d3618b821e3e8500ed8df23ac79d639f.ppt
- Количество слайдов: 72

POSTER INTRODUCTION CREL Annual Meeting October 28, 2004 Chemical Reaction Engineering Laboratory Department of Chemical Engineering St. Louis, MO 63130 CHEMICAL REACTION ENGINEERING LABORATORY

(SLURRY) BUBBLE COLUMN AND GAS-LIQUID STIRRED TANK REACTORS A. Experimental Techniques and Measurements

Ashfaq Shaikh Bubble Column Reactors Hydrodynamics Flow Regime Transition Scale-up Hydrodynamics ü Hydrodynamics of High Pressure Bubble Column Slurry Reactor ü Combination of two single modal tomographic techniques for three dynamic phase flow imaging Flow Regime Transition ü Evaluation of CT for regime identification ü New technique and its ‘flow regime identifiers’ developed Scale-up A new hypothesis proposed ü Experimental evaluation of proposed hypothesis ü Development of ANN correlations for hydrodynamic parameters

Characterization of Hydrodynamic Flow regime in Bubble Column via Computed Tomography Homogeneous/Bubbly Flow Heterogeneous/Churnturbulent Flow Different hydrodynamic characteristics ü Explored the potential of CT for flow regime delineation in bubble column ü Evaluated the developed approach with traditional methods such as Drift Flux method ü Investigated the effect of operating pressure on flow regime transition

Mass Transfer Measurement Techniques for Slurry Bubble Column Reactors Lu Han, Muthanna Al-Dahhan. CREL, Oct. 2004 Slurry Bubble Column Reactors • • • Vertical cylindrical vessels, three-phase gas-liquid-solid systems with solid particle sizes in the range 5 -150µm and solids loading up to 50% by volume Simple to construct and do not involve any mechanically moving parts Exhibit excellent heat and mass transfer characteristics Applications: – Fischer-Tropsch (FT) Synthesis – oxidation and hydrogenation – chlorination and alkylation – polymerization, methanol synthesis – waste water treatment – bio and biochemical processes G – Reactant L – Reactant and/or Product S – Catalyst G L+S G The goal of this work is to measure the gas-liquid volumetric mass transfer coefficient, k. La, in SBC with high gas velocity/pressure/solid loading, with assistance of hydrodynamic information obtained using CARPT/CT methodology.
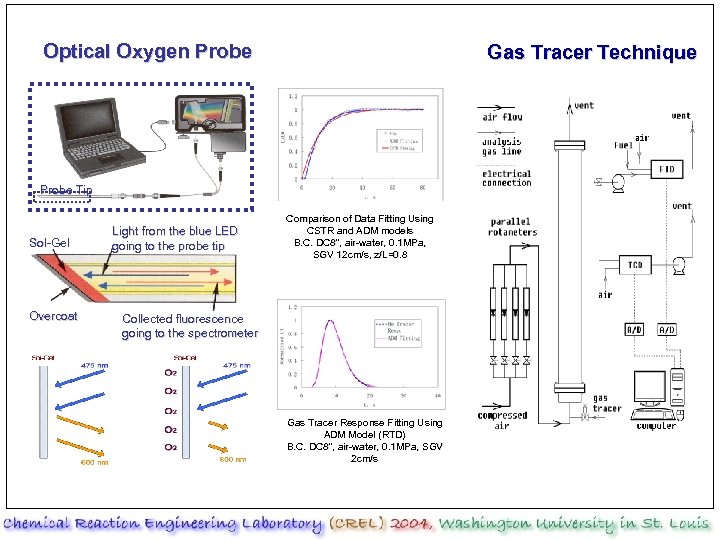
Optical Oxygen Probe Gas Tracer Technique Probe Tip Sol-Gel Overcoat Light from the blue LED going to the probe tip Comparison of Data Fitting Using CSTR and ADM models B. C. DC 8”, air-water, 0. 1 MPa, SGV 12 cm/s, z/L=0. 8 Collected fluorescence going to the spectrometer Gas Tracer Response Fitting Using ADM Model (RTD) B. C. DC 8”, air-water, 0. 1 MPa, SGV 2 cm/s

A Novel Modeling Approach for Predictions of the Dynamic Growth of Microalgae in Multiphase Photo-bioreactors Flows Dynamics in An Internal Loop Airlift Column Bioreactor Producing And Carbonylating of Dimethyl Carbonate: A Process Development Study Hu-Ping Luo, Muthanna H. Al-Dahhan Chemical Reaction Engineering Laboratory (CREL) Bioprocess & Bioreactor Engineering Laboratory (BBEL) Chemical Engineering Department Washington University in St. Louis CREL Annual Meeting October 2004 CHEMICAL REACTION ENGINEERING LABORATORY
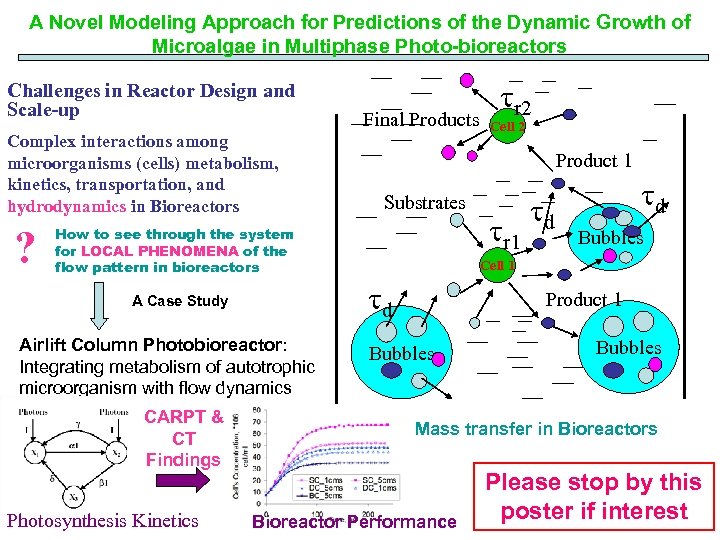
A Novel Modeling Approach for Predictions of the Dynamic Growth of Microalgae in Multiphase Photo-bioreactors Challenges in Reactor Design and Scale-up Complex interactions among microorganisms (cells) metabolism, kinetics, transportation, and hydrodynamics in Bioreactors ? How to see through the system for LOCAL PHENOMENA of the flow pattern in bioreactors A Case Study Airlift Column Photobioreactor: Integrating metabolism of autotrophic microorganism with flow dynamics CARPT & CT Findings Photosynthesis Kinetics Final Products tr 2 Cell 2 Product 1 Substrates tr 1 td td Bubbles Cell 1 td Product 1 Bubbles Mass transfer in Bioreactors Bioreactor Performance Please stop by this poster if interest
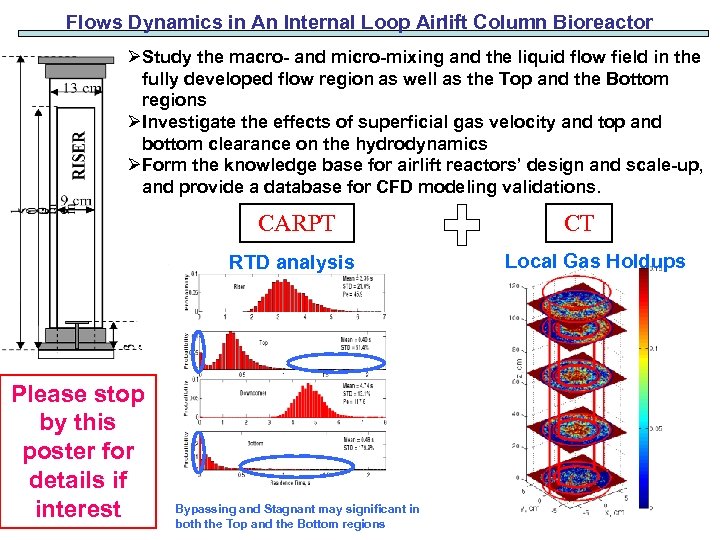
Flows Dynamics in An Internal Loop Airlift Column Bioreactor ØStudy the macro- and micro-mixing and the liquid flow field in the fully developed flow region as well as the Top and the Bottom regions ØInvestigate the effects of superficial gas velocity and top and bottom clearance on the hydrodynamics ØForm the knowledge base for airlift reactors’ design and scale-up, and provide a database for CFD modeling validations. CARPT RTD analysis Please stop by this poster for details if interest Bypassing and Stagnant may significant in both the Top and the Bottom regions CT Local Gas Holdups
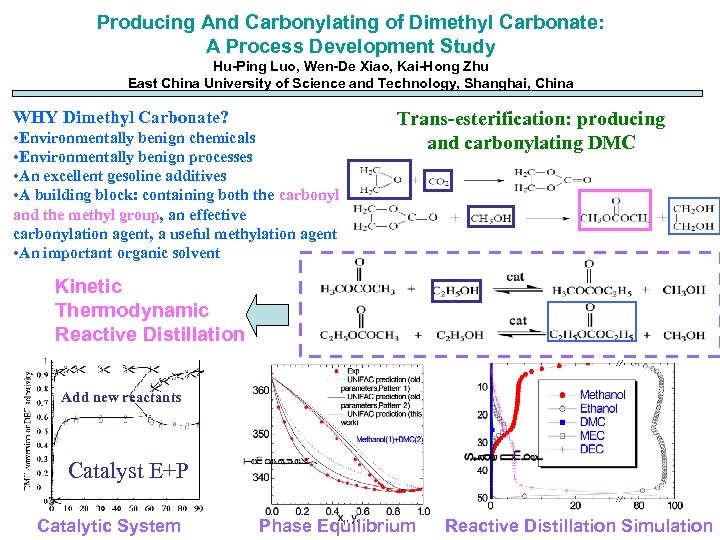
Producing And Carbonylating of Dimethyl Carbonate: A Process Development Study Hu-Ping Luo, Wen-De Xiao, Kai-Hong Zhu East China University of Science and Technology, Shanghai, China WHY Dimethyl Carbonate? • Environmentally benign chemicals • Environmentally benign processes • An excellent gesoline additives • A building block: containing both the carbonyl and the methyl group, an effective carbonylation agent, a useful methylation agent • An important organic solvent Trans-esterification: producing and carbonylating DMC Kinetic Thermodynamic Reactive Distillation Add new reactants Catalyst E+P Catalytic System Phase Equilibrium Reactive Distillation Simulation

Heat Transfer Coefficient Measurement Technique in High Pressure Slurry Bubble Column Chengtian Wu, Muthanna Al-Dahhan • The instantaneous heat transfer coefficient(hi) can be obtained from the heat transfer flux(Q) and temperature difference between the probe surface(Ts) and the bulk(Tb). – The probe measures the instantaneous local heat flux(Q) and the surface temperature(Ts). – Three thermocouples are used to measure the bulk temperature(Tb). CHEMICAL REACTION ENGINEERING LABORATORY
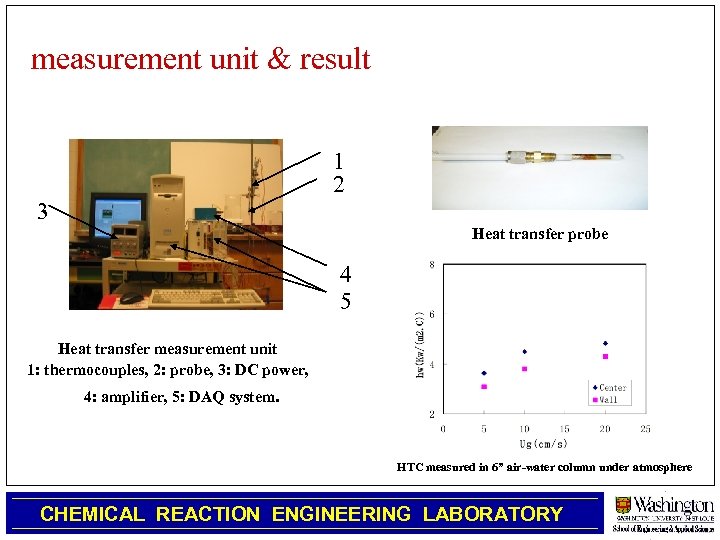
measurement unit & result 1 2 3 Heat transfer probe 4 5 Heat transfer measurement unit 1: thermocouples, 2: probe, 3: DC power, 4: amplifier, 5: DAQ system. HTC measured in 6” air-water column under atmosphere CHEMICAL REACTION ENGINEERING LABORATORY

Bubble Velocity, Chord Length and Specific Interfacial Area Measurements in Bubble Columns Using Four-point Optical Probe Junli Xue, M. H. Al-Dahhan, M. P. Dudukovic, R. F. Mudde The performance of (slurry) bubble columns is governed dfiber=0. 2 mm by the hydrodynamics. Validation of Computational Fluid Dynamics (CFD) codes requires also local information on bubble properties. The measurement of bubble properties is difficult, especially in churn-turbulent flow. A four-point optical probe is employed in this study to measure the bubble properties. Cofiguration of the Four-Point Optical Probe CHEMICAL REACTION ENGINEERING LABORATORY

Four-Point Optical Probe Measurements in a 16. 2 cm (6. 4”) Bubble Column: The operating conditions span from bubbly flow to churn-turbulent flow. Superficail gas velocity: 2~60 cm/s Pressure: 1~10 bar The probe was positioned both upwards and downwards. So both bubbles moving upwards and downwards are measured. CHEMICAL REACTION ENGINEERING LABORATORY

3 D VIEW OF BIAZZI HYDROGENATION REACTOR CONFIDENTIAL References: Projects realized: • 41 plants built • 16 of which c. GMP • Maximum 110 bar and 300°C Customers: • Fine Chemicals • Pharmaceuticals • Resins and Intermediaries • Speciality sugars • Edible oils Operation modes: • Continuous • Dedicated c. GMP and regular • Multipurpose c. GMP and regular Countries: • Europe: Italy, Belgium, Austria, Switzerland, Netherlands, Germany, England, Spain, France, • Americas: Brasil, USA, • Asia: South Korea, India, Japan, Taiwan R. O. C. , China, Russia 25, Ch de la Tavallaz, CH-1816 Chailly s/Montreux, Switzerland - Tel. : +41 21 989 2121 - Telefax: +41 21 989 2120 - www. biazzi. com
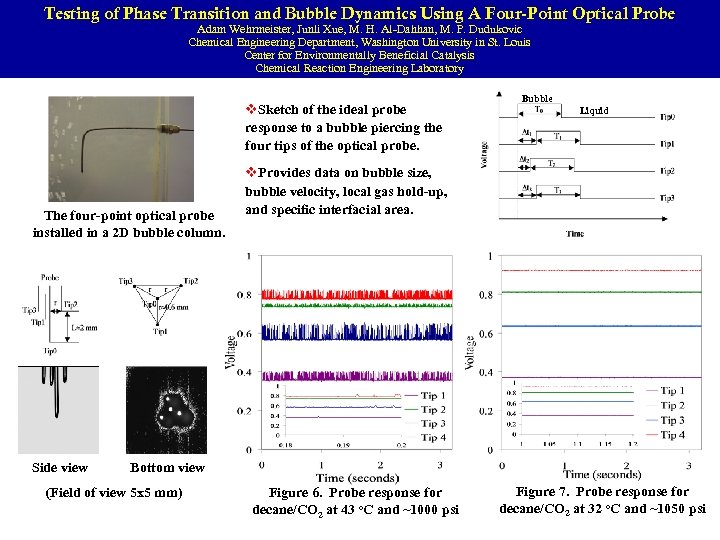
Testing of Phase Transition and Bubble Dynamics Using A Four-Point Optical Probe Adam Wehrmeister, Junli Xue, M. H. Al-Dahhan, M. P. Dudukovic Chemical Engineering Department, Washington University in St. Louis Center for Environmentally Beneficial Catalysis Chemical Reaction Engineering Laboratory v. Sketch of the ideal probe response to a bubble piercing the four tips of the optical probe. The four-point optical probe installed in a 2 D bubble column. Side view Bubble Liquid v. Provides data on bubble size, bubble velocity, local gas hold-up, and specific interfacial area. Bottom view (Field of view 5 x 5 mm) Figure 6. Probe response for decane/CO 2 at 43 o. C and ~1000 psi Figure 7. Probe response for decane/CO 2 at 32 o. C and ~1050 psi

(SLURRY) BUBBLE COLUMN AND GAS-LIQUID STIRRED TANK REACTORS B. Modeling and Computational Fluid Dynamics (CFD)

Predicting Gas Holdup, Liquid Velocity Profiles and Mixing in Bubble Column Flows Accounting for Coalescence-Breakup Peng Chen and M. P. Dudukovic CREL Meeting, 2004 CHEMICAL REACTION ENGINEERING LABORATORY
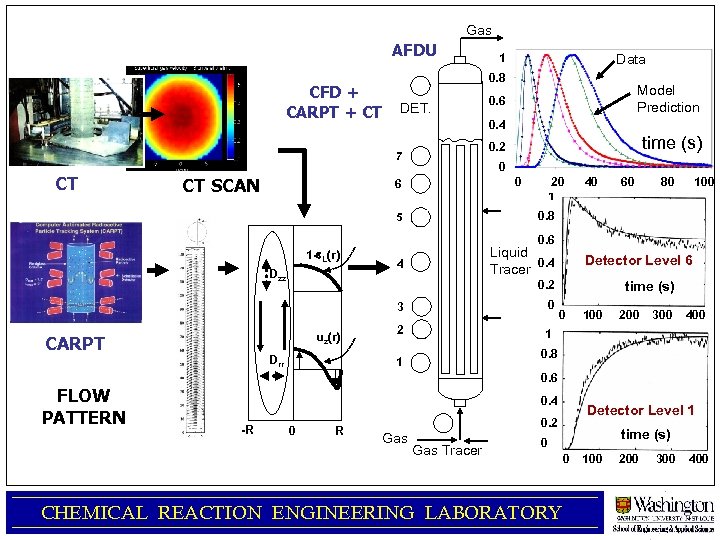
Gas AFDU CFD + CARPT + CT CT SCAN Data 0. 8 DET. Model Prediction 0. 6 0. 4 time (s) 0. 2 7 CT 1 0 0 6 20 1 40 60 80 100 0. 8 5 0. 6 1 -e. L(r) Dzz Liquid Tracer 0. 4 4 0. 2 0 3 uz(r) CARPT Detector Level 6 Drr 2 time (s) 0 100 200 300 400 1 0. 8 1 0. 6 FLOW PATTERN 0. 4 -R 0 R Detector Level 1 0. 2 Gas Tracer time (s) 0 0 CHEMICAL REACTION ENGINEERING LABORATORY 100 200 300 400
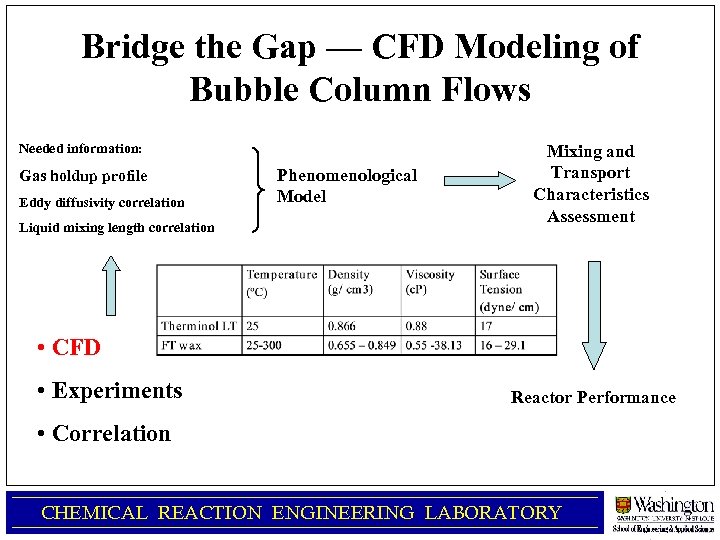
Bridge the Gap — CFD Modeling of Bubble Column Flows Needed information: Gas holdup profile Eddy diffusivity correlation Liquid mixing length correlation Phenomenological Model Mixing and Transport Characteristics Assessment • CFD • Experiments Reactor Performance • Correlation CHEMICAL REACTION ENGINEERING LABORATORY

Computational Modeling of Gas-Liquid Flow in Bubble Columns P. Chen, M. Rafique and M. P. Dudukovic Outlines • Hydrodynamics of bubble columns • Eulerian-Eulerian Two-Fluid model • Algebraic Slip Mixture Model (ASMM) • Hydrodynamics of (passive) tracers (gas/liquid) in bubble column flows

CFD-based Compartmental Modeling of Single Phase Stirred Tank Reactors Debangshu Guha, M. P. Dudukovic & P. A. Ramachandran CREL Annual Meeting, 2004

Motivation n Reactor Performance = f (kinetics, flow pattern and mixing) n Mixing = f (flow pattern and turbulence characteristics) n Most available phenomenological models for mixing do not account for the flow pattern and the turbulence inhomogeneities in the reactor n The performance prediction can be improved if flows and turbulence characteristics can be used from CFD
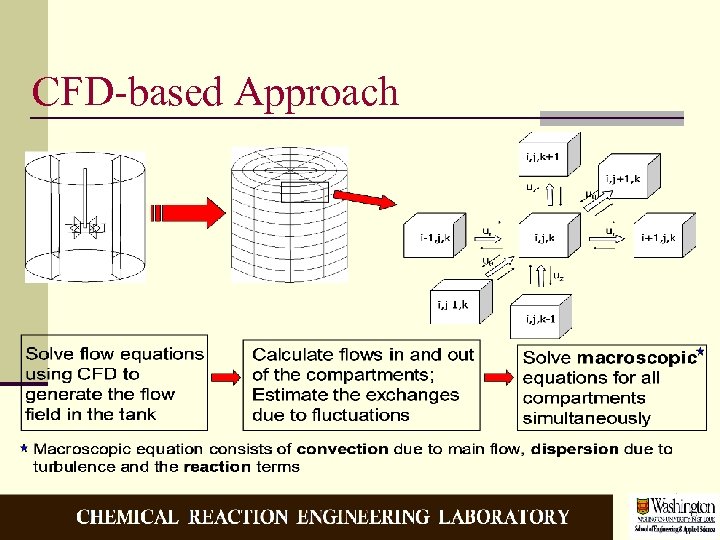
CFD-based Approach
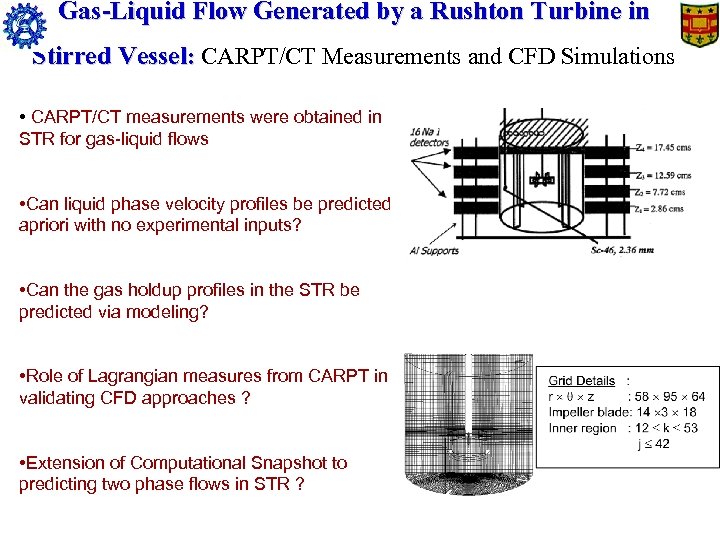
Gas-Liquid Flow Generated by a Rushton Turbine in Stirred Vessel: CARPT/CT Measurements and CFD Simulations • CARPT/CT measurements were obtained in STR for gas-liquid flows • Can liquid phase velocity profiles be predicted apriori with no experimental inputs? • Can the gas holdup profiles in the STR be predicted via modeling? • Role of Lagrangian measures from CARPT in validating CFD approaches ? • Extension of Computational Snapshot to predicting two phase flows in STR ?

PACKED BED, STRUCTURED BED, CRICULATING FLUIDIZED BED

Poster 1 Solids Flow Mapping in a Fast Fluid Bed Satish Bhusarapu, M. H. Al-Dahhan and M. P. Duduković CREL Annual Meeting October 28, 2004 Chemical Reaction Engineering Laboratory Department of Chemical Engineering St. Louis, MO 63130 CHEMICAL REACTION ENGINEERING LABORATORY
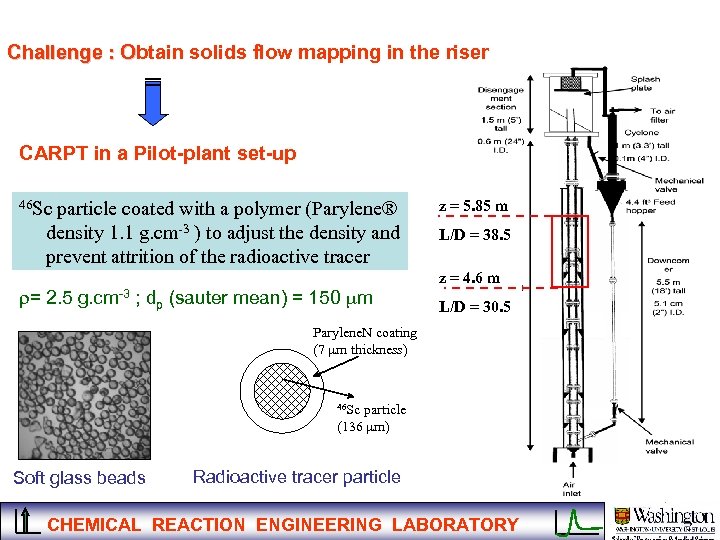
Challenge : Obtain solids flow mapping in the riser O CARPT in a Pilot-plant set-up 46 Sc particle coated with a polymer (Parylene® density 1. 1 g. cm-3 ) to adjust the density and prevent attrition of the radioactive tracer r= 2. 5 g. cm-3 ; dp (sauter mean) = 150 mm z = 5. 85 m L/D = 38. 5 z = 4. 6 m L/D = 30. 5 Parylene. N coating (7 mm thickness) 46 Sc particle (136 mm) Soft glass beads Radioactive tracer particle CHEMICAL REACTION ENGINEERING LABORATORY

Poster 2 Solids RTD in a Gas-Solid Riser at Low and High Fluxes: Single Radioactive Particle Tracking Satish Bhusarapu, M. H. Al-Dahhan and M. P. Duduković CREL Annual Meeting October 28, 2004 Chemical Reaction Engineering Laboratory Department of Chemical Engineering St. Louis, MO 63130 CHEMICAL REACTION ENGINEERING LABORATORY
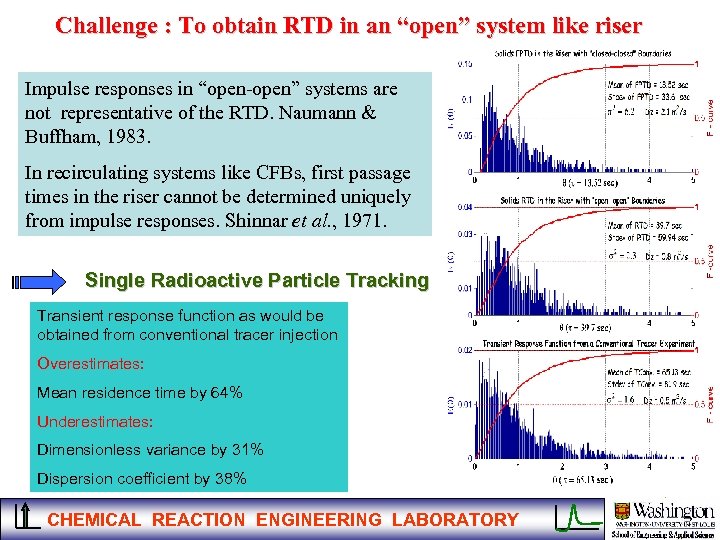
Challenge : To obtain RTD in an “open” system like riser Impulse responses in “open-open” systems are not representative of the RTD. Naumann & Buffham, 1983. In recirculating systems like CFBs, first passage times in the riser cannot be determined uniquely from impulse responses. Shinnar et al. , 1971. Single Radioactive Particle Tracking Transient response function as would be obtained from conventional tracer injection Overestimates: Mean residence time by 64% Underestimates: Dimensionless variance by 31% Dispersion coefficient by 38% CHEMICAL REACTION ENGINEERING LABORATORY

Poster 3 An Alternating Minimization Algorithm for Image Reconstruction in Computed Tomography Satish Bhusarapu, M. H. Al-Dahhan and M. P. Duduković CREL Annual Meeting October 28, 2004 Chemical Reaction Engineering Laboratory Department of Chemical Engineering St. Louis, MO 63130 CHEMICAL REACTION ENGINEERING LABORATORY

Challenge: To improve image quality of the CT data Beer Lambert’s Law - Estimation - Maximization An approximation is made in the solution which is true only for low attenuation values Phase holdup profiles at various axial positions Implement an Alternating Minimization (AM) algorithm (O’Sullivan and Benac, 2001), where each step of minimization is exact. CHEMICAL REACTION ENGINEERING LABORATORY

Trickle Bed Reactors Maxime Capitaine M. P. Dudukovic, M. H. Al-Dahhan Chemical Reaction Engineering Laboratory (CREL) Washington University in St. Louis, MO J. Bousquet, D. Védrine, P. Tanguy Centre Européen de Recherche et Technique, TOTAL Harfleur, FRANCE

Hydrodynamics Parameters • Liquid Distribution • Pressure Drop • Liquid Hold Up Measurement Methods • Collector Tray • Computed Tomography Results • Effects of liquid and gas superficial velocities and packed bed height

Cell Network Modeling For Catalytic Trickle-Bed Reactors J. Guo, Y. Jiang, P. A. Ramachandran, M. Al-Dahhan, M. P. Dudukovic Washington University St. Louis, Missouri CREL Annual Meeting 10. 28. 2004 CHEMICAL REACTION ENGINEERING LABORATORY
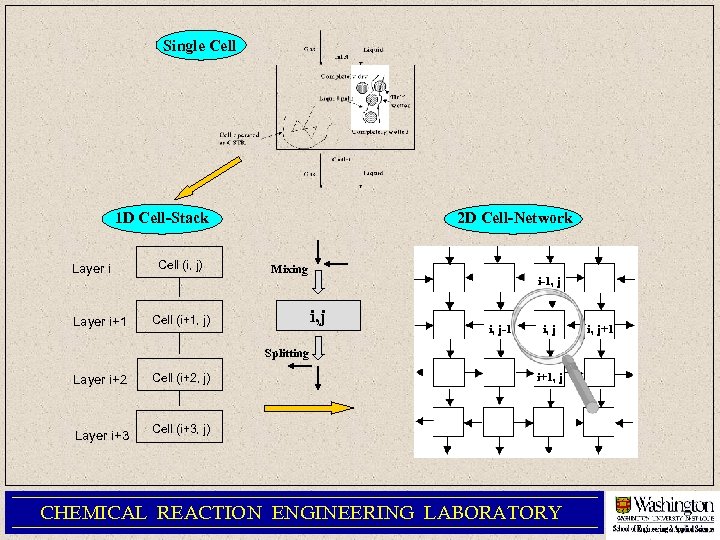
Single Cell 1 D Cell-Stack Layer i+1 Cell (i, j) 2 D Cell-Network Mixing i-1, j i, j Cell (i+1, j) i, j-1 i, j Splitting Layer i+2 Layer i+3 Cell (i+2, j) i+1, j Cell (i+3, j) CHEMICAL REACTION ENGINEERING LABORATORY i, j+1

Dynamics of Coupling Exothermic & Endothermic Reactions in Directly Coupled Adiabatic Reactors R C Ramaswamy Advisors P A Ramachandran, M P Duduković CREL Annual Meeting Fall, 2004 CHEMICAL REACTION ENGINEERING LABORATORY
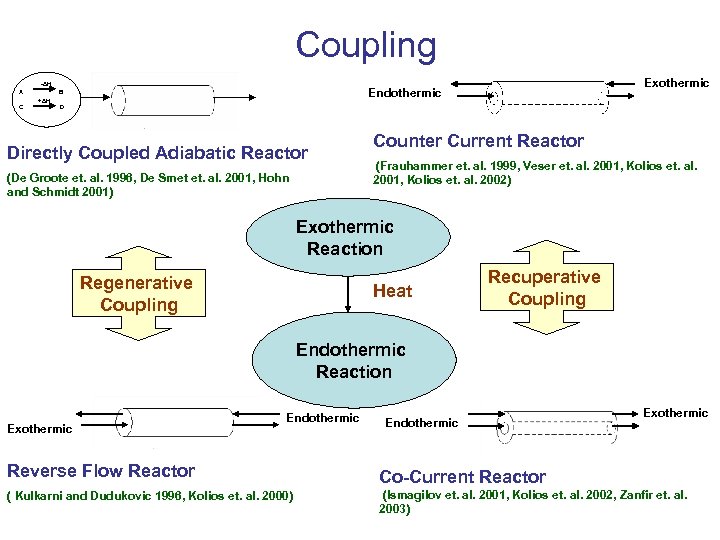
Coupling -ΔH A C +ΔH Exothermic Endothermic B D Directly Coupled Adiabatic Reactor (De Groote et. al. 1996, De Smet et. al. 2001, Hohn and Schmidt 2001) Counter Current Reactor (Frauhammer et. al. 1999, Veser et. al. 2001, Kolios et. al. 2002) Exothermic Reaction Regenerative Coupling Heat Recuperative Coupling Endothermic Reaction Exothermic Endothermic Reverse Flow Reactor ( Kulkarni and Dudukovic 1996, Kolios et. al. 2000) Endothermic Exothermic Co-Current Reactor (Ismagilov et. al. 2001, Kolios et. al. 2002, Zanfir et. al. 2003)
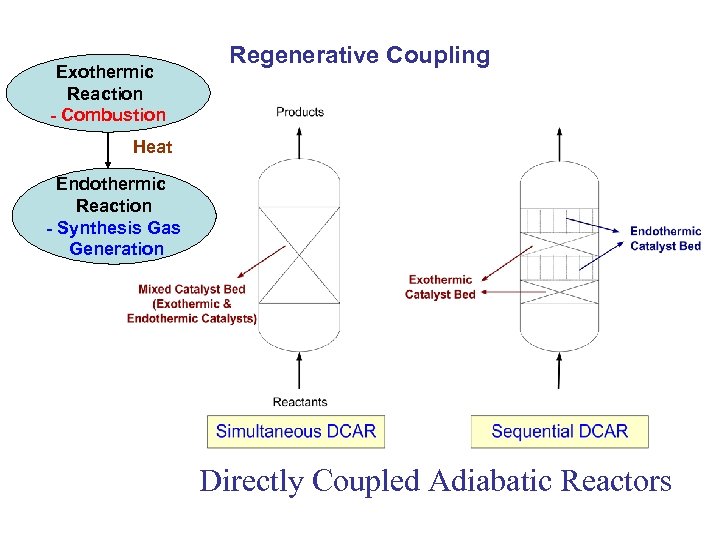
Exothermic Reaction - Combustion Regenerative Coupling Heat Endothermic Reaction - Synthesis Gas Generation Directly Coupled Adiabatic Reactors
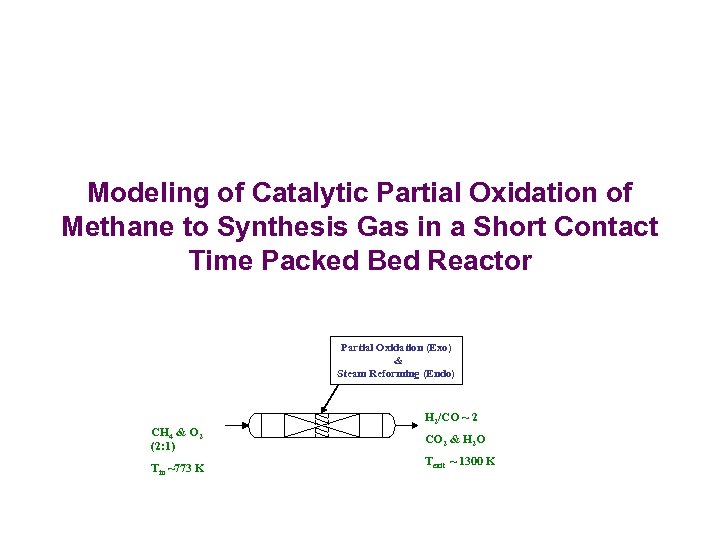
Modeling of Catalytic Partial Oxidation of Methane to Synthesis Gas in a Short Contact Time Packed Bed Reactor Partial Oxidation (Exo) & Steam Reforming (Endo) CH 4 & O 2 (2: 1) Tin ~773 K H 2/CO ~ 2 CO 2 & H 2 O Texit ~ 1300 K

Synthesis Gas (mixture of H 2 and CO) (Pena et. al. 1996) – Feed stock for synthesis of liquid fuels, methanol – Source of hydrogen for fuel cells – Feed stock for ammonia plant, hydrogenation plant etc Catalytic Partial Oxidation of Methane to Syngas (De Smet et. al. , CES 56 , 2001) High Active Catalysts (Rh) Short Contact Time Reactors (4 -15 milli seconds) Hohn & Schmidt, 2001
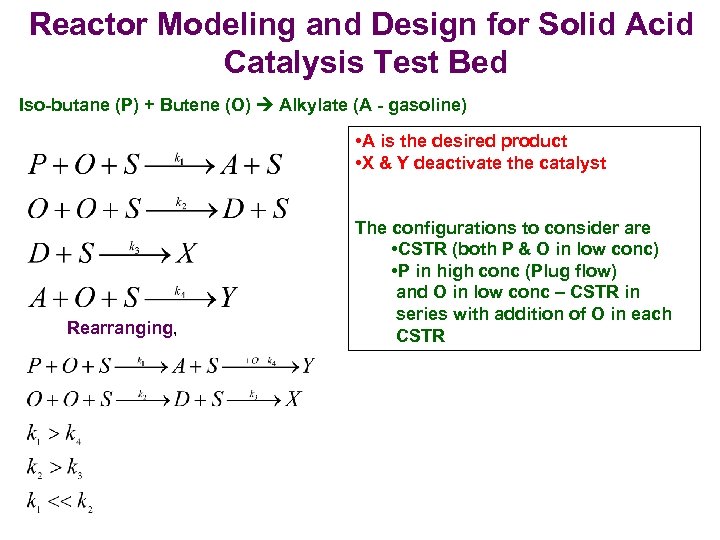
Reactor Modeling and Design for Solid Acid Catalysis Test Bed Iso-butane (P) + Butene (O) Alkylate (A - gasoline) • A is the desired product • X & Y deactivate the catalyst Rearranging, The configurations to consider are • CSTR (both P & O in low conc) • P in high conc (Plug flow) and O in low conc – CSTR in series with addition of O in each CSTR
Performance studies of a solid-catalyzed gas-liquid monolith reactor: Effect of flow maldistribution Shaibal Roy Muthanna Al-Dahhan CREL Annual Meeting 28 th October 2004 Introduction • Multiphase reactors (for solid catalyzed gasliquid reaction) used extensively in petroleum, petrochemical, biochemical, material, and environmental industries • Catalytic monolith reactor have shown promise to overcome some of the drawbacks of conventional reactors as well as give higher productivity (Krautzer et al. 2003, Nijhaus et al. , 2001) CHEMICAL REACTION ENGINEERING LABORATORY

Background Previous researches have assumed uniform flow distribution across a monolith cross-section in the Taylor flow regime. Experimental performance studies Monolith reactor performance (small diameter reactor) modeling (single channel model) Nijhaus et al. , 2001 Edvinsson et al. 1994 Krautzer et al. , 2003 Cybulski et al. 1999 Liu, 2001 Nijhaus et al. , 2003 However, this is not always the case as demonstrated by recent non-invasive flow measurement techniques (Mewes et al. , 1999; Gladden et al. , 2003) Objectives Gladden et al 2003 using MRI Mewes et al. 1999 using Capa. Tomo. • What is the effect of the following operating parameters on the flow distribution: • Gas and liquid velocities • Type of liquid distributor • Cell density and void fraction of monolith • How is the performance of monolith reactor (conducted in a large diameter reactor) affected by flow distribution • How does monolith reactor performance compare with trickle bed reactor • Does monolith scale reactor model (integrating flow distribution in the model) fare better than single tube model

Developing User Friendly Modules for Modeling Multiphase Reactors By Canan Tunca

Two friendly user simulation packages have been developed. User specifies several parameters needed in reactor design calculations. Liquid-solid circulating bed reactor for alkylation process Trickle bed reactor for phenol oxidation process

MICROREACTORS

Micro Reactors Evaluation for Environmentally Benign Processes • Radmila Jevtic, Milorad Dudukovic, and Muthanna Al-Dahhan (taken from http: //www. mikroglas. com) CHEMICAL REACTION ENGINEERING LABORATORY

Introduction The potential advantages of using microreactors instead of conventional reactors are (Jensen, 2001): • Higher surface to volume ratio • Higher mass and heat transfer rates • More aggressive reaction conditions with higher yields • Safer operation • Higher throughputs • Minimal environmental hazards CHEMICAL REACTION ENGINEERING LABORATORY

Test Reaction and Methodology 4% conversion of cyclohexane ; 80% selectivity is for cyclohexanol and cyclohexanone. The reaction has been performed under atmospheric pressure, both at room and the elevated temperatures (up to 90 o. C), with or without catalyst (cobalt naphthenate), and with various oxidants (air, oxygen, ozone, and hydrogen peroxide). CHEMICAL REACTION ENGINEERING LABORATORY

CATALYSIS CHARACTERIZATION AND DEVELOPMENT

Atomic Tailoring of Catalyst Surfaces for High Selectivity: Partial Oxidation of Propane Funded by the National Science Foundation’s GOALI (Grant Opportunities for Academic Liaison with Industry) Initiative Research Personnel: Professor John Gleaves Professor Gregory Yablonsky Dr. Anne Gaffney Mrs. Rebecca Fushimi Mr. Mike Rude Mr. David French Miss. Pam Buzzetta Mr. Sean Mueller Mr. Joseph Swisher Mr. Josh Searcy Heterogeneous Kinetics and Particle Chemistry Laboratory Department of Chemical Engineering Washington University, St. Louis MO Monomers Research 727 Norristown Road, PO Box 904 Spring House, Pennsylvania
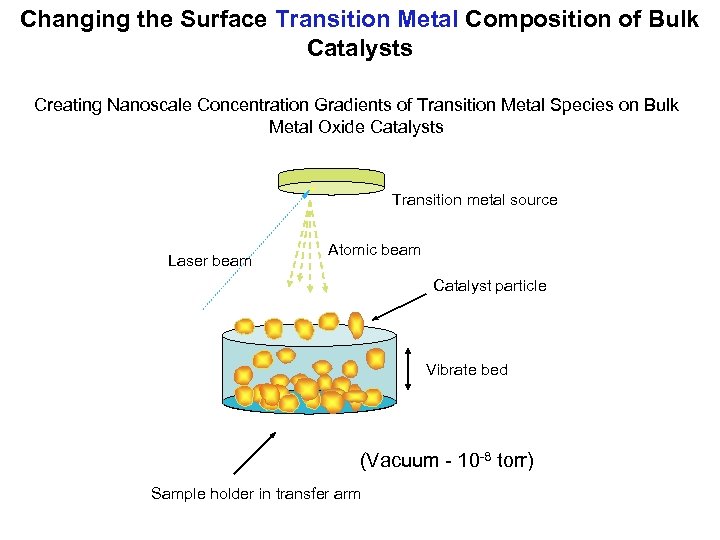
Changing the Surface Transition Metal Composition of Bulk Catalysts Creating Nanoscale Concentration Gradients of Transition Metal Species on Bulk Metal Oxide Catalysts Transition metal source Laser beam Atomic beam Catalyst particle Vibrate bed (Vacuum - 10 -8 torr) Sample holder in transfer arm
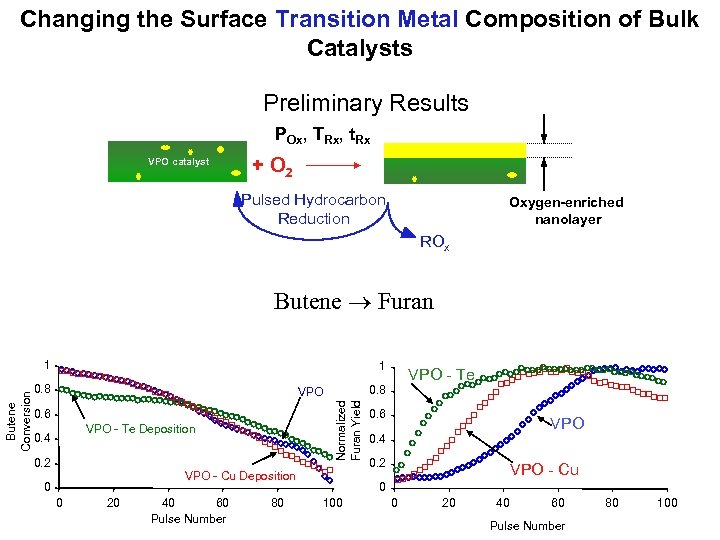
Changing the Surface Transition Metal Composition of Bulk Catalysts Preliminary Results POx, TRx, t. Rx VPO catalyst + O 2 Pulsed Hydrocarbon Reduction Oxygen-enriched nanolayer ROx Butene Furan 1 0. 8 VPO Normalized Furan Yield Butene Conversion 1 0. 6 VPO - Te Deposition 0. 4 0. 2 VPO - Cu Deposition 0 0 20 40 60 Pulse Number 80 VPO - Te 0. 6 VPO 0. 4 0. 2 VPO - Cu 0 100 0 20 40 60 Pulse Number 80 100

TAP Vacuum Pulse Response and Normal Pressure Studies of Propane Oxidation over Mo. VTe. Nb Oxide Catalyst Rebecca Fushimi 1, Sergiy O. Shekhtman 1, Michael Rude 1, Anne Gaffney 2, Scott Han 2, Gregory S. Yablonsky 1, John T. Gleaves 1 1 Dept. of Chem. Eng. , Washington University & Haas Company 2 Rohm
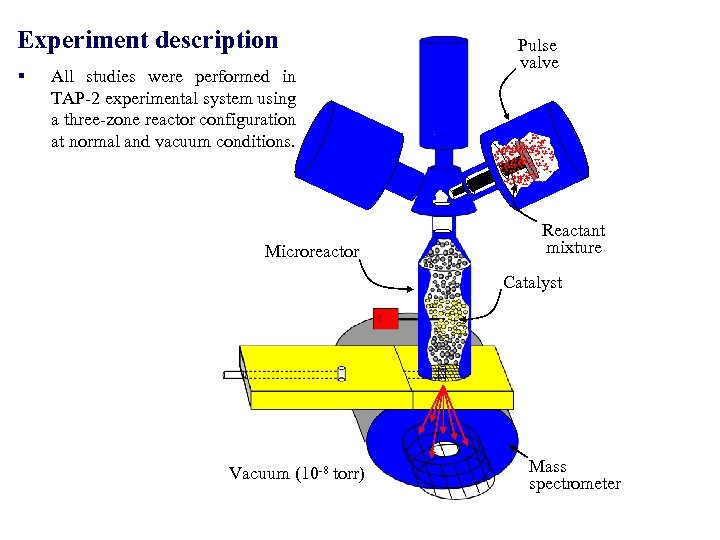
Experiment description § All studies were performed in TAP-2 experimental system using a three-zone reactor configuration at normal and vacuum conditions. Microreactor Pulse valve Reactant mixture Catalyst Vacuum (10 -8 torr) Mass spectrometer
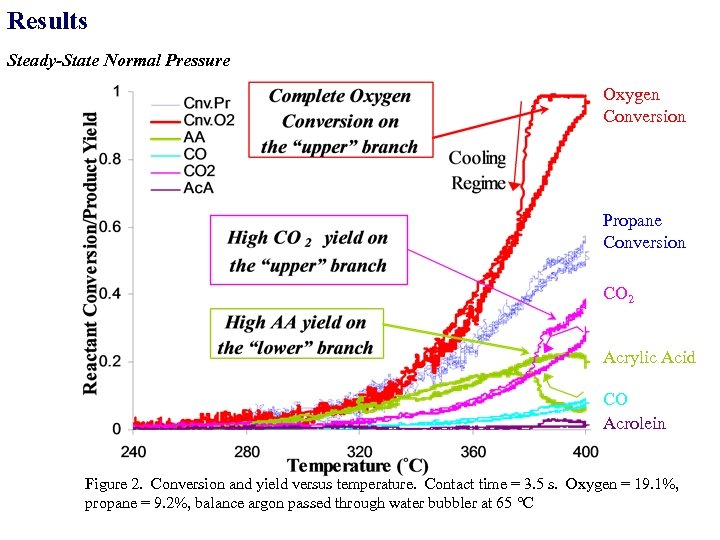
Results Steady-State Normal Pressure Oxygen Conversion Propane Conversion CO 2 Acrylic Acid CO Acrolein Figure 2. Conversion and yield versus temperature. Contact time = 3. 5 s. Oxygen = 19. 1%, propane = 9. 2%, balance argon passed through water bubbler at 65 C

Statistical Analysis of Complex Diffusion-Reaction Process in a Temporal Analysis of Product (TAP) System Elizabeth Maroon, Zhengjun Zhang, Michael Rude, Gregory S. Yablonsky Department of Mathematics, Washington University Department of Chemical. Engineering, Washington University
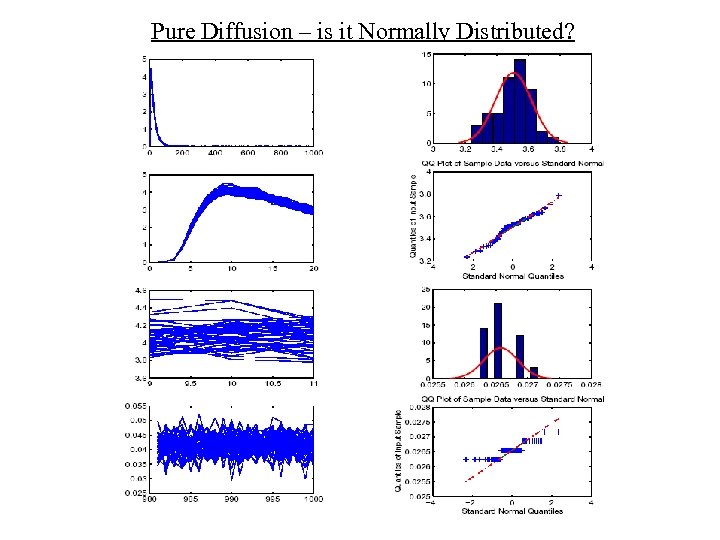
Pure Diffusion – is it Normally Distributed?

BIO-REACTORS, ENVIRONMENTAL

A Bioenergy-Based Bench-Scale Experiment for Undergraduate Engineering Students Using Fermiol Super HA® Bia Henriques, Fan Mei, Kursheed Karim, Muthanna Al-Dahhan Objectives: To create an experiment for undergraduate chemical engineering students that exposes them to bioprocesses and biofuels To give the students hands-on experience and knowledge about the dry grind corn to ethanol process To determine the effects of different sets of parameters on the fermentation process To study the effect of initial substrate concentration on ethanol production and yeast growth To examine the following: 1) Effects of different yeast strains on fermentation 2) Optimization of parameters of a kinetic model and prediction of fermentation performance 3) Effects of various design and operation parameters on corn syrup fermentation and product inhibition Accomplishments: Studied the effect of substrate concentration on corn syrup fermentation using a specific strain of Saccharomyces cerevisiae Collecting and analyzing data to validate existing kinetic models with and without the product inhibition term Optimized batch model parameters using experimental data Chemical Reaction Engineering Laboratory

Simulation and Design of a Process Control System for a Pilot Plant-Scale Distillation Unit Bia Henriques, Jonathan Lowe, Robert Heider, Terry Tolliver, Rachel Vazzi, Kwaku Opoku-Mensah Objectives: To simulate the distillation unit of SIU-E corn to ethanol pilot plant in HYSYS To study the design of the distillation unit by configuring its process control system in Delta. V To interface HYSYS and Delta. V to provide optimum process and process control design to SIU-E To study the behavior of the distillation unit’s control system and devise the best tuning method for the system To create an interactive model of the distillation unit in order to teach operators how the system works for better use of controls Accomplishments: Created an interactive learning model of SIU-Es pilot plant distillation unit Studied the effect of different tuning methods on the distillation process control Developed interface to use process simulation in HYSYS to control the system in Delta. V Optimized process performance by studying the behavior of the process control system Modeled all piping and instrumentation equipment found in SIU-E’s distillation unit Chemical Reaction Engineering Laboratory

Anaerobic Digestion of Animal Waste Rebecca Hoffmann, Khursheed Karim, Muthanna Al-Dahhan, Lars Angenent 2004 CREL Annual Meeting October 28, 2004 Chemical Reaction Engineering Laboratory (CREL) Bioprocessing and Bioreactor Engineering Laboratory (BELL)

Background n Anaerobic Digestion n Breakdown of organic molecules by microorganisms to produce methane gas which can be used as an energy source Waste management option that is a renewable energy source Role of mixing n n n Substrate and microorganism distribution throughout the reactor Ensures uniform p. H and temperature Prevents stratification and scum accumulation in dilute waste slurry Prevents accumulation of inert solids which decreases the active volume of a digester Effect of mixing is not well understood. Past research shows contradictory findings

Effect of Shear on Performance and Microbial Community in Anaerobic Digesters Treating Cow Manure Objective: Study the effect of mixing intensity, or applied shear, on digester performance, microbial ecology, and syntrophic relationships Hypotheses: n Higher mixing intensities have a detrimental effect upon reactor stability. n Different mixing intensities selectively create different microbial communities within each reactor. n Higher mixing intensities break up and/or prevent the formation of larger flocs of syntrophic microorganisms

Evaluation of Upflow Anaerobic Solids Removal (UASR) Digester for Animal Waste Digestion Objective: Evaluate the UASR as a new approach for animal waste slurry digestion and bioenergy production, focusing on the effect of increased solids concentration on digester performance

Development of a Predictive Model for Distiller’s Dried Grains/Solubles M. N. May and R. L. Heider
Introduction • DDGS – Co-product of dry grind ethanol process – Used in animal feed • Goal – Develop predictive models for chemical and physical properties of DDGS • Improve quality of DDGS product • Neural Networks – Derive meaning from complicated data and detect trends – Applicable in any industry to gain insight and answers to process questions

Gas Holdup Studies With CT In Anaerobic Bioreactors Rajneesh Varma Muthanna Al-Dahhan Chemical Reaction Engineering Laboratory (CREL) Bioprocess and Bioreactors Engineering Laboratory (BBEL) CREL Annual Meeting October 28 th 2004 CHEMICAL REACTION ENGINEERING LABORATORY
ANIMAL WASTE : Environmental Perspective and motivation for Treatment Unsafe and improperly disposed Gas injection Surface & groundwater contamination port Ammonia leaching 334 mm Methane emission Odors 50 m m n 153 mm n Level 2 n 50 m m Waste can be used to generate Methane 100 mm 140 mm 100 mm 3 biogas = 1. 7 k. Wh of electricity Methane = Energy, 1 m n 50 m m n 0 38 mm Level 1 140 mm 153 mm Diameter 220 mm n 1 Level 38 mm Level 1 mm Biomass has applications ofmm fertilizer and land fill 40 26 mm 25 Angle n 153 mm 334 mm n 150 mm n 40 mm 26 mm 0 25 Angle Gas mixed anaerobic bioreactors are found to most the popular choice. CHEMICAL REACTION ENGINEERING LABORATORY

Structural and magnetic properties of flame aerosol synthesized nanoparticles as a function of size Prakash Kumar, M. P. Dudukovic, Da-Ren Chen, Richard Axelbaum, Ronald Indeck, Pratim Biswas Biomedical Applications o Biocompatible ferromagnetic particles for targeted drug delivery o Selective deposition of magnetic particles for Tumor necrosis o Magnetic particles guided by external magnetic field for Aneurysm treatment SEM –γ Fe 2 O 3 Flame Reactor Modified DMA Key Results o X-ray diffraction and VSM results of the powder collected show the presence of pure Fe 2 O 3 with high saturation magnetization. o Flame pyrolysis of Iron pentacarbonyl gives Maghemite, Ferrocene gives Magnetite; whereas Iron nitrate gives Hematite. o. Post heat treatment of maghemite and magnetite showed to gradually transform to hematite at 500 o C.

Product Development in Ch. E: • New Courses Added: – New Product and Process Development (Ch. E 450) – Product Development Methodologies (Ch. E 452) • The Instructor: – Nick Nissing, Adjunct Faculty – Ex-P&G product development – Patent agent – Corporate IP Consultant • Unconventional Topics: – Creativity and Innovation – Intellectual Property – Theory of Inventive Problem Solving (TRIZ) – Design of Experiments – Impact of the Customer/Consumer – Fermi Problems – Product Focused Economics • How could we be useful to industry? – Brainstorming? – Consumer testing? – New Product Ideas? – In class or outside of class?
d3618b821e3e8500ed8df23ac79d639f.ppt