4e4b6ffbcd9f5205c18bc055e4f30a64.ppt
- Количество слайдов: 30
Practical Blood Bank Lab 11 Cord Blood

Umbilical cord • In placental mammals, the umbilical cord (also called the birth cord or funiculus umbilicalis) is the connecting cord from the developing embryo or fetus to the placenta. • During prenatal development, the umbilical cord is physiologically and genetically part of the fetus and (in humans) normally contains two arteries (the umbilical arteries) and one vein (the umbilical vein), buried within Wharton's jelly. • The umbilical vein supplies the fetus with oxygenated, nutrient-rich blood from the placenta. Conversely, the fetal heart pumps deoxygenated, nutrient-depleted blood through the umbilical arteries back to the placenta.

Wharton's jelly • Wharton's jelly is a gelatinous substance within the umbilical cord, largely made up of mucopolysaccharides (hyaluronic acid and chondroitin sulfate). It also contains some fibroblasts and macrophages. • As a mucous tissue it protects and insulates umbilical blood vessels. • Cells in Wharton's jelly express several stem cell genes, including telomerase. They can be extracted, cultured, and induced to differentiate into mature cell types such as neurons. Wharton's jelly is therefore a potential source of adult stem cells (also see the more common method of storing cord blood).
Cord blood testing • Immediately after the birth of the baby, the umbilical cord is clamped and cut. If cord blood is to be drawn, another clamp is placed 8 to 10 inches away from the first, then the isolated section is cut and a blood sample is collected into a specimen tube. • Cord blood testing is done to determine the following: – – – Bilirubin levels Blood culture (if an infection is suspected) Blood gases, to evaluate the oxygen, carbon dioxide, and p. H levels Blood sugar level Blood type and Rh Complete blood count (CBC)

Abnormal Results Mean • A low p. H (less than 7. 04 to 7. 10) means there are higher levels of acids in the baby's blood. This might occur when the baby does not get enough oxygen during labor. One reason for this could be that the umbilical cord was compressed during labor or delivery. • A blood culture that is positive for bacteria indicates septicemia. • High levels of blood sugar (glucose) in the cord blood may be found if the mother has diabetes. The newborn will be watched for hypoglycemia (low blood sugar) after delivery.

• High levels of bilirubin could be due to: – Infections that the baby gets before birth, including: • • Congenital CMV Congenital hepatitis Congenital rubella Congenital toxoplasmosis – Other possible causes include: • • Dubin-Johnson syndrome Jaundice in the mother Mother taking sulfa drugs during pregnancy Rh incompatibility

Collection of Cord Blood • A cord sample (about 5 to 10 mls) should be taken from the babies of Rh. D negative women, women with known antibodies or in cases where there is insufficient documentation of maternal blood group or antibody status. • The cord sample should be tested for blood group and direct antiglobulin test. • Elution studies may be useful. • Haemoglobin and bilirubin estimation should also be performed if DAT is positive. • When the cord blood sample of the baby of an Rh. D negative woman is Rh. D positive, Rh. D immunoglobulin administration is indicated.

• When the cord blood is Rh. D negative, it is recommended that testing for the presence of the weak Rh. D antigen by the indirect antiglobulin test be performed. If positive, Rh. D immunoglobulin is indicated. If the direct antiglobulin test is positive, it may indicate fetomaternal incompatibility • Antibody elution from the neonatal red cells can be performed to confirm the identity of the antibody coating the cord red blood cells. • When haemolytic disease of the newborn is suspected, Hb, blood group, direct Coombs and bilirubin must be checked. Otherwise cord blood samples are not routinely tested though some collect the samples and store them for up to a week at 4°C in case they should be needed.

• When the direct Coombs is positive, Rh typing must be confirmed and the coating antibody identified. • The initial Rh typing will sometimes be found to be incorrect when the direct Coombs is positive, due to interference by the antibody.

Considerations • Most hospitals routinely collect cord blood for testing at birth, since it is relatively convenient and the only time possible to collect such blood. Besides cord blood testing, cord blood can be used to treat certain types of bone marrow-related cancers. Some parents may choose to save (bank) their child's cord blood for this and other, future medical purposes. • Cord blood banking for personal use is done by private companies, which charge for the service. Cord blood can also be donated to your local blood bank for use by others as needed to treat leukemia and other cancers.

Cord Blood Banking • Cord blood is rich in potentially life-saving stem cells. When they are transplanted, those stem cells are effective for the treatment of patients with diseases of the immune system (e. g. , immunodeficiencies), blood (e. g. , leukemia and sickle cell anemia) and selected metabolic disorders. • The umbilical cord, placenta, and the blood they contain have no function after birth and they end up in waste. If exploited, this resource can be used for the good of the whole community. • Stem cells in the cord blood similar to those found in bone marrow. This is why cord blood can be used for transplantation as an alternative to bone marrow.

• Stem-cell transplants have advantages over bone marrow transplants in the treatment, with a lower risk of tissue rejection (Gv. HD). The collection process is noninvasive, and involves a simple blood transfusion. • As per the commitment of the Islamic University to serve the Palestinian community, this project is to set up the first National Umbilical Cord Blood Bank in Palestine.

Qualification of Cord Blood Units for the NYBC's National Cord Blood Program Inventory. • A cord blood unit must meet all of the criteria (at a minimum) below to qualify for inclusion in the NCBP Search Inventory and be available for transplantation: 1. The unit contains 1. 1 billion total nucleated cells (TNC) or more before processing. 2. The mother has given consent for donation 3. The unit has been typed for HLA-A, -B and –DRB 1 (A and B at low resolution and DRB 1 at high resolution) and for the blood group antigens A, B and Rh: 1 (D); 4. The unit had acceptable results for CD 34+ cells and CFU (colony forming units). Note: CFU evaluation has not been performed in all units;

5. 6. 7. 8. 9. There is no bacterial contamination of the unit; The infant has no identified risks of genetic or infectious disease that might be transmitted in the cord blood; The unit and the mother’s blood are negative for infectious disease markers; The cord blood unit was processed and frozen following the NCBP protocol validated to maintain nucleated cell viability. A sample of cord blood is available in a segment of the tubing integral to the bag containing the cord blood unit, for future confirmatory HLA typing.

What is cord blood used for? • The most common disease category has been leukemia. The next largest group is inherited diseases (of red blood cells, the immune system and certain metabolic abnormalities. ) • Patients with lymphoma, myelodysplasia and severe aplastic anemia have also been successfully transplanted with cord blood.
How long do cord blood cells remain viable? • It is not known how long cord blood can be stored in liquid nitrogen and its cells remain viable. • NCBP's earliest units were stored in 1993. In checking the viability of cells in cord blood units that will not be used for transplantation, they did not detect any deterioration in the quality of the cells in cord blood units stored for up to 16 years. • Units stored for up to 13 years have been used in transplants and the outcomes have been similar to those of newly collected units.

Checking Donor's and Blood Tests? • Normal procedure includes – Asking the mother and father for their health related history – Blood tests to rule out • HIV infections, other blood diseases human immunodeficiency virus (HIV, the cause of AIDS), • hepatitis B and C viruses (HBV and HCV), • human T-lymphotropic virus (HTVL), • syphilis, • West Nile Virus (WNV) • and Chagas disease (a disease common in South America). • test the baby's blood for Cytomegalovirus (CMV) • and for hemoglobin abnormalities such as sickle cell disease. . – Signing of a consent form.

Methodology • Collection • Processing • Storage

How is cord blood collected? • The cord blood collection process is simple, safe, and painless. It is usually completed in less than five minutes by your health care provider. Cord blood collection does not interfere with delivery and is possible with vaginal or cesarean deliveries. Your health care provider will use one of two options for cord blood collection: syringe method or bag method. – Syringe method: a syringe is used to draw blood from the umbilical cord shortly after the umbilical cord has been cut. The process is basically the same as drawing blood for a blood test. – Bag method: the umbilical cord is elevated to cause the blood to drain into a bag. • The syringe or bag should be pre-labeled with a unique number that represents your baby. Cord blood may only be collected during the first 15 minutes following the birth, and should be processed by the laboratory within 48 hours.

• The Cord Blood Bank is responsible for collection of the blood. • However, the related authorities (delivery doctors, nurses) must be aware of the mothers’ wish to donate or store Cord Blood after the birth. • Minutes after a child’s birth and after the umbilical cord is cut, the placenta and its attached portion of the cord are delivered With maternal assent to collect the cord blood, • staff move the placenta to an adjacent laboratory, where they collect the cord blood into a sterile plastic bag, much like those used for blood transfusion. • Because they collect cord blood from the delivered placenta, no risk is incurred by mother or child. • explains all aspects of the Program to the mother and requests her consent to donate the cord blood. Donation of the blood from a single placenta becomes a unique cord blood unit. • The volume collected varies but usually ranges from 50 to 200 ml (about one-half to one cup).
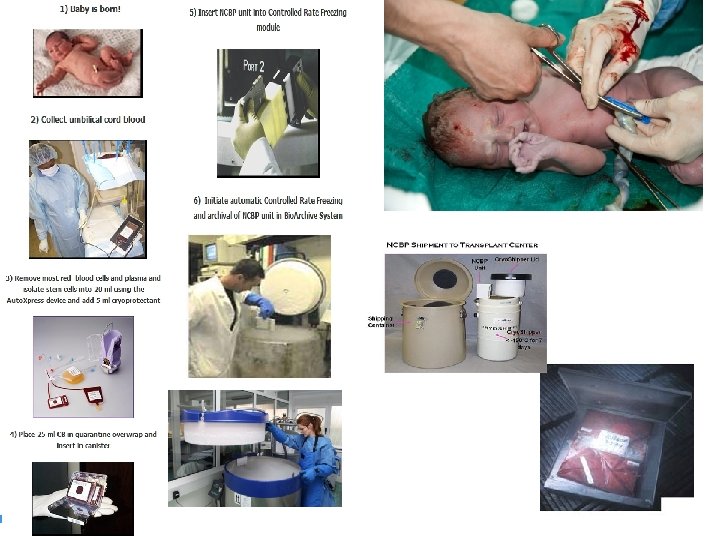

Processing: • to remove excess red blood cells (RBCs) and plasma and bring each collection down to a uniform single unit volume of 20 ml. 1. Upon arrival in the Processing Laboratory, cord blood units are "logged-in" to the database and weighed to estimate the collected blood volume. 2. The cord blood is transferred using a sterile-docking device, from the collection bag into a sterile, disposable processing bag set in preparation for its automated processing using the AXP (Auto. Xpress) platform (510 k-approved by FDA). 3. Before and after processing, small samples are removed to count the total nucleated cells (TNC) and the number of nucleated cells that are positive for CD 34. These samples are removed within plastic bubbles (pre-set in the tubing), sealed off from the processing bag set without exposing the products to possible infectious agents in the air of the laboratory.

4. 5. 6. The bag set containing the cord blood unit is placed into the computerized AXP device, which is automatically centrifuged twice. The first centrifugation stratifies the cellular components [RBCs, white blood cells (WBC) and platelets] separated from the plasma. The second spin harvests the blood components into the three different bags of the AXP sterile bag set: RBC bag, freezing bag [containing the WBCs, predominantly the mononuclear cells (MNCs)] and plasma bag. The freezing bag contains the 20 ml final product (on average, more than 90% of the original MNCs and stem cells in some of the plasma, along with some red cells). Cold, sterile, Dimethylsulfoxide (DMSO, 5 ml of a 50% solution) is then added slowly. DMSO is a kind of "anti-freeze" or cryoprotectant that prevents ice crystals from forming inside of and destroying cells during freezing. It is also protective when the unit is eventually thawed.

Storage: • The cord blood is frozen and stored in liquid nitrogen freezers designed especially for cord blood units. • The Bio. Archive is a robotic freezer that controls and monitors the rate of freezing, then stores the unit in liquid nitrogen and provides quick access to stored units • whenever needed, all under computer control and with complete documentation. • When a transplant center requests a matched cord blood unit for a patient, the available inventory of cord blood units is “searched” for matches to the patient. The best matching unit (or units) can be selected, reported, confirmed and delivered rapidly upon request.

Freezing: • Each cord blood unit is frozen (one at a time) in an individual “controlled-rate freezer” module within the Bio. Archive™ system. We use a freezing rate documented to maintain cell viability. A record of the freezing curve is printed and maintained in the Bio. Archive™ computer memory.
Testing of cord blood units: 1. Typing for HLA-A, -B and -DRB 1. Typing is performed at low intermediate resolution for HLA-A and -B and at high resolution (sequence based typing) for HLA-DRB 1. In addition, before releasing a cord blood unit for transplantation, confirmatory HLA typing is performed on cells from a continuous segment for HLAA, -B, -C at low intermediate resolution and HLA-DRB 1 at high resolution. 2. Total nucleated cell enumeration is performed on all cord blood units before and after processing. Since mid-2000, cell counts have been done with an automated hematology analyzer (Sysmex XE-2100), that includes a count of nucleated red blood cells. The total nucleated cell count (TNC) of a unit includes all nucleated cells: white blood cells and nucleated red cells.

3. CD 34+ cell number has been assessed on all units since February 2000. Currently, CD 34+ cell count and viability are assessed in all cord blood units by flow cytometry before and after cord blood processing. 4. Colony forming unit (CFU) assay. , this assay is very reproducible and does not depend on the operator (laboratory technician). In addition, images of CFU from each cord blood unit are stored electronically and can be reviewed at later times. 5. Bacterial culture (of the unit after processing) for both aerobic and anaerobic organisms. using the ESP Culture System (Versa TREK). The system screens a sample of the cord blood unit for bacterial infection, and if a positive signal is detected, the sample is sent to a Microbiology Reference Laboratory for bacterial identification and antibiotic sensitivity. 6. Infectious disease testing of both the mother's blood and cord. Testing includes antibodies to HIV-1 and 2 (with a test that detects antibodies to HIV-O also), HTLV-1 and 2, HCV, HBV (anti-HBc), syphilis and hepatitis B virus surface antigen (HBs. Ag). Since 2008, mother’s blood is also tested for antibodies to Chagas’ disease.

7. Nucleic Acid Testing (NAT) for HIV, HCV and West Nile Virus are done on the maternal blood sample only 8. Cytomegalovirus (CMV) testing. Mother’s and cord blood samples are tested for total antibody to CMV, indicating past or recent exposure to the virus. In addition, perform CMV culture of the baby’s saliva. test the cord blood sample by PCR amplification of the viral DNA, in all cases that the mother’s antibody testing is positive for CMV. 9. ABO blood group and Rh type 10. Hemoglobinopathies screening. Sickle cell hemoglobin and hemoglobins C and E are detected by high performance liquid chromatography (HPLC) 11. Stored Samples. Aliquoted samples of plasma, viable white cells and DNA from the cord blood unit and from the mother's blood are kept frozen and are available for special testing in the future, as needed.

Finding a Match • Several studies indicate that cord blood transplants do not need to be as perfectly HLA-matched to the recipient as bone marrow transplants do. However, patients have a better chance to survive and regain health if the cord blood graft is better matched (6/6 or 5/6) and provides cell doses of 20 million nucleated cells per kg or more. • Cord Blood Unit Match Reports: An individual Match Report gives detailed information about the cord blood unit, including number of total nucleated and CD 34+ cells, ABO and Rh blood groups and results of other laboratory tests. • The match referred to is required between the donor’s and recipient’s HLA antigens. The closer the match is, the better is the body’s chance of accepting the new stem cell.

Samples are saved and may be used for further testing in the future. • Cord and mother’s blood samples are kept, frozen, and may be used in the future, even years after the baby’s birth. We must plan for the possibility that new tests for infectious or genetic diseases will be developed for which the cord blood must be tested in order to protect a transplant recipient from risk.
4e4b6ffbcd9f5205c18bc055e4f30a64.ppt