9ede1e56bc07d2c8e31ec1c445bd09a7.ppt
- Количество слайдов: 26
 PREQUALIFICATION General overview and procedures for the procurement agencies involved in procurement Procurement and Supply Management for HIV/AIDS, TB and Malaria Workshop 13 -16 March 2006, Moscow, RF Alexander Polishchuk Special Project on Pharmaceutical for NIS, Health Technologies and Pharmaceuticals World Health Organization Regional Office for Europe E-mail: apo@euro. who. int
PREQUALIFICATION General overview and procedures for the procurement agencies involved in procurement Procurement and Supply Management for HIV/AIDS, TB and Malaria Workshop 13 -16 March 2006, Moscow, RF Alexander Polishchuk Special Project on Pharmaceutical for NIS, Health Technologies and Pharmaceuticals World Health Organization Regional Office for Europe E-mail: apo@euro. who. int
 Prequalification of essential medicines q The UN prequalification program is an action plan for expanding access for the hardest hit by q q Tuberculosis q q HIV/AIDS Malaria for ensuring quality, efficacy and safety of medicines all the way through the medicines supply chain. Health Technologies and Pharmaceuticals 2 WHO - PSM
Prequalification of essential medicines q The UN prequalification program is an action plan for expanding access for the hardest hit by q q Tuberculosis q q HIV/AIDS Malaria for ensuring quality, efficacy and safety of medicines all the way through the medicines supply chain. Health Technologies and Pharmaceuticals 2 WHO - PSM
 Why the prequalification is needed q Problems • • q Millions of people living with HIV/AIDS, tuberculosis and malaria, have no or limited access to treatment Procurement and supply of substandard and counterfeit products in different countries Weak/absent QA systems of medicines supply chain Lot of money invested in procurement no harmonized quality assurance system available for procurement organizations Risks • Sourcing of poor quality products or even counterfeit medicines risk to patients, treatment failure, resistance Health Technologies and Pharmaceuticals 3 WHO - PSM
Why the prequalification is needed q Problems • • q Millions of people living with HIV/AIDS, tuberculosis and malaria, have no or limited access to treatment Procurement and supply of substandard and counterfeit products in different countries Weak/absent QA systems of medicines supply chain Lot of money invested in procurement no harmonized quality assurance system available for procurement organizations Risks • Sourcing of poor quality products or even counterfeit medicines risk to patients, treatment failure, resistance Health Technologies and Pharmaceuticals 3 WHO - PSM
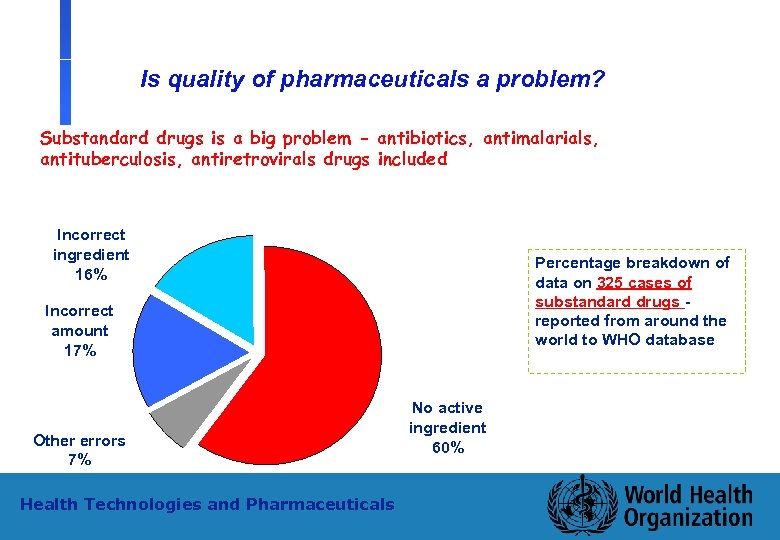 Is quality of pharmaceuticals a problem? Substandard drugs is a big problem - antibiotics, antimalarials, antituberculosis, antiretrovirals drugs included Incorrect ingredient 16% Percentage breakdown of data on 325 cases of substandard drugs reported from around the world to WHO database Incorrect amount 17% Other errors 7% Health Technologies and Pharmaceuticals 4 No active ingredient 60% WHO - PSM
Is quality of pharmaceuticals a problem? Substandard drugs is a big problem - antibiotics, antimalarials, antituberculosis, antiretrovirals drugs included Incorrect ingredient 16% Percentage breakdown of data on 325 cases of substandard drugs reported from around the world to WHO database Incorrect amount 17% Other errors 7% Health Technologies and Pharmaceuticals 4 No active ingredient 60% WHO - PSM
 Expected outcome of prequalification q List of products and manufacturers/CROs • q Harmonization • q Meeting international norms and standards on quality, safety, and efficacy (Q, S & E) Co-operation, training, capacity building – NDRAs, WHO, NGOs Facilitate access to treatment • • • Procurement mechanisms (e. g. tender, competition) Ongoing monitoring of Quality, Safety & Efficacy WHO commitment to developing Health Technologies and Pharmaceuticals 5 WHO - PSM
Expected outcome of prequalification q List of products and manufacturers/CROs • q Harmonization • q Meeting international norms and standards on quality, safety, and efficacy (Q, S & E) Co-operation, training, capacity building – NDRAs, WHO, NGOs Facilitate access to treatment • • • Procurement mechanisms (e. g. tender, competition) Ongoing monitoring of Quality, Safety & Efficacy WHO commitment to developing Health Technologies and Pharmaceuticals 5 WHO - PSM
 Steps of prequalification 1. Expression of interest (EOI) from a prospective supplier interested in a voluntary participation in the program. 2. Receipt of the dossier at UNICEF in Copenhagen and the Site Master File in WHO Geneva. q Explicative notes and guidelines are published on the WEB in order to explain how to bring together a product dossier meeting the requirements for prequalification. 3. Screening of the dossier, "Quality" part, "Clinical" part and samples. possible inspection 4. Assessment of the dossier and writing of the assessment report and assessment letter. 5. Outcome of the evaluation communicated to supplier Health Technologies and Pharmaceuticals 6 WHO - PSM
Steps of prequalification 1. Expression of interest (EOI) from a prospective supplier interested in a voluntary participation in the program. 2. Receipt of the dossier at UNICEF in Copenhagen and the Site Master File in WHO Geneva. q Explicative notes and guidelines are published on the WEB in order to explain how to bring together a product dossier meeting the requirements for prequalification. 3. Screening of the dossier, "Quality" part, "Clinical" part and samples. possible inspection 4. Assessment of the dossier and writing of the assessment report and assessment letter. 5. Outcome of the evaluation communicated to supplier Health Technologies and Pharmaceuticals 6 WHO - PSM
 Steps of prequalification cont… 6. Inspection of the site (s) of manufacturing and follow-up inspection when necessary GMP compliant list of manufacturers 7. Inspection of the Research Laboratory or Contract Research Laboratory (CRO) where the bioequivalence study has been performed GCP compliant list of CROs 8. Conclusion and listing of the product in the prequalification list 9. Publication of the Public Assessment (WHOPAR) and Inspection (WHOPIR) Reports 10. Assessment of the variation when submitted, market survey, de-listing if necessary 11. Re-qualification after 3 years Health Technologies and Pharmaceuticals 7 WHO - PSM
Steps of prequalification cont… 6. Inspection of the site (s) of manufacturing and follow-up inspection when necessary GMP compliant list of manufacturers 7. Inspection of the Research Laboratory or Contract Research Laboratory (CRO) where the bioequivalence study has been performed GCP compliant list of CROs 8. Conclusion and listing of the product in the prequalification list 9. Publication of the Public Assessment (WHOPAR) and Inspection (WHOPIR) Reports 10. Assessment of the variation when submitted, market survey, de-listing if necessary 11. Re-qualification after 3 years Health Technologies and Pharmaceuticals 7 WHO - PSM
 Assessment procedure q Assessment of products dossiers i. e. quality, specifications, pharmaceutical development, bioequivalence etc. • teams of professionals from national drug regulatory authorities (DRA): • Copenhagen assessment week – 8 to 12 assessors together during one week at least every two months at UNICEF in Copenhagen – Every dossier is assessed by at least two assessors. – An assessment report is issued; signed by two assessors – Letter summarizing the findings and asking for clarification and additional data if necessary. • Letter is sent first by e-mail to the applicant followed by surface mail Brazil, Canada, Denmark, Estonia, Finland, France, Germany, Hungary, Indonesia, Malaysia, Philippines, Spain, South-Africa, Sweden, Switzerland, Tanzania, Zimbabwe. . . Health Technologies and Pharmaceuticals 8 WHO - PSM
Assessment procedure q Assessment of products dossiers i. e. quality, specifications, pharmaceutical development, bioequivalence etc. • teams of professionals from national drug regulatory authorities (DRA): • Copenhagen assessment week – 8 to 12 assessors together during one week at least every two months at UNICEF in Copenhagen – Every dossier is assessed by at least two assessors. – An assessment report is issued; signed by two assessors – Letter summarizing the findings and asking for clarification and additional data if necessary. • Letter is sent first by e-mail to the applicant followed by surface mail Brazil, Canada, Denmark, Estonia, Finland, France, Germany, Hungary, Indonesia, Malaysia, Philippines, Spain, South-Africa, Sweden, Switzerland, Tanzania, Zimbabwe. . . Health Technologies and Pharmaceuticals 8 WHO - PSM
 Assessment procedure-Product dossiers q Innovator products • • q Multisource products (generics) • • q Assessment report from DRAs WHO type Certificate of Pharmaceutical Product (CPP) Batch certificate Update on changes. Full dossier with data and information Quality : information on starting materials and finished product including API details, specifications, stability data, formulation, manufacturing method, packaging, labelling etc Efficacy: Bio-equivalence study or clinical study report US FDA tentative approvals – recognition based on information exchange (Confidentiality agreement) Commercial sample Health Technologies and Pharmaceuticals 9 WHO - PSM
Assessment procedure-Product dossiers q Innovator products • • q Multisource products (generics) • • q Assessment report from DRAs WHO type Certificate of Pharmaceutical Product (CPP) Batch certificate Update on changes. Full dossier with data and information Quality : information on starting materials and finished product including API details, specifications, stability data, formulation, manufacturing method, packaging, labelling etc Efficacy: Bio-equivalence study or clinical study report US FDA tentative approvals – recognition based on information exchange (Confidentiality agreement) Commercial sample Health Technologies and Pharmaceuticals 9 WHO - PSM
 Inspection procedure Inspections Manufacturing site (FPP, packaging) q Active pharmaceutical ingredient (API) q Research laboratory/Contract Research Organization (CRO) q Teamwork of inspectors • WHO representative (qualified GMP inspector) • Inspector from well-established inspectorate (Pharmaceutical Inspection Convention Scheme PIC/S countries) • National inspector(s): Canada, India, China, France, Italy, Switzerland, South-Africa, Thailand… Quality control analysis - upon need but not always necessarily before prequalification and supply; increasingly as part of follow-up q Health Technologies and Pharmaceuticals 10 WHO - PSM
Inspection procedure Inspections Manufacturing site (FPP, packaging) q Active pharmaceutical ingredient (API) q Research laboratory/Contract Research Organization (CRO) q Teamwork of inspectors • WHO representative (qualified GMP inspector) • Inspector from well-established inspectorate (Pharmaceutical Inspection Convention Scheme PIC/S countries) • National inspector(s): Canada, India, China, France, Italy, Switzerland, South-Africa, Thailand… Quality control analysis - upon need but not always necessarily before prequalification and supply; increasingly as part of follow-up q Health Technologies and Pharmaceuticals 10 WHO - PSM
 Current status – December 2005 q Started with HIV/AIDS products in 2001 – malaria and TB products joined later q Prequalified products (Dec 2005) • • 105 8 2 115 HIV related medicines anti-tuberculosis medicines anti-malarial medicines Dossiers arrived - 316 (Aug-05) - 156 - 48 520 343 (Dec -05) 165 49 557 q Ongoing assessments and follow-up • • • Products Manufacturing sites CROs Health Technologies and Pharmaceuticals 11 WHO - PSM
Current status – December 2005 q Started with HIV/AIDS products in 2001 – malaria and TB products joined later q Prequalified products (Dec 2005) • • 105 8 2 115 HIV related medicines anti-tuberculosis medicines anti-malarial medicines Dossiers arrived - 316 (Aug-05) - 156 - 48 520 343 (Dec -05) 165 49 557 q Ongoing assessments and follow-up • • • Products Manufacturing sites CROs Health Technologies and Pharmaceuticals 11 WHO - PSM
 Current status – Manufacturers of finished products q In the prequalification list: 15 sites of generics q q q Asia: 9 sites Europe: 4 sites Africa: 2 sites Health Technologies and Pharmaceuticals 12 WHO - PSM
Current status – Manufacturers of finished products q In the prequalification list: 15 sites of generics q q q Asia: 9 sites Europe: 4 sites Africa: 2 sites Health Technologies and Pharmaceuticals 12 WHO - PSM
 Ongoing monitoring and requalification q q q Samples taken after supply Routine inspections and additional inspections Changes and variations controlled • q q Products and manufacturers Requalification (re-assessment) every 3 years World Health Assembly resolution: WHA 57. 14 of May 2004 Public reports requested • • WHOPIRs (WHO Public inspection report) and WHOPARs (WHO Public Assessment Report) are now on the prequalification web site Increasing interest in WHO Public Inspection Reports (WHOPIR) Health Technologies and Pharmaceuticals 13 WHO - PSM
Ongoing monitoring and requalification q q q Samples taken after supply Routine inspections and additional inspections Changes and variations controlled • q q Products and manufacturers Requalification (re-assessment) every 3 years World Health Assembly resolution: WHA 57. 14 of May 2004 Public reports requested • • WHOPIRs (WHO Public inspection report) and WHOPARs (WHO Public Assessment Report) are now on the prequalification web site Increasing interest in WHO Public Inspection Reports (WHOPIR) Health Technologies and Pharmaceuticals 13 WHO - PSM
 PQ web site information for procurement q List of prequalified products q List of manufacturers for APIs (Active Pharmaceutical Ingredient) and FFPs (Finished Pharmaceutical Product), which are GMP compliant List of GCP compliant Contract Research Organisations (CROs), bio-equivalence centers and research laboratories, which are GCP/GLP compliant WHOPIRs (WHO Public Inspection Report) WHOPARs (WHO Public Assessment Report) q q q Health Technologies and Pharmaceuticals 14 WHO - PSM
PQ web site information for procurement q List of prequalified products q List of manufacturers for APIs (Active Pharmaceutical Ingredient) and FFPs (Finished Pharmaceutical Product), which are GMP compliant List of GCP compliant Contract Research Organisations (CROs), bio-equivalence centers and research laboratories, which are GCP/GLP compliant WHOPIRs (WHO Public Inspection Report) WHOPARs (WHO Public Assessment Report) q q q Health Technologies and Pharmaceuticals 14 WHO - PSM
 Important in the PQ process related to Quality and Efficacy q Dossier evaluation and Inspections (GMP = manufacturing and GCP for bioequivalence studies) Go hand in the PQ process Dossier assessment Inspections GMP/GCP Sometimes "GMP compliance" alone does not tell the whole truth!! Health Technologies and Pharmaceuticals 15 WHO - PSM
Important in the PQ process related to Quality and Efficacy q Dossier evaluation and Inspections (GMP = manufacturing and GCP for bioequivalence studies) Go hand in the PQ process Dossier assessment Inspections GMP/GCP Sometimes "GMP compliance" alone does not tell the whole truth!! Health Technologies and Pharmaceuticals 15 WHO - PSM
 This is misleading and/or misunderstood q WHO type certificate (by WHO Certification Scheme) is used worldwide But it q is not WHO certificate GMP compliance? ? National GMP requirements are not always those of WHO or stringent authority requirements Does not tell everything about the product Health Technologies and Pharmaceuticals 16 WHO - PSM
This is misleading and/or misunderstood q WHO type certificate (by WHO Certification Scheme) is used worldwide But it q is not WHO certificate GMP compliance? ? National GMP requirements are not always those of WHO or stringent authority requirements Does not tell everything about the product Health Technologies and Pharmaceuticals 16 WHO - PSM
 !!!!!!!!!!!!!!!!This is correct!!!!!!!!!!!!!!!! q WHO Prequalification does not give GMP/GCP certificates, but the GMP/GCP compliant companies are listed in our web site WHO GMP/GCP CERTIFICATE Health Technologies and Pharmaceuticals 17 WHO - PSM
!!!!!!!!!!!!!!!!This is correct!!!!!!!!!!!!!!!! q WHO Prequalification does not give GMP/GCP certificates, but the GMP/GCP compliant companies are listed in our web site WHO GMP/GCP CERTIFICATE Health Technologies and Pharmaceuticals 17 WHO - PSM
 Summary and conclusion q Good news; quality of generics exists ä ä ä q Relatively large number of products and suppliers comply with the standards Many potential suppliers appreciating feedback and willing to improve Unique technical knowledge obtained of products, especially generic antiretrovirals and antimalarials Bad news: quality assurance has the price ä ä ä Only limited number of products have met the required standards Takes time to get into compliance • Data to be generated, tests carried out • GMP upgrade needed Bad quality generics may undermine the public confidence in generics Health Technologies and Pharmaceuticals 18 WHO - PSM
Summary and conclusion q Good news; quality of generics exists ä ä ä q Relatively large number of products and suppliers comply with the standards Many potential suppliers appreciating feedback and willing to improve Unique technical knowledge obtained of products, especially generic antiretrovirals and antimalarials Bad news: quality assurance has the price ä ä ä Only limited number of products have met the required standards Takes time to get into compliance • Data to be generated, tests carried out • GMP upgrade needed Bad quality generics may undermine the public confidence in generics Health Technologies and Pharmaceuticals 18 WHO - PSM
 Summary and conclusion CONT… However… REMEMBER Quality can not be assessed, tested or inspected into the product, BUT has to be built into the product in all the steps … development phase, production, QC, BE study etc. with the help of guidelines, regulatory requirements etc. !! More technical help to manufacturers in developing countries is needed!! Health Technologies and Pharmaceuticals 19 WHO - PSM
Summary and conclusion CONT… However… REMEMBER Quality can not be assessed, tested or inspected into the product, BUT has to be built into the product in all the steps … development phase, production, QC, BE study etc. with the help of guidelines, regulatory requirements etc. !! More technical help to manufacturers in developing countries is needed!! Health Technologies and Pharmaceuticals 19 WHO - PSM
 http: //mednet 3. who. int/prequal/ Health Technologies and Pharmaceuticals 20 WHO - PSM
http: //mednet 3. who. int/prequal/ Health Technologies and Pharmaceuticals 20 WHO - PSM
 Health Technologies and Pharmaceuticals 21 WHO - PSM
Health Technologies and Pharmaceuticals 21 WHO - PSM
 Health Technologies and Pharmaceuticals 22 WHO - PSM
Health Technologies and Pharmaceuticals 22 WHO - PSM
 Health Technologies and Pharmaceuticals 23 WHO - PSM
Health Technologies and Pharmaceuticals 23 WHO - PSM
 Health Technologies and Pharmaceuticals 24 WHO - PSM
Health Technologies and Pharmaceuticals 24 WHO - PSM
 Что делать в этой ситуации? Health Technologies and Pharmaceuticals 25 WHO - PSM
Что делать в этой ситуации? Health Technologies and Pharmaceuticals 25 WHO - PSM
 THANK YOU VERY MUCH FOR YOUR ATTENTION!! Health Technologies and Pharmaceuticals 26 WHO - PSM
THANK YOU VERY MUCH FOR YOUR ATTENTION!! Health Technologies and Pharmaceuticals 26 WHO - PSM


