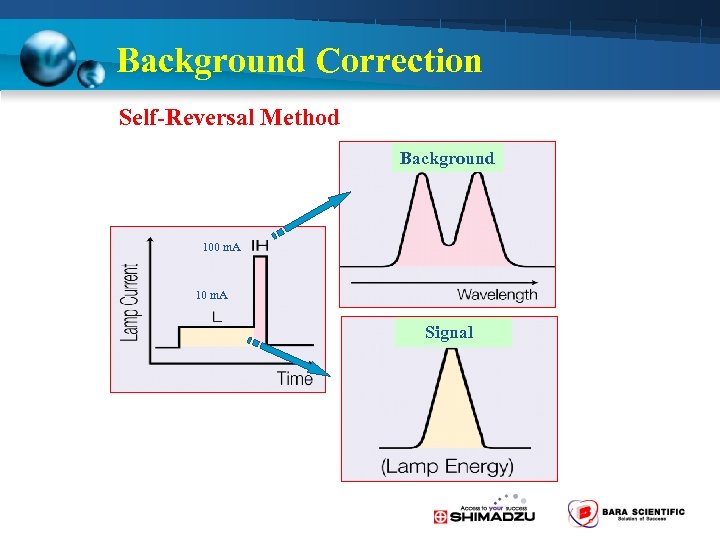
Background Correction Self-Reversal Method Background 100 m. A 10 m. A Signal

Atomic Absorption Spectrophotometer AA-6300
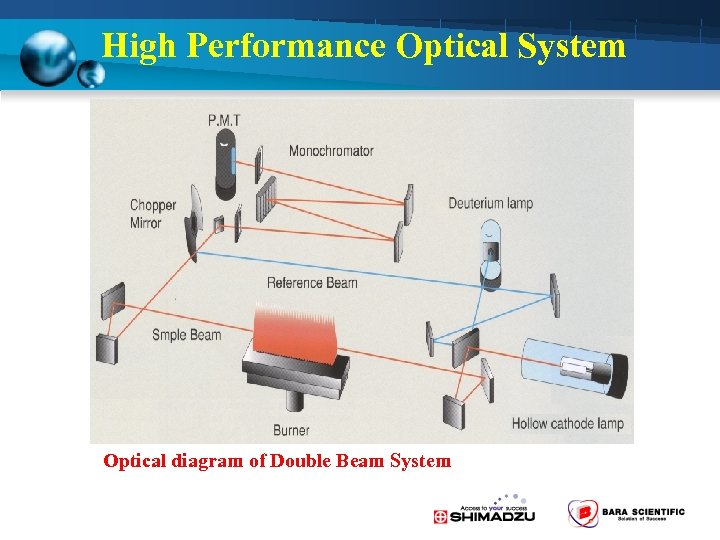
High Performance Optical System Optical diagram of Double Beam System

Easy Switching between Flame and Furnace Flame -> Furnace: All that is involved is to remove the burner head, place the furnace unit, and fix it with the screw. No tools are required. Remove the burner head. Fit the furnace. Fit the burner head. Remove the furnace.
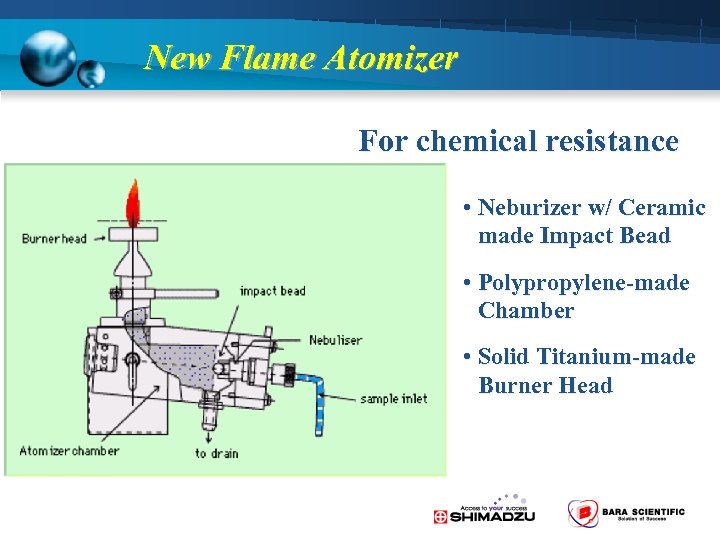
New Flame Atomizer For chemical resistance • Neburizer w/ Ceramic made Impact Bead • Polypropylene-made Chamber • Solid Titanium-made Burner Head

High Productivity • Full Auto ASC - Auto measurement up to 60 samples - Reagent addition 8 position - Automatic dilution • Optimize Flame analysis - Automatic search the best fuel gas flow rate - Automatic search the Optimize Flame analysis best burner height

Enhanced Safety • Auto Gas Leak Check • Gas pressure monitoring to prevent flashback • Automatic flame monitoring • Automatic flame extinguish when power failure High Temp. Burner • Safety interlock for burner misuse • Auto Air/N 2 O flame changeover • Drain level sensor
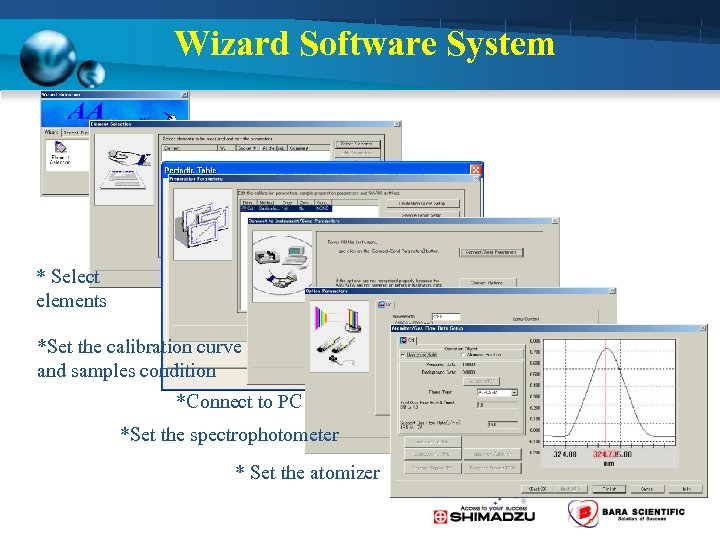
Wizard Software System * Select elements *Set the calibration curve and samples condition *Connect to PC *Set the spectrophotometer * Set the atomizer

Automated/ Optimized Effectiveness of the automatic Line Search/Beam Balance

Automated/ Optimized Effectiveness of the automatic burner height (Cr : 4 ppm standard solution used) Burner height & Sensitivity (Cr)

Automated/ Optimized Search for the optimal fuel flow rate (Cu : 4 ppm standard solution used)
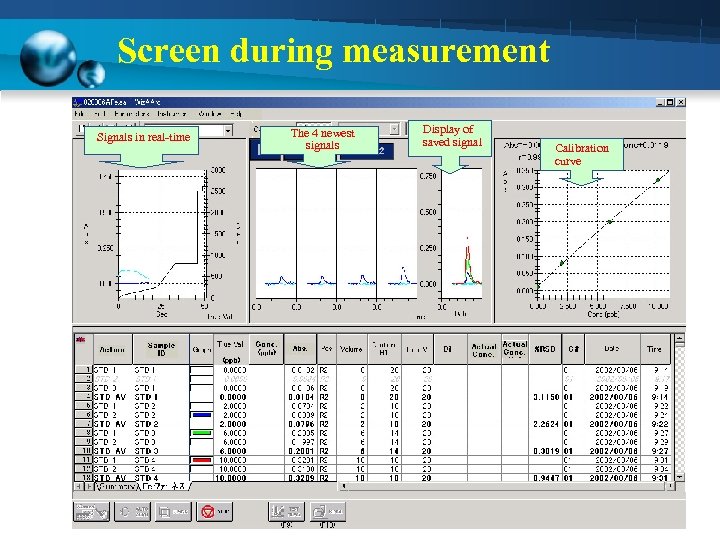
Screen during measurement Signals in real-time The 4 newest signals Display of saved signal Calibration curve
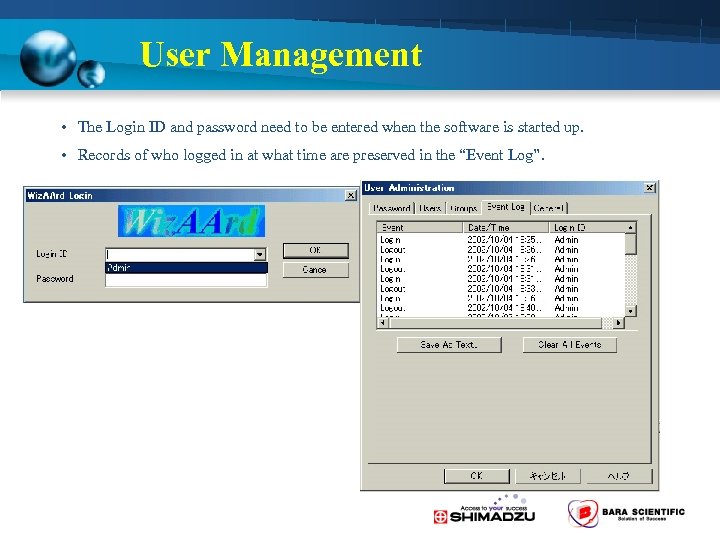
User Management • The Login ID and password need to be entered when the software is started up. • Records of who logged in at what time are preserved in the “Event Log”.
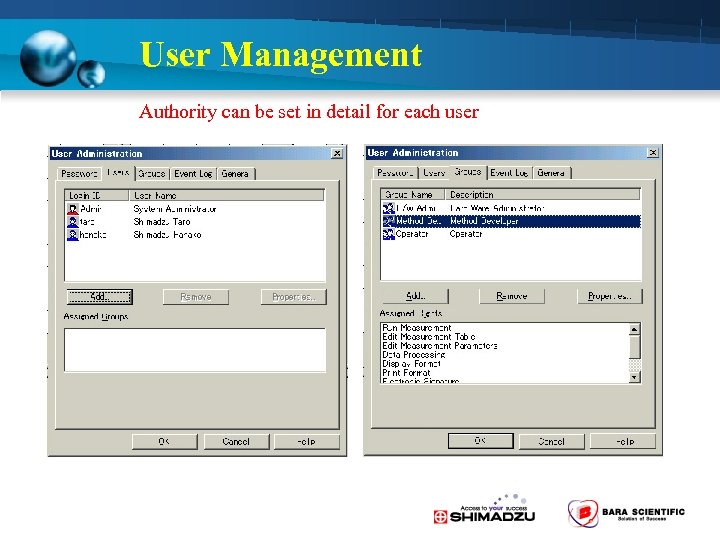
User Management Authority can be set in detail for each user
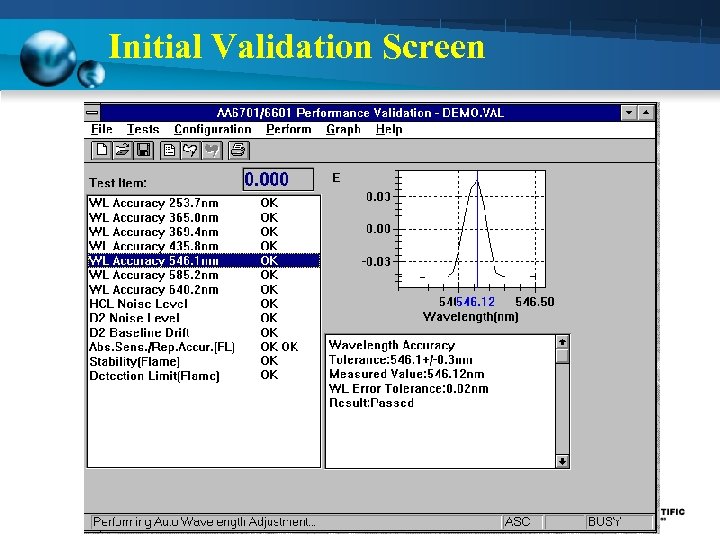
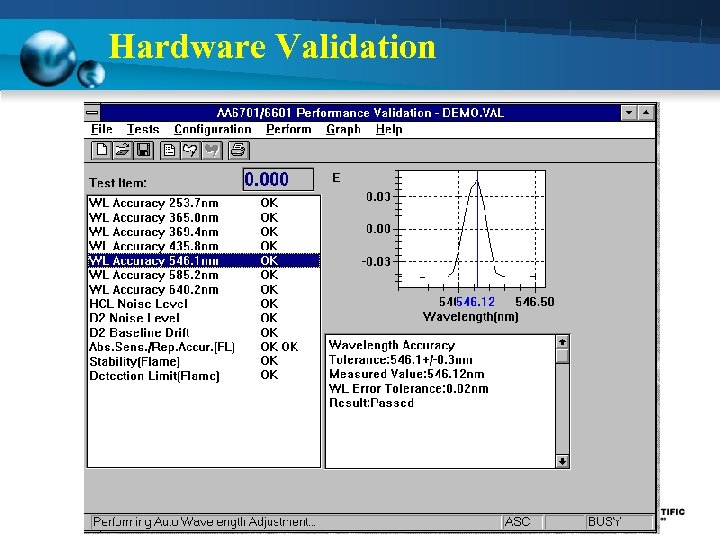
Initial Validation Screen
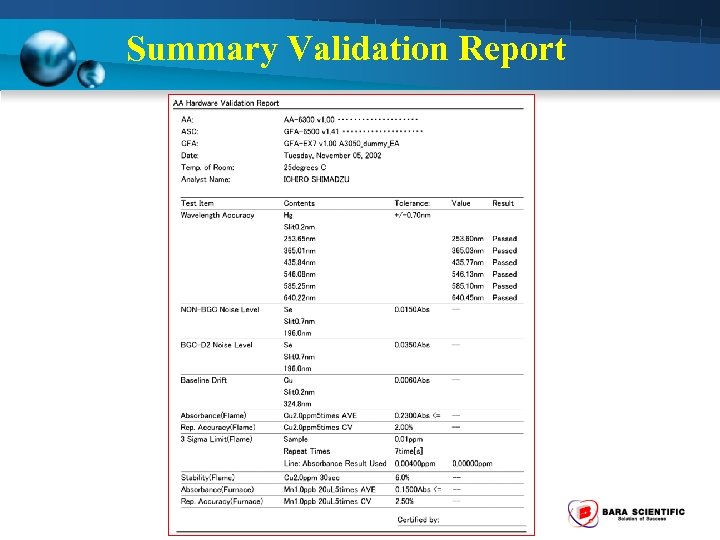
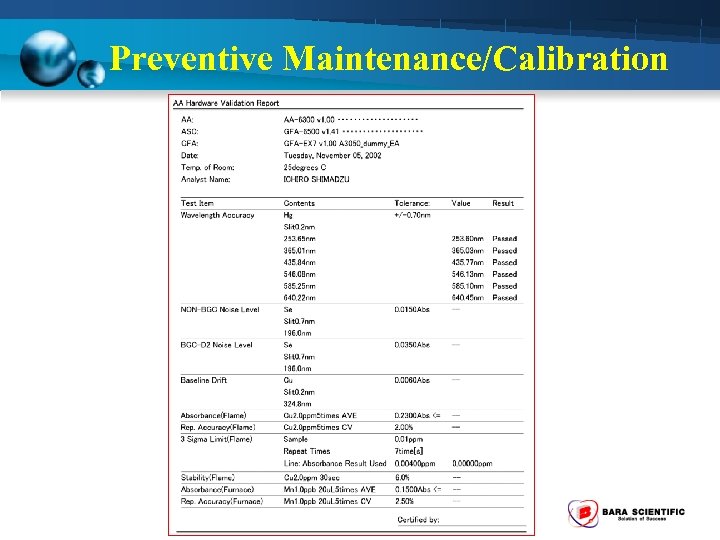
Summary Validation Report

Application of Atomic Absorption Spectrophotometry
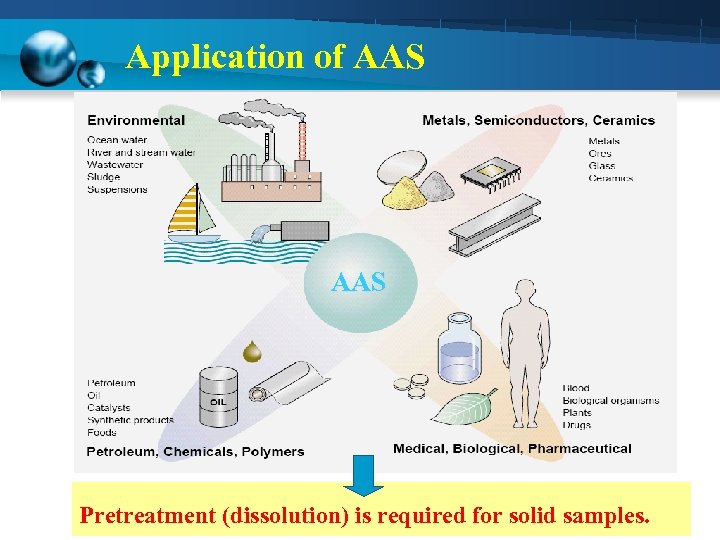
Application of AAS Pretreatment (dissolution) is required for solid samples.

Pretreatment u. Precautions for pretreatment: v Dissolve all the elements into the same solution evenly. (Check with certified reference material. ) v Ensure that elements are not lost in the solution. i. e. , due to vaporization or sedimentation (Check with recovery test. ) v Contamination : Purified water, reagent (e. g. , acid), container, environment. (Check with blank operation. ) v Ensure that the solution to be analyzed is stable for a long time (i. e. , no hydrolysis or sedimentation). v Consider the interference effect of the reagent on the analysis values.

Types of Pretreatment u Dilution Dilute the sample with purified water, dilute acid, or organic solvents. Examples: food products (e. g. , dairy products), pharmaceuticals, and biological samples (e. g. , blood, urine). u Dry Decomposition Heat the sample to a high temperature (400 to 500 C), Decomposition is possible in a short time (a few hours) and operation is simple. Elements with low boiling points (e. g. , Hg, As, Se, Te, and Sb) will vaporize u Wet Decomposition Heat the sample together with acid to a low temperature (approx. 300 C). Suitable for volatile elements. A long time is required for the decomposition of organic substances. u Microwave Decomposition Decompose the sample at high pressure by heating it together with acid to a temperature in the range 100 to 200 C in a sealed Teflon container. The decomposition process is sealed; there is little vaporization of elements with low boiling points; the decomposition time is short; there is little contamination from the operating environment and the reagent; and only a small amount of acid is required. Examples: Sediment, soil, dust, ceramics, living organisms, food products, etc.

Wet Decomposition Method Kjeldahl flask wet decomposition method Waste gas Cooling tube Nitric acid Simple method (no cooling) Sample+ Sulfuric acid Heating

Pretreatment Microwave Decomposition u Decompose the sample together with an acid in a sealed container. u Decomposition possible in a short time with little vaporization or contamination. - Ideal for the pretreatment of trace elements and trace samples. - Food products, living organisms, pharmaceuticals, airborne dust, soil, etc. High-pressure Decomposition Container Microwave Digestion
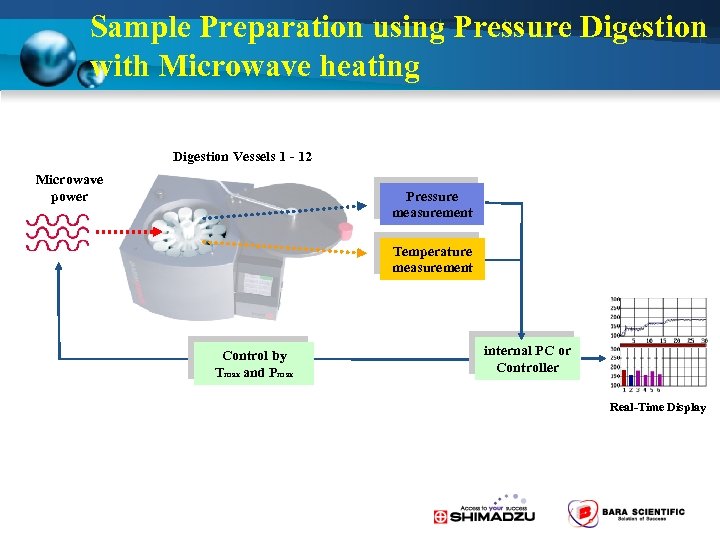
Sample Preparation using Pressure Digestion with Microwave heating Digestion Vessels 1 - 12 Microwave power Pressure measurement Temperature measurement Control by Tmax and Pmax internal PC or Controller Real-Time Display

Pretreatment Solubility of Elements in Samples Total Content Simple water-soluble ions Simple soluble metals & compounds Organic compounds Inorganic compounds with low solubility Sulfides, oxides, silicates, etc. Carbonates, oxides, etc. Pretreatment Methods Dilution, Elution Wet Decomposition Purified water, Hydrochloric acid, nitric acid, etc. solvents, etc. Dry/Wet Decomposition Microwave Decomposition Nitric acid, sulfuric acid, etc. Wet/High-pressure Decomposition Hydrofluoric acid, nitric acid, etc.

Example Application of AAS

EU Regulation for Hazardous Substances

EU Regulation for Hazardous Substances IEC Recommendation for Ro. HS : Restriction of Hazardous Substance in Electrical and Electronic equipment. Substances PBB/PBDE : 1000 ppm Cr 6+ : 1000 ppm Polymers Metals Electronics GC-MS NA GC-MS Colorimetric Method Spot-test (Spectrophotometer) procedure/boiling=water. Extraction procedure (Clause 8) Hg : 1000 ppm Pb : 1000 ppm Cd : 100 ppm Colorimetric Method (Spectrophotometer) Cold Vapor-AAS, ICP

Preparation of circuit boards Pre-Cutting with the Heavy Duty Cutting Mill , bottom sieve 6 mm Heavy Duty Cutting Mill SM 2000 after a grinding time of 2 min. endfineness 90 % < 125 µm Vibratory Disc Mill RS 100

Sample Preparation Targ et Elem ent Hg Pretreatment Methods Polymer Metals Electronics Microwave digestion )HNO+3 HBF+4 H 2 O(2 a) Common method Microwave (HCl : HNO 3 : water ; 2 : Microwave digestion Step A 1 : 2) b) If Cd digestion (HNO 3+HBF 4+H 2 O 2 containing Zr, Hf, Ti, )HNO+3 H 2 O (2 ( Ta, Nb, W (HNO 3 : Pretreatment method, which follow by IEC 62321 Pb )If contain ing Si, Microwave HF ; 1 : 3) digestion Step B Ti add HF( c) If containing Sn (add HCl( (HCl : HNO 3 ; 3 : 1

Analyzing Cadmium (Cd) in Rice Pretreatment Using Wet Decomposition Level suggested by FAO/WHO Codex Committee ; 0. 2 ppm max. in polished rice (proposed) Put 5 g of the sample in a beaker. Add 30 m. L of nitric acid (1+1) and 0. 5 m. L of sulfuric acid. Warm on a hot plate until the violent reaction subsides. ↓ Perform thermal decomposition until the contents approach a hardened and dried state. When the contents turn dark brown, add 1 m. L of nitric acid. Repeat this process. When the contents turn light yellow or become transparent, expel the white smoke of the sulfuric acid and leave to cool. Add nitric acid. Heat on the hot plate to dissolve the salt content. Leave to cool. Dilute for measurement.
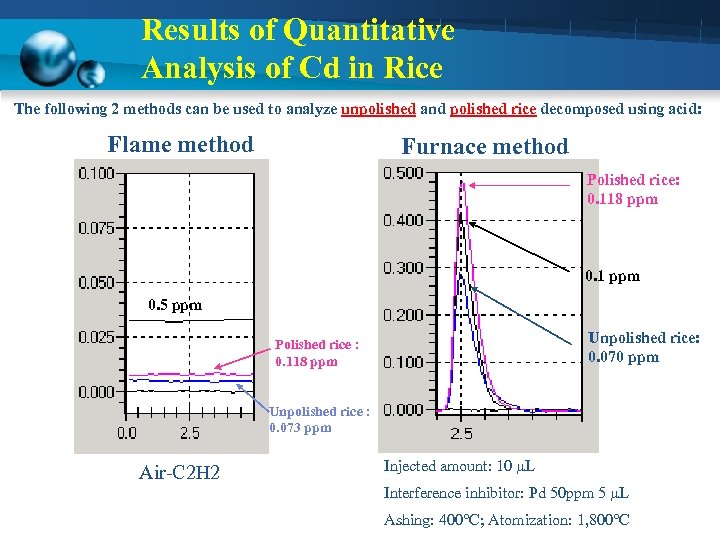
Results of Quantitative Analysis of Cd in Rice The following 2 methods can be used to analyze unpolished and polished rice decomposed using acid: Flame method Furnace method Polished rice: 0. 118 ppm 0. 1 ppm 0. 5 ppm Unpolished rice: 0. 070 ppm Polished rice : 0. 118 ppm Unpolished rice : 0. 073 ppm Air-C 2 H 2 Injected amount: 10 µL Interference inhibitor: Pd 50 ppm 5 µL Ashing: 400 C; Atomization: 1, 800 C

Summary of Methods for Analyzing Cd in Rice â Comparison of Pretreatment Methods Wet oxidation: 3 to 5 hours; Dry ashing: 5 to 10 hours; Microwave: 1 hour; Acid extraction: 2 hours â Expected Lower Limits for Quantitative Measurement Flame method : 0. 100 ppm Furnace method : 0. 001 ppm â Comparison of Measurement Times for Each Measurement Method (n=3) Flame method : 30 s Furnace method : 360 s

Preventive Maintenance / Calibration of Atomic Absorption Spectrophotometry

Preventive Maintenance/Calibration Maintenance Weekly check 1. Cleaning the Burner head Clogged (by carbide or salt etc. ) Normal

Preventive Maintenance/Calibration Maintenance Weekly check 2. Cleaning the Chamber with diluted water or alcohol Burner Head Nebulizer Construction Sample Solution O-ring Chamber Disperser O-ring Fixing Plate Air Nebulizer Drain Sample Suction

Preventive Maintenance/Calibration Maintenance Weekly check 3. Cleaning the Nebulizer Cleaning wire Nebulizer Do not apply the ultrasonic cleaner to the nebulizer
Hardware Validation

Preventive Maintenance/Calibration (12 Month) 1. Wavelength Accuracy - Using Hg hollow cathode lamp set at Emission mode - Measure peaks should be within + 0. 7 nm (253. 6 nm 365. 0 nm 435. 8 nm 546. . 1 nm 585. 2 640. 2 nm) 2. Noise Level - Using Se hollow cathode lamp ( 196 nm) - NON-BGC Noise level should be < 0. 015 Abs. - BGC-D 2 Noise level should be < 0. 035 Abs. 3. Baseline Drift - Using Cu hollow cathode lamp ( 324. 8 nm) - Measuring time 1800 sec - Measured value less than 0. 006 Abs

Preventive Maintenance/Calibration (12 Month) 4. Absorption - Using Cu hollow cathode lamp - Standard Cu 2 ppm - Measured value more than 0. 23 Abs 5. Repeatability - Using Cu hollow cathode lamp - Standard Cu 2 ppm - Measure 5 time and CV value < 2%

Preventive Maintenance/Calibration (12 Month) 6. Detection Limit - Using Cu hollow cathode lamp - Standard Cu 2 ppm - Measure Standard is 3 -5 time and calculate the mean value (A) - Measure Blank solution is 3 -5 time and calculate the standard deviation (S) - Take the obtained value as the detection limit < 0. 004 Abs. Detection limit = (2. 0 x 3 x S) / A

Preventive Maintenance/Calibration (12 Month) 7. Stability - Using Se and Cu hollow cathode lamp - Standard Cu 2 ppm - Measure std. Cu around 5 sec (B) - Measure std. Cu continuous around 30 sec and measure amplitude of Abs. value (W) - Take the ratio of W to B Stability = W/B < 6. 0 %
Preventive Maintenance/Calibration

c6e6b712805de86b13ff03981a121b3d.ppt