da18d3fb6622874ebb9dd34cc9def9b3.ppt
- Количество слайдов: 16

Preparation of HBV DNA reference standards and the experience of HBV NAT in Taiwan Dr. Hwei-Fang Cheng Department of Health, Taiwan

HBV DNA Reference Standards § National HBV DNA standard : 106 IU/m. L, 0. 5 m. L/vial (relative to NIBSC HBV NAT standard) § National HBV DNA working reagent : 103 IU/m. L, 1 m. L/vial (relative to NIBSC HBV NAT working reagent) § Intended use for HBV NAT testing of blood and pooled plasma and biological research

n High titer (~109 IU/ml) HBV positive plasma n Diluents: pooled human plasma (negative for anti-HIV, anti-HCV and NAT of HIV, HCV , HAV , and B 19) a) genotype B b) virus inactivation: 60 o. C for 1 hr c) dilute into ~106 IU/m. L and ~103 IU/m. L HBs. Ag, anti-HIV, anti-HCV and NAT of HIV, HCV, HBV, HAV, and B 19)

Collaborative Study n Participating Labs (11 Labs) n n n US CBER/FDA National Institute of Infectious Diseases (NIID) , Japan Canadian Blood Services National Institute for Biological Standards and Control (NIBSC) Victorian Infectious Diseases Reference Laboratory (VDRL), Australia NTUH Hepatitis Research Center, Taiwan Center of Disease Control, Taiwan BFDA, Taiwan Roche Diagnostics Artus Gmb. H Digene Diagnostic Technology
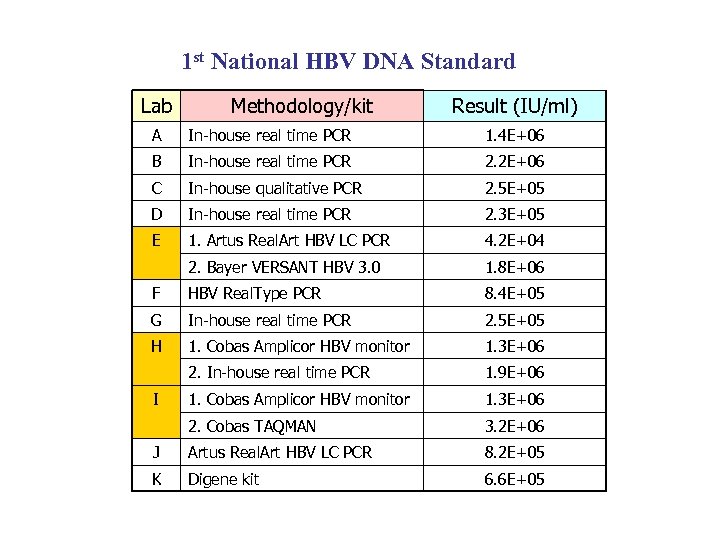
1 st National HBV DNA Standard Lab Methodology/kit Result (IU/ml) A In-house real time PCR 1. 4 E+06 B In-house real time PCR 2. 2 E+06 C In-house qualitative PCR 2. 5 E+05 D In-house real time PCR 2. 3 E+05 E 1. Artus Real. Art HBV LC PCR 4. 2 E+04 2. Bayer VERSANT HBV 3. 0 1. 8 E+06 F HBV Real. Type PCR 8. 4 E+05 G In-house real time PCR 2. 5 E+05 H 1. Cobas Amplicor HBV monitor 1. 3 E+06 2. In-house real time PCR 1. 9 E+06 1. Cobas Amplicor HBV monitor 1. 3 E+06 2. Cobas TAQMAN 3. 2 E+06 J Artus Real. Art HBV LC PCR 8. 2 E+05 K Digene kit 6. 6 E+05 I
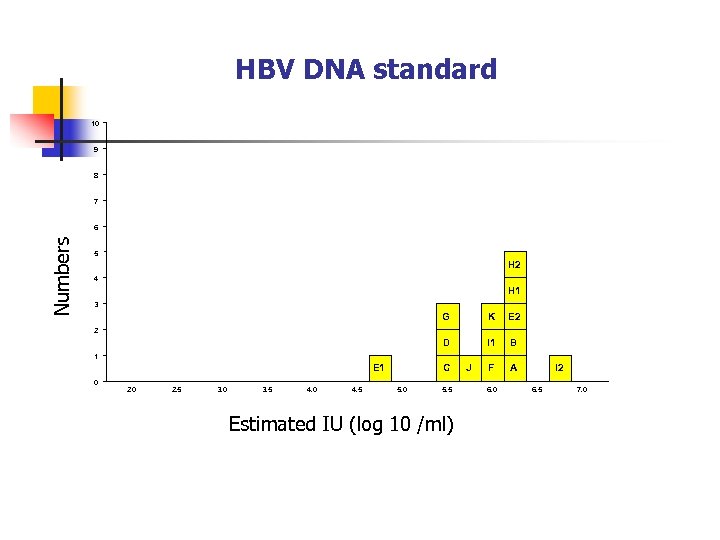
HBV DNA standard 10 9 8 7 Numbers 6 5 H 2 4 H 1 3 G K E 2 D I 1 B F A 2 1 E 1 0 2. 5 3. 0 3. 5 4. 0 4. 5 C 5. 0 5. 5 Estimated IU (log 10 /ml) J 6. 0 I 2 6. 5 7. 0

+2 SD Average 5. 955 -2 SD
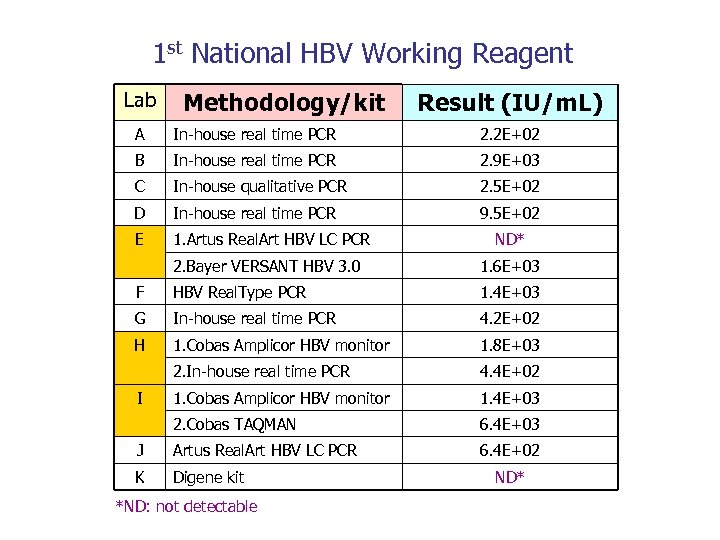
1 st National HBV Working Reagent Lab Methodology/kit Result (IU/m. L) A In-house real time PCR 2. 2 E+02 B In-house real time PCR 2. 9 E+03 C In-house qualitative PCR 2. 5 E+02 D In-house real time PCR 9. 5 E+02 E 1. Artus Real. Art HBV LC PCR ND* 2. Bayer VERSANT HBV 3. 0 1. 6 E+03 F HBV Real. Type PCR 1. 4 E+03 G In-house real time PCR 4. 2 E+02 H 1. Cobas Amplicor HBV monitor 1. 8 E+03 2. In-house real time PCR 4. 4 E+02 1. Cobas Amplicor HBV monitor 1. 4 E+03 2. Cobas TAQMAN 6. 4 E+03 J Artus Real. Art HBV LC PCR 6. 4 E+02 K Digene kit I *ND: not detectable ND*
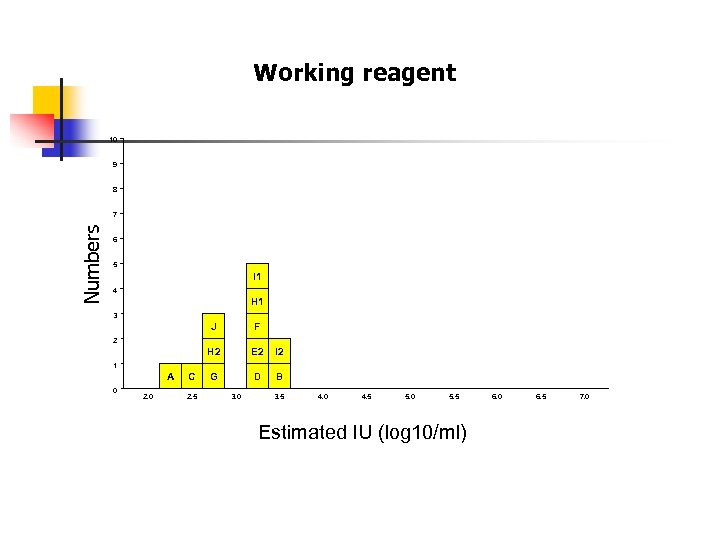
Working reagent 10 9 8 Numbers 7 6 5 I 1 4 H 1 3 J F H 2 E 2 I 2 G D B 2 1 A 0 2. 0 C 2. 5 3. 0 3. 5 4. 0 4. 5 5. 0 5. 5 Estimated IU (log 10/ml) 6. 0 6. 5 7. 0

+2 SD Average 2. 953 -2 SD
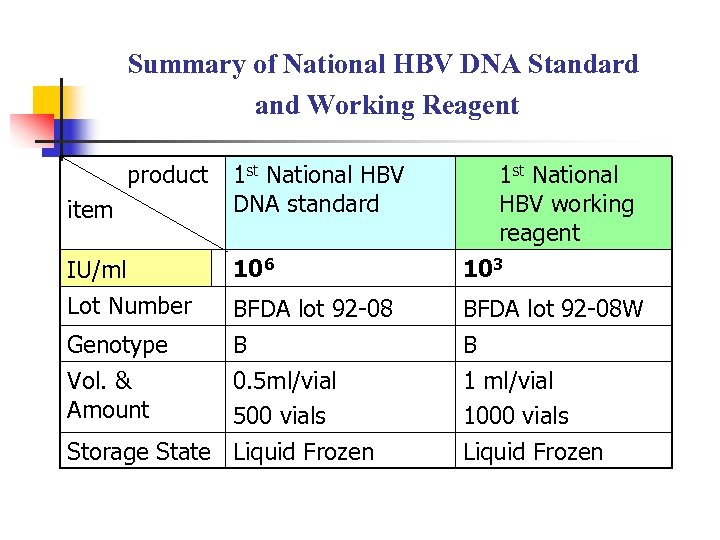
Summary of National HBV DNA Standard and Working Reagent product item 1 st National HBV DNA standard IU/ml 106 103 Lot Number BFDA lot 92 -08 W Genotype Vol. & Amount B 0. 5 ml/vial 500 vials Storage State Liquid Frozen 1 st National HBV working reagent B 1 ml/vial 1000 vials Liquid Frozen

Stability Test – HBV DNA Standard at 5 ± 3 o. C days

Stability Test – HBV DNA Working Reagent at 5 ± 3 o. C Cobas Amplicor days

Summary § The assigned potency of HBV DNA standard and HBV working reagent are 106 IU/ml and 103 IU/ml, respectively. § Genotype of national HBV DNA standard and working reagents is genotype B; and the serotype is adw. § National HBV DNA standard and working reagent are stable at 4 ℃ for 4 weeks.

HBV NAT Experience in Taiwan
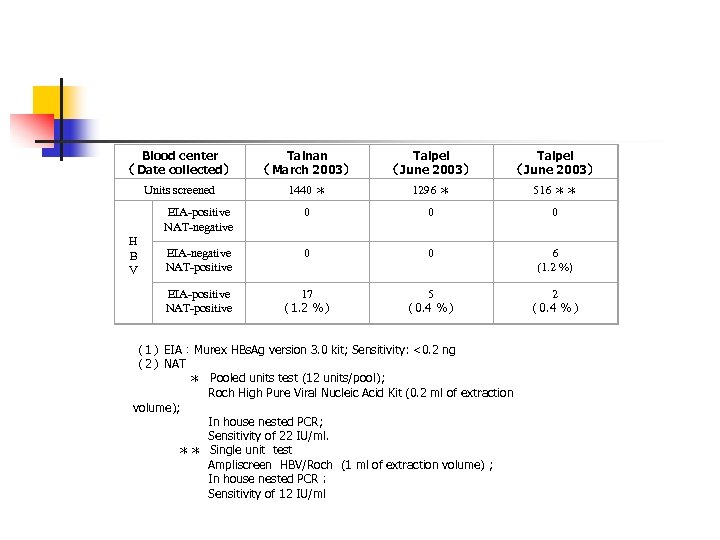
Blood center (Date collected) Tainan (March 2003) Taipei (June 2003) Units screened 1440 * 1296 * 516 ** EIA-positive NAT-negative 0 0 0 EIA-negative NAT-positive 0 0 6 (1. 2 %) EIA-positive NAT-positive 17 (1. 2 %) 5 (0. 4 %) 2 (0. 4 %) H B V (1)EIA:Murex HBs. Ag version 3. 0 kit; Sensitivity: <0. 2 ng (2)NAT * Pooled units test (12 units/pool); Roch High Pure Viral Nucleic Acid Kit (0. 2 ml of extraction volume); In house nested PCR; Sensitivity of 22 IU/ml. ** Single unit test Ampliscreen HBV/Roch (1 ml of extraction volume) ; In house nested PCR; Sensitivity of 12 IU/ml
da18d3fb6622874ebb9dd34cc9def9b3.ppt