d485a086409bc1b12ae34dd212667c3e.ppt
- Количество слайдов: 30
 PK/PD Modeling in Support of Drug Development Alan Hartford, Ph. D. Associate Director Scientific Staff Clinical Pharmacology Statistics Merck Research Laboratories, Inc. alan_hartford@merck. com
PK/PD Modeling in Support of Drug Development Alan Hartford, Ph. D. Associate Director Scientific Staff Clinical Pharmacology Statistics Merck Research Laboratories, Inc. alan_hartford@merck. com
 Outline • • • Introduction Purpose of PK/PD modeling The Modeling Procedure Example from literature: Bevacizumab 2
Outline • • • Introduction Purpose of PK/PD modeling The Modeling Procedure Example from literature: Bevacizumab 2
 Introduction • Pharmacokinetics is the study of what an organism does with a dose of a drug – kinetics = motion – Absorbs, Distributes, Metabolizes, Excretes • Pharmacodynamics is the study of what the drug does to the body – dynamics = change 3
Introduction • Pharmacokinetics is the study of what an organism does with a dose of a drug – kinetics = motion – Absorbs, Distributes, Metabolizes, Excretes • Pharmacodynamics is the study of what the drug does to the body – dynamics = change 3
 Pharmacokinetics • Endpoints – AUC, Cmax, Tmax, half-life (terminal), C_trough • The effect of the drug is assumed to be related to some measure of exposure. (AUC, Cmax, C_trough) 4
Pharmacokinetics • Endpoints – AUC, Cmax, Tmax, half-life (terminal), C_trough • The effect of the drug is assumed to be related to some measure of exposure. (AUC, Cmax, C_trough) 4
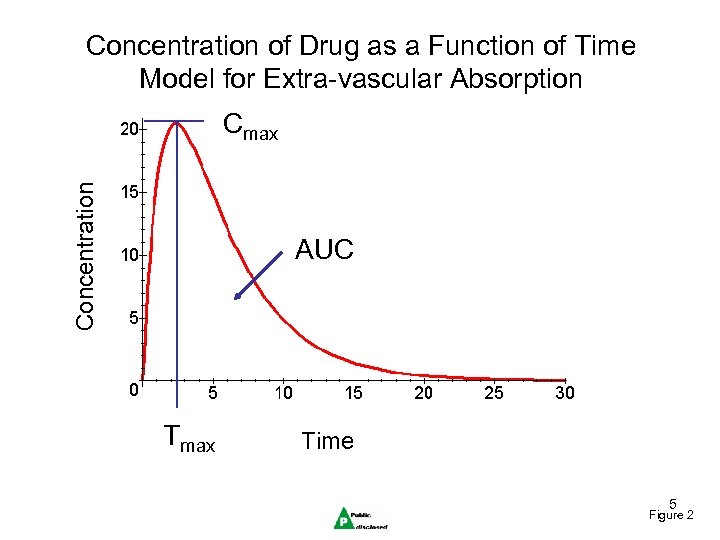 Concentration of Drug as a Function of Time Model for Extra-vascular Absorption Concentration Cmax AUC Tmax Time 5 Figure 2
Concentration of Drug as a Function of Time Model for Extra-vascular Absorption Concentration Cmax AUC Tmax Time 5 Figure 2
 PK/PD Modeling • Procedure: – Estimate exposure and examine correlation between PD other endpoints (including AE rates) – Use mechanistic models • Purpose: – – Estimate therapeutic window Dose selection Identify mechanism of action Model probability of AE as function of exposure (and covariates) – Inform the label of the drug 6
PK/PD Modeling • Procedure: – Estimate exposure and examine correlation between PD other endpoints (including AE rates) – Use mechanistic models • Purpose: – – Estimate therapeutic window Dose selection Identify mechanism of action Model probability of AE as function of exposure (and covariates) – Inform the label of the drug 6
 Drug Label • Additional negotiation after drug approval • Need information for prescribing doctors and pharmacists • Need instructions for patients • Aim for clear summary of PK, efficacy, and safety information • If instructions are complicated, may reduce patient ability to properly dose 7
Drug Label • Additional negotiation after drug approval • Need information for prescribing doctors and pharmacists • Need instructions for patients • Aim for clear summary of PK, efficacy, and safety information • If instructions are complicated, may reduce patient ability to properly dose 7
 Observed or Predicted PK? • Exposure (AUC) not measured – only modeled • Concentration in blood or plasma is a biomarker for concentration at site of action • PK parameters are not directly measured 8
Observed or Predicted PK? • Exposure (AUC) not measured – only modeled • Concentration in blood or plasma is a biomarker for concentration at site of action • PK parameters are not directly measured 8
 The Nonlinear Mixed Effects Model Pharmacokineticists use the term ”population” model when the model involves random effects. 9
The Nonlinear Mixed Effects Model Pharmacokineticists use the term ”population” model when the model involves random effects. 9
 Compartmental Modeling • A person’s body is modeled with a system of differential equations, one for each “compartment” • If each equation represents a specific organ or set of organs with similar perfusion rates, then called Physiologically Based PK (PBPK) modeling. • The mean function f is a solution of this system of differential equations. • Each equation in the system describes the flow of drug into and out of a specific compartment. 10
Compartmental Modeling • A person’s body is modeled with a system of differential equations, one for each “compartment” • If each equation represents a specific organ or set of organs with similar perfusion rates, then called Physiologically Based PK (PBPK) modeling. • The mean function f is a solution of this system of differential equations. • Each equation in the system describes the flow of drug into and out of a specific compartment. 10
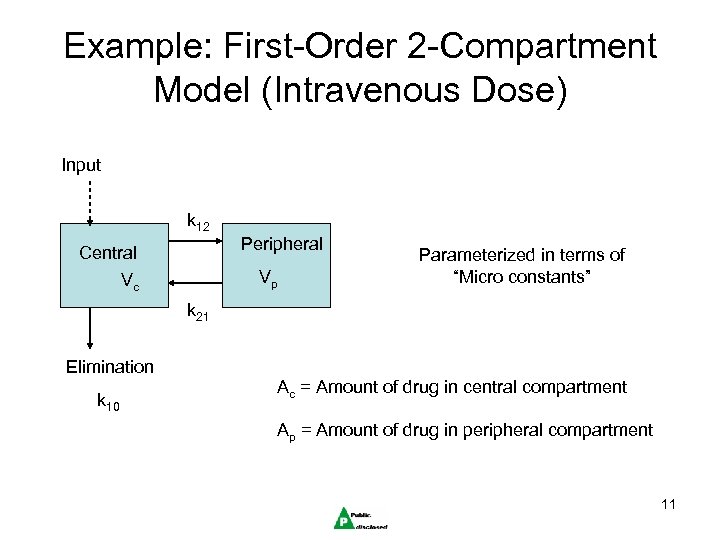 Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc Parameterized in terms of “Micro constants” k 21 Elimination k 10 Ac = Amount of drug in central compartment Ap = Amount of drug in peripheral compartment 11
Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc Parameterized in terms of “Micro constants” k 21 Elimination k 10 Ac = Amount of drug in central compartment Ap = Amount of drug in peripheral compartment 11
 Web Demonstration • http: //vam. anest. ufl. edu/simulations/simula tionportfolio. php 12
Web Demonstration • http: //vam. anest. ufl. edu/simulations/simula tionportfolio. php 12
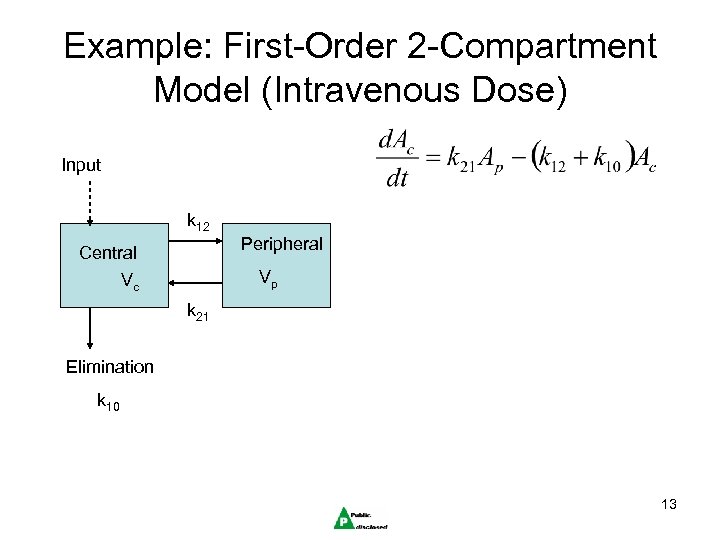 Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 13
Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 13
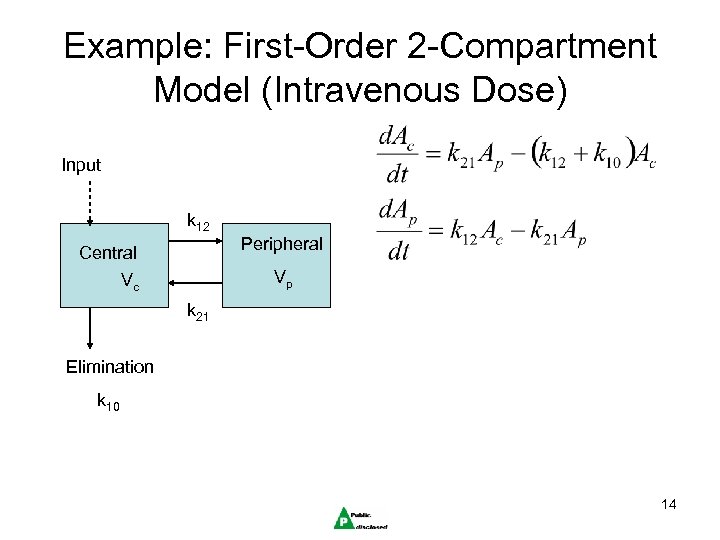 Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 14
Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 14
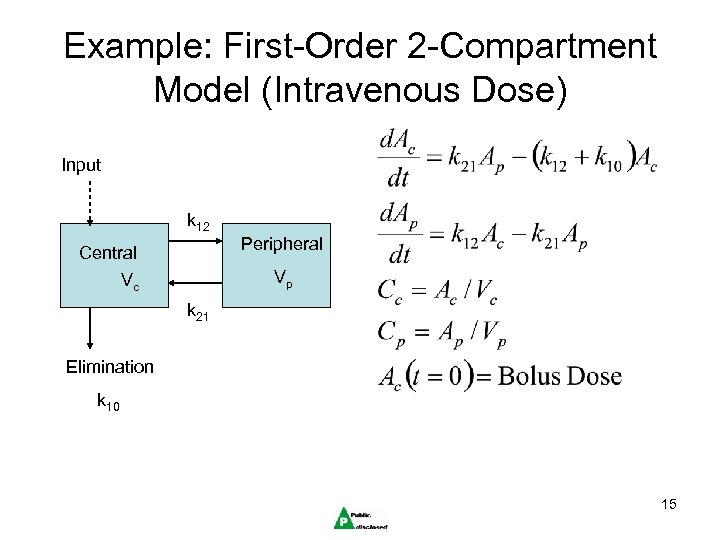 Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 15
Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 15
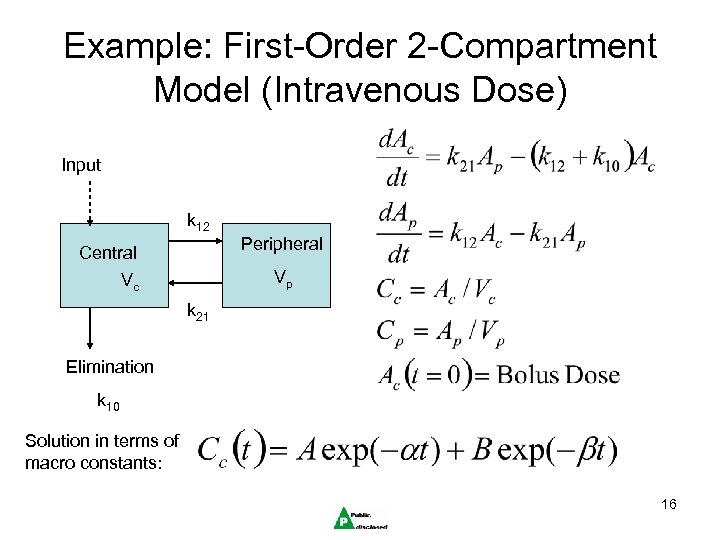 Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 Solution in terms of macro constants: 16
Example: First-Order 2 -Compartment Model (Intravenous Dose) Input k 12 Peripheral Central Vp Vc k 21 Elimination k 10 Solution in terms of macro constants: 16
 Modeling Covariates Assumed: PK parameters vary with respect to a patient’s weight or age. Covariates can be added to the model in a secondary structure (hierarchical model). “Population Pharmacokinetics” refers specifically to these mixed effects models with covariates included in the secondary, hierarchical structure 17
Modeling Covariates Assumed: PK parameters vary with respect to a patient’s weight or age. Covariates can be added to the model in a secondary structure (hierarchical model). “Population Pharmacokinetics” refers specifically to these mixed effects models with covariates included in the secondary, hierarchical structure 17
 Nonlinear Mixed Effects Model With secondary structure for covariates: Often, is a vector of log Cl, log V, and log ka 18
Nonlinear Mixed Effects Model With secondary structure for covariates: Often, is a vector of log Cl, log V, and log ka 18
 Pharmacodynamic Model • PK: nonlinear mixed effect model (mechanistic) • PD: – now assume predicted PK parameters are true – less PD data per subject – nonlinear fixed effect model (mechanistic) 19
Pharmacodynamic Model • PK: nonlinear mixed effect model (mechanistic) • PD: – now assume predicted PK parameters are true – less PD data per subject – nonlinear fixed effect model (mechanistic) 19
 Next Step: Simulations • Using the PK/PD model, clinical trial simulations can be performed to: – Inform adaptive design – Determine good dose or dosing regimen for future trial – Satisfy regulatory agencies in place of additional trials – Surrogate for trials for testing biomarkers to discriminate doses 20
Next Step: Simulations • Using the PK/PD model, clinical trial simulations can be performed to: – Inform adaptive design – Determine good dose or dosing regimen for future trial – Satisfy regulatory agencies in place of additional trials – Surrogate for trials for testing biomarkers to discriminate doses 20
 Example 1: Bevacizumab • Recombinant humanized Ig. G 1 antibody • Binds and inhibits effects induced by vascular endothelial growth factor (VEGF) • (stops tumors from growing by cutting off supply of blood) • Approved for use with chemotherapy for colorectal cancer 21
Example 1: Bevacizumab • Recombinant humanized Ig. G 1 antibody • Binds and inhibits effects induced by vascular endothelial growth factor (VEGF) • (stops tumors from growing by cutting off supply of blood) • Approved for use with chemotherapy for colorectal cancer 21
 Paper: Clinical PK of bevacizumab in patients with solid tumors (Lu et al 2007) • Objective stated in paper: To characterize the population PK and the influence of demographic factors, disease severity, and concomitantly used chemotherapy agents on it’s PK behavior. • Purpose: to make conclusions about PK to confirm dosing strategy is appropriate 22
Paper: Clinical PK of bevacizumab in patients with solid tumors (Lu et al 2007) • Objective stated in paper: To characterize the population PK and the influence of demographic factors, disease severity, and concomitantly used chemotherapy agents on it’s PK behavior. • Purpose: to make conclusions about PK to confirm dosing strategy is appropriate 22
 Patients and Methods • 4629 bevacizumab concentration samples • 491 patients with solid tumors • Doses from 1 to 20 mg/kg from weekly to every 3 weeks • NONMEM software used to fit nonlinear mixed effects model 23
Patients and Methods • 4629 bevacizumab concentration samples • 491 patients with solid tumors • Doses from 1 to 20 mg/kg from weekly to every 3 weeks • NONMEM software used to fit nonlinear mixed effects model 23
 Demographic Variables • Gender (male/female) • Race (caucasian, Black, Hispanic, Asian, Native American, Other) • ECOG Performance Status (0, 1, 2) • Chemotherapy (6 different therapies) • Weight • Height • Body Surface Area • Lean Body Mass 24
Demographic Variables • Gender (male/female) • Race (caucasian, Black, Hispanic, Asian, Native American, Other) • ECOG Performance Status (0, 1, 2) • Chemotherapy (6 different therapies) • Weight • Height • Body Surface Area • Lean Body Mass 24
 Other Covariates • • Serum-asparate aminotransferase (SGPT) Serum-alanine aminotransferase (SGOT) Serum-alkaline phosphatase (ALK) Serum-bilirubin Total protein Albumin Creatinine clearance 25
Other Covariates • • Serum-asparate aminotransferase (SGPT) Serum-alanine aminotransferase (SGOT) Serum-alkaline phosphatase (ALK) Serum-bilirubin Total protein Albumin Creatinine clearance 25
 Results • First-order, two-compartment model fitted data well • Weight, gender, and albumin had largest effects on CL • ALK and SGOT also significantly effected CL • Weight, gender, and Albumin had significant effects on Vc 26
Results • First-order, two-compartment model fitted data well • Weight, gender, and albumin had largest effects on CL • ALK and SGOT also significantly effected CL • Weight, gender, and Albumin had significant effects on Vc 26
 Results (cont. ) • Bevacizumab CL was 26% faster in males than females • Subjects with low serum albumin have 19% faster CL than typical patients • Subjects with higher ALK have a 23% faster CL than typical patients • CL was different for different chemo regimens 27
Results (cont. ) • Bevacizumab CL was 26% faster in males than females • Subjects with low serum albumin have 19% faster CL than typical patients • Subjects with higher ALK have a 23% faster CL than typical patients • CL was different for different chemo regimens 27
 Ex 1: Conclusions • Population PK parameters for Bevacizumab similar to other IGg antibodies • Weight and gender effects from modeling support weight based dosing • Linear PK suggest similar exposures can be achieved with flexible dosage regimens (Q 2 or Q 3 weekly dosing) 28
Ex 1: Conclusions • Population PK parameters for Bevacizumab similar to other IGg antibodies • Weight and gender effects from modeling support weight based dosing • Linear PK suggest similar exposures can be achieved with flexible dosage regimens (Q 2 or Q 3 weekly dosing) 28
 Review • PK/PD modeling performed to help better understand the drug: – Estimate therapeutic window – Dose selection – Identify mechanism of action – Model probability of AE as function of exposure (and covariates) 29
Review • PK/PD modeling performed to help better understand the drug: – Estimate therapeutic window – Dose selection – Identify mechanism of action – Model probability of AE as function of exposure (and covariates) 29
 Reference • Clinical pharmacokinetics of bevacizumab in patients with solid tumors, Jian-Feng Lu, Rene Bruno, Steve Eppler, William Novotny, Bert Lum, and Jacques Gaudreault, Cancer Chemother Pharmacol. , 2008 Jan 19. 30
Reference • Clinical pharmacokinetics of bevacizumab in patients with solid tumors, Jian-Feng Lu, Rene Bruno, Steve Eppler, William Novotny, Bert Lum, and Jacques Gaudreault, Cancer Chemother Pharmacol. , 2008 Jan 19. 30


