c63ccd6d1383638bbe5df40050490824.ppt
- Количество слайдов: 33

Prenatal Care and Obstetrical Management of HIV+ Women Deborah Cohan, MD, MPH Bay Area Perinatal AIDS Center National Perinatal HIV Consultation and Referral Service UCSF

Overview: l Antepartum management £ Antiretroviral therapy: Benefits, Risks l Intrapartum management £ L&D management £ Mode of delivery l Post-partum management

Perinatal HIV in the U. S.
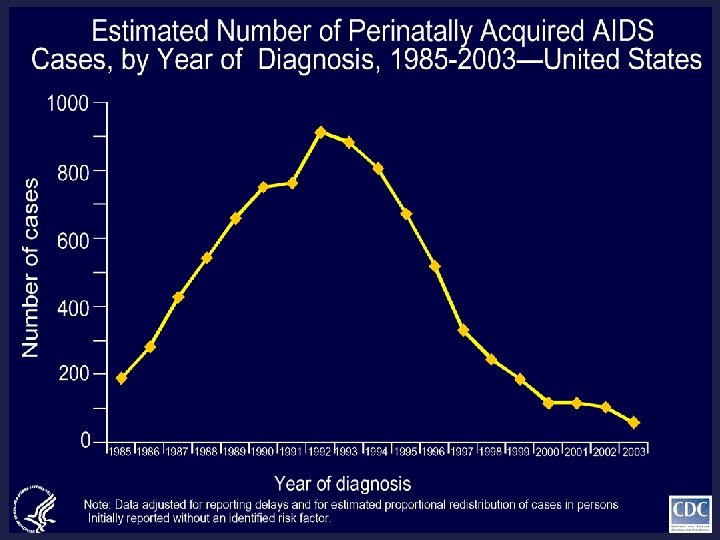
Number of cases

Perinatal HIV testing: the key to prevention

Prenatal HIV Testing Strategies l Opt-in: voluntary, women sign consent to test l Opt-out: voluntary, informed that test is standard, sign if decline testing (Tennessee, Canada) l Mandatory newborn screening: regardless of maternal consent (NY, Connecticut) l Uptake of HIV testing Opt-in (25 - 69%) vs. Opt-out (71 -98%) approach £ CA law mandates prenatal providers to offer HIV testing (opt-in) and explain that testing is routinely done unless pt declines £ Likely change in CA law Jan 2008: opt-out £ DHHS 2002; CDC 1998; CDC 2001; CDC 2002

Antepartum management

Goals of prenatal care l Optimize woman’s health and psychosocial situation ART: total viral suppression £ Opportunistic Infection (OI) prophylaxis prn £ Immunization prn £ l Prevent vertical transmission of HIV £ ART, c/section in specific situations, Bottle-feeding l Minimize maternal risks £ Viral resistance, Obstetrical outcomes l Minimize/assess risks to fetus/neonate £ Teratogenicity, Genetic testing l Prepare for or prevent subsequent pregnancies

Maternal Risk Factors l Plasma viral load @ delivery £ per log : w OR 3. 4 (1. 7 -6. 8) £ VL <1000: w 0. 7%-0. 9% transmission l Genital VL @ delivery £ Cell-associated w per log : OR 2. 3 (1. 1 -4. 8) £ Cell-free w OR 3. 4 (p=0. 001) l CD 4 count l Drug-resistant HIV £ £ ZDV GT resist OR 5. 16 ZDV PT resist OR 1. 25 Other possible risk factors STIs Drug Use Smoking Anemia Vitamin A deficiency Clade D virus (vs. clade A) Monocyte/macrophage tropism Viral homogeneity Class I HLA concordance Certain HLA-B alleles Rapid replication kinetics p 24 antigenemia Primary HIV infection Landesman 1996; Thea 1997; Shapiro 2002; Tuomala 2003; Chuachoowong 2000; Goedert 2001; O'Shea 1998; Mofeson 1999; Shapiro 1999; Monforte 1991; Ometto 1995; Mac. Donald 1998; Arroyo 2002; Winchester 2004; Yang 2003
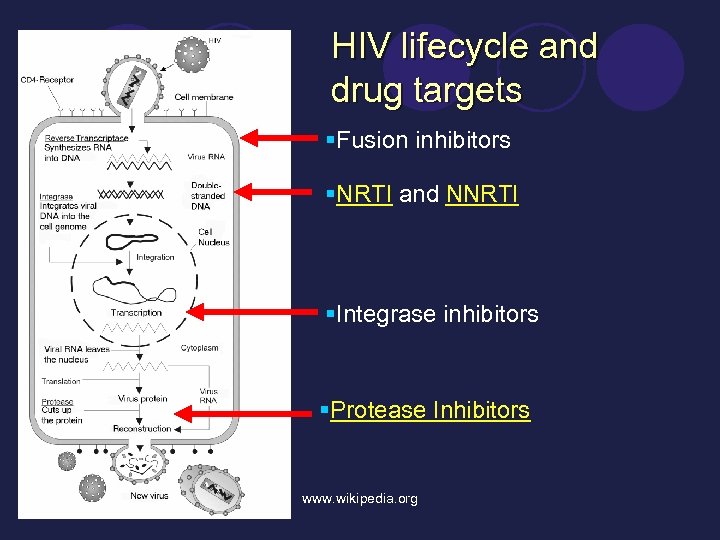
HIV lifecycle and drug targets §Fusion inhibitors §NRTI and NNRTI §Integrase inhibitors §Protease Inhibitors www. wikipedia. org

When and How Should a non-pregnant Adult Be Treated? l When Symptomatic, at any CD 4 count £ CD 4 count <200 (AIDS) £ CD 4 count 200 -350: Treatment offered £ l How £ HAART: Highly Active Antiretroviral Therapy w 2 NRTI’s plus w PI or NNRTI l Monotherapy, dual therapy, and triple NRTI regimens no longer standard of care DHHS Guidelines for the Use of Antiretrovirals in HIV-Infected Adults and Adolescents, May 2006

Antiretrovirals in pregnancy l All HIV+ pregnant women should get ART regardless of CD 4 count and viral load. l But… £ When to start £ What to choose £ What to avoid

ART: when to start l Goal: viral suppression by 3 rd trimester l Typically start in 2 nd trimester l Exceptions to starting in 2 nd trimester £ Continuing preconception regimen and nonteratogenic £ Needs ARV immediately for own health l If not tolerating preconception regimen in 1 st trimester despite anti-emetics, d/c all at once £ Stagger d/c of NVP-based ART Wright, SMFM, 2003; Thorne CROI 2005

ART: what to choose l Same principles as non-pregnant HIV+ adults £ Resistance/prior regimens, adherence/pill burden, S/E profile, degree of immunosuppression, viral hepatitis status l Except consider… l AZT-containing regimen unless contraindicated l Purpose of ART: her health vs. prophylaxis £ If not needed for own health, less potent regimens may be acceptable w Triple NRTI regimens w AZT monotherapy for baseline viral load <1000?
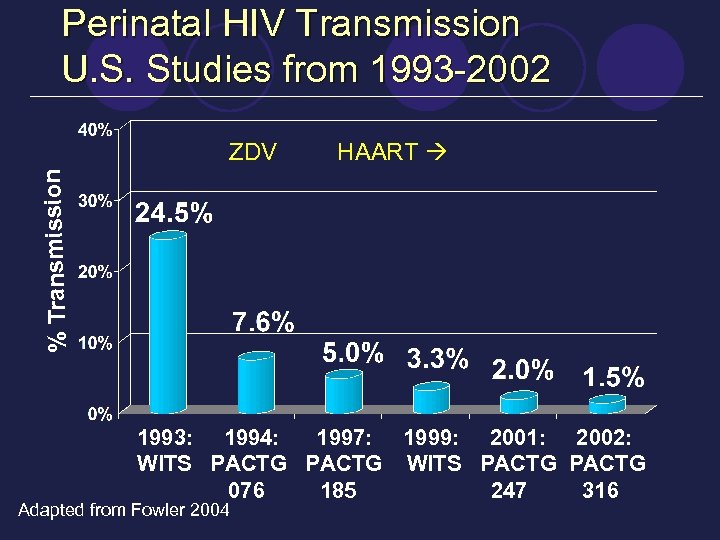
Perinatal HIV Transmission U. S. Studies from 1993 -2002 HAART % Transmission ZDV 1993: 1994: 1997: 1999: 2001: 2002: WITS PACTG 076 185 247 316 Adapted from Fowler 2004

Adverse effects of antiretrovirals in pregnancy

Maternal Risks and ARVs l Lactic acidosis and d 4 T (and dd. I) £ £ £ 12 reports of maternal LA (3 fatal) Avoid d 4 T and dd. I if possible Think of LA if w N/V, abdominal pain, SOB, leg and arm weakness l Hepatic Toxicity and NVP £ £ £ 1 st 6 wks NVP, may persist even when d/c NVP Distinguished from other etiologies (ob and non-ob) Avoid starting NVP if CD 4 > 250 l Gestational DM and PIs £ £ Conflicting data, most studies don’t find association Not a reason to avoid using PIs

Obstetrical Risks and ARVs l Preterm delivery and ARVs? £ Conflicting data; all based on observational cohorts w Europ Collaborative & Swiss Mother+Child HIV: yes w U. S. Collaborative (n=2123): no w Meta-analysis: PTD only if preconception or 1 st trimester ARV l Pre-Eclampsia and ARVs? Conflicting preliminary data £ ARVs increase risk? £ ARVs restore immune system to allow Pre-E to occur? £ Euro Collaborative Study and Swiss Mother+Child 2000; Thorne CROI 2004; Tuomala 2002; Cotter JID 2006; Wimalasundera Lancet 2002; Suy AIDS 2006

Fetal/Neonatal Risks

FDA Drug Classification l A l B £ £ £ NRTI: dd. I, FTC, TDF (monkey osteomalacia @ high dose) PI: ATV, NFV, RTV, SQV FI: T-20 l C £ £ £ NRTI: ABC (rats 35 x dose), 3 TC, d 4 T, dd. C, ZDV NNRTI: NVP PI: APV (rat thymic elongation/ skeletal ossification), f-APV, IDV, LPV/r l D £ EFV (monkey 15% CNS malformations; 3 human NTD, 1 Dandy Walker) l Avoid using preconception/1 st trimester EFV l 2 nd/3 rd trimester EFV only if no other options DHHS 2005

Nelfinavir l Sept. 2007, Pfizer sent a letter to providers regarding the presence of low levels of ethyl methane sulfonate (EMS) in nelfinavir. EMS is teratogenic, carcinogenic, and mutagenic in animals. No human data exist. l Not recommended unless no other alternative is available.

Benefits >>>> Potential Risks

Intrapartum Management l Shorten duration of ruptured membranes l No evidence of c/section to shorten ROM l Minimize # exams to risk of chorio l Avoid FSE, fetal scalp sampling l PPROM? ? ? £ Balancing MTCT vs. prematurity £ Management should be based on maternal viral load and NICU capabilities

Standard Intrapartum ART l Intrapartum AZT regardless of antepartum ART £ 2 mg/kg IV load, then 1 mg/kg IV qhr until delivery £ Loading dose can be given over 20 min-1 hr £ D/C d 4 T when receiving AZT £ Give 3 -4 hrs of IV AZT prior to elective c-section l Continue oral ART, even if getting cesarean Dorenbaum JAMA 2002

Cesarean Delivery and MTCT

Elective Cesarean and MTCT l 38 weeks, no labor, no ROM l Benefit seen in early studies £ AZT alone, observ studies didn’t adjust for VL l Studies in the HAART era: limited benefit £ £ PACTG 367 cohort, 1998 -2001; 72 U. S. sites, n=2875 singleton births Transmission 2. 9% overall MTCT by pre-delivery maternal viral load <1000: 0. 7% vs. 1000 -9999: 2. 1% vs. 10, 000+: 5. 9% l Elective c/s vs. vaginal delivery by maternal VL £ <1000: 0. 8% vs. 0. 7% £ 1000 -9999: 2. 8% vs. 1. 9%: OR 1. 5 (0. 4 -5. 0) £ 10, 000+: 4. 1% vs. 7. 3%: OR 0. 5 (0. 2 -1. 5) £ No RNA in chart: 8. 3% vs. 22. 4%: OR 0. 3 (0. 1 -0. 9) The European Mode of Delivery Collaboration, 1999; International Perinatal HIV Group 1999; Shapiro CROI 2004

Elective Cesarean and MTCT: Cochrane Collaboration l “Elective c/section is a good intervention for the prevention of MTCT among HIV-infected women not taking antiretrovirals or taking only zidovudine… l Among women with less advanced or wellcontrolled HIV disease…the short-term risk of the intervention may exceed the long-term benefit. ” Read and Newell 2005

Post-partum maternal care l For those continuing on ART post-partum: £ Reinforce medication adherence £ Dose maternal and neonatal ART on similar schedules l Remove breastfeeding literature from educational packs l Contraception

Post-partum vaccination l l Tdap Complete hepatitis A/B series prn Flu vax (if didn’t get antepartum) Rubella vax MMR: live-attenuated vaccine £ Case report of measles pneumonitis £ Advisory Committee on Immunization Practices: £ w Recommends in susceptible, asymptomatic HIV w Not recommended if cd 4 <200 or <14% w Check titers at 3 months and revaccinate prn Advisory Committee on Immunization Practices 1998; Brady CROI 2002

Conclusions l Prevent perinatal HIV transmission through 1° prevention l l l among women Ensure access to HIV testing: preconception and during pregnancy Ensure access to contraception and abortion services Keep woman healthy and preserve future ART options HIV-specific prenatal care Consider Cesarean £ if high viral load, no HAART, no labor/rupture of membranes l Avoid intrapartum interventions l Bottle feed (formula or banked human milk)

Resources l Clinical consultation £ National Perinatal HIV Consultation and Referral Service (NCCC) w 24/7 coverage, based at SFGH w 1 -888 -448 -8765 (1 -888 -HIV-8765) £ Bay Area Perinatal AIDS Center (BAPAC) w 415 -206 -8919 (M-F, 8 a-5 p) £ Reproductive Infectious Disease Fellows w 719 -8726 (24/7 coverage) l Web-based resources www. aidsinfo. nih. gov (Perinatal HIV Guidelines) £ www. womenchildrenhiv. org £

Thank you
c63ccd6d1383638bbe5df40050490824.ppt