3c756063de6d771ff3e905fea3671056.ppt
- Количество слайдов: 60
 Pre-Exposure Prophylaxis: The Time is Now Carlos Malvestutto, MD, MPH Medical Director FACES Clinic Assistant Professor OSU Medical Center ………………. . …………………………………………………………………. .
Pre-Exposure Prophylaxis: The Time is Now Carlos Malvestutto, MD, MPH Medical Director FACES Clinic Assistant Professor OSU Medical Center ………………. . …………………………………………………………………. .
 Disclosures • No disclosures to report
Disclosures • No disclosures to report
 Objectives • Define Pr. EP • Review the scientific evidence supporting Pr. EP – Review clinical trial data – Review recent “real world” experience with Pr. EP • Review guidelines for Pr. EP • Implementation of Pr. EP in Columbus • What are the next steps?
Objectives • Define Pr. EP • Review the scientific evidence supporting Pr. EP – Review clinical trial data – Review recent “real world” experience with Pr. EP • Review guidelines for Pr. EP • Implementation of Pr. EP in Columbus • What are the next steps?
 What Is Pr. EP? • Pre-exposure Prophylaxis: A pharmacologic HIV prevention intervention for persons at high risk of becoming infected with HIV. • An HIV-uninfected individual takes antiretroviral medication(s) before potential HIV exposure • The use of medication for prophylaxis is well established: – Use of contraceptive methods to prevent pregnancy – Use of antimalaria medications before traveling to endemic areas
What Is Pr. EP? • Pre-exposure Prophylaxis: A pharmacologic HIV prevention intervention for persons at high risk of becoming infected with HIV. • An HIV-uninfected individual takes antiretroviral medication(s) before potential HIV exposure • The use of medication for prophylaxis is well established: – Use of contraceptive methods to prevent pregnancy – Use of antimalaria medications before traveling to endemic areas
 Use of Antiretrovirals for HIV Prevention: • Prevention of mother-to-child transmission – Antiretrovirals given to the mother during pregnancy, labor, and delivery and to the infant postpartum[1] – PMTCT has nearly eliminated perinatal HIV infection in the US and other developed countries • Postexposure prophylaxis – Antiretrovirals given within hours of a known or suspected HIV exposure (eg, needle stick injury, rape, unprotected sexual intercourse with someone whose HIV status is unknown ) 1. DHHS. Perinatal Guidelines. 2014. 2013; 34: 875 -892. 2. MMWR. 2005; 54(RR-2): 1 -20.
Use of Antiretrovirals for HIV Prevention: • Prevention of mother-to-child transmission – Antiretrovirals given to the mother during pregnancy, labor, and delivery and to the infant postpartum[1] – PMTCT has nearly eliminated perinatal HIV infection in the US and other developed countries • Postexposure prophylaxis – Antiretrovirals given within hours of a known or suspected HIV exposure (eg, needle stick injury, rape, unprotected sexual intercourse with someone whose HIV status is unknown ) 1. DHHS. Perinatal Guidelines. 2014. 2013; 34: 875 -892. 2. MMWR. 2005; 54(RR-2): 1 -20.
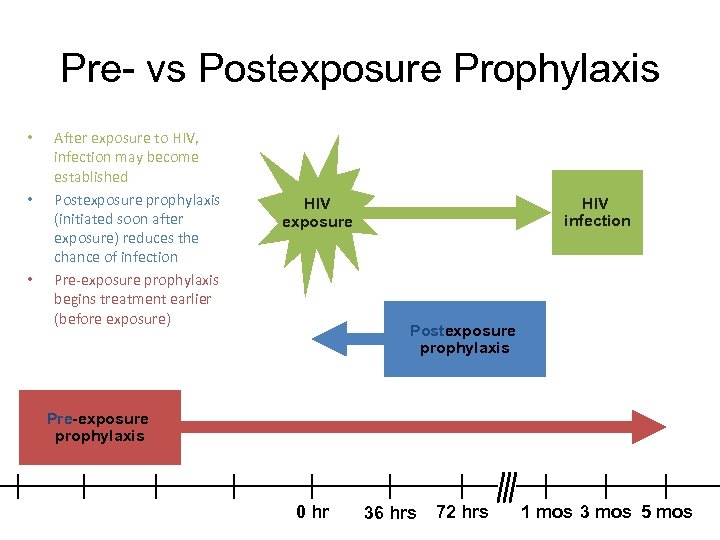 Pre- vs Postexposure Prophylaxis • • • After exposure to HIV, infection may become established Postexposure prophylaxis (initiated soon after exposure) reduces the chance of infection Pre-exposure prophylaxis begins treatment earlier (before exposure) HIV infection HIV exposure Postexposure prophylaxis Pre-exposure prophylaxis 0 hr 36 hrs 72 hrs 1 mos 3 mos 5 mos
Pre- vs Postexposure Prophylaxis • • • After exposure to HIV, infection may become established Postexposure prophylaxis (initiated soon after exposure) reduces the chance of infection Pre-exposure prophylaxis begins treatment earlier (before exposure) HIV infection HIV exposure Postexposure prophylaxis Pre-exposure prophylaxis 0 hr 36 hrs 72 hrs 1 mos 3 mos 5 mos
 How were the drugs used in Pr. EP selected? • Tenofovir disiproxil fumarate (TDF) and emtricitabine (FTC) well established nucleoside reverse transcriptase inhibitors already used for treatment of HIV-1 • Clinical trials have evaluated oral TDF, oral TDF/FTC combination and TDF vaginal gel – Safe, potent and well tolerated – Available co-formulated in single pill (Truvada) – Both FTC and TDF have long plasma (10 to 17 hours) and intracellular (391 and 1502 hours) half-lives – Have high penetration in vaginal and rectal tissues 1. Duwal et al, PLo. S One 2012; 7(7): e 40382 2. Jackson et al, JAIDS 2013; 62(3): 275 -281
How were the drugs used in Pr. EP selected? • Tenofovir disiproxil fumarate (TDF) and emtricitabine (FTC) well established nucleoside reverse transcriptase inhibitors already used for treatment of HIV-1 • Clinical trials have evaluated oral TDF, oral TDF/FTC combination and TDF vaginal gel – Safe, potent and well tolerated – Available co-formulated in single pill (Truvada) – Both FTC and TDF have long plasma (10 to 17 hours) and intracellular (391 and 1502 hours) half-lives – Have high penetration in vaginal and rectal tissues 1. Duwal et al, PLo. S One 2012; 7(7): e 40382 2. Jackson et al, JAIDS 2013; 62(3): 275 -281
 What is the scientific evidence supporting Pr. EP?
What is the scientific evidence supporting Pr. EP?
 i. Pr. Ex: Eligibility • • Male sex at birth (N = 2499) 18 yrs of age or older HIV-seronegative status Evidence of risk for acquisition of HIV infection Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599.
i. Pr. Ex: Eligibility • • Male sex at birth (N = 2499) 18 yrs of age or older HIV-seronegative status Evidence of risk for acquisition of HIV infection Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599.
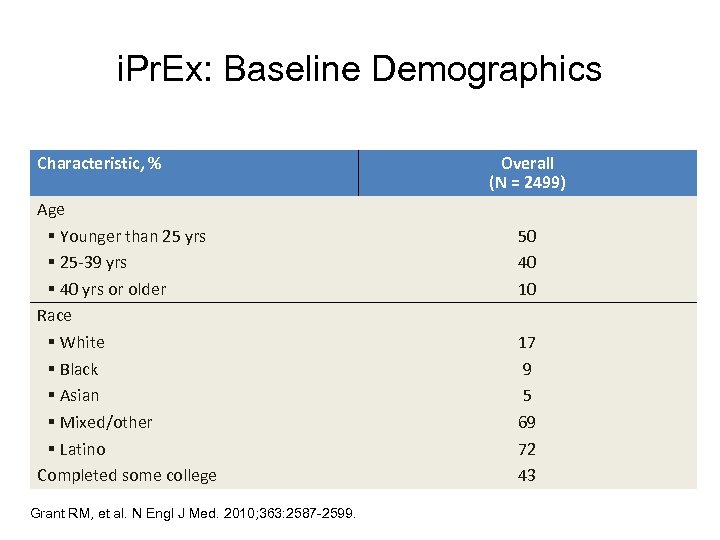 i. Pr. Ex: Baseline Demographics Characteristic, % Age § Younger than 25 yrs § 25 -39 yrs § 40 yrs or older Race § White § Black § Asian § Mixed/other § Latino Completed some college Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. Overall (N = 2499) 50 40 10 17 9 5 69 72 43
i. Pr. Ex: Baseline Demographics Characteristic, % Age § Younger than 25 yrs § 25 -39 yrs § 40 yrs or older Race § White § Black § Asian § Mixed/other § Latino Completed some college Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. Overall (N = 2499) 50 40 10 17 9 5 69 72 43
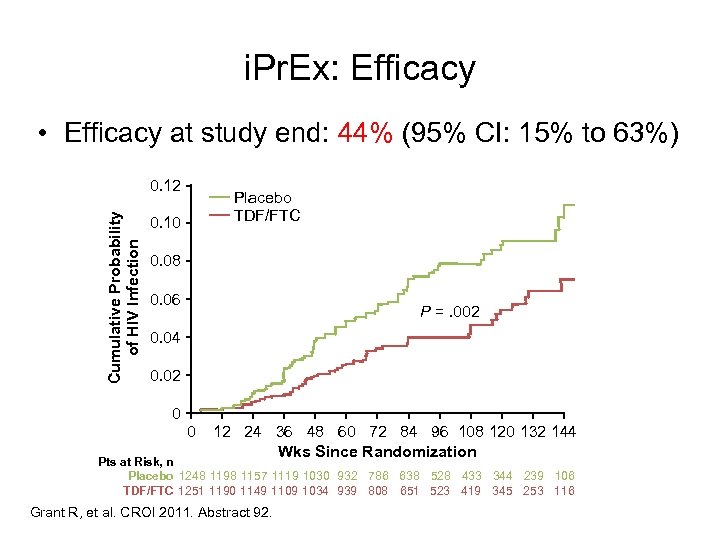 i. Pr. Ex: Efficacy • Efficacy at study end: 44% (95% CI: 15% to 63%) Cumulative Probability of HIV Infection 0. 12 Placebo TDF/FTC 0. 10 0. 08 0. 06 P =. 002 0. 04 0. 02 0 0 12 24 36 48 60 72 84 96 108 120 132 144 Wks Since Randomization Pts at Risk, n Placebo 1248 1198 1157 1119 1030 932 786 638 528 433 344 239 106 TDF/FTC 1251 1190 1149 1109 1034 939 808 651 523 419 345 253 116 Grant R, et al. CROI 2011. Abstract 92.
i. Pr. Ex: Efficacy • Efficacy at study end: 44% (95% CI: 15% to 63%) Cumulative Probability of HIV Infection 0. 12 Placebo TDF/FTC 0. 10 0. 08 0. 06 P =. 002 0. 04 0. 02 0 0 12 24 36 48 60 72 84 96 108 120 132 144 Wks Since Randomization Pts at Risk, n Placebo 1248 1198 1157 1119 1030 932 786 638 528 433 344 239 106 TDF/FTC 1251 1190 1149 1109 1034 939 808 651 523 419 345 253 116 Grant R, et al. CROI 2011. Abstract 92.
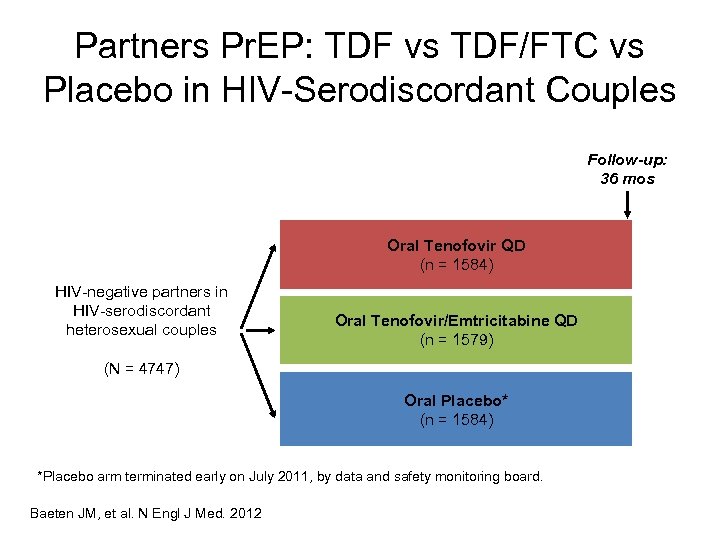 Partners Pr. EP: TDF vs TDF/FTC vs Placebo in HIV-Serodiscordant Couples Follow-up: 36 mos Oral Tenofovir QD (n = 1584) HIV-negative partners in HIV-serodiscordant heterosexual couples Oral Tenofovir/Emtricitabine QD (n = 1579) (N = 4747) Oral Placebo* (n = 1584) *Placebo arm terminated early on July 2011, by data and safety monitoring board. Baeten JM, et al. N Engl J Med. 2012
Partners Pr. EP: TDF vs TDF/FTC vs Placebo in HIV-Serodiscordant Couples Follow-up: 36 mos Oral Tenofovir QD (n = 1584) HIV-negative partners in HIV-serodiscordant heterosexual couples Oral Tenofovir/Emtricitabine QD (n = 1579) (N = 4747) Oral Placebo* (n = 1584) *Placebo arm terminated early on July 2011, by data and safety monitoring board. Baeten JM, et al. N Engl J Med. 2012
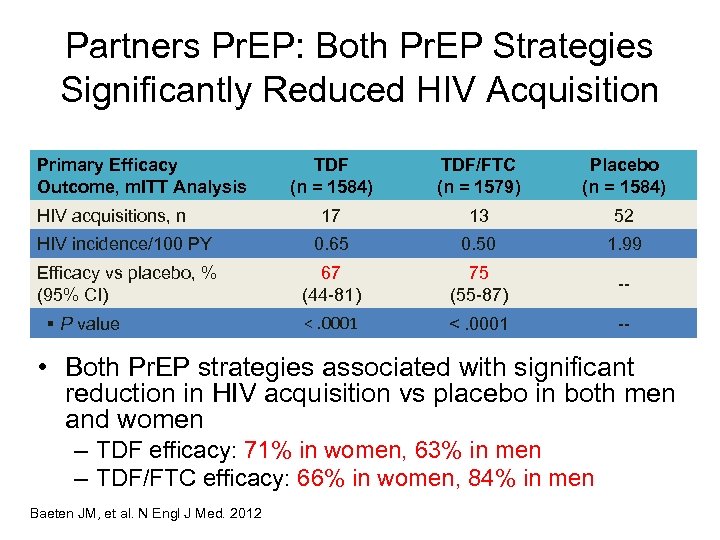 Partners Pr. EP: Both Pr. EP Strategies Significantly Reduced HIV Acquisition Primary Efficacy Outcome, m. ITT Analysis TDF (n = 1584) TDF/FTC (n = 1579) Placebo (n = 1584) 17 13 52 HIV incidence/100 PY 0. 65 0. 50 1. 99 Efficacy vs placebo, % (95% CI) 67 (44 -81) 75 (55 -87) -- <. 0001 -- HIV acquisitions, n § P value • Both Pr. EP strategies associated with significant reduction in HIV acquisition vs placebo in both men and women – TDF efficacy: 71% in women, 63% in men – TDF/FTC efficacy: 66% in women, 84% in men Baeten JM, et al. N Engl J Med. 2012
Partners Pr. EP: Both Pr. EP Strategies Significantly Reduced HIV Acquisition Primary Efficacy Outcome, m. ITT Analysis TDF (n = 1584) TDF/FTC (n = 1579) Placebo (n = 1584) 17 13 52 HIV incidence/100 PY 0. 65 0. 50 1. 99 Efficacy vs placebo, % (95% CI) 67 (44 -81) 75 (55 -87) -- <. 0001 -- HIV acquisitions, n § P value • Both Pr. EP strategies associated with significant reduction in HIV acquisition vs placebo in both men and women – TDF efficacy: 71% in women, 63% in men – TDF/FTC efficacy: 66% in women, 84% in men Baeten JM, et al. N Engl J Med. 2012
 TDF 2: Pr. EP With TDF/FTC in HIVNegative Heterosexuals in Botswana ≥ 12 -mo follow-up HIV-uninfected adults, heterosexually active, aged 18 -39 yrs Oral Tenofovir/Emtricitabine (n = 601) (N =1219)* Thigpen MC, et al N Engl J Med. 2012; [Epub ahead of print]. Oral Placebo (n = 599)
TDF 2: Pr. EP With TDF/FTC in HIVNegative Heterosexuals in Botswana ≥ 12 -mo follow-up HIV-uninfected adults, heterosexually active, aged 18 -39 yrs Oral Tenofovir/Emtricitabine (n = 601) (N =1219)* Thigpen MC, et al N Engl J Med. 2012; [Epub ahead of print]. Oral Placebo (n = 599)
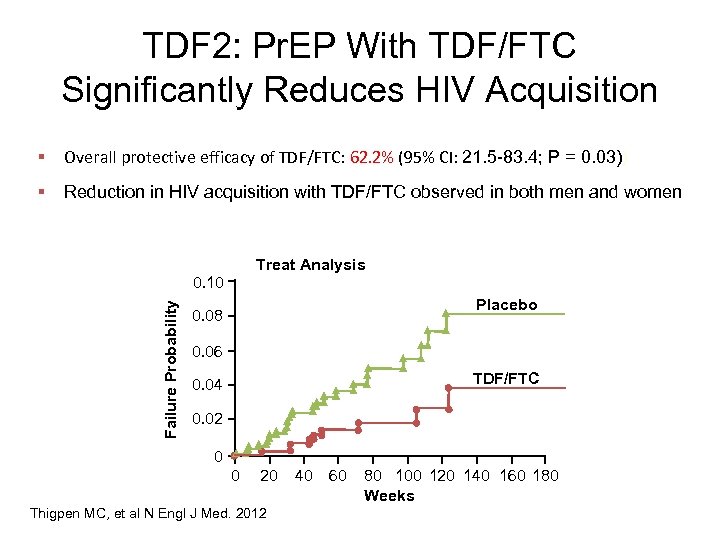 TDF 2: Pr. EP With TDF/FTC Significantly Reduces HIV Acquisition § Overall protective efficacy of TDF/FTC: 62. 2% (95% CI: 21. 5 -83. 4; P = 0. 03)) § Reduction in HIV acquisition with TDF/FTC observed in both men and women Treat Analysis Failure Probability 0. 10 Placebo 0. 08 0. 06 TDF/FTC 0. 04 0. 02 0 0 20 Thigpen MC, et al N Engl J Med. 2012 40 60 80 100 120 140 160 180 Weeks
TDF 2: Pr. EP With TDF/FTC Significantly Reduces HIV Acquisition § Overall protective efficacy of TDF/FTC: 62. 2% (95% CI: 21. 5 -83. 4; P = 0. 03)) § Reduction in HIV acquisition with TDF/FTC observed in both men and women Treat Analysis Failure Probability 0. 10 Placebo 0. 08 0. 06 TDF/FTC 0. 04 0. 02 0 0 20 Thigpen MC, et al N Engl J Med. 2012 40 60 80 100 120 140 160 180 Weeks
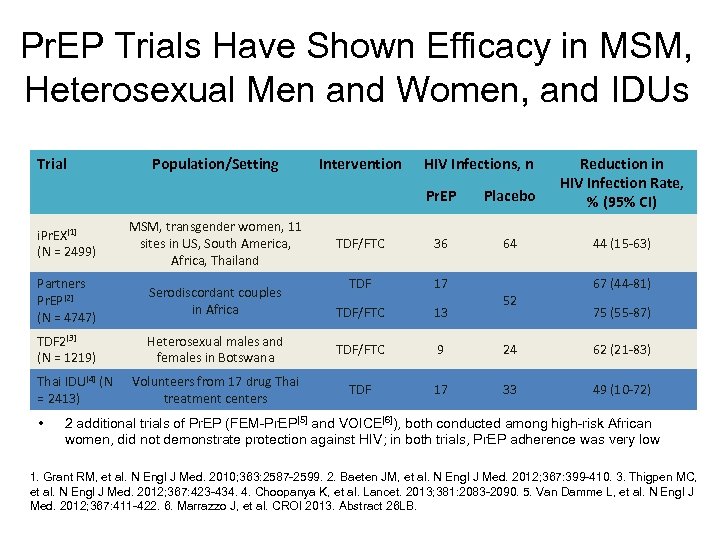 Pr. EP Trials Have Shown Efficacy in MSM, Heterosexual Men and Women, and IDUs Trial Population/Setting Intervention HIV Infections, n Pr. EP Placebo Reduction in HIV Infection Rate, % (95% CI) TDF/FTC 36 64 44 (15 -63) TDF 17 TDF/FTC 13 i. Pr. EX[1] (N = 2499) MSM, transgender women, 11 sites in US, South America, Africa, Thailand Partners Pr. EP[2] (N = 4747) Serodiscordant couples in Africa TDF 2[3] (N = 1219) Heterosexual males and females in Botswana TDF/FTC 9 24 62 (21 -83) Volunteers from 17 drug Thai treatment centers TDF 17 33 49 (10 -72) Thai IDU[4] (N = 2413) • 52 67 (44 -81) 75 (55 -87) 2 additional trials of Pr. EP (FEM-Pr. EP[5] and VOICE[6]), both conducted among high-risk African women, did not demonstrate protection against HIV; in both trials, Pr. EP adherence was very low 1. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 2. Baeten JM, et al. N Engl J Med. 2012; 367: 399 -410. 3. Thigpen MC, et al. N Engl J Med. 2012; 367: 423 -434. 4. Choopanya K, et al. Lancet. 2013; 381: 2083 -2090. 5. Van Damme L, et al. N Engl J Med. 2012; 367: 411 -422. 6. Marrazzo J, et al. CROI 2013. Abstract 26 LB.
Pr. EP Trials Have Shown Efficacy in MSM, Heterosexual Men and Women, and IDUs Trial Population/Setting Intervention HIV Infections, n Pr. EP Placebo Reduction in HIV Infection Rate, % (95% CI) TDF/FTC 36 64 44 (15 -63) TDF 17 TDF/FTC 13 i. Pr. EX[1] (N = 2499) MSM, transgender women, 11 sites in US, South America, Africa, Thailand Partners Pr. EP[2] (N = 4747) Serodiscordant couples in Africa TDF 2[3] (N = 1219) Heterosexual males and females in Botswana TDF/FTC 9 24 62 (21 -83) Volunteers from 17 drug Thai treatment centers TDF 17 33 49 (10 -72) Thai IDU[4] (N = 2413) • 52 67 (44 -81) 75 (55 -87) 2 additional trials of Pr. EP (FEM-Pr. EP[5] and VOICE[6]), both conducted among high-risk African women, did not demonstrate protection against HIV; in both trials, Pr. EP adherence was very low 1. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 2. Baeten JM, et al. N Engl J Med. 2012; 367: 399 -410. 3. Thigpen MC, et al. N Engl J Med. 2012; 367: 423 -434. 4. Choopanya K, et al. Lancet. 2013; 381: 2083 -2090. 5. Van Damme L, et al. N Engl J Med. 2012; 367: 411 -422. 6. Marrazzo J, et al. CROI 2013. Abstract 26 LB.
 Disappointing Results of Pr. EP in Women: FEM-Pr. EP and VOICE § FEM-Pr. EP: Study of oral TDF/FTC for 3900 § high-risk women in Africa (2120 randomized) – Study ended early by DSMB due to lack of efficacy – 35 vs 33 new HIV infections in the placebo and TDF/FTC arms[1] – TDF blood levels show that adherence was too low (< 40%) 1. 2. VOICE: Phase IIB placebo-controlled trial of > 5000 women in South Africa, Uganda, and Zimbabwe[2] – Daily oral TDF; daily oral TDF/FTC; daily vaginal TFV 1% gel – DSMB stopped the daily oral TDF arm and daily vaginal gel arm for lack of efficacy – Daily oral TDF/FTC arm continued but recently shown to have low efficacy due to poor treatment adherence Van Damme L, et al N Engl J Med. 2012 Jul 11. [Epub ahead of print]. Marrazzo J, et al. CROI 2013. Abstract 26 LB.
Disappointing Results of Pr. EP in Women: FEM-Pr. EP and VOICE § FEM-Pr. EP: Study of oral TDF/FTC for 3900 § high-risk women in Africa (2120 randomized) – Study ended early by DSMB due to lack of efficacy – 35 vs 33 new HIV infections in the placebo and TDF/FTC arms[1] – TDF blood levels show that adherence was too low (< 40%) 1. 2. VOICE: Phase IIB placebo-controlled trial of > 5000 women in South Africa, Uganda, and Zimbabwe[2] – Daily oral TDF; daily oral TDF/FTC; daily vaginal TFV 1% gel – DSMB stopped the daily oral TDF arm and daily vaginal gel arm for lack of efficacy – Daily oral TDF/FTC arm continued but recently shown to have low efficacy due to poor treatment adherence Van Damme L, et al N Engl J Med. 2012 Jul 11. [Epub ahead of print]. Marrazzo J, et al. CROI 2013. Abstract 26 LB.
 Importance of Pr. EP Adherence
Importance of Pr. EP Adherence
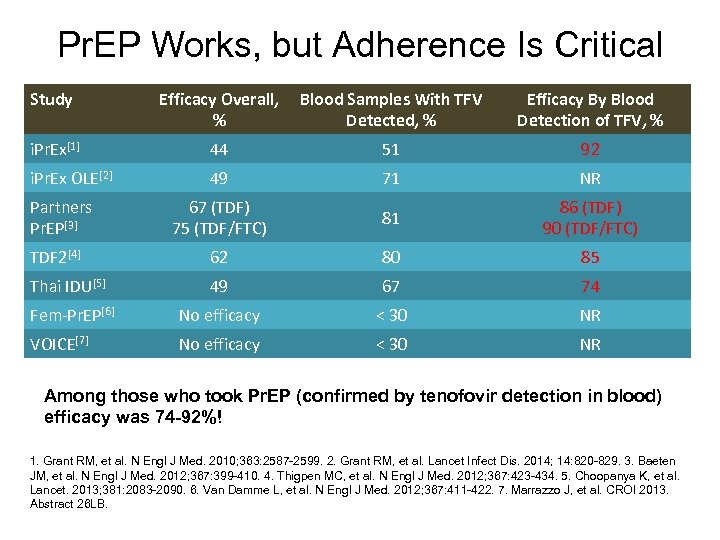 Pr. EP Works, but Adherence Is Critical Study Efficacy Overall, % Blood Samples With TFV Detected, % Efficacy By Blood Detection of TFV, % i. Pr. Ex[1] 44 51 92 i. Pr. Ex OLE[2] 49 71 NR 67 (TDF) 75 (TDF/FTC) 81 86 (TDF) 90 (TDF/FTC) TDF 2[4] 62 80 85 Thai IDU[5] 49 67 74 Fem-Pr. EP[6] No efficacy < 30 NR VOICE[7] No efficacy < 30 NR Partners Pr. EP[3] Among those who took Pr. EP (confirmed by tenofovir detection in blood) efficacy was 74 -92%! 1. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 2. Grant RM, et al. Lancet Infect Dis. 2014; 14: 820 -829. 3. Baeten JM, et al. N Engl J Med. 2012; 367: 399 -410. 4. Thigpen MC, et al. N Engl J Med. 2012; 367: 423 -434. 5. Choopanya K, et al. Lancet. 2013; 381: 2083 -2090. 6. Van Damme L, et al. N Engl J Med. 2012; 367: 411 -422. 7. Marrazzo J, et al. CROI 2013. Abstract 26 LB.
Pr. EP Works, but Adherence Is Critical Study Efficacy Overall, % Blood Samples With TFV Detected, % Efficacy By Blood Detection of TFV, % i. Pr. Ex[1] 44 51 92 i. Pr. Ex OLE[2] 49 71 NR 67 (TDF) 75 (TDF/FTC) 81 86 (TDF) 90 (TDF/FTC) TDF 2[4] 62 80 85 Thai IDU[5] 49 67 74 Fem-Pr. EP[6] No efficacy < 30 NR VOICE[7] No efficacy < 30 NR Partners Pr. EP[3] Among those who took Pr. EP (confirmed by tenofovir detection in blood) efficacy was 74 -92%! 1. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 2. Grant RM, et al. Lancet Infect Dis. 2014; 14: 820 -829. 3. Baeten JM, et al. N Engl J Med. 2012; 367: 399 -410. 4. Thigpen MC, et al. N Engl J Med. 2012; 367: 423 -434. 5. Choopanya K, et al. Lancet. 2013; 381: 2083 -2090. 6. Van Damme L, et al. N Engl J Med. 2012; 367: 411 -422. 7. Marrazzo J, et al. CROI 2013. Abstract 26 LB.
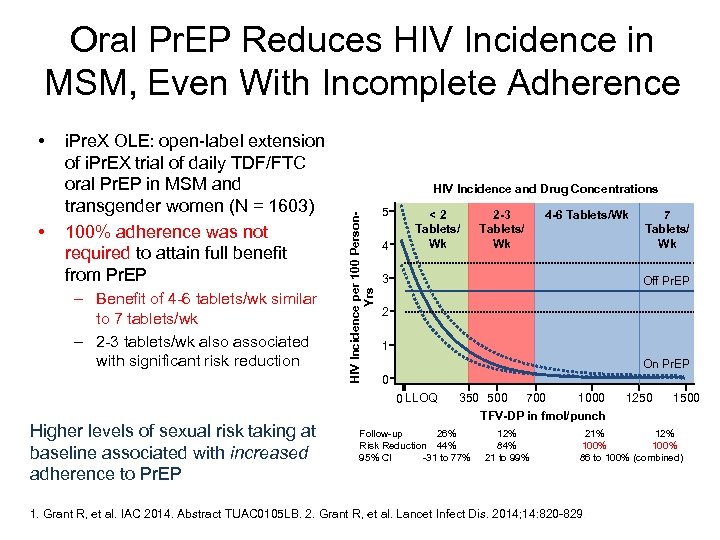 Oral Pr. EP Reduces HIV Incidence in MSM, Even With Incomplete Adherence • i. Pre. X OLE: open-label extension of i. Pr. EX trial of daily TDF/FTC oral Pr. EP in MSM and transgender women (N = 1603) 100% adherence was not required to attain full benefit from Pr. EP – Benefit of 4 -6 tablets/wk similar to 7 tablets/wk – 2 -3 tablets/wk also associated with significant risk reduction HIV Incidence and Drug Concentrations HIV Incidence per 100 Person. Yrs • 5 4 <2 Tablets/ Wk 4 -6 Tablets/Wk 3 7 Tablets/ Wk Off Pr. EP 2 1 On Pr. EP 0 0 LLOQ Higher levels of sexual risk taking at baseline associated with increased adherence to Pr. EP 2 -3 Tablets/ Wk 350 500 700 1000 TFV-DP in fmol/punch Follow-up 26% Risk Reduction 44% 95% Cl -31 to 77% 12% 84% 21 to 99% 1250 1500 21% 12% 100% 86 to 100% (combined) 1. Grant R, et al. IAC 2014. Abstract TUAC 0105 LB. 2. Grant R, et al. Lancet Infect Dis. 2014; 14: 820 -829.
Oral Pr. EP Reduces HIV Incidence in MSM, Even With Incomplete Adherence • i. Pre. X OLE: open-label extension of i. Pr. EX trial of daily TDF/FTC oral Pr. EP in MSM and transgender women (N = 1603) 100% adherence was not required to attain full benefit from Pr. EP – Benefit of 4 -6 tablets/wk similar to 7 tablets/wk – 2 -3 tablets/wk also associated with significant risk reduction HIV Incidence and Drug Concentrations HIV Incidence per 100 Person. Yrs • 5 4 <2 Tablets/ Wk 4 -6 Tablets/Wk 3 7 Tablets/ Wk Off Pr. EP 2 1 On Pr. EP 0 0 LLOQ Higher levels of sexual risk taking at baseline associated with increased adherence to Pr. EP 2 -3 Tablets/ Wk 350 500 700 1000 TFV-DP in fmol/punch Follow-up 26% Risk Reduction 44% 95% Cl -31 to 77% 12% 84% 21 to 99% 1250 1500 21% 12% 100% 86 to 100% (combined) 1. Grant R, et al. IAC 2014. Abstract TUAC 0105 LB. 2. Grant R, et al. Lancet Infect Dis. 2014; 14: 820 -829.
 Why poor results in VOICE and FEM-Pr. EP? • Why were there differences between these studies and the other TDF-based studies? – Very poor adherence. – Poor adherence in MSM may be more forgiving due to high TDF concentration in rectal tissue.
Why poor results in VOICE and FEM-Pr. EP? • Why were there differences between these studies and the other TDF-based studies? – Very poor adherence. – Poor adherence in MSM may be more forgiving due to high TDF concentration in rectal tissue.
 Safety and Tolerability
Safety and Tolerability
![Adverse Events • Very few and mild AEs observed in Pr. EP trials[1] – Adverse Events • Very few and mild AEs observed in Pr. EP trials[1] –](https://present5.com/presentation/3c756063de6d771ff3e905fea3671056/image-23.jpg) Adverse Events • Very few and mild AEs observed in Pr. EP trials[1] – i. Pr. Ex: small but significant early nausea and weight loss[2] • Potential bone and renal toxicity – Known risk associated with TDF • Potential for drug-resistant HIV infection – Infrequent in clinical trials but must exclude HIV infection 1. CDC. Pr. EP Guideline. 2014. 2. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599.
Adverse Events • Very few and mild AEs observed in Pr. EP trials[1] – i. Pr. Ex: small but significant early nausea and weight loss[2] • Potential bone and renal toxicity – Known risk associated with TDF • Potential for drug-resistant HIV infection – Infrequent in clinical trials but must exclude HIV infection 1. CDC. Pr. EP Guideline. 2014. 2. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599.
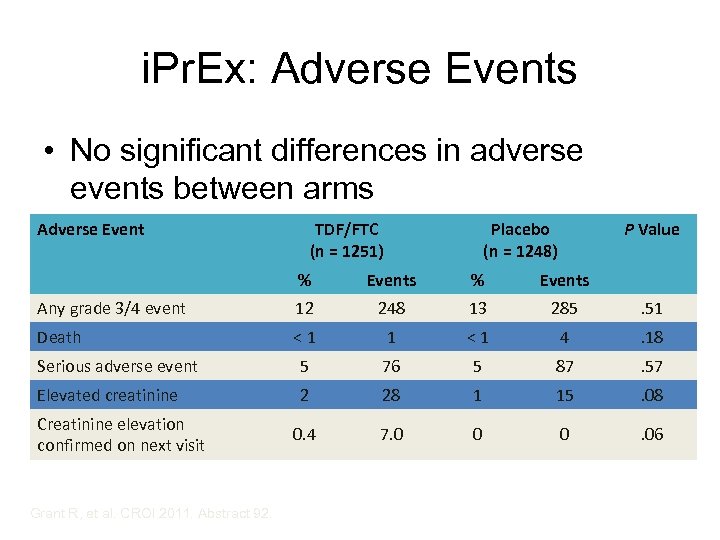 i. Pr. Ex: Adverse Events • No significant differences in adverse events between arms Adverse Event TDF/FTC (n = 1251) Placebo (n = 1248) P Value % Events Any grade 3/4 event 12 248 13 285 . 51 Death <1 1 <1 4 . 18 Serious adverse event 5 76 5 87 . 57 Elevated creatinine 2 28 1 15 . 08 0. 4 7. 0 0 0 . 06 Creatinine elevation confirmed on next visit Grant R, et al. CROI 2011. Abstract 92.
i. Pr. Ex: Adverse Events • No significant differences in adverse events between arms Adverse Event TDF/FTC (n = 1251) Placebo (n = 1248) P Value % Events Any grade 3/4 event 12 248 13 285 . 51 Death <1 1 <1 4 . 18 Serious adverse event 5 76 5 87 . 57 Elevated creatinine 2 28 1 15 . 08 0. 4 7. 0 0 0 . 06 Creatinine elevation confirmed on next visit Grant R, et al. CROI 2011. Abstract 92.
 i. Pr. Ex: Bone Mineral Density Changes and Fractures § Bone mineral density changes were small (~1%); no clear negative effect on health[1] § No differences in fracture rates between groups[1, 2] § All fractures were trauma related 1. Liu AY, et al. PLo. S One. 2011; 6: e 23688. 2. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599.
i. Pr. Ex: Bone Mineral Density Changes and Fractures § Bone mineral density changes were small (~1%); no clear negative effect on health[1] § No differences in fracture rates between groups[1, 2] § All fractures were trauma related 1. Liu AY, et al. PLo. S One. 2011; 6: e 23688. 2. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599.
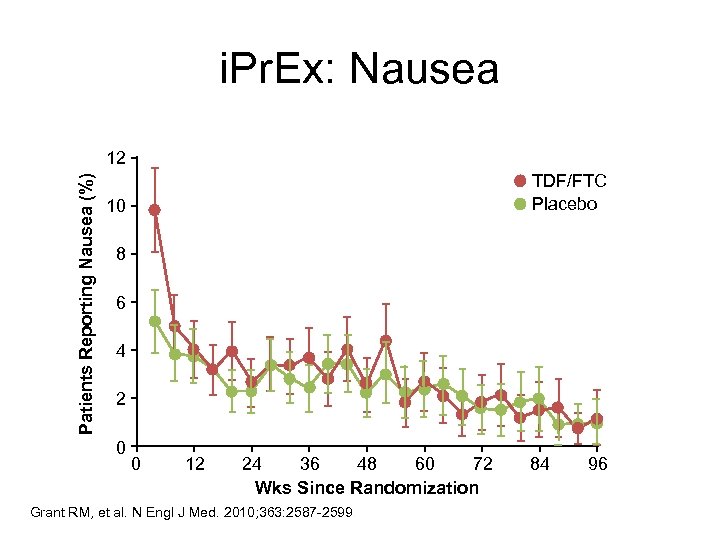 i. Pr. Ex: Nausea Patients Reporting Nausea (%) 12 TDF/FTC Placebo 10 8 6 4 2 0 0 12 24 36 48 60 72 Wks Since Randomization Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 84 96
i. Pr. Ex: Nausea Patients Reporting Nausea (%) 12 TDF/FTC Placebo 10 8 6 4 2 0 0 12 24 36 48 60 72 Wks Since Randomization Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 84 96
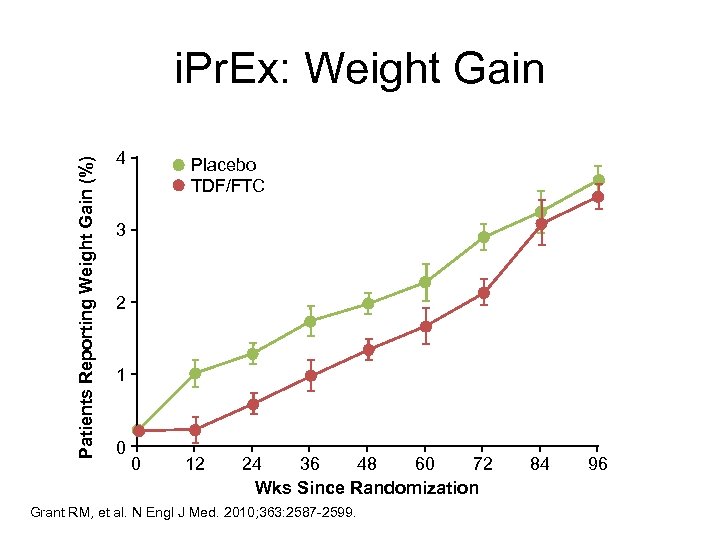 Patients Reporting Weight Gain (%) i. Pr. Ex: Weight Gain 4 Placebo TDF/FTC 3 2 1 0 0 12 24 36 48 60 72 Wks Since Randomization Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 84 96
Patients Reporting Weight Gain (%) i. Pr. Ex: Weight Gain 4 Placebo TDF/FTC 3 2 1 0 0 12 24 36 48 60 72 Wks Since Randomization Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 84 96
 Risk Behavior
Risk Behavior
![Pr. EP Trials Found Decreasing Risk Behavior Over Time i. Pr. Ex[1] Placebo TDF/FTC Pr. EP Trials Found Decreasing Risk Behavior Over Time i. Pr. Ex[1] Placebo TDF/FTC](https://present5.com/presentation/3c756063de6d771ff3e905fea3671056/image-29.jpg) Pr. EP Trials Found Decreasing Risk Behavior Over Time i. Pr. Ex[1] Placebo TDF/FTC 80 60 40 20 Placebo TDF/FTC TDF 50 Subjects Reporting Unprotected Sex (%) Subjects Reporting Unprotected Receptive Anal Sex (%) 100 Partners Pr. EP[2] 0 40 30 20 10 0 0 24 48 72 96 120 144 Wks Since Randomization 1. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 2. Baeten JM, et al. N Engl J Med. 2012; 367: 399 -410. 0 3 6 9 12 15 18 21 24 27 30 33 Follow-up Time (Mos)
Pr. EP Trials Found Decreasing Risk Behavior Over Time i. Pr. Ex[1] Placebo TDF/FTC 80 60 40 20 Placebo TDF/FTC TDF 50 Subjects Reporting Unprotected Sex (%) Subjects Reporting Unprotected Receptive Anal Sex (%) 100 Partners Pr. EP[2] 0 40 30 20 10 0 0 24 48 72 96 120 144 Wks Since Randomization 1. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 2. Baeten JM, et al. N Engl J Med. 2012; 367: 399 -410. 0 3 6 9 12 15 18 21 24 27 30 33 Follow-up Time (Mos)
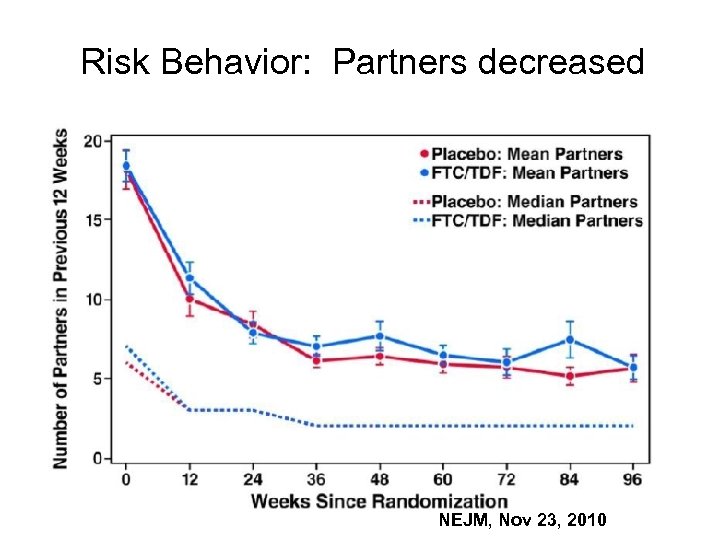 Risk Behavior: Partners decreased NEJM, Nov 23, 2010
Risk Behavior: Partners decreased NEJM, Nov 23, 2010
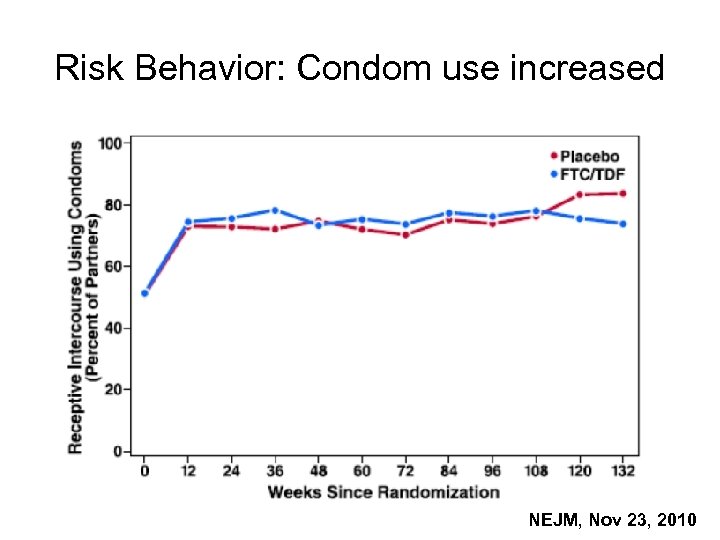 Risk Behavior: Condom use increased NEJM, Nov 23, 2010
Risk Behavior: Condom use increased NEJM, Nov 23, 2010
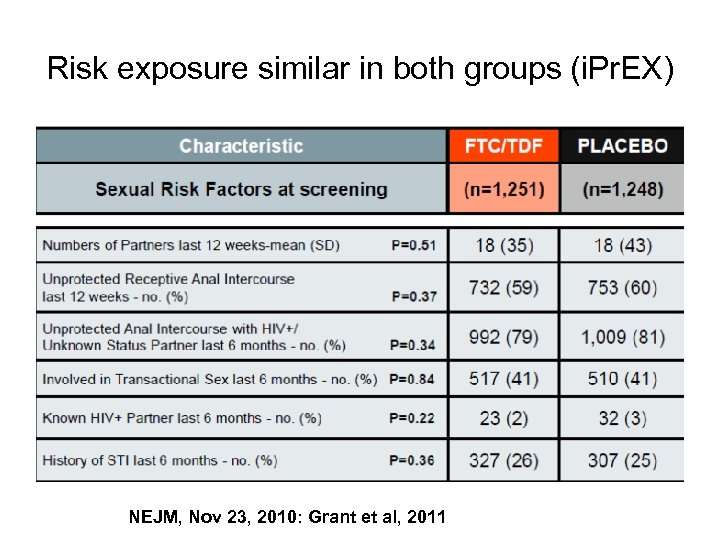 Risk exposure similar in both groups (i. Pr. EX) NEJM, Nov 23, 2010: Grant et al, 2011
Risk exposure similar in both groups (i. Pr. EX) NEJM, Nov 23, 2010: Grant et al, 2011
 Drug Resistance
Drug Resistance
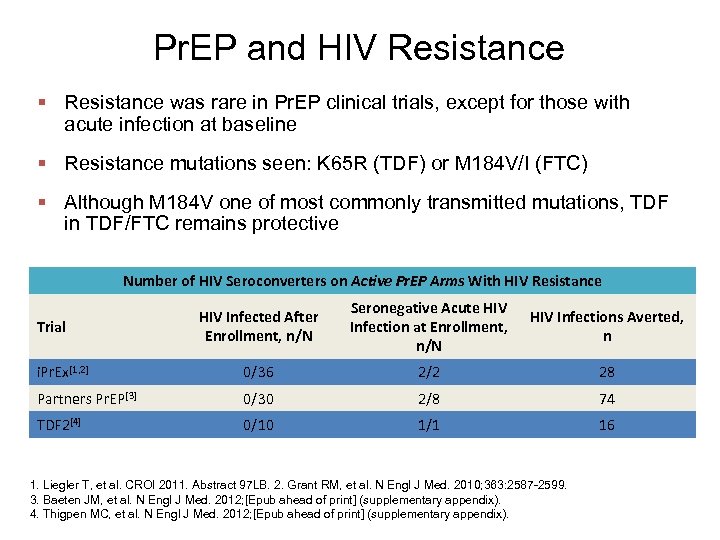 Pr. EP and HIV Resistance § Resistance was rare in Pr. EP clinical trials, except for those with acute infection at baseline § Resistance mutations seen: K 65 R (TDF) or M 184 V/I (FTC) § Although M 184 V one of most commonly transmitted mutations, TDF in TDF/FTC remains protective Number of HIV Seroconverters on Active Pr. EP Arms With HIV Resistance HIV Infected After Enrollment, n/N Seronegative Acute HIV Infection at Enrollment, n/N HIV Infections Averted, n i. Pr. Ex[1, 2] 0/36 2/2 28 Partners Pr. EP[3] 0/30 2/8 74 TDF 2[4] 0/10 1/1 16 Trial 1. Liegler T, et al. CROI 2011. Abstract 97 LB. 2. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 3. Baeten JM, et al. N Engl J Med. 2012; [Epub ahead of print] (supplementary appendix). 4. Thigpen MC, et al. N Engl J Med. 2012; [Epub ahead of print] (supplementary appendix).
Pr. EP and HIV Resistance § Resistance was rare in Pr. EP clinical trials, except for those with acute infection at baseline § Resistance mutations seen: K 65 R (TDF) or M 184 V/I (FTC) § Although M 184 V one of most commonly transmitted mutations, TDF in TDF/FTC remains protective Number of HIV Seroconverters on Active Pr. EP Arms With HIV Resistance HIV Infected After Enrollment, n/N Seronegative Acute HIV Infection at Enrollment, n/N HIV Infections Averted, n i. Pr. Ex[1, 2] 0/36 2/2 28 Partners Pr. EP[3] 0/30 2/8 74 TDF 2[4] 0/10 1/1 16 Trial 1. Liegler T, et al. CROI 2011. Abstract 97 LB. 2. Grant RM, et al. N Engl J Med. 2010; 363: 2587 -2599. 3. Baeten JM, et al. N Engl J Med. 2012; [Epub ahead of print] (supplementary appendix). 4. Thigpen MC, et al. N Engl J Med. 2012; [Epub ahead of print] (supplementary appendix).
 “Real World” Experience
“Real World” Experience
 PROUD: “Real World” Use of Pr. EP was highly effective. • Randomized, open-label trial of daily oral TDF/FTC Pr. EP in HIV- MSM in 13 clinics in London – Immediate (n = 267) vs § § – Deferred for 12 mos (n = 256) • Primary endpoint: HIV infection in first 12 mos • 86% reduction in risk seen over 60 wks with immediate Pr. EP (90% CI: 58% to 96%, P =. 0002) – Rate difference: 7. 6 (90% CI: 4. 1 -11. 2) – Number needed to treat to prevent 1 infection: 13 (90% CI: 9 -25) Mc. Cormack S, et al. CROI 2015. Abstract 22 LB. 2 of 3 infected persons in immediate group seroconverting at study entry or shortly after first dose of Pr. EP M 184 V/I observed in 3/6 patients who seroconverted – No K 65 R mutation against TDF § § High rate of STIs seen in both groups (suggests no risk compensation!) DMSB stopped trial; recommended that all participants be offered Pr. EP
PROUD: “Real World” Use of Pr. EP was highly effective. • Randomized, open-label trial of daily oral TDF/FTC Pr. EP in HIV- MSM in 13 clinics in London – Immediate (n = 267) vs § § – Deferred for 12 mos (n = 256) • Primary endpoint: HIV infection in first 12 mos • 86% reduction in risk seen over 60 wks with immediate Pr. EP (90% CI: 58% to 96%, P =. 0002) – Rate difference: 7. 6 (90% CI: 4. 1 -11. 2) – Number needed to treat to prevent 1 infection: 13 (90% CI: 9 -25) Mc. Cormack S, et al. CROI 2015. Abstract 22 LB. 2 of 3 infected persons in immediate group seroconverting at study entry or shortly after first dose of Pr. EP M 184 V/I observed in 3/6 patients who seroconverted – No K 65 R mutation against TDF § § High rate of STIs seen in both groups (suggests no risk compensation!) DMSB stopped trial; recommended that all participants be offered Pr. EP
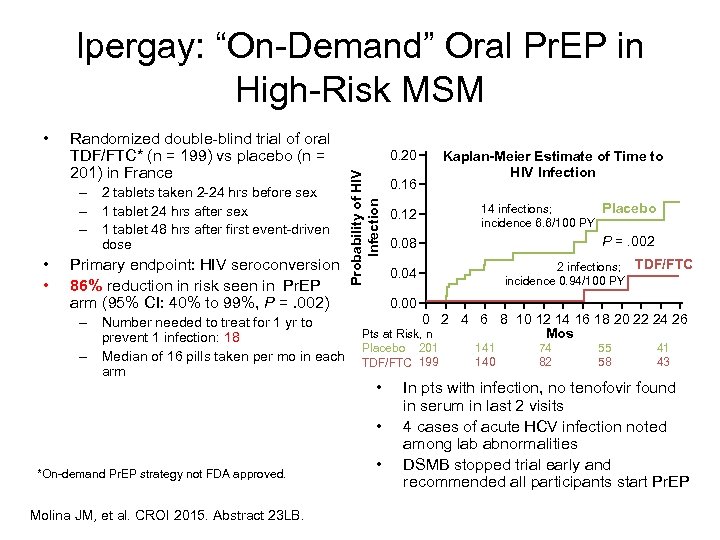 Ipergay: “On-Demand” Oral Pr. EP in High-Risk MSM Randomized double-blind trial of oral TDF/FTC* (n = 199) vs placebo (n = 201) in France – 2 tablets taken 2 -24 hrs before sex – 1 tablet 24 hrs after sex – 1 tablet 48 hrs after first event-driven dose • • Primary endpoint: HIV seroconversion 86% reduction in risk seen in Pr. EP arm (95% CI: 40% to 99%, P =. 002) – Number needed to treat for 1 yr to prevent 1 infection: 18 – Median of 16 pills taken per mo in each arm 0. 20 Probability of HIV Infection • Molina JM, et al. CROI 2015. Abstract 23 LB. 0. 12 Placebo 14 infections; incidence 6. 6/100 PY 0. 08 P =. 002 2 infections; TDF/FTC incidence 0. 94/100 PY 0. 04 0. 00 0 2 4 6 8 10 12 14 16 18 20 22 24 26 Mos Pts at Risk, n Placebo 201 TDF/FTC 199 • • *On-demand Pr. EP strategy not FDA approved. 0. 16 Kaplan-Meier Estimate of Time to HIV Infection • 141 140 74 82 55 58 41 43 In pts with infection, no tenofovir found in serum in last 2 visits 4 cases of acute HCV infection noted among lab abnormalities DSMB stopped trial early and recommended all participants start Pr. EP
Ipergay: “On-Demand” Oral Pr. EP in High-Risk MSM Randomized double-blind trial of oral TDF/FTC* (n = 199) vs placebo (n = 201) in France – 2 tablets taken 2 -24 hrs before sex – 1 tablet 24 hrs after sex – 1 tablet 48 hrs after first event-driven dose • • Primary endpoint: HIV seroconversion 86% reduction in risk seen in Pr. EP arm (95% CI: 40% to 99%, P =. 002) – Number needed to treat for 1 yr to prevent 1 infection: 18 – Median of 16 pills taken per mo in each arm 0. 20 Probability of HIV Infection • Molina JM, et al. CROI 2015. Abstract 23 LB. 0. 12 Placebo 14 infections; incidence 6. 6/100 PY 0. 08 P =. 002 2 infections; TDF/FTC incidence 0. 94/100 PY 0. 04 0. 00 0 2 4 6 8 10 12 14 16 18 20 22 24 26 Mos Pts at Risk, n Placebo 201 TDF/FTC 199 • • *On-demand Pr. EP strategy not FDA approved. 0. 16 Kaplan-Meier Estimate of Time to HIV Infection • 141 140 74 82 55 58 41 43 In pts with infection, no tenofovir found in serum in last 2 visits 4 cases of acute HCV infection noted among lab abnormalities DSMB stopped trial early and recommended all participants start Pr. EP
 Pr. EP + ART as Prevention in Serodiscordant Couples • Partners Demonstration Project in Uganda – Oral daily TDF/FTC Pr. EP for HIVuninfected partner in serodiscordant couple continued 6 mos beyond initiation of ART for infected partner – High-risk couples: younger age, fewer children, uncircumcised HIV-negative male, cohabitating, unprotected sex in past mo, high HIV-1 RNA in HIVpositive partner § – > 95% of HIV-negative partners using Pr. EP – 80% of HIV-positive partners have initiated ART; of these, > 90% with suppression § Expected Actual § Infected, n Incidence/100 PY (95% CI) 39. 7 5. 2 (3. 7 -6. 9) 2 0. 2 (0 -0. 9) Baeten J, et al. CROI 2015. Abstract 24. 96% reduction in expected infections ‒ HIV Incidence, Actual vs Expected Group Interim analysis IRR, expected vs observed: 0. 04 (95% CI: 0. 01 -0. 19; P <. 0001 In pts with seroconversion, no tenofovir detectable in plasma at time of seroconversion – HIV-positive partner in 1 couple not on ART (high CD 4+ count) – Other couple dissolved and HIV-negative partner in new relationship
Pr. EP + ART as Prevention in Serodiscordant Couples • Partners Demonstration Project in Uganda – Oral daily TDF/FTC Pr. EP for HIVuninfected partner in serodiscordant couple continued 6 mos beyond initiation of ART for infected partner – High-risk couples: younger age, fewer children, uncircumcised HIV-negative male, cohabitating, unprotected sex in past mo, high HIV-1 RNA in HIVpositive partner § – > 95% of HIV-negative partners using Pr. EP – 80% of HIV-positive partners have initiated ART; of these, > 90% with suppression § Expected Actual § Infected, n Incidence/100 PY (95% CI) 39. 7 5. 2 (3. 7 -6. 9) 2 0. 2 (0 -0. 9) Baeten J, et al. CROI 2015. Abstract 24. 96% reduction in expected infections ‒ HIV Incidence, Actual vs Expected Group Interim analysis IRR, expected vs observed: 0. 04 (95% CI: 0. 01 -0. 19; P <. 0001 In pts with seroconversion, no tenofovir detectable in plasma at time of seroconversion – HIV-positive partner in 1 couple not on ART (high CD 4+ count) – Other couple dissolved and HIV-negative partner in new relationship
 Pr. EP Implementation: Pre. Pr. EP Counseling, Testing, and Clinical Evaluation
Pr. EP Implementation: Pre. Pr. EP Counseling, Testing, and Clinical Evaluation
 CDC Pr. EP Guideline: For Which Patients Is Pr. EP Recommended? • Pr. EP is recommended as one prevention option for the following adults at substantial risk of HIV acquisition – Sexually active MSM – Heterosexually active men and women – Injection drug users MSM Potential indicators of substantial risk of acquiring HIV infection § HIV-positive sexual partner § Recent bacterial STI § High number of sex partners § History of inconsistent or no condom use § Commercial sex work CDC. Pr. EP Guideline. 2014. Heterosexual Women and Men HIV-positive sexual partner Recent bacterial STI High number of sex partners History of inconsistent or no condom use § Commercial sex work § In high-prevalence area or network § § Injection Drug Users § HIV-positive injecting partner § Sharing injection equipment § Recent drug treatment (but currently injecting)
CDC Pr. EP Guideline: For Which Patients Is Pr. EP Recommended? • Pr. EP is recommended as one prevention option for the following adults at substantial risk of HIV acquisition – Sexually active MSM – Heterosexually active men and women – Injection drug users MSM Potential indicators of substantial risk of acquiring HIV infection § HIV-positive sexual partner § Recent bacterial STI § High number of sex partners § History of inconsistent or no condom use § Commercial sex work CDC. Pr. EP Guideline. 2014. Heterosexual Women and Men HIV-positive sexual partner Recent bacterial STI High number of sex partners History of inconsistent or no condom use § Commercial sex work § In high-prevalence area or network § § Injection Drug Users § HIV-positive injecting partner § Sharing injection equipment § Recent drug treatment (but currently injecting)
 CDC Guideline: Clinical Eligibility for Pr. EP • The following are required before prescribing Pr. EP to identify patients for whom Pr. EP would be harmful or may present risks to health: – Documented negative HIV test result – No signs or symptoms of acute HIV infection – Normal renal function; no use of contraindicated medications – Documented HBV infection status and vaccination status CDC. Pr. EP Guideline. 2014.
CDC Guideline: Clinical Eligibility for Pr. EP • The following are required before prescribing Pr. EP to identify patients for whom Pr. EP would be harmful or may present risks to health: – Documented negative HIV test result – No signs or symptoms of acute HIV infection – Normal renal function; no use of contraindicated medications – Documented HBV infection status and vaccination status CDC. Pr. EP Guideline. 2014.
![HIV Screening • Exclude acute and chronic HIV infection[1, 2] – May need to HIV Screening • Exclude acute and chronic HIV infection[1, 2] – May need to](https://present5.com/presentation/3c756063de6d771ff3e905fea3671056/image-42.jpg) HIV Screening • Exclude acute and chronic HIV infection[1, 2] – May need to use 4 th-generation HIV Ag/Ab or HIV-1 RNA using nucleic acid–based tests if acute infection is suspected – Document negative antibody test within the week before starting Pr. EP – Do not accept patient-reported results – Avoid use of oral rapid HIV testing due to lower sensitivity 1. CDC. Pr. EP Guideline. 2014. 2. Daar ES, et al. Curr Opin HIV AIDS. 2008; 3: 10 -15.
HIV Screening • Exclude acute and chronic HIV infection[1, 2] – May need to use 4 th-generation HIV Ag/Ab or HIV-1 RNA using nucleic acid–based tests if acute infection is suspected – Document negative antibody test within the week before starting Pr. EP – Do not accept patient-reported results – Avoid use of oral rapid HIV testing due to lower sensitivity 1. CDC. Pr. EP Guideline. 2014. 2. Daar ES, et al. Curr Opin HIV AIDS. 2008; 3: 10 -15.
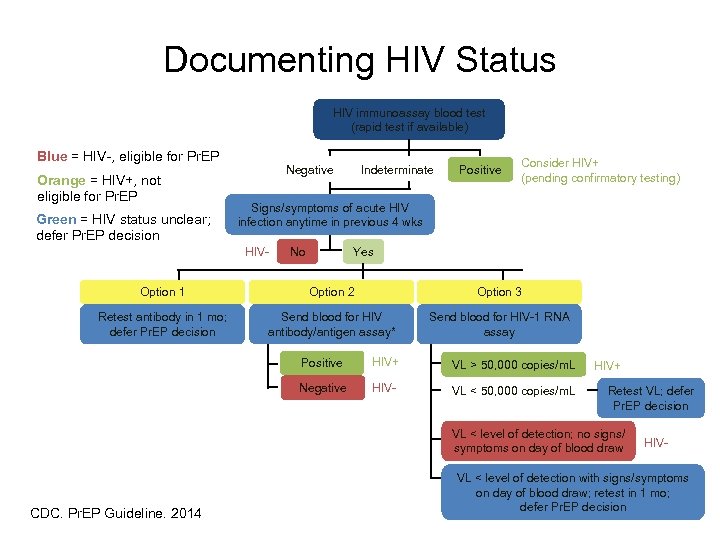 Documenting HIV Status HIV immunoassay blood test (rapid test if available) Blue = HIV-, eligible for Pr. EP Orange = HIV+, not eligible for Pr. EP Green = HIV status unclear; defer Pr. EP decision Indeterminate Negative Positive Consider HIV+ (pending confirmatory testing) Signs/symptoms of acute HIV infection anytime in previous 4 wks HIV- No Yes Option 1 Option 2 Option 3 Retest antibody in 1 mo; defer Pr. EP decision Send blood for HIV antibody/antigen assay* Send blood for HIV-1 RNA assay Positive HIV+ VL > 50, 000 copies/m. L Negative HIV- VL < 50, 000 copies/m. L HIV+ Retest VL; defer Pr. EP decision VL < level of detection; no signs/ symptoms on day of blood draw CDC. Pr. EP Guideline. 2014. HIV- VL < level of detection with signs/symptoms on day of blood draw; retest in 1 mo; defer Pr. EP decision
Documenting HIV Status HIV immunoassay blood test (rapid test if available) Blue = HIV-, eligible for Pr. EP Orange = HIV+, not eligible for Pr. EP Green = HIV status unclear; defer Pr. EP decision Indeterminate Negative Positive Consider HIV+ (pending confirmatory testing) Signs/symptoms of acute HIV infection anytime in previous 4 wks HIV- No Yes Option 1 Option 2 Option 3 Retest antibody in 1 mo; defer Pr. EP decision Send blood for HIV antibody/antigen assay* Send blood for HIV-1 RNA assay Positive HIV+ VL > 50, 000 copies/m. L Negative HIV- VL < 50, 000 copies/m. L HIV+ Retest VL; defer Pr. EP decision VL < level of detection; no signs/ symptoms on day of blood draw CDC. Pr. EP Guideline. 2014. HIV- VL < level of detection with signs/symptoms on day of blood draw; retest in 1 mo; defer Pr. EP decision
 Acute HIV Infection • Patients who are candidates for Pr. EP are at substantial risk of HIV infection • Acute HIV infection should be suspected in patients with recent HIV exposure[1] – Signs and symptoms include fever, rash, pharyngitis, lymphadenopathy, myalgia, headache, diarrhea, arthralgia[2] • All Pr. EP candidates with a negative or indeterminate HIV antibody test MUST be asked about symptoms of viral illness in the previous month or on the day of evaluation – Additional confirmatory testing is needed in patients reporting recent signs or symptoms suggestive of acute HIV 1. CDC. Pr. EP Guideline. 2014. 2. Daar ES, et al. Curr Opin HIV AIDS. 2008; 3: 10 -15.
Acute HIV Infection • Patients who are candidates for Pr. EP are at substantial risk of HIV infection • Acute HIV infection should be suspected in patients with recent HIV exposure[1] – Signs and symptoms include fever, rash, pharyngitis, lymphadenopathy, myalgia, headache, diarrhea, arthralgia[2] • All Pr. EP candidates with a negative or indeterminate HIV antibody test MUST be asked about symptoms of viral illness in the previous month or on the day of evaluation – Additional confirmatory testing is needed in patients reporting recent signs or symptoms suggestive of acute HIV 1. CDC. Pr. EP Guideline. 2014. 2. Daar ES, et al. Curr Opin HIV AIDS. 2008; 3: 10 -15.
 Additional Clinical and Laboratory Evaluation • Evaluate for comorbidities that may complicate Pr. EP use, such as renal disease or hepatitis, and drug interactions – Any person with an estimated Cr. Cl of < 60 m. L/min should not be prescribed Pr. EP with TDF/FTC – Document HBV and HCV status before Pr. EP use; vaccinate patients who are HBV susceptible • Optional assessments – Metabolic panel, urinalysis, STI, and pregnancy screening CDC. Pr. EP Guideline. 2014.
Additional Clinical and Laboratory Evaluation • Evaluate for comorbidities that may complicate Pr. EP use, such as renal disease or hepatitis, and drug interactions – Any person with an estimated Cr. Cl of < 60 m. L/min should not be prescribed Pr. EP with TDF/FTC – Document HBV and HCV status before Pr. EP use; vaccinate patients who are HBV susceptible • Optional assessments – Metabolic panel, urinalysis, STI, and pregnancy screening CDC. Pr. EP Guideline. 2014.
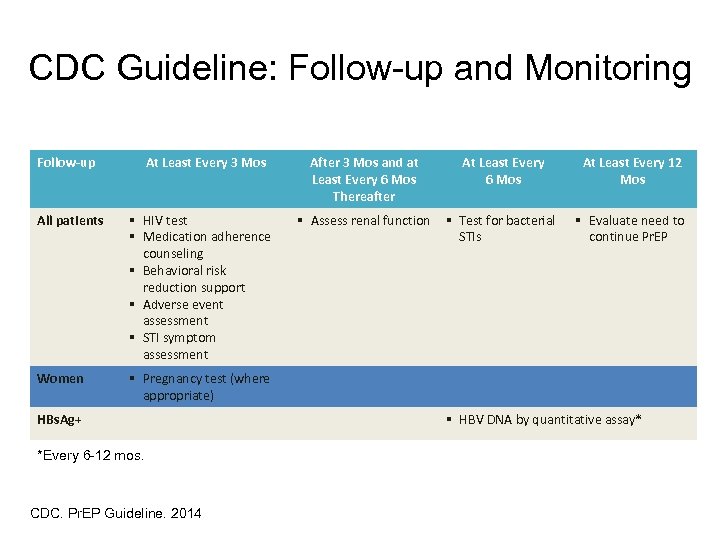 CDC Guideline: Follow-up and Monitoring Follow-up At Least Every 3 Mos All patients § HIV test § Medication adherence counseling § Behavioral risk reduction support § Adverse event assessment § STI symptom assessment Women After 3 Mos and at Least Every 6 Mos Thereafter At Least Every 6 Mos At Least Every 12 Mos § Assess renal function § Test for bacterial STIs § Evaluate need to continue Pr. EP § Pregnancy test (where appropriate) HBs. Ag+ *Every 6 -12 mos. CDC. Pr. EP Guideline. 2014. § HBV DNA by quantitative assay*
CDC Guideline: Follow-up and Monitoring Follow-up At Least Every 3 Mos All patients § HIV test § Medication adherence counseling § Behavioral risk reduction support § Adverse event assessment § STI symptom assessment Women After 3 Mos and at Least Every 6 Mos Thereafter At Least Every 6 Mos At Least Every 12 Mos § Assess renal function § Test for bacterial STIs § Evaluate need to continue Pr. EP § Pregnancy test (where appropriate) HBs. Ag+ *Every 6 -12 mos. CDC. Pr. EP Guideline. 2014. § HBV DNA by quantitative assay*
 Opportunity for Engagement/Education of High Risk Individual § Patient with knowledge of impending risk § Seeks out provider recommendation and/or prescription § Opportunity for education – Risk avoidance techniques – Risks of exposure to disease if non-compliant
Opportunity for Engagement/Education of High Risk Individual § Patient with knowledge of impending risk § Seeks out provider recommendation and/or prescription § Opportunity for education – Risk avoidance techniques – Risks of exposure to disease if non-compliant
![Medication Adherence Counseling • Address adverse events[1] • Identify barriers to adherence[1] • Respond Medication Adherence Counseling • Address adverse events[1] • Identify barriers to adherence[1] • Respond](https://present5.com/presentation/3c756063de6d771ff3e905fea3671056/image-48.jpg) Medication Adherence Counseling • Address adverse events[1] • Identify barriers to adherence[1] • Respond to missed doses in nonjudgemental manner, and stress importance of adherence[1] • Patient self-reporting may not reflect actual adherence[2, 3] 1. CDC. Pr. EP Guideline. 2014. 2. Van Der Straten A, et al. CROI 2014. Abstract 44. 3. Baxi SM, et al. CROI 2014. Abstract 953.
Medication Adherence Counseling • Address adverse events[1] • Identify barriers to adherence[1] • Respond to missed doses in nonjudgemental manner, and stress importance of adherence[1] • Patient self-reporting may not reflect actual adherence[2, 3] 1. CDC. Pr. EP Guideline. 2014. 2. Van Der Straten A, et al. CROI 2014. Abstract 44. 3. Baxi SM, et al. CROI 2014. Abstract 953.
 Stopping Pr. EP • Pr. EP is not meant to be a “permanent” intervention. Pr. EP should be used during periods of high risk. • Reasons to stop Pr. EP: – – – Evidence of HIV infection Adverse events Chronic nonadherence Change in level of risk Patient choice • If restarting Pr. EP after stopping, repeat standard pre. Pr. EP evaluation CDC. Pr. EP Guideline. 2014.
Stopping Pr. EP • Pr. EP is not meant to be a “permanent” intervention. Pr. EP should be used during periods of high risk. • Reasons to stop Pr. EP: – – – Evidence of HIV infection Adverse events Chronic nonadherence Change in level of risk Patient choice • If restarting Pr. EP after stopping, repeat standard pre. Pr. EP evaluation CDC. Pr. EP Guideline. 2014.
 Pr. EP: An Opportunity for Engagement/Education of High Risk Individual • Open discussion about risk behavior • Patient goes through “seasons of risk” which may require different prevention tools • Opportunity for education/STI testing – – Disease avoidance techniques Risks of exposure to disease if non-compliant Presentation of disease if prophylaxis not efficacious Frequent STI testing
Pr. EP: An Opportunity for Engagement/Education of High Risk Individual • Open discussion about risk behavior • Patient goes through “seasons of risk” which may require different prevention tools • Opportunity for education/STI testing – – Disease avoidance techniques Risks of exposure to disease if non-compliant Presentation of disease if prophylaxis not efficacious Frequent STI testing
 Pr. EP in Clinical Practice: What Are the Barriers to Pr. EP Uptake? • Users – Unaware of HIV risk, Pr. EP availability, or how to access it – No or delayed access to clinical preventive care – Lack of knowledge about insurance coverage – Adherence challenges – Concern about disclosure and stigma • Providers – Unaware of intervention – Wary of complexity and time involved – Discomfort with assessing risk – Uncertain how to bill for intervention
Pr. EP in Clinical Practice: What Are the Barriers to Pr. EP Uptake? • Users – Unaware of HIV risk, Pr. EP availability, or how to access it – No or delayed access to clinical preventive care – Lack of knowledge about insurance coverage – Adherence challenges – Concern about disclosure and stigma • Providers – Unaware of intervention – Wary of complexity and time involved – Discomfort with assessing risk – Uncertain how to bill for intervention
 Family AIDS Clinic and Education Services (FACES) Pr. EP Clinic • Weekly Pr. EP Clinic started in December 2014 • Resources – Social worker to help with insurance coverage/enrollment – Nursing – Peer navigators help with adherence counseling • Enrolled Patients – – – Uninfected sexual partners of recently diagnosed HIV patients Uninfected partners in serodiscordant couples wishing to become pregnant High number of missed initial visits 11 Patients started on Pr. EP since December 2014 Referrals from adolescent medicine clinic, internally from HIV clinic and word of mouth.
Family AIDS Clinic and Education Services (FACES) Pr. EP Clinic • Weekly Pr. EP Clinic started in December 2014 • Resources – Social worker to help with insurance coverage/enrollment – Nursing – Peer navigators help with adherence counseling • Enrolled Patients – – – Uninfected sexual partners of recently diagnosed HIV patients Uninfected partners in serodiscordant couples wishing to become pregnant High number of missed initial visits 11 Patients started on Pr. EP since December 2014 Referrals from adolescent medicine clinic, internally from HIV clinic and word of mouth.
 Promotional Card
Promotional Card
 FACES Experience with Pr. EP • Access: – Many new referrals are uninsured. SW works with patient to enroll in health insurance – Health insurance plans cover Pr. EP – Copay assistance offered by manufacturer – Uninsured with income up to 500% above poverty line covered by manufacturer’s Patient Assistance • Next Steps – – Advertising and Promotion of clinic in the community Development of information materials Addition of weekly Pr. EP clinic at Neighborhood House Training and support of primary care providers within NCH
FACES Experience with Pr. EP • Access: – Many new referrals are uninsured. SW works with patient to enroll in health insurance – Health insurance plans cover Pr. EP – Copay assistance offered by manufacturer – Uninsured with income up to 500% above poverty line covered by manufacturer’s Patient Assistance • Next Steps – – Advertising and Promotion of clinic in the community Development of information materials Addition of weekly Pr. EP clinic at Neighborhood House Training and support of primary care providers within NCH
 Educational Materials: Pamphlets Projectinform. org
Educational Materials: Pamphlets Projectinform. org
 NYC DOH Pr. EP Posters
NYC DOH Pr. EP Posters
 Pr. EP in the Future § Rectal/vaginal gels § Availability of Tenofovir Alafenamide (TAF) reduced risk of BMD and renal complications § New dosing protocols for Truvada and other oral Pr. EP formulations § Long-acting injectable Pr. EP (cabotegravir and rilpivirine injectables animal studies and early human studies very promising)!
Pr. EP in the Future § Rectal/vaginal gels § Availability of Tenofovir Alafenamide (TAF) reduced risk of BMD and renal complications § New dosing protocols for Truvada and other oral Pr. EP formulations § Long-acting injectable Pr. EP (cabotegravir and rilpivirine injectables animal studies and early human studies very promising)!
 Recommendations for Pr. EP Implementation • • Coordinated effort to educate potential users Visible marketing campaign needed Need to engage and educate providers Pr. EP works in a continuum with PEP and HIV testing • Increase education and availability of PEP • Promote HIV testing in adherence with CDC guidelines (opt out, universal for all 13 -64, no pre-test counseling)
Recommendations for Pr. EP Implementation • • Coordinated effort to educate potential users Visible marketing campaign needed Need to engage and educate providers Pr. EP works in a continuum with PEP and HIV testing • Increase education and availability of PEP • Promote HIV testing in adherence with CDC guidelines (opt out, universal for all 13 -64, no pre-test counseling)
 Conclusions/Recommendations • Pr. EP Works! • Most efficacious if we coordinate our efforts in the community • We all have a role to play: public health officials, community activists, educators, providers. • Don’t forget other pillars of prevention: – HIV Testing that adheres to CDC/USPTF guidelines – Treatment as Prevention – PEP • Provider education and buy-in is key • Raise awareness in the community to increase uptake and reduce stigma
Conclusions/Recommendations • Pr. EP Works! • Most efficacious if we coordinate our efforts in the community • We all have a role to play: public health officials, community activists, educators, providers. • Don’t forget other pillars of prevention: – HIV Testing that adheres to CDC/USPTF guidelines – Treatment as Prevention – PEP • Provider education and buy-in is key • Raise awareness in the community to increase uptake and reduce stigma



