1d655b89894087b07b25c7f284bdb7a7.ppt
- Количество слайдов: 38
 Prenatal screening: state of the art in 2015 François Audibert
Prenatal screening: state of the art in 2015 François Audibert
 Conflicts of interest l None!
Conflicts of interest l None!
 Objectives l l l To review the advantages and disadvantages of different prenatal screening options Describe the objectives and results of first trimester ultrasound Discuss the evolution of prenatal screening programs with the availability fetal DNA analysis
Objectives l l l To review the advantages and disadvantages of different prenatal screening options Describe the objectives and results of first trimester ultrasound Discuss the evolution of prenatal screening programs with the availability fetal DNA analysis
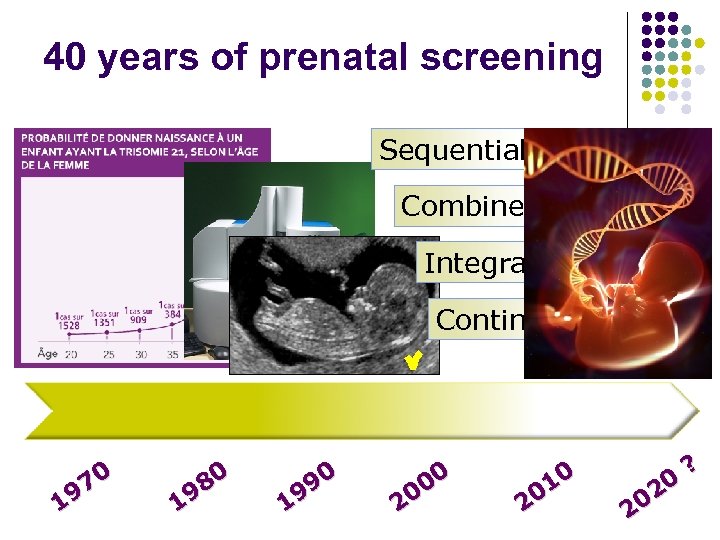 40 years of prenatal screening Sequential Combined Integrated Contingent 70 19 80 19 90 19 00 20 10 20 20 20 ?
40 years of prenatal screening Sequential Combined Integrated Contingent 70 19 80 19 90 19 00 20 10 20 20 20 ?
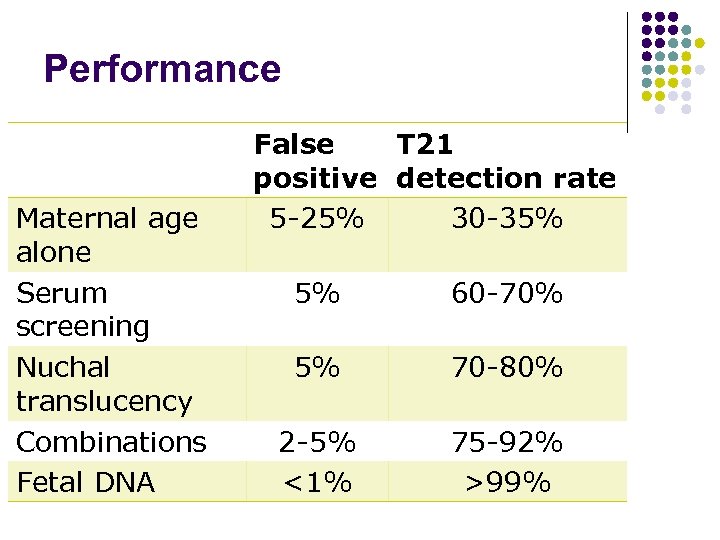 Performance Maternal age alone Serum screening Nuchal translucency Combinations Fetal DNA False T 21 positive detection rate 5 -25% 30 -35% 5% 60 -70% 5% 70 -80% 2 -5% <1% 75 -92% >99%
Performance Maternal age alone Serum screening Nuchal translucency Combinations Fetal DNA False T 21 positive detection rate 5 -25% 30 -35% 5% 60 -70% 5% 70 -80% 2 -5% <1% 75 -92% >99%
 SOGC guidelines – 2007 - 2011 1. All pregnant women in Canada, regardless of age, should be offered, through an informed counselling process, the option of a prenatal screening test for the most common clinically significant fetal aneuploidies… (I-A) 2. Maternal age alone is a poor minimum standard for prenatal screening for aneuploidy, and it should not be used a basis for recommending invasive testing when noninvasive prenatal screening for aneuploidy is available… (II-2 A)
SOGC guidelines – 2007 - 2011 1. All pregnant women in Canada, regardless of age, should be offered, through an informed counselling process, the option of a prenatal screening test for the most common clinically significant fetal aneuploidies… (I-A) 2. Maternal age alone is a poor minimum standard for prenatal screening for aneuploidy, and it should not be used a basis for recommending invasive testing when noninvasive prenatal screening for aneuploidy is available… (II-2 A)
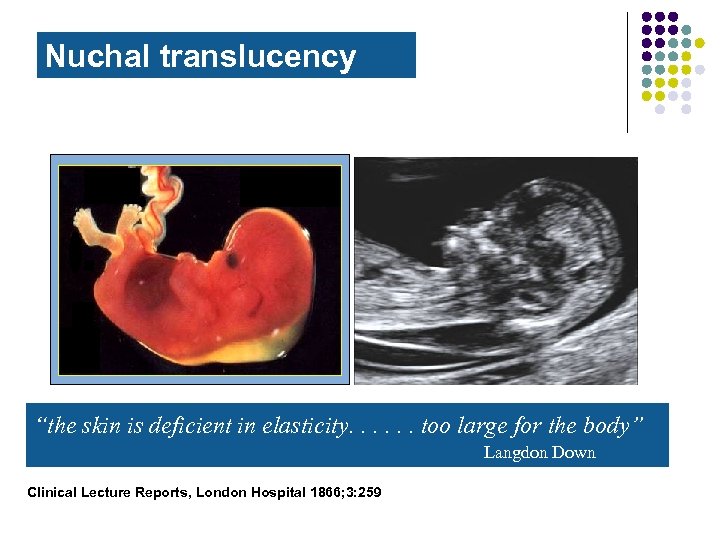 Nuchal translucency “the skin is deficient in elasticity. . . too large for the body” Langdon Down Clinical Lecture Reports, London Hospital 1866; 3: 259
Nuchal translucency “the skin is deficient in elasticity. . . too large for the body” Langdon Down Clinical Lecture Reports, London Hospital 1866; 3: 259
 Nuchal translucency l l l Most efficient ultrasound screening tool 11 -13 weeks Very dependent on technique and equipment Should be performed with adequate quality control Should be combined to serum screening Continuous audit of results should be in place
Nuchal translucency l l l Most efficient ultrasound screening tool 11 -13 weeks Very dependent on technique and equipment Should be performed with adequate quality control Should be combined to serum screening Continuous audit of results should be in place
 Standardized training International guidelines Free training and licensing NT criterias 1. 2. 3. 4. 5. 6. Sagittal view Neutral position Adequate zoom Largest area Calipers placement Thin and clear membrane www. fetalmedicine. com
Standardized training International guidelines Free training and licensing NT criterias 1. 2. 3. 4. 5. 6. Sagittal view Neutral position Adequate zoom Largest area Calipers placement Thin and clear membrane www. fetalmedicine. com
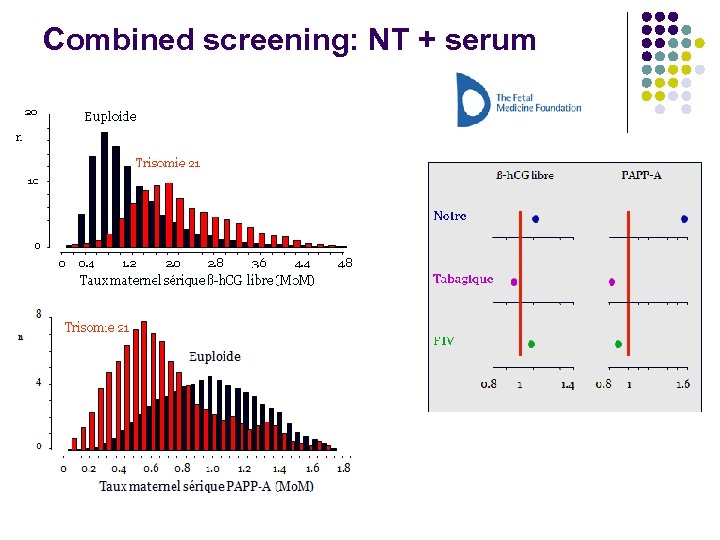 Combined screening: NT + serum
Combined screening: NT + serum
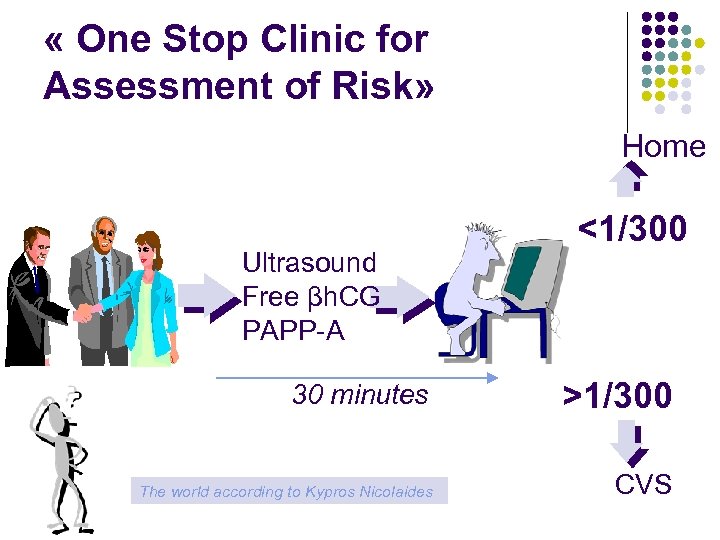 « One Stop Clinic for Assessment of Risk» Home Ultrasound Free βh. CG PAPP-A <1/300 30 minutes >1/300 The world according to Kypros Nicolaides CVS
« One Stop Clinic for Assessment of Risk» Home Ultrasound Free βh. CG PAPP-A <1/300 30 minutes >1/300 The world according to Kypros Nicolaides CVS
 OSCAR clinic : results l l l 1998 -2002 N=32, 372 women High risk (≥ 1/300) = 5. 8% 117 T 21 / 127 identified by the test Sensitivity = 92% Spencer and Nicolaides, Ultrasound Obstet Gyneco
OSCAR clinic : results l l l 1998 -2002 N=32, 372 women High risk (≥ 1/300) = 5. 8% 117 T 21 / 127 identified by the test Sensitivity = 92% Spencer and Nicolaides, Ultrasound Obstet Gyneco
 2 nd trimester serum markers l l l 15 -20 weeks AFP Estriol h. CG Inhibine A
2 nd trimester serum markers l l l 15 -20 weeks AFP Estriol h. CG Inhibine A
 «Integrated screening » l Wald et al. , N Engl J Med 1999 l Estimation of FPR and DR, by various strategies (modelisation) l l l Triple or quadruple serum screening NT + MSS 1 st trimester (Combined Test) NT + MSS 1 st and 2 nd trimester (Integrated Test)
«Integrated screening » l Wald et al. , N Engl J Med 1999 l Estimation of FPR and DR, by various strategies (modelisation) l l l Triple or quadruple serum screening NT + MSS 1 st trimester (Combined Test) NT + MSS 1 st and 2 nd trimester (Integrated Test)
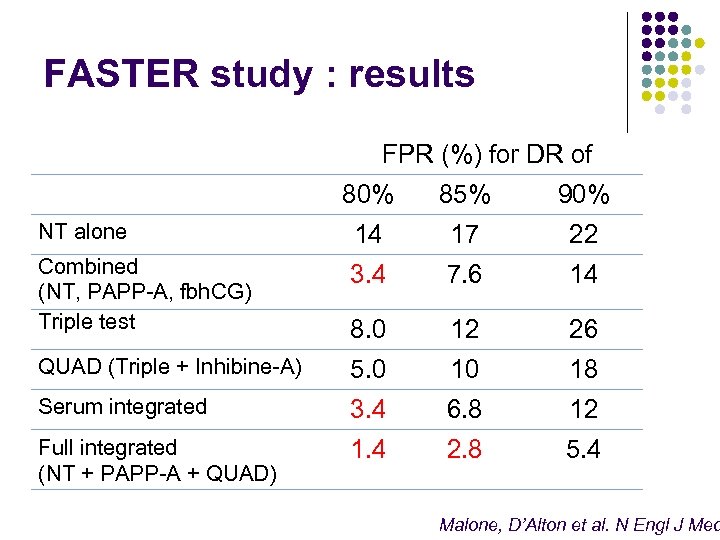 FASTER study : results FPR (%) for DR of 80% 85% 90% NT alone 14 17 22 Combined (NT, PAPP-A, fbh. CG) Triple test 3. 4 7. 6 14 8. 0 12 26 QUAD (Triple + Inhibine-A) 5. 0 10 18 Serum integrated 3. 4 6. 8 12 Full integrated (NT + PAPP-A + QUAD) 1. 4 2. 8 5. 4 Malone, D’Alton et al. N Engl J Med
FASTER study : results FPR (%) for DR of 80% 85% 90% NT alone 14 17 22 Combined (NT, PAPP-A, fbh. CG) Triple test 3. 4 7. 6 14 8. 0 12 26 QUAD (Triple + Inhibine-A) 5. 0 10 18 Serum integrated 3. 4 6. 8 12 Full integrated (NT + PAPP-A + QUAD) 1. 4 2. 8 5. 4 Malone, D’Alton et al. N Engl J Med
 Combined or integrated screening? l COMBINED l l l Earlier diagnosis One single blood sample Organisation more simple l l INTEGRATED Two-step process Lower FPR Cost effectiveness? Same sensitivity More complex for women and clinicians
Combined or integrated screening? l COMBINED l l l Earlier diagnosis One single blood sample Organisation more simple l l INTEGRATED Two-step process Lower FPR Cost effectiveness? Same sensitivity More complex for women and clinicians
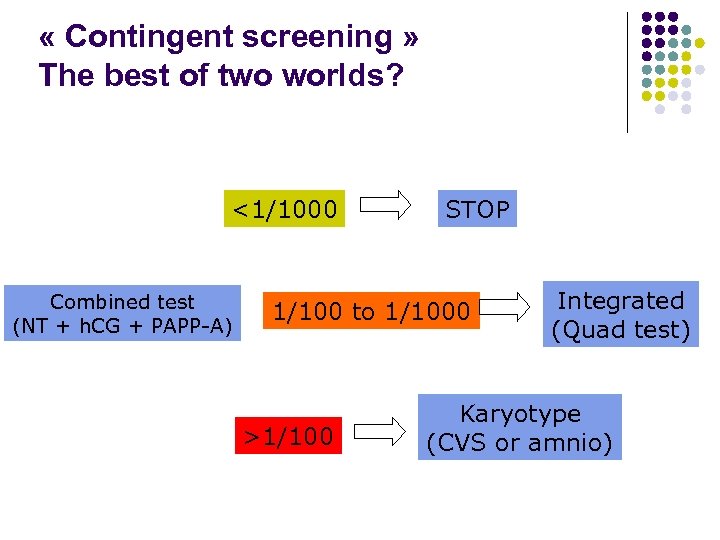 « Contingent screening » The best of two worlds? <1/1000 Combined test (NT + h. CG + PAPP-A) STOP 1/100 to 1/1000 >1/100 Integrated (Quad test) Karyotype (CVS or amnio)
« Contingent screening » The best of two worlds? <1/1000 Combined test (NT + h. CG + PAPP-A) STOP 1/100 to 1/1000 >1/100 Integrated (Quad test) Karyotype (CVS or amnio)
 Conclusion ( « conventional » screening) l l l Maternal age alone has a false positive rate of 10 -15% (detection rate 30 -50%) Quality assurance is more important than the type of screening Ultrasound (dating at least, but ideally NT) is of paramount importance for a good screening
Conclusion ( « conventional » screening) l l l Maternal age alone has a false positive rate of 10 -15% (detection rate 30 -50%) Quality assurance is more important than the type of screening Ultrasound (dating at least, but ideally NT) is of paramount importance for a good screening
 Non Invasive Prenatal Testing (NIPT) Cell-free Fetal DNA
Non Invasive Prenatal Testing (NIPT) Cell-free Fetal DNA
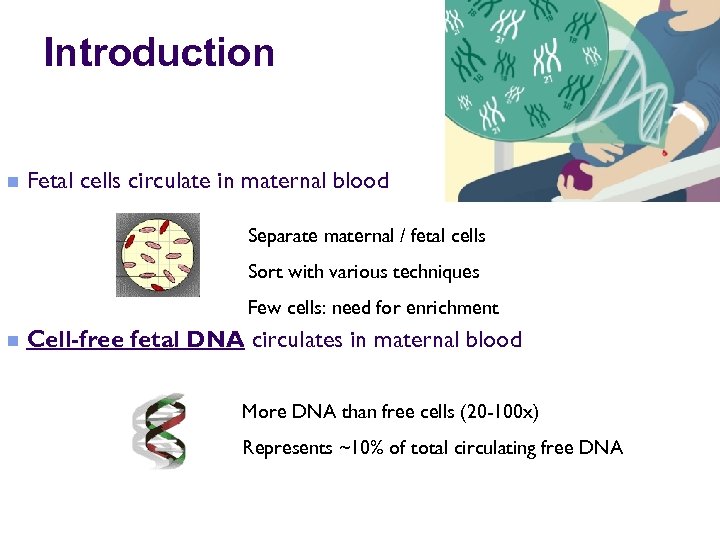 Introduction n Fetal cells circulate in maternal blood Separate maternal / fetal cells Sort with various techniques Few cells: need for enrichment n Cell-free fetal DNA circulates in maternal blood More DNA than free cells (20 -100 x) Represents ~10% of total circulating free DNA
Introduction n Fetal cells circulate in maternal blood Separate maternal / fetal cells Sort with various techniques Few cells: need for enrichment n Cell-free fetal DNA circulates in maternal blood More DNA than free cells (20 -100 x) Represents ~10% of total circulating free DNA
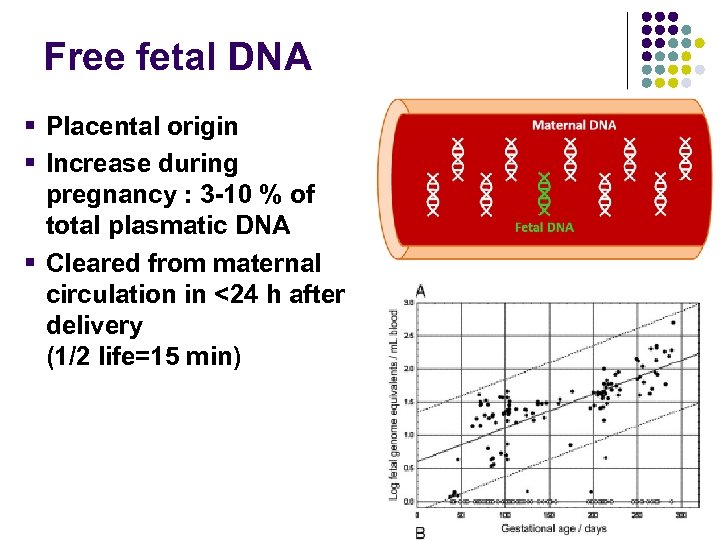 Free fetal DNA § Placental origin § Increase during pregnancy : 3 -10 % of total plasmatic DNA § Cleared from maternal circulation in <24 h after delivery (1/2 life=15 min)
Free fetal DNA § Placental origin § Increase during pregnancy : 3 -10 % of total plasmatic DNA § Cleared from maternal circulation in <24 h after delivery (1/2 life=15 min)
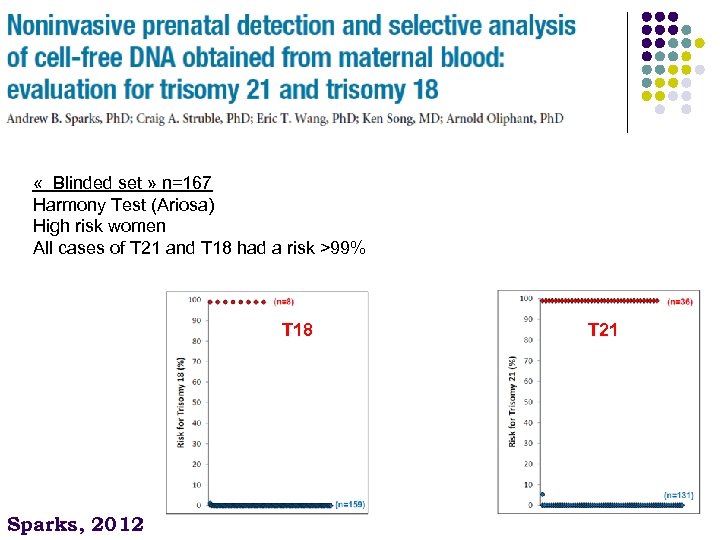 « Blinded set » n=167 Harmony Test (Ariosa) High risk women All cases of T 21 and T 18 had a risk >99% T 18 Sparks, 2012 T 21
« Blinded set » n=167 Harmony Test (Ariosa) High risk women All cases of T 21 and T 18 had a risk >99% T 18 Sparks, 2012 T 21
 l l l Test « MPSS » (Verinata/Illumina), N=1912 Prenatal screening, unselected population PPV 45%, NPV 100% 5 T 21 2 T 18
l l l Test « MPSS » (Verinata/Illumina), N=1912 Prenatal screening, unselected population PPV 45%, NPV 100% 5 T 21 2 T 18
 Large-scale study in unselected population NEJM, April 2015 N=15, 841 pregnancies Routine screening T 21 Harmony Test Sens= 100% (38 of 38) FPR= 0. 06% PPV= 80. 9%
Large-scale study in unselected population NEJM, April 2015 N=15, 841 pregnancies Routine screening T 21 Harmony Test Sens= 100% (38 of 38) FPR= 0. 06% PPV= 80. 9%
 $$$. . .
$$$. . .
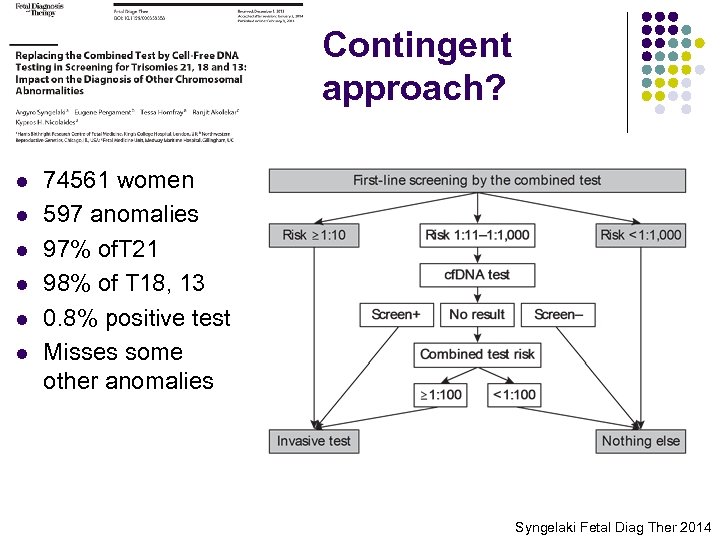 Contingent approach? l l l 74561 women 597 anomalies 97% of. T 21 98% of T 18, 13 0. 8% positive test Misses some other anomalies Syngelaki Fetal Diag Ther 2014
Contingent approach? l l l 74561 women 597 anomalies 97% of. T 21 98% of T 18, 13 0. 8% positive test Misses some other anomalies Syngelaki Fetal Diag Ther 2014
 First line test? l l l 0. 9% False positive 98. 6% T 21 95. 7% T 18, 13 No other anomaly detected (Turner, 46 XXY, triploïdies…) High cost
First line test? l l l 0. 9% False positive 98. 6% T 21 95. 7% T 18, 13 No other anomaly detected (Turner, 46 XXY, triploïdies…) High cost

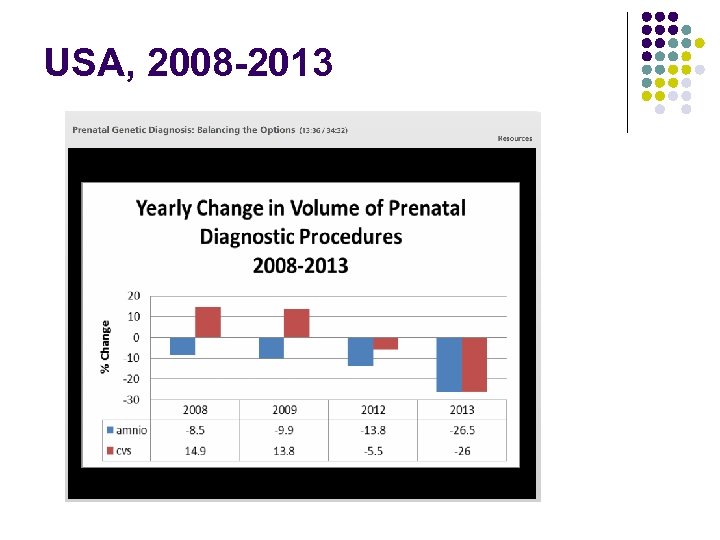 USA, 2008 -2013
USA, 2008 -2013
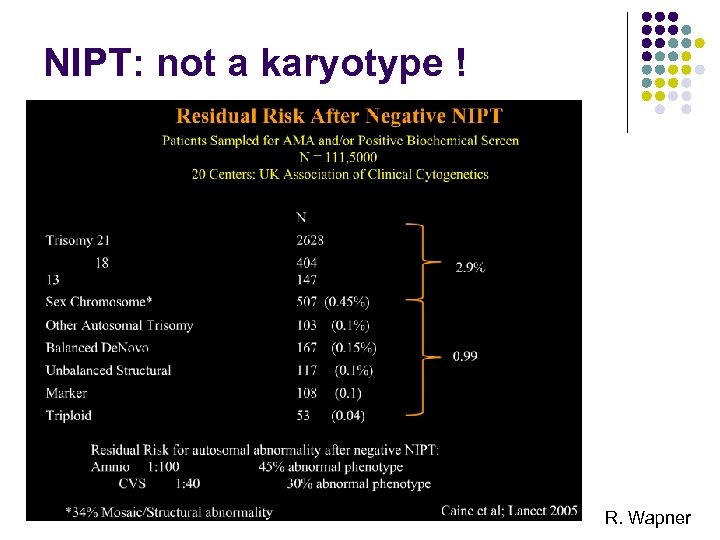 NIPT: not a karyotype ! R. Wapner
NIPT: not a karyotype ! R. Wapner
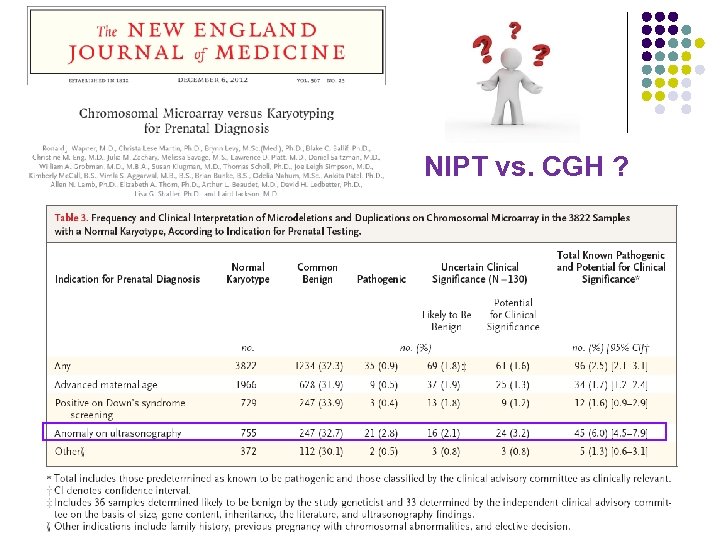 NIPT vs. CGH ?
NIPT vs. CGH ?
 Some limitations Depending on the methodology used, reasons for discordancy between cf. DNA results and fetal karyotype can include: • • • true fetal mosaicism confined placental mosaicism maternal karyotype abnormality insufficient counting due to low fetal fraction vanishing twin
Some limitations Depending on the methodology used, reasons for discordancy between cf. DNA results and fetal karyotype can include: • • • true fetal mosaicism confined placental mosaicism maternal karyotype abnormality insufficient counting due to low fetal fraction vanishing twin

 Guidelines / Genetics committee SOGC Feb 2013 1. Non-invasive prenatal testing using massive parallel sequencing of cell-free fetal DNA to test for trisomies 21, 18, and 13 should be an option available to women at increased risk in lieu of amniocentesis. Pretest counselling of these women should include a discussion of the limitations of non-invasive prenatal testing. (II-2 A)
Guidelines / Genetics committee SOGC Feb 2013 1. Non-invasive prenatal testing using massive parallel sequencing of cell-free fetal DNA to test for trisomies 21, 18, and 13 should be an option available to women at increased risk in lieu of amniocentesis. Pretest counselling of these women should include a discussion of the limitations of non-invasive prenatal testing. (II-2 A)
 Guidelines SOGC Feb 2013 2. No irrevocable obstetrical decision should be made in pregnancies with a positive noninvasive prenatal testing result without confirmatory invasive diagnostic testing. (II 2 A)
Guidelines SOGC Feb 2013 2. No irrevocable obstetrical decision should be made in pregnancies with a positive noninvasive prenatal testing result without confirmatory invasive diagnostic testing. (II 2 A)
 Guidelines SOGC Feb 2013 3. Although testing of cell-free fetal DNA in maternal plasma appears very promising as a screening test for Down syndrome and other trisomies, studies in average-risk pregnancies and a significant reduction in the cost of the technology are needed before this can replace the current maternal screening approach using biochemical serum markers with or without fetal nuchal translucency ultrasound. (III-A)
Guidelines SOGC Feb 2013 3. Although testing of cell-free fetal DNA in maternal plasma appears very promising as a screening test for Down syndrome and other trisomies, studies in average-risk pregnancies and a significant reduction in the cost of the technology are needed before this can replace the current maternal screening approach using biochemical serum markers with or without fetal nuchal translucency ultrasound. (III-A)
 The PEGASUS study…
The PEGASUS study…
 Take home messages § NIPT is no longer a « research » tool § NIPT is performed by sequencing cell-free fetal DNA § Validated applications: § Trisomies 13, 18 and 21 in high-risk women § Second-line test after positive screening § More research needed for screening in low risk women / integration with existing screening strategies
Take home messages § NIPT is no longer a « research » tool § NIPT is performed by sequencing cell-free fetal DNA § Validated applications: § Trisomies 13, 18 and 21 in high-risk women § Second-line test after positive screening § More research needed for screening in low risk women / integration with existing screening strategies


