003cc16e307147a6da3688efdaf7f7e9.ppt
- Количество слайдов: 22

Post - ASCO 2007 Unresectable Stage IIIA/B NSCLC CONTENT • Take Home Message . . . 2 • Key Issues . . . 4 • Chemotherapy and RT. . . 7 • Navelbine and RT . . 15 . . 18 . . • Targeted agents and RT. ASCO ‘ 08 NSCLC IIIA/IIIB 1

ASCO 08 NSCLC unresectable Stage IIIA/B Take Home Message • Many data on CT-RT without major results - Phases II with Taxanes, Pemetrexed - Integration of targeted agents (Cetuximab, Bevacizumab, Erlotinib) in concurrent CT-RT • • Docetaxel as consolidation not recommended Low MS with paclitaxel-CBDCA (known data…) New data on NVB Oral and concurrent CT-RT Focus on RT & CT-RT-induced toxicities (lung) (#7555 p, #7573 p, #7602 p) ASCO ‘ 08 NSCLC IIIA/IIIB 2

ASCO 08 NSCLC unresectable Stage IIIA/B RT ant then… Induction, Sequential or Consolidation CT? • Induction CT before concurrent CT-RT: • GEM-platin alone(#7599 p)+ Taxane(#7589 p) (#7592 p) before D-platin-RT “limited impact on OS and PFS” or “GPG concurrently with GEM 225 mg/m 2/w too toxic” but GEM-CDDP before CDDP-RT more favourable(#7558 p) • Consolidation CT after concurrent CT-RT: • HOG LUN 01 update: docetaxel not recommended • GEM vs GEM-CBDCA(#7559 p): no benefit from the combination (46 vs 42% 2 y-OS) • GEM vs GEM-D after EP-RT(#7597 p): better PFS for the doublet (24 vs 8 mo) but at the price of higher toxicity • Sequential CT: • Meta-analysis on 13 rando trials, 2776 pts (Bozcuk #7575 p): The shorter is the time to RT after the onset of induction CT, the greater is the benefit ASCO ‘ 08 NSCLC IIIA/IIIB 3

ASCO 08 NSCLC unresectable Stage IIIA/B Key Issues: Chemotherapy (1/2) • Docetaxel (D): - Ph III DP vs MVP with concur. RT(#7515 p): benefit of 3 rd generation CT - HOG-LUN 01 update (#7519 p): refer to consolidation section - Weekly D in CT-RT schemes: • D-CDDP induction wkly D-RT (#7561 p): MS 21 mo & 2 y OS 34% • D-CDDP induction D-CDDP+concur. RT (#P 7585): 65% RR • Concurrent wkly D-CBDCA-RT (20 mg/m 2, AUC 2, 60 Gy) consol. DCBDCA (#7565 p): 2 y OS of 20%!, 21% G 3 esophagitis & pneumonitis scheme not recommended • Concurrent D-RT (20 mg/m 2, #7581 p): significant G 3 -4 tox. (20% esophagitis & 9% pneumonitis) + 5. 5% related-death, MS 12. 2 mo • Paclitaxel-CDDP: sequential vs induction + RT (#7520 p): - Low median survival in both arms: 13. 2 and 15. 4 m • 3 rd generation CT with CDDP–RT likely superior to 2 nd generation • Unimproved outcome from taxanes as concurrent or consolidation CT in CT-RT ASCO ‘ 08 NSCLC IIIA/IIIB 4

ASCO 08 NSCLC unresectable Stage IIIA/B Key Issues: Chemotherapy (2/2) • Pemetrexed (PEM) : - Concurrent Pem-CDDP-RT consol. D (#7569 p): 53% RR - Pilot study of CBDCA + dose-dense Pem (#7571 p) - Ph I concur. Pem-CDDP-RT(#7550 p) • Other CT agents & concurrent RT: - S 1 (Oral Fluoropyrim. ) (Japan) + P + RT Ph II (#7556 p): • 87% RR (46% IIIA), PFS 13. 4 mo, 6% FN, 24% G 3 neutropenia, 12% G 3 esophagitis and 4% pneumonitis, - Oral platinum (#7560 p): MTD to be confirmed 30 mg /m 2/d ASCO ‘ 08 NSCLC IIIA/IIIB 5

ASCO 08 NSCLC unresectable Stage IIIA/B Key Issues : Navelbine Oral/i. v. • Navelbine Oral is effective in elderly pts • 70 % ORR and 40% 2 y-OS in “curative” intent (60 Gy RT) concurrent CT-RT setting (Silvano #20698 p) • Navelbine iv or Oral: • A retrospective experience (Portalone #18507 p) Effective and safe concurrently with RT, even in elderly pts ASCO ‘ 08 NSCLC IIIA/IIIB 6

ASCO 08 NSCLC unresectable Stage IIIA/B Key Issues : Targeted Therapies • Cetuximab • Appears safe and promising when grafted onto various chemoradiation regimens (#7516 p) (#7607 p) • Bevacizumab and Erlotinib • Addition seems to be feasible but squamous histology requires major caution (pulmonary hemorrage G 3 -5) (#7517 p) • Erlotinib added to RT, no convincing early results (#7563 p) ASCO ‘ 08 NSCLC IIIA/IIIB 7

ASCO 2008 Chemotherapy and RT ASCO ‘ 08 NSCLC IIIA/IIIB 8
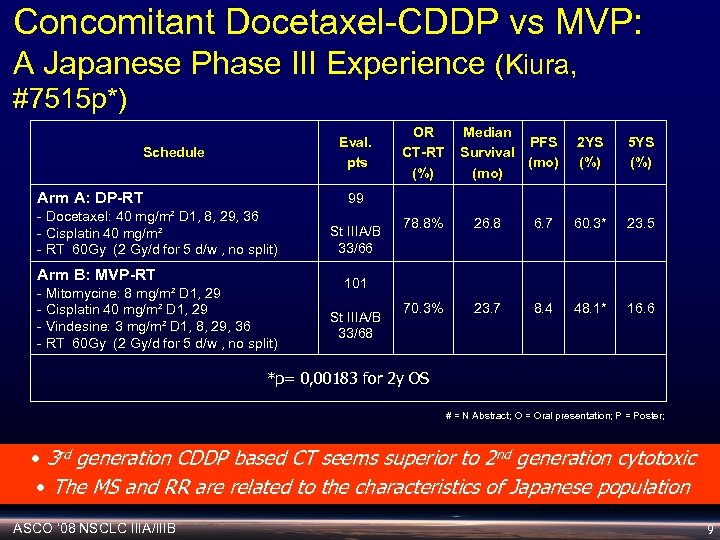
Concomitant Docetaxel-CDDP vs MVP: A Japanese Phase III Experience (Kiura, #7515 p*) Eval. pts Schedule Arm A: DP-RT OR CT-RT (%) Median PFS Survival (mo) 2 YS (%) 5 YS (%) 99 - Docetaxel: 40 mg/m² D 1, 8, 29, 36 - Cisplatin 40 mg/m² - RT 60 Gy (2 Gy/d for 5 d/w , no split) Arm B: MVP-RT - Mitomycine: 8 mg/m² D 1, 29 - Cisplatin 40 mg/m² D 1, 29 - Vindesine: 3 mg/m² D 1, 8, 29, 36 - RT 60 Gy (2 Gy/d for 5 d/w , no split) St IIIA/B 33/66 78. 8% 26. 8 6. 7 60. 3* 23. 5 70. 3% 23. 7 8. 4 48. 1* 16. 6 101 St IIIA/B 33/68 *p= 0, 00183 for 2 y OS # = N Abstract; O = Oral presentation; P = Poster; • 3 rd generation CDDP based CT seems superior to 2 nd generation cytotoxic • The MS and RR are related to the characteristics of Japanese population ASCO ‘ 08 NSCLC IIIA/IIIB 9
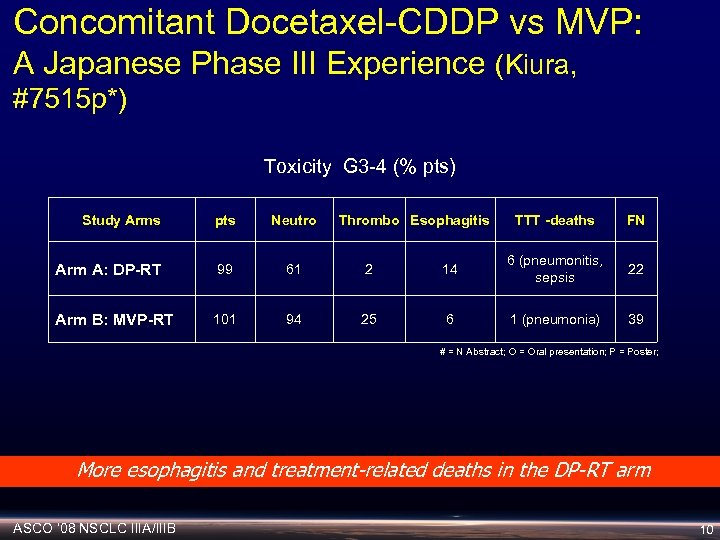
Concomitant Docetaxel-CDDP vs MVP: A Japanese Phase III Experience (Kiura, #7515 p*) Toxicity G 3 -4 (% pts) Study Arms pts Neutro Thrombo Esophagitis Arm A: DP-RT 99 61 2 Arm B: MVP-RT 101 94 25 TTT -deaths FN 14 6 (pneumonitis, sepsis 22 6 1 (pneumonia) 39 # = N Abstract; O = Oral presentation; P = Poster; More esophagitis and treatment-related deaths in the DP-RT arm ASCO ‘ 08 NSCLC IIIA/IIIB 10
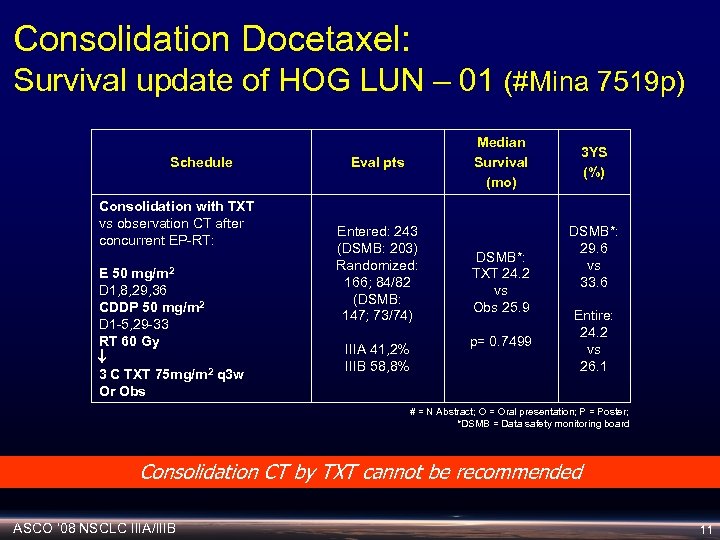
Consolidation Docetaxel: Survival update of HOG LUN – 01 (#Mina 7519 p) Schedule Consolidation with TXT vs observation CT after concurrent EP-RT: mg/m 2 E 50 D 1, 8, 29, 36 CDDP 50 mg/m 2 D 1 -5, 29 -33 RT 60 Gy 3 C TXT 75 mg/m 2 q 3 w Or Obs Median Survival (mo) Eval pts Entered: 243 (DSMB: 203) Randomized: 166; 84/82 (DSMB: 147; 73/74) IIIA 41, 2% IIIB 58, 8% DSMB*: TXT 24. 2 vs Obs 25. 9 p= 0. 7499 3 YS (%) DSMB*: 29. 6 vs 33. 6 Entire: 24. 2 vs 26. 1 # = N Abstract; O = Oral presentation; P = Poster; *DSMB = Data safety monitoring board Consolidation CT by TXT cannot be recommended ASCO ‘ 08 NSCLC IIIA/IIIB 11
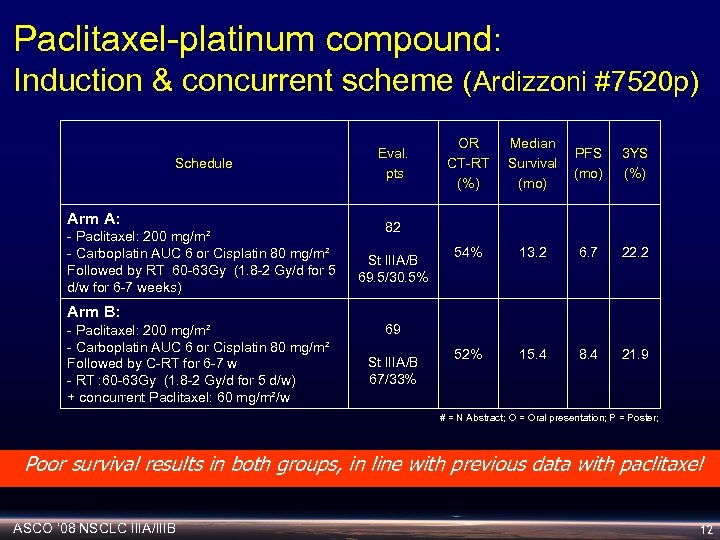
Paclitaxel-platinum compound: Induction & concurrent scheme (Ardizzoni #7520 p) Schedule Arm A: - Paclitaxel: 200 mg/m² - Carboplatin AUC 6 or Cisplatin 80 mg/m² Followed by RT 60 -63 Gy (1. 8 -2 Gy/d for 5 d/w for 6 -7 weeks) Eval. pts OR CT-RT (%) Median Survival (mo) PFS (mo) 3 YS (%) 54% 13. 2 6. 7 22. 2 52% 15. 4 8. 4 21. 9 82 St IIIA/B 69. 5/30. 5% Arm B: - Paclitaxel: 200 mg/m² - Carboplatin AUC 6 or Cisplatin 80 mg/m² Followed by C-RT for 6 -7 w - RT : 60 -63 Gy (1. 8 -2 Gy/d for 5 d/w) + concurrent Paclitaxel: 60 mg/m²/w 69 St IIIA/B 67/33% # = N Abstract; O = Oral presentation; P = Poster; Poor survival results in both groups, in line with previous data with paclitaxel ASCO ‘ 08 NSCLC IIIA/IIIB 12
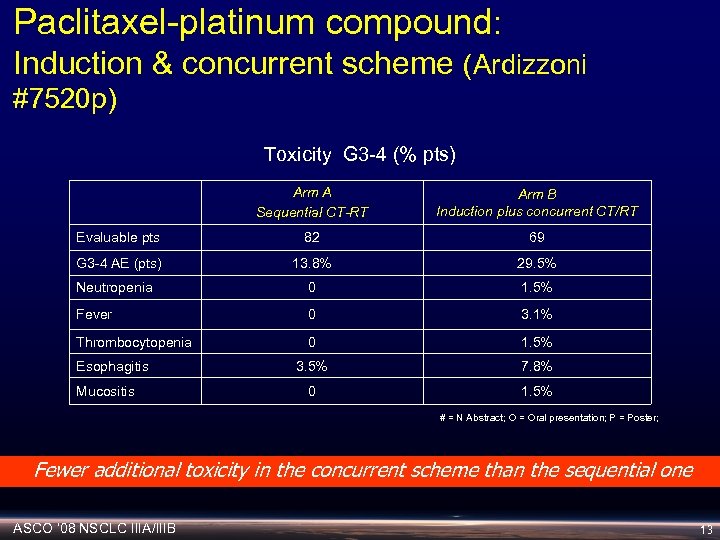
Paclitaxel-platinum compound: Induction & concurrent scheme (Ardizzoni #7520 p) Toxicity G 3 -4 (% pts) Arm A Sequential CT-RT Arm B Induction plus concurrent CT/RT Evaluable pts 82 69 G 3 -4 AE (pts) 13. 8% 29. 5% Neutropenia 0 1. 5% Fever 0 3. 1% Thrombocytopenia 0 1. 5% 3. 5% 7. 8% 0 1. 5% Esophagitis Mucositis # = N Abstract; O = Oral presentation; P = Poster; Fewer additional toxicity in the concurrent scheme than the sequential one ASCO ‘ 08 NSCLC IIIA/IIIB 13
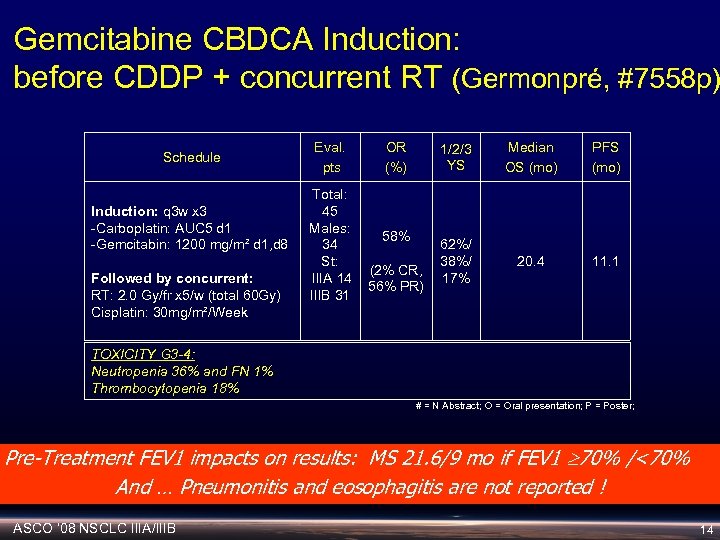
Gemcitabine CBDCA Induction: before CDDP + concurrent RT (Germonpré, #7558 p) Schedule Induction: q 3 w x 3 -Carboplatin: AUC 5 d 1 -Gemcitabin: 1200 mg/m² d 1, d 8 Followed by concurrent: RT: 2. 0 Gy/fr x 5/w (total 60 Gy) Cisplatin: 30 mg/m²/Week Eval. pts Total: 45 Males: 34 St: IIIA 14 IIIB 31 OR (%) 1/2/3 YS 58% (2% CR, 56% PR) Median OS (mo) PFS (mo) 62%/ 38%/ 17% 20. 4 11. 1 TOXICITY G 3 -4: Neutropenia 36% and FN 1% Thrombocytopenia 18% # = N Abstract; O = Oral presentation; P = Poster; Pre-Treatment FEV 1 impacts on results: MS 21. 6/9 mo if FEV 1 70% /<70% And … Pneumonitis and eosophagitis are not reported ! ASCO ‘ 08 NSCLC IIIA/IIIB 14

ASCO 2008: Navelbine and RT ASCO ‘ 08 NSCLC IIIA/IIIB 15

Navelbine Oral concurrently with RT in Elderly Stage IIIA/B NSCLC (Silvano, #20698 p) • • • Oral VRB : 20 mg/m 2 twice weekly concurrently with RT 25 pts > 65 y, ECOG < 2, Stage IIIA/B 30/70% > 1 poor risk inclusion criteria: 60% 10 pts received 60 Gy as “curative” intent RT 15 pts received 45 Gy as palliative RT Palliative RT: improvement of PS ECOG in 65% pts ORR 60 Gy RT(10 pts) 2 y OS Toxicity 70% (40% CR) 40% No G 3/4 hematologic 1 G 4 esophagitis # = N Abstract; O = Oral presentation; P = Poster; NBVo concurrently with 60 Gy RT effective in elderly population ASCO ‘ 08 NSCLC IIIA/IIIB 16

Navelbine concurrently with RT A retrospective analysis in Stage IIIA/B (Portalone P#18507) • 31 pts, 16 males, median age 60 y, Stage IIIA/B 80. 7/19. 3% • Induction: CDDP 80 mg/ m 2 or CBDCA AUC 5 plus either GEM 1200 mg/ m 2 or NVBiv 25 mg/ m 2 D 1, 8, 21 • CT-RT: NVB iv 5 mg/ m 2 or NVBo 12 mg/ m 2 x 3 / week 2 hours before RT(2 Gy /d for 6 w) Induction RR CT-RT RR TTP (mo) 38. 7% 43. 3% 5 underwent surgery (2 IIIB) 10 MS (mo) OS 1 y 15. 3 65 % (21. 6 IIIA, 14. 6 IIIB) OS 2 y Toxicity 35 % 8% G 3 & 1. 3% G 4 hemato Esophagitis: 1 G 3 + 1 G 4 No toxic death # = N Abstract; O = Oral presentation; P = Poster; NVB concurrently with RT effective and safe ASCO ‘ 08 NSCLC IIIA/IIIB 17

ASCO 2008: Targeted Therapies and RT ASCO ‘ 08 NSCLC IIIA/IIIB 18

Erlotinib & concurrent 3 D-RT vs 3 D-RT Randomized Phase II-Efficacy (Martinez, #7563 p*) Data updated since published abstract Schedule A: 3 DRT(66 Gy, 33 fr for 6, 5 w). B: 3 DRT(66 Gy 33 fr for 6, 5 w). Erlotinib: 150 mg/day p. o. continued up to 6 months OR (%) Eval. pts Median Survival (mo) PFS (mo) A: 70 B: 70 A: 15. 3 B: NR NA 36 enrolled (13 arm A, 23 arm B), 34 for safety &22), (12 20 for efficacy (10 &10) St I/II: 36% St III: 64% Arm B : moderate skin rash (54. 5%) and diarrhea (22. 7%) G 3 -4 toxicities: 4. 5% radiodermitis and skin rash; 9. 1% renal failure # = N Abstract; O = Oral presentation; P = Poster; • Erlotinib is safe & reported similar OR added to RT with OS not yet mature ASCO ‘ 08 NSCLC IIIA/IIIB 19
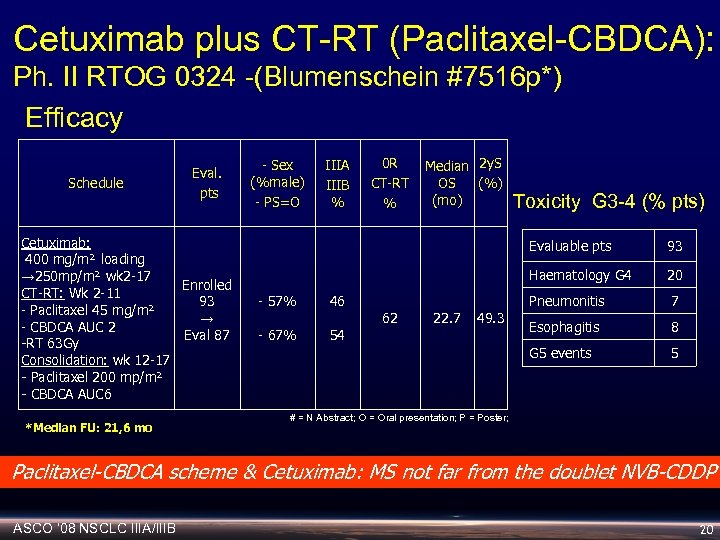
Cetuximab plus CT-RT (Paclitaxel-CBDCA): Ph. II RTOG 0324 -(Blumenschein #7516 p*) Efficacy Schedule Eval. pts Cetuximab: 400 mg/m² loading → 250 mp/m² wk 2 -17 Enrolled CT-RT: Wk 2 -11 93 - Paclitaxel 45 mg/m² → - CBDCA AUC 2 Eval 87 -RT 63 Gy Consolidation: wk 12 -17 - Paclitaxel 200 mp/m² - CBDCA AUC 6 *Median FU: 21, 6 mo - Sex (%male) - PS=O IIIA IIIB % 0 R CT-RT % Median 2 y. S OS (%) (mo) Toxicity G 3 -4 (% pts) Evaluable pts Haematology G 4 46 - 67% 54 Pneumonitis 62 22. 7 49. 3 20 7 Esophagitis 8 G 5 events - 57% 93 5 # = N Abstract; O = Oral presentation; P = Poster; Paclitaxel-CBDCA scheme & Cetuximab: MS not far from the doublet NVB-CDDP ASCO ‘ 08 NSCLC IIIA/IIIB 20
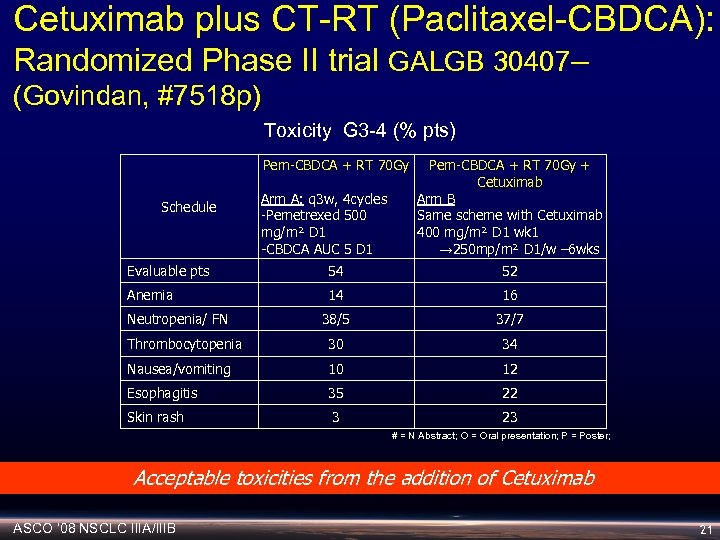
Cetuximab plus CT-RT (Paclitaxel-CBDCA): Randomized Phase II trial GALGB 30407– (Govindan, #7518 p) Toxicity G 3 -4 (% pts) Pem-CBDCA + RT 70 Gy Schedule Arm A: q 3 w, 4 cycles -Pemetrexed 500 mg/m² D 1 -CBDCA AUC 5 D 1 Pem-CBDCA + RT 70 Gy + Cetuximab Arm B Same scheme with Cetuximab 400 mg/m² D 1 wk 1 → 250 mp/m² D 1/w – 6 wks Evaluable pts 54 52 Anemia 14 16 38/5 37/7 Thrombocytopenia 30 34 Nausea/vomiting 10 12 Esophagitis 35 22 Skin rash 3 23 Neutropenia/ FN # = N Abstract; O = Oral presentation; P = Poster; Acceptable toxicities from the addition of Cetuximab ASCO ‘ 08 NSCLC IIIA/IIIB 21
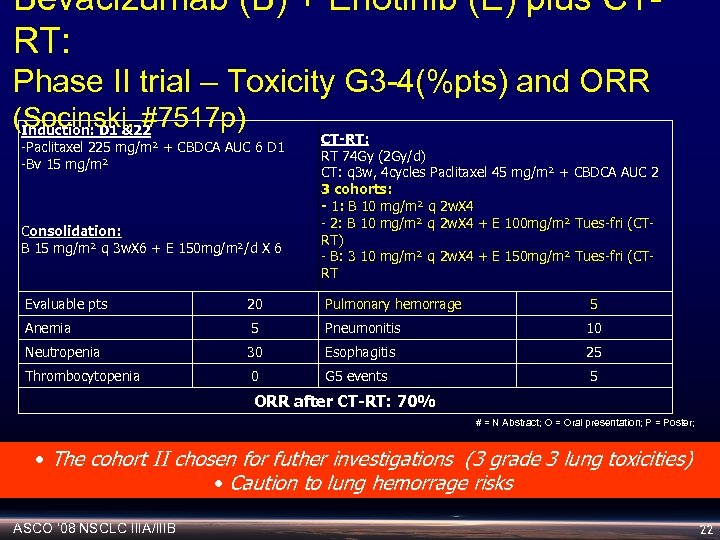
Bevacizumab (B) + Erlotinib (E) plus CTRT: Phase II trial – Toxicity G 3 -4(%pts) and ORR (Socinski, #7517 p) Induction: D 1 &22 -Paclitaxel 225 mg/m² + CBDCA AUC 6 D 1 -Bv 15 mg/m² Consolidation: B 15 mg/m² q 3 w. X 6 + E 150 mg/m²/d X 6 CT-RT: RT 74 Gy (2 Gy/d) CT: q 3 w, 4 cycles Paclitaxel 45 mg/m² + CBDCA AUC 2 3 cohorts: - 1: B 10 mg/m² q 2 w. X 4 - 2: B 10 mg/m² q 2 w. X 4 + E 100 mg/m² Tues-fri (CTRT) - B: 3 10 mg/m² q 2 w. X 4 + E 150 mg/m² Tues-fri (CTRT Evaluable pts 20 Pulmonary hemorrage 5 Anemia 5 Pneumonitis 10 Neutropenia 30 Esophagitis 25 Thrombocytopenia 0 G 5 events 5 ORR after CT-RT: 70% # = N Abstract; O = Oral presentation; P = Poster; • The cohort II chosen for futher investigations (3 grade 3 lung toxicities) • Caution to lung hemorrage risks ASCO ‘ 08 NSCLC IIIA/IIIB 22
003cc16e307147a6da3688efdaf7f7e9.ppt