a3d74a5b0f6ae6861f01b2ff0d443983.ppt
- Количество слайдов: 178

Physiology of Neurons By Lincoln Edwards, D. D. S. ; Ph. D. Ext 42923 email: lpedwards@llu. edu

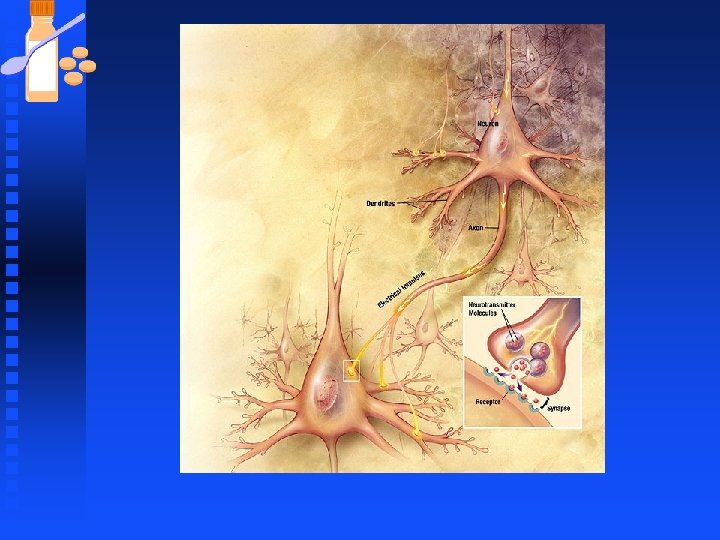
Neurons u Functional unit of the nervous system, they are specialized for F Communication F signaling. u Typical neuron consists of a cell body (soma) with branches (dendrites) and a single axon that extends from the soma and branches multiple times.
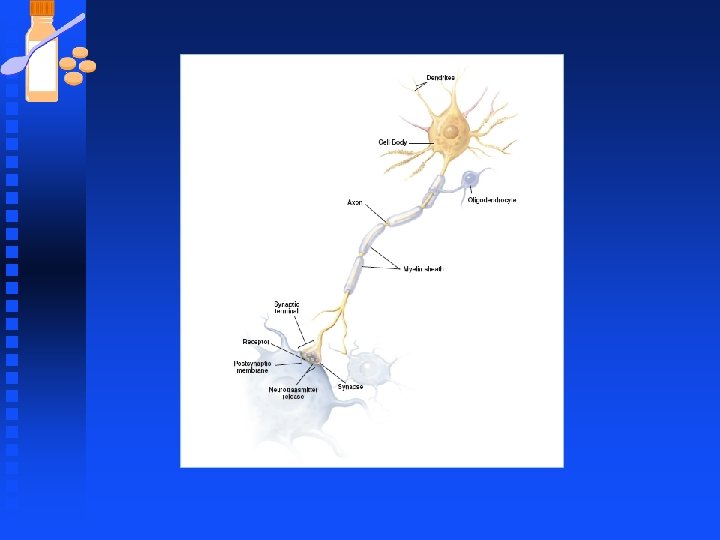

Neurons Each branch ends at a presynaptic terminal that contacts another cell. n The branching set of dendrites is called the dendritic tree. n Neurons have an enormous variety n u shapes and size and morphologic variations among neurons is produced by differences in the branching patterns of dendrites and the axons.

Types of Neurons n Unipolar neurons Have one process extending from the cell body u Single, very short process splits into longer processes (a dendrite plus an axon) u Functionally, unipolar neurons are sensory neurons - conducting impulses into the central nervous system u
Types of Neurons n Bipolar neurons u These have two processes - one axon & one dendrite u These neurons are sensory u Biopolar neurons can be found in the retina of the eye.

Types of Neurons n Multipolar neurons u Have many (multi-) processes that extend from the cell body u Several dendrites plus a single axon u Functionally, these neurons are either motor (conducting impulses that lead to action e. g. Muscle contraction) or association (conducting impulses and permitting 'communication' between neurons within the central nervous system).
Neuroglial or Glial Cells n Function u Form myelin sheaths. Cells produce large membranous extensions that ensheath the axons in successive layers that are then compacted by exclusion of cytoplasm to form the myelin sheath. u The thickness of the myelin sheath (the number of wraps around the axon) is proportional to the axon's diameter.

Neuroglial or Glial Cells n Function continued, n n n Protect neurons (via phagocytosis) Regulating the internal environment of neurons in the central nervous system Glial cells are called Schwann cells in the PNS and oligodendrocytes in the CNS.

Neurons Dendrites normally conduct information towards the cell body. n A single neuron may receive input from tens of thousands of other neurons, and it simplifies these ionic changes and transmits a single new message through its axons to other cells. n

Neurons n Signals in the form of voltage changes normally flow from dendrites to soma to axon and finally to synapses on the next set of cells.
![Membrane Potential n The Nernst Equation u Veq = RT ln ([X]out) z. F Membrane Potential n The Nernst Equation u Veq = RT ln ([X]out) z. F](https://present5.com/presentation/a3d74a5b0f6ae6861f01b2ff0d443983/image-15.jpg)
Membrane Potential n The Nernst Equation u Veq = RT ln ([X]out) z. F u FR ([X]in) is the universal gas constant (8. 314 J. K-1. mol-1), T is the temperature in Kelvin F z is the valence of the ionic species. For example, z is +1 for Na+, +1 for K+, +2 for Ca 2+, -1 for Cl-, F F is the Faraday's constant (96485 C. mol-1). F [X]out is the concentration of the ionic species X in the extracellular fluid. [X]in is the concentration in the intracellular fluid. Note that the concentration unit must match that of [X]out. n
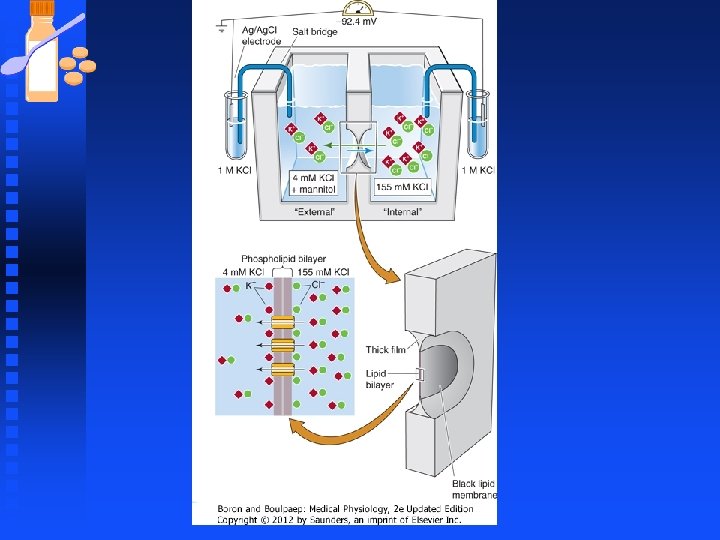

Membrane Potential n The Nernst equation u calculates the equilibrium potential (also referred to as the Nernst potential) for an ion based on the charge on the ion (i. e. , its valence) and the concentrations across the membrane u Temperature also influences the Nernst potential.

Membrane Potential n Nernst potential u will develop across a membrane if two criteria are met: (i) a concentration gradient exists across the membrane F (ii) if selective permeation pathways (i. e. , selective ion channels) exist that allow transmembrane movement of ions. F

Membrane Potential n The Goldman-Hodgkin-Katz Equation F This equation is used to determine the resting membrane potential in real cells, in which K+, Na+, and Cl- are the major contributors to the membrane potential

Membrane Potential u R is the universal gas constant (8. 314 J. K-1. mol 1). u T is the temperature in Kelvin (°K = °C + 273. 15). u F is the Faraday's constant (96485 C. mol-1). u p. K is the membrane permeability for K u For a typical neuron at rest, p. K : p. Na : p. Cl = 1 : 0. 05 : 0. 45.

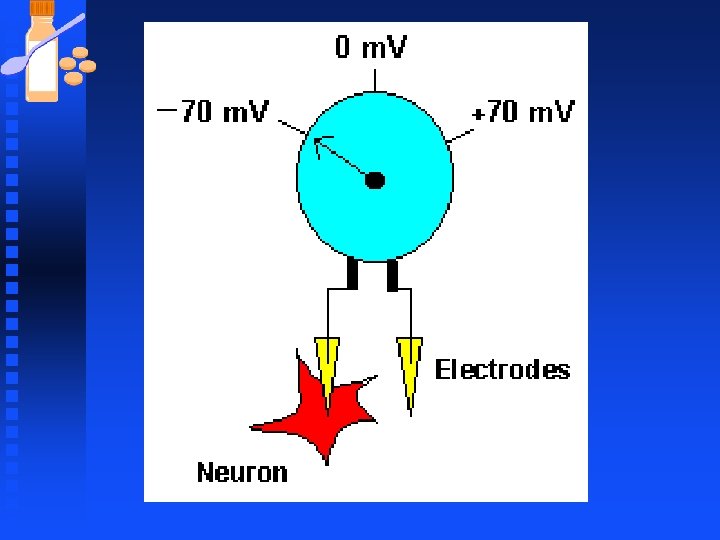
Membrane Potential n Determined mainly by Na+ and K+ u Established by sodium-potassium pump u Leaky potassium channels n (: How the Sodium-Potassium Pump. Animation Works).

Neurons n Excitatory input to a neuron usually generates an inward flow of positive charges (inward current) across dendritic membrane.

Neurons Since inside the neuron is negatively charged with respect to the outside, the inward flow of positive charges makes the membrane less negative and we say the membrane is depolarized. n Inhibitory input to the cell generates an outward current and we say the cell is hyperpolarized. n

Neurons n Sensory neurons are activated by environmental stimuli chemicals u mechanical deformation u light u n These stimuli activate ion channels which causes a flow of current.

Neurons n The flow of charge causes a change in membrane potential u n postsynaptic potential (PSP) if it generated at the postsynaptic membrane by a neurotransmitter. If it is generated at a sensory nerve ending by an external stimuli u called a receptor potential.

Neurons n If the neurotransmitter is excitatory and produces a depolarizing PSP u call an excitatory postsynaptic potential (EPSP). n If the neurotransmitter is inhibitory and produces a hyperpolarizing PSP u call an inhibitory postsynaptic potential (IPSP).

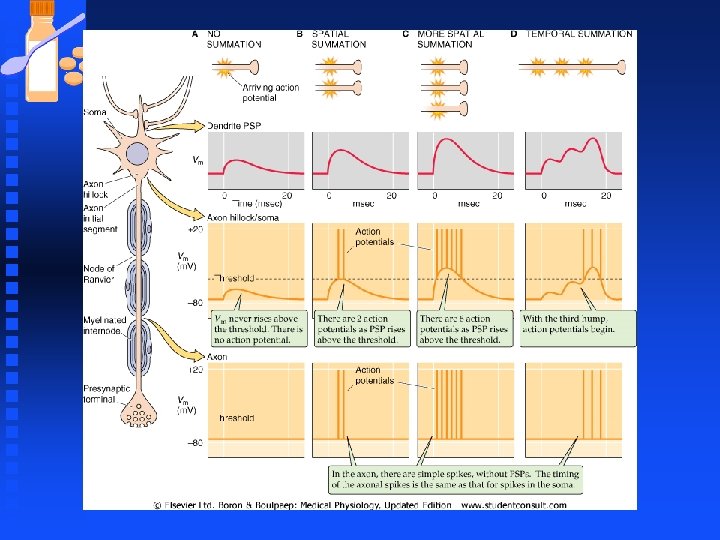
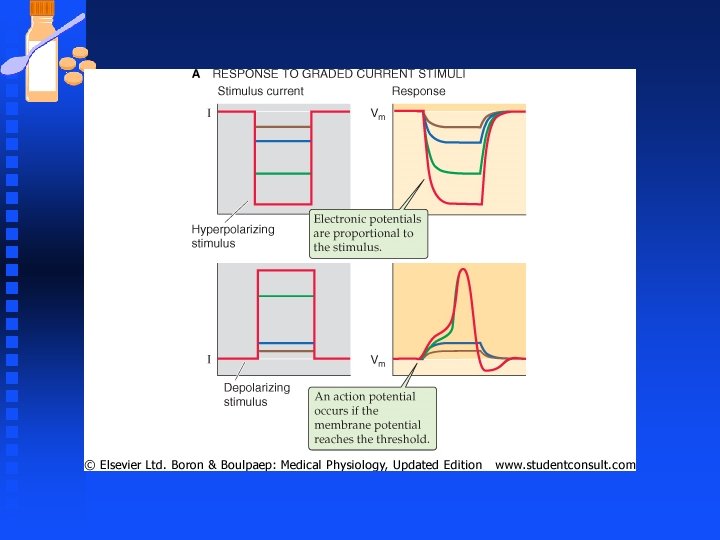
Neurons n Voltage changes are graded from small to large depending on the strength or quantity of input signal. n Stronger sensory signals generate larger receptor potentials , similarly more synapses activated together generate larger PSP. u Graded responses are a form of neural coding whereby the size and duration of the input are encoded as the size and duration of dendritic Vm

Neurons Synaptic potential generated at dendrites are communicated to the soma but with substantial attenuation of signals (leaky cables). n EPSPs from one dendrite may combine with EPSPs arriving by other dendrites on the cell; this behavior is a type of spatial summation and can lead to EPSPs that are substantially larger than those generated by a single synapse n
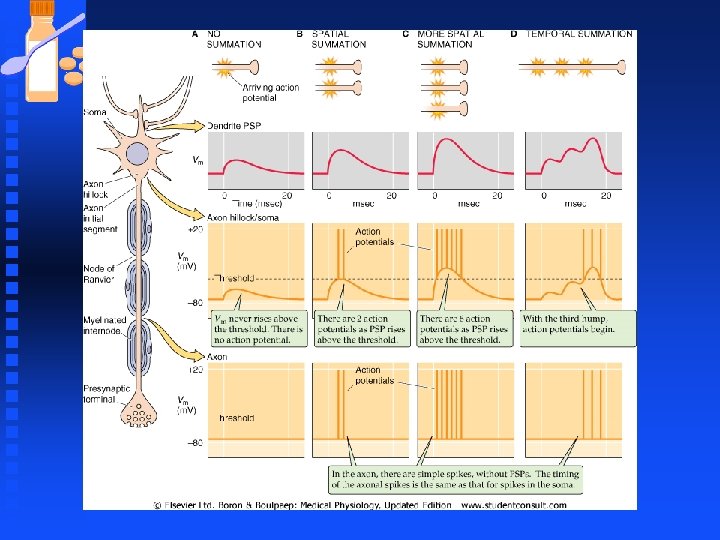

Neurons n Temporal summation u occurs when EPSPs arrive rapidly in succession when the first EPSP has not yet dissipated, a subsequent EPSP tends to add its amplitude to the residual of the preceding EPSP.
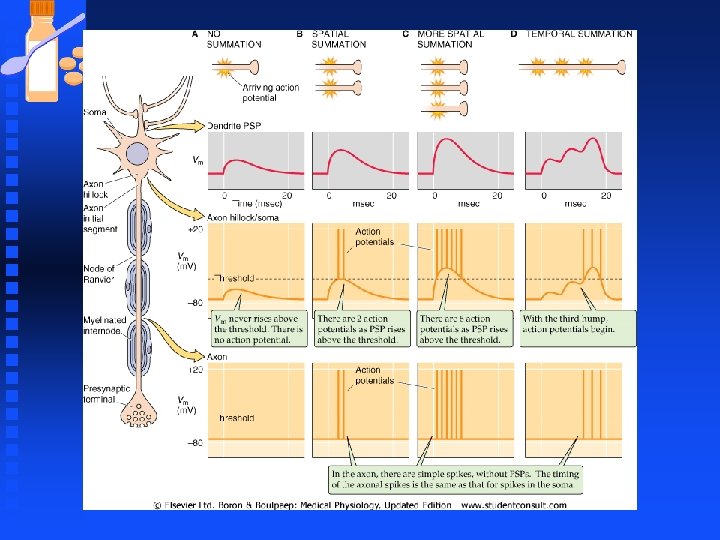

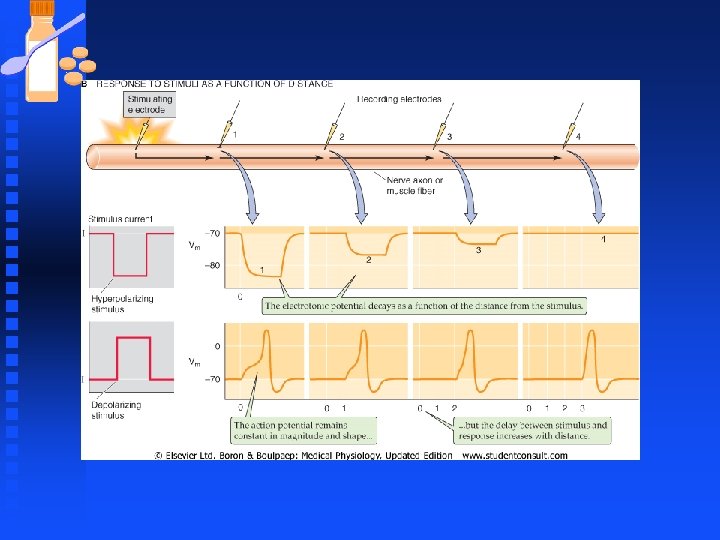
Neurons n Dendritic potential u dwindle with distance, if nothing else happens the depolarizing potential would dwindle back to resting membrane potential as they spread through the soma and down the axon. n Dendritic signal u Carried only a few millimeters, not adequate to wiggling a toe when the axon of the motor neuron stretches from spinal cord to the foot (1000 mm)

Neurons n Amplification u needed for certain inputs to generate effective signals to and from the CNS u provided in the form of regenerating action potentials if the Vm change in the soma is large enough to reach the threshold voltage. u Depolarization may trigger more than one action potential between soma and axon.

Neurons n Action Potentials large rapid fluctuation in Vm u Efficient, rapid and reliable way to carry a signal over long distances u Neurons convert the graded-voltage code of the dendrites (PSPs) to a temporal code of action potential in axons u
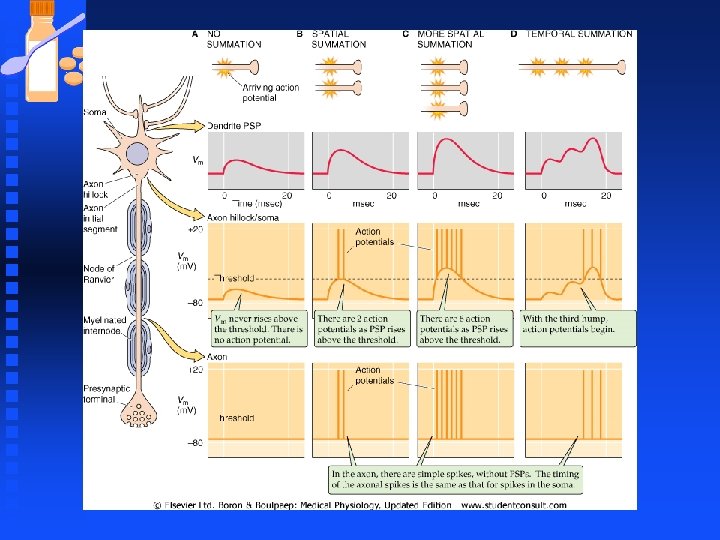

Neurons n Action Potentials u fixed amplitude not graded u have uniform shape u Neurons code info by varying number of spikes and timing so info may be encoded by the average rate of action potential firing, the total number of action potentials, their temporal pattern, or some combination. F Larger synaptic potentials in soma produce more frequent AP that last longer
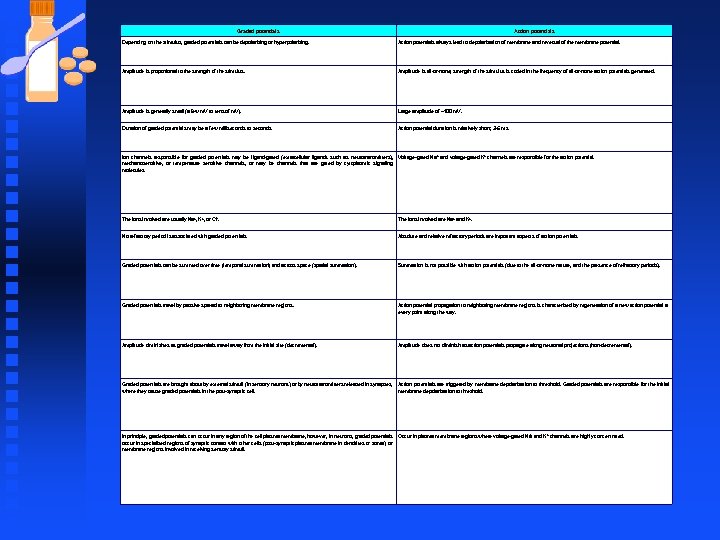
Graded potentials Action potentials Depending on the stimulus, graded potentials can be depolarizing or hyperpolarizing. Action potentials always lead to depolarization of membrane and reversal of the membrane potential. Amplitude is proportional to the strength of the stimulus. Amplitude is all-or-none; strength of the stimulus is coded in the frequency of all-or-none action potentials generated. Amplitude is generally small (a few m. V to tens of m. V). Large amplitude of ~100 m. V. Duration of graded potentials may be a few milliseconds to seconds. Action potential duration is relatively short; 3 -5 ms. Ion channels responsible for graded potentials may be ligand-gated (extracellular ligands such as neurotransmitters), Voltage-gated Na+ and voltage-gated K+ channels are responsible for the action potential. mechanosensitive, or temperature sensitive channels, or may be channels that are gated by cytoplasmic signaling molecules. The ions involved are usually Na+, K+, or Cl-. The ions involved are Na+ and K+. No refractory period is associated with graded potentials. Absolute and relative refractory periods are important aspects of action potentials. Graded potentials can be summed over time (temporal summation) and across space (spatial summation). Summation is not possible with action potentials (due to the all-or-none nature, and the presence of refractory periods). Graded potentials travel by passive spread to neighboring membrane regions. Action potential propagation to neighboring membrane regions is characterized by regeneration of a new action potential at every point along the way. Amplitude diminishes as graded potentials travel away from the initial site (decremental). Amplitude does not diminish as action potentials propagate along neuronal projections (non-decremental). Graded potentials are brought about by external stimuli (in sensory neurons) or by neurotransmitters released in synapses, where they cause graded potentials in the post-synaptic cell. Action potentials are triggered by membrane depolarization to threshold. Graded potentials are responsible for the initial membrane depolarization to threshold. In principle, graded potentials can occur in any region of the cell plasma membrane, however, in neurons, graded potentials occur in specialized regions of synaptic contact with other cells (post-synaptic plasma membrane in dendrites or soma), or membrane regions involved in receiving sensory stimuli. + Occur in plasma membrane regions where voltage-gated Na and K+ channels are highly concentrated.

Neurons n Action Potentials u The final output of the neuron is entirely encoded in the action potential u When APs reach axonal terminals, they may trigger the release of a neurotransmitter at the next set of synapses and the cycle begins again.
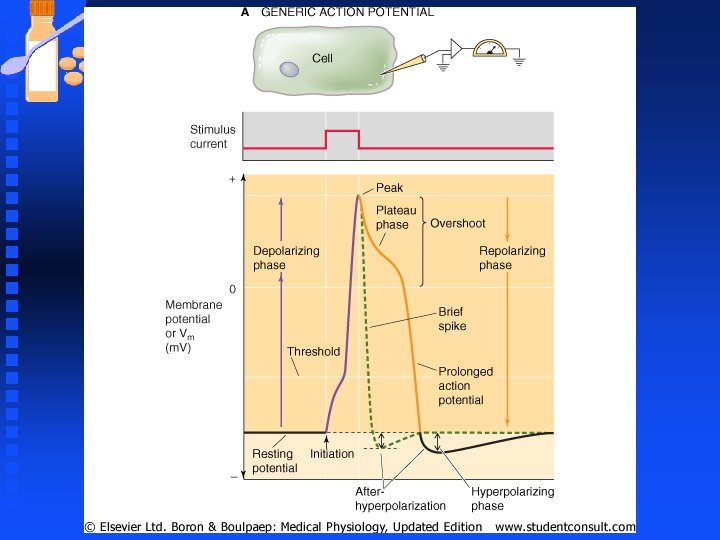

Phases of the Action Potential Resting phase n Depolarization to and beyond threshold n u The uncontrolled depolarization that takes place (spike phase of the action potential); is strictly a function of voltage-gated Na+ channels in neurons. u At rest (-70 m. V), the voltage-gated Na+ channels are closed, but begin to open at membrane potentials ranging from -40 to -50 m. V (threshold voltage, Vth).

Phases of the Action Potential n Depolarization phase continued, u Opening of Na+ channels leads to the entry of a large amount of Na+ ions into the cell. u Na+ entry favored by the Na+ concentration gradient, and the inside negative membrane potential. u Entry of Na+ into the cell brings about further depolarization. u Membrane depolarization further activates additional Na+ channels which, in turn, leads to the entry of more Na+ into the cell.

Phases of the Action Potential n Depolarization phase continued, u Therefore, a positive feedback loop is established, which leads to increasing entry of Na+ into the cell. This positive feedback loop is called the Hodgkin cycle

Phases of the Action Potential n Overshoot u Continued entry of Na+ into the cell leads to rapid depolarization of the cell (< 1 ms). Because rapid opening of Na+ channels leads to a rapid rise in membrane permeability to Na+, the membrane potential reverses its sign (goes from negative to positive) and approaches the equilibrium potential for Na+ (about +50 m. V). Reversal of the sign where the membrane potential becomes positive is referred to as overshoot

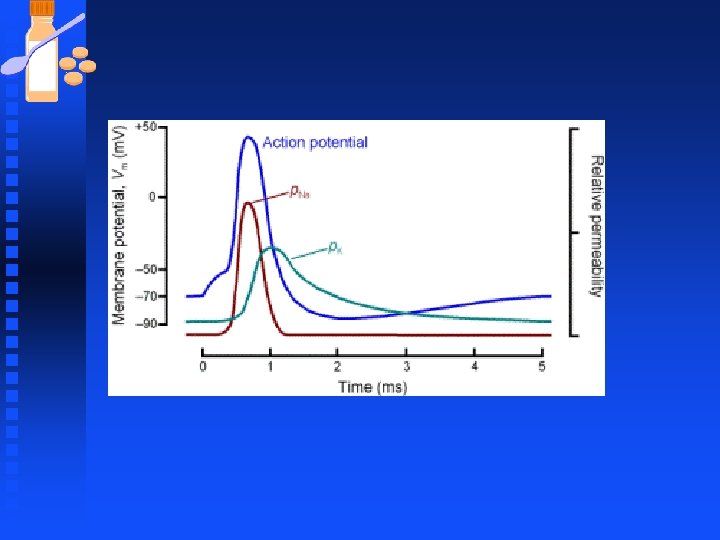
Phases of the Action Potential n Peak of the action potential u At the peak of the action potential, the membrane potential is close to VNa, but it never reaches VNa F voltage-gated Na+ channels begin to inactivate spontaneously very rapidly after opening F voltage-gated K+ channels become activated by membrane depolarization (also at around the threshold voltage of -40 to -50 m. V).

Phases of the Action Potential n Peak continued, u Activation of the voltage-gated K+ channels, however, is much slower than that of voltagegated Na+ channels (delayed rectifiers). Therefore, at the peak of the action potential, p. K is greater than its value when the neuron is at rest, and movement of K+ out of the cell opposes the depolarization caused by the movement of Na+ into the cell.

Phases of the Action Potential n Hyperpolarization u Once the peak of the action potential is reached, Na+ channels inactivate, and as a result p. Na falls rapidly with time, and approaches its value at rest u p. K is still becoming larger because of Delayed opening of voltage-gated K+ channels u Balance of ion flow across the membrane is in favor of K+ moving out of the cell. u Movement of K+ out of the cell brings about rapid repolarization of the membrane back to the resting value

Phases of the Action Potential n Hyperpolarization phase u p. K remains elevated for some time even after Vm has reached the resting value. Therefore, continued movement of K+ out of the cell causes a membrane hyperpolarization (i. e. , more negative than Vrest). This phase is commonly referred to as the hyperpolarizing afterpotential or simply hyperpolarization (or undershoot)

Phases of the Action Potential n Return to resting phase u K+ channels do not inactivate. They close simply because the membrane potential becomes more negative than the threshold potential (the potential at which Na+ and K+ channels become activated). u Thus, the repolarization and hyperpolarization that is caused by movement of K+ out of the cell through the voltage-gated K+ channels, also causes the closing of the same voltage-gated K+ channels.

Animation: Voltage-gated channels)

Refractory periods: n ABSOLUTE – (1 -2 ms) u The period from the initiation of the action potential to immediately after the peak is referred to as the absolute refractory period (ARP) u During this period a second stimulus (no matter how strong) will not produce an action potential u Inactivation of Na+ channels is solely responsible for the absolute refractory. u When Na+ channels are inactivated, they cannot be immediately opened again (3 -4 ms for all Na+ channels to come out of inactivation), so additional depolarizing stimuli do not lead to new action potentials.
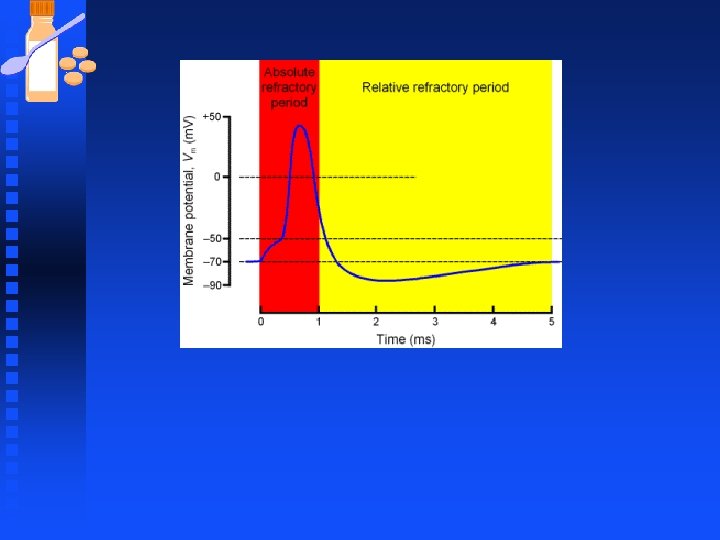

Refractory periods: n RELATIVE u Period during which a stronger than normal stimulus is needed in order to elicit an action potential u Both Na+ channel inactivation and the greater than resting p. K value are responsible for the relative refractory period. Open potassium channels allow continued K+ flow out of the cell and this tend to oppose any depolarization caused by opening of Na+ channels that have recovered from inactivation. Hence stronger than normal depolarization signal is needed to get AP.

Refractory periods: n RELATIVE u the nerve cell membrane becomes progressively more 'sensitive' (easier to stimulate) as the relative refractory period proceeds u So, it takes a very strong stimulus to cause an action potential at the beginning of the relative refractory period, but only a slightly above threshold stimulus to cause an action potential near the end of the relative refractory period
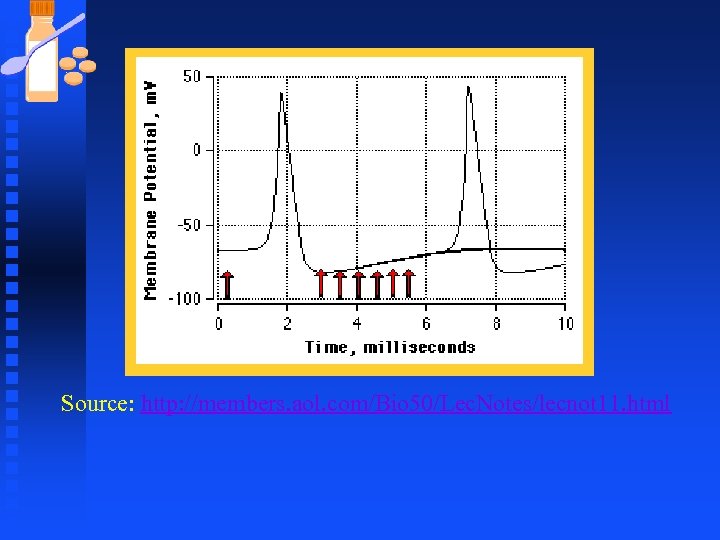
Source: http: //members. aol. com/Bio 50/Lec. Notes/lecnot 11. html

Refractory periods: n ABSOLUTE u The absolute refractory period places a limit on the rate at which a neuron can conduct impulses n RELATIVE u The relative refractory period permits variation in the rate at which a neuron conducts impulses

Refractory periods: n Such variation is important because it is one of the ways by which our nervous system recognizes differences in stimulus strength, e. g. , dim light = retinal cells conduct fewer impulses per second vs. u brighter light = retinal cells conduct more impulses per second u

Neurons n Signal Conduction in Dendrites u Dendrites arborize through brain tissue to collect information in the form of synaptic input. They have a great diversity of shape and account for up to 99% of a neuron’s membrane. n Dendrites Attenuate Synaptic Potentials

Neurons n Dendritic membranes u have Voltage-Gated Ion Channels influence their signaling properties u Most dendrites have a low density of voltagegated channels that may amplify synaptic signals by adding additional inward current as the signal propagate from distal dendrite towards the soma u

Neurons n Dendritic membranes u If the membrane has voltage-gated channels that are able to carry more inward current (Na+ or Ca 2+) under depolarizing conditions, a sufficiently strong EPSP would drive Vm into the activation range of the voltage-gated channels. u These channels would open and the additional inward currents would add to that generated initially by the synaptic channels

Neurons n Dendritic membranes u Thus synaptic signals would fall off much less steeply than in a passive dendrite u voltage gated channels can be distributed all along the dendrite and thus amplify the signal along the entire dendritic length or they can cluster at particular sites.

Neurons n Dendritic membranes u Some dendrites such as those of the Purkinje cell ( large output neuron of the cerebellum) have such a high density of voltage gated ion channels that they can produce action potentials just like axons can u The action potentials are mediated by voltagegated calcium channels.

Neurons n Dendritic membranes u The calcium spikes can sometimes propagate toward or even into the soma but they do not continue down the axons u They trigger fast Na+-dependent AP that are generated by voltage-gated Na+ channels in the soma and initial segment. The Na+ spikes carry the signal along the axon in the conventional way

Neurons n Dendritic membranes u Dendrites of pyramidal cells of the cortex can also generate spikes dependent on Ca, Na or both u Another advantage of voltage-gated channels in dendrites may be the selective boosting of highfrequency synaptic input u Passive dendrites attenuate signals of high frequency more than those of low frequency. Dendrites with the appropriate voltage-gated channels will be better able to communicate high -frequency synaptic input.

Neurons n Dendritic membranes u Electrical signals from dendrites converge and summate at the soma. Action potential often appear first at the axon hillock and initial segment of the axon.

Neurons n Neurons can transform a simple input into a variety of output patterns. u No two types of neurons behave the same u There is variability in the shape and height of the action potential. Most neurons generate AP by fast voltage-gated Na channels generate a strong inward current that depolarizes the membrane from rest, usually in the range of -60 to -80 m. V, to a peak that is usually between +10 and +40 m. V.

Neurons This is the upstroke of the Ap. The Na+ channel quickly inactivate and close, and certain K+ channels (often voltage-gated, delayed outward rectifier channels) open and thus cause Vm to fall and terminate the spike. n In some neurons there is an additional fast Ca 2+ current that frequently occur and if it is large enough, broaden the spike duration. n

Neurons The greatest variability occur in the repolarization phase n Many neurons are repolarized by other voltage-gated K+ currents in addition to the delayed outward rectifier K+ current, and some also have a K+ current carried by channels that are rapidly inactivated by the combination of membrane depolarization and a rise in [Ca 2+]i. n

Neurons n More dramatic variations occur in the repetitive spiking patterns of neurons, observed when the duration of a stimulus is long. u Apply a simple, continuous stimulus ( a current pulse) to each neuron and measure its output ( number and pattern of AP fired at the soma) u The current pulse is the equivalent of a steady, strong input of excitatory synaptic currents. The transformation from stimulus input to spiking output can take many different forms.

Neurons u Some cells generate a rapid train of AP that do not adapt (spikes occur at a regular interval throughout the current pulse). u Other cells fire rapidly at first but then adapt strongly i. e. spikes gradually become less frequent during the current pulse u Some cells fire a burst of AP and then stop firing altogether, while others generate rhythmic bursts of AP that continue as long as the stimulus. cell.

Neurons n These behavior are distinctive to each neuron type. The neuron firing pattern is determined by the membrane properties of the cell u Rhythmically bursting cells may be involved in central circuits that generate rhythmic motor output for locomotion or respiration u Cells that secrete peptide hormone in hypothalamus have rhythmic behavior.

Neurons n Spiking patterns in neurons determined by a large set of ion channel types that have slow kinetics compared to the fast Na+ and K+ channels that shape the spike. u A neuron that has only fast Na+ channels and delayed rectifier channels will generate repetitive spikes when presented with a long stimulus u Pattern of spikes is regular over time (cerebral cortical interneurons) u
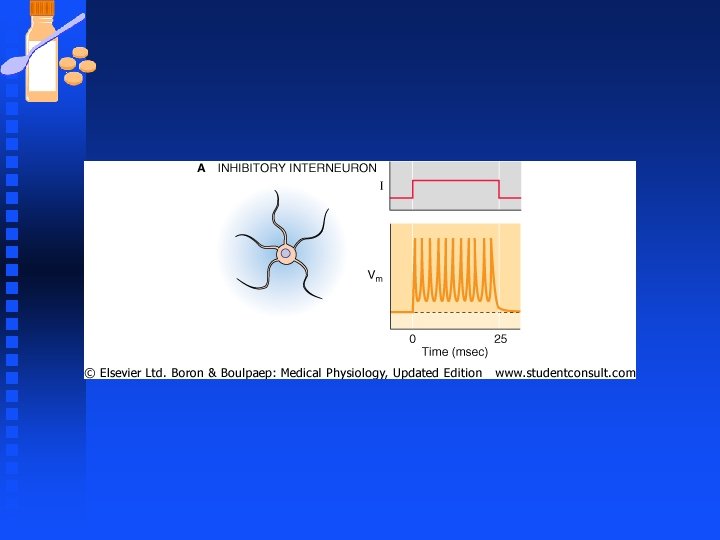

Neurons n Spiking patterns in neurons u If the neuron also have another set of K+ channels that activate only very slowly, the spiking pattern becomes more time dependent: the spike frequency may initially be very high but it adapts to progressively lower rates as a slow K+ current turns on to counteract the stimulus (small pyramidal cells)
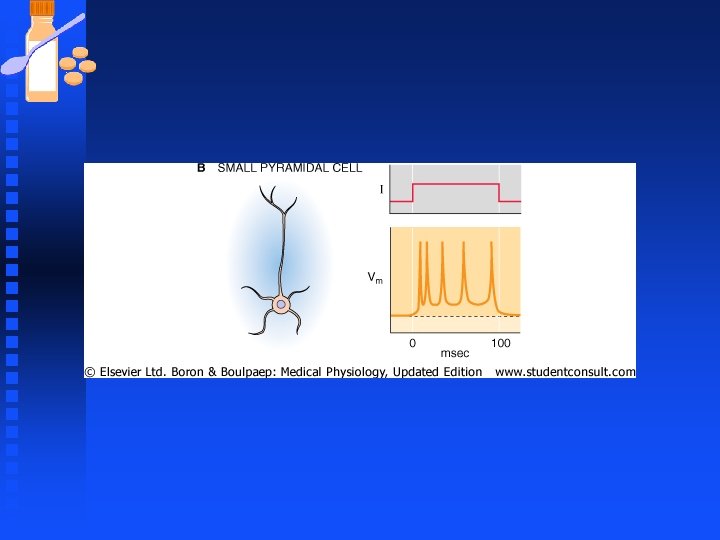

Neurons n Spiking patterns in neurons u A neuron by exploiting the interplay between two or more voltage gated currents can generate spontaneous rhythmic bursting even without ongoing synaptic activity to drive it (large pyramidal neurons).
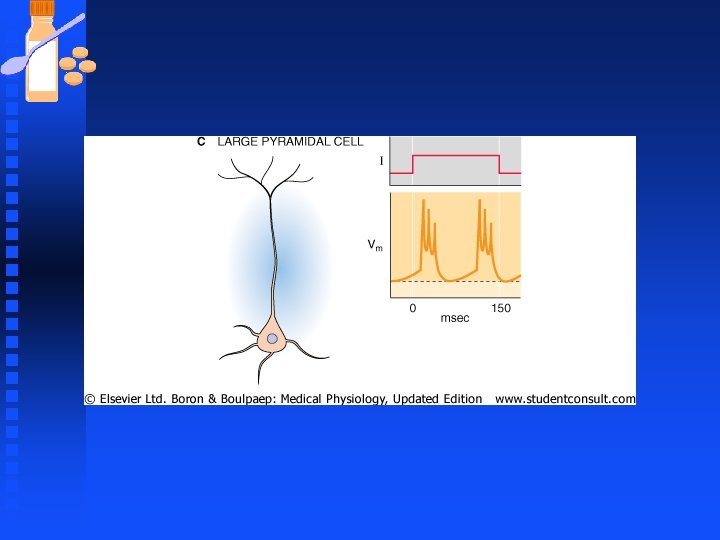

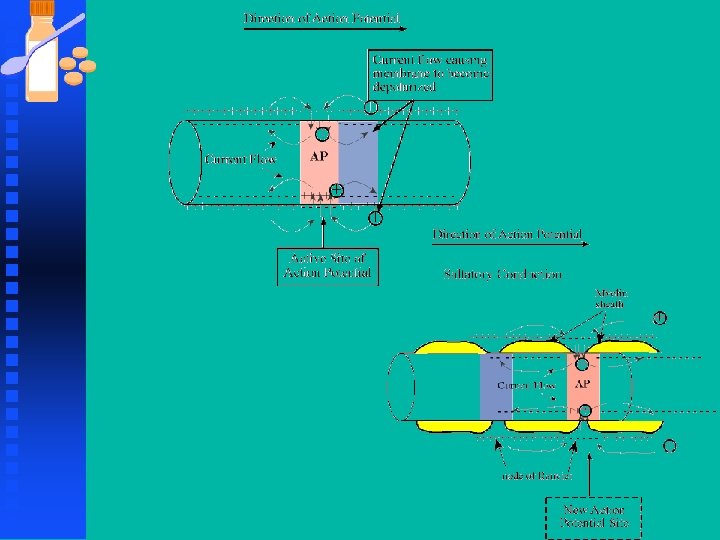
Neurons n Axonal Conduction u Axons carry the computed signals ( series of AP) from one place in the brain to the next rapidly, efficiently and reliably. u Some axons are thin and unmyelinated and slow u Others are myelinated with nodes of Ranvier for fast and reliable conduction over long distances.
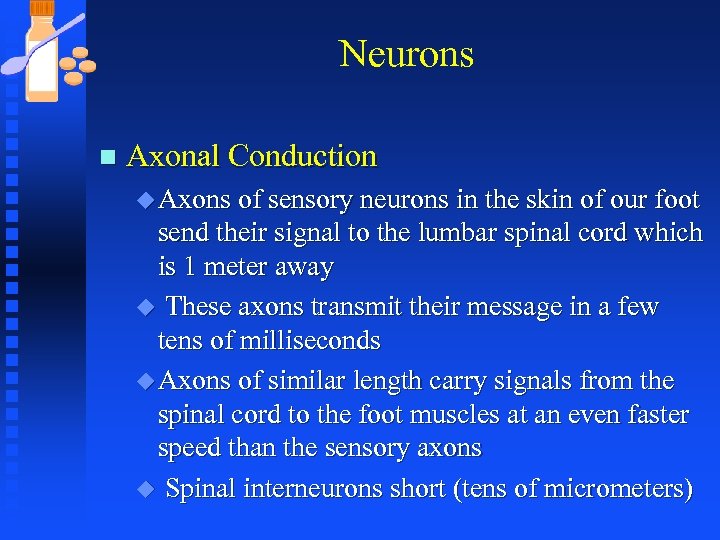
Neurons n Axonal Conduction u Axons of sensory neurons in the skin of our foot send their signal to the lumbar spinal cord which is 1 meter away u These axons transmit their message in a few tens of milliseconds u Axons of similar length carry signals from the spinal cord to the foot muscles at an even faster speed than the sensory axons u Spinal interneurons short (tens of micrometers)

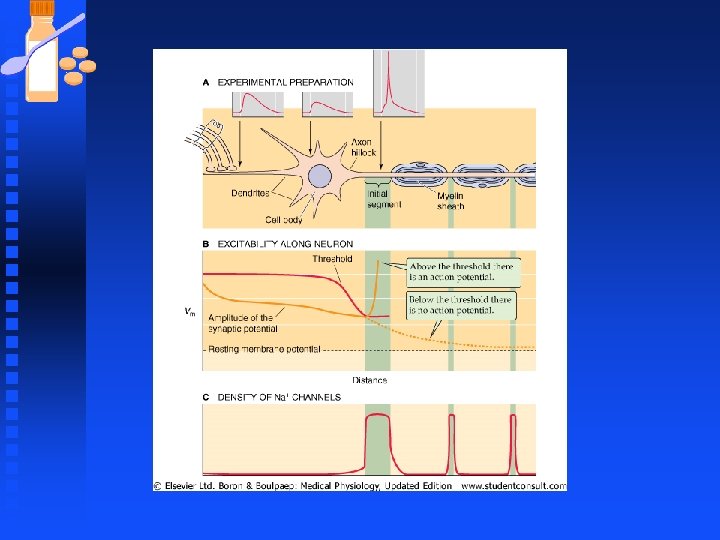
Neurons n Action potential initiated at the initial segment u The many graded synaptic potentials carried by numerous dendrites converge at the soma and generate one electrical signal. EPSPs evoked in the dendrites propagate down to and through the soma and trigger an action potential in the initial segment

Neurons n Action potential initiated at the initial segment u The action potential can propagate in two directions: forward (orthodromically) into the axon with no loss of amplitude, and backward into the soma and dendrite with strong attenuation. Forward propagation carries the signal to the next set of neurons.

Neurons n Action potential u Action potential is usually triggered in the initial segment because threshold is uniquely low there since there is a high density of voltagegated Na+ channels (in comparison to the soma and dendrites) u There is approximately 1000 fold higher density of channels in the initial segment.

Neurons n Action Potential u Conduction velocity of a myelinated axon increases linearly with diameter. u The larger the diameter of an axon, the faster its conduction velocity. u

Neurons n Action Potential u Myelination confers speed advantages and efficiency. u An unmyelinated axon has to increase its diameter ~ 50 X to obtain the speed of a myelinated axon u More myelinated axon can be packed into the volume occupied by an unmyelinated axon.

Neurons n Action Potential u At diameters below 1µm unmyelinated axons in the peripheral nervous system conduct more rapidly than myelinated ones do u The thinnest axons of the peripheral sensory nerves (C fibers) are ~1µm or less and are all ummyelinated. u Axons with diameter larger than 1µm are all myelinated.

Neurons n Action Potential u The largest axons occupy more room and are expensive to synthesize and maintain metabolically and so these are used sparingly u They are used only to carry sensory information about the most rapidly changing stimuli over the longest distances (e. g. stretch receptors in muscles, mechanoreceptors in tendons and skin) or finely coordinated contraction of muscles.

Neurons n Action Potential u The thin C fibers are slow and are mainly sensory axons related to chronic pain (eg. dental pain) and temperature for which speed of message is not as critical u In the CNS form and function is not so closely related u Many central myelinated axons are as thin as 0. 2 µm, while very few myelinated central axons are larger than 4 µm in diameter.

Neurons n Action Potential u In normal central axons Nav 1. 6 channels populate the nodes of Ranvier at a density of 1000 to 2000 channels/ µm 2 u Between 2 and 200 channels/µm 2 in unmyelinated axons u K+ channels are less important in myelinated axons than they are in most other excitable membranes, and very few are present in the nodal membranes.

Neurons n Action Potential u Some clinical disorders selectively damage or destroy the myelin sheath and leave the axonal membranes intact but bare u Most common demyelinating disease of the CNS is multiple sclerosis which is characterized by zones of demyelination in the brain and spinal cord. u Landry-Guillain-Barre’ syndrome is an inflammatory disorder of the PNS that may rapidly incapacitate but may end in recovery.

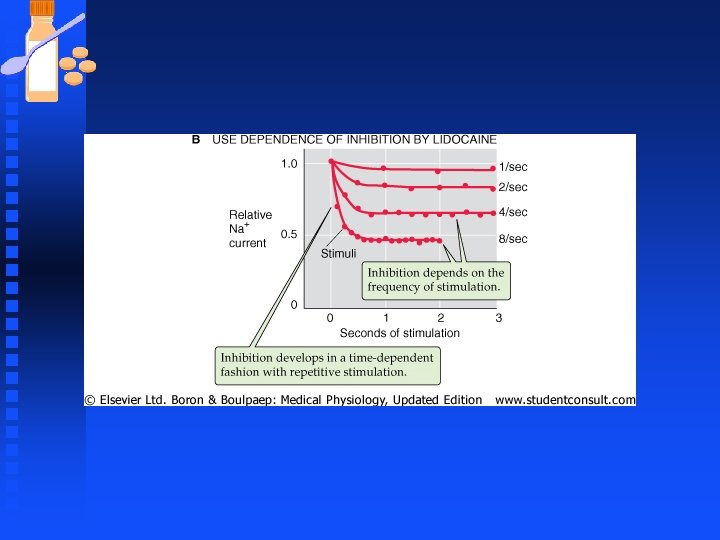
Pharmacological Inhibition of Na and K Channels n Voltage-gated Sodium channels u Blocked by Tetrodotoxin (TTX) obtained from ovaries of puffer fish u Local anesthetics such as lidocaine (Xylocaine®) and procaine (Novacaine®) F Local anesthetics reversibly block nerve impulse generation and propagation in a “use dependent manner F Use dependence occurs because the drug binds most effectively only after the Na channels have already opened. Enhance inhibition of impulses at sites where repetitive firing of AP takes place.

Pharmacological Inhibition of Na and K Channels n Voltage-gated Sodium channels u Saxitoxin (STX) from “red tide” dinoflagellates F Agent responsible for paralytic shellfish poisoning, caused by ingestion of toxic shellfish that have accumulated STX-producing plankton u μ-conotoxin blocks muscle Na channels

Pharmacological Inhibition of Na and K Channels n Potassium Channels u Tetraethylammonium (TEA) blocks potassium currents. u 4 -aminopyridine blocks A-type K+ currents
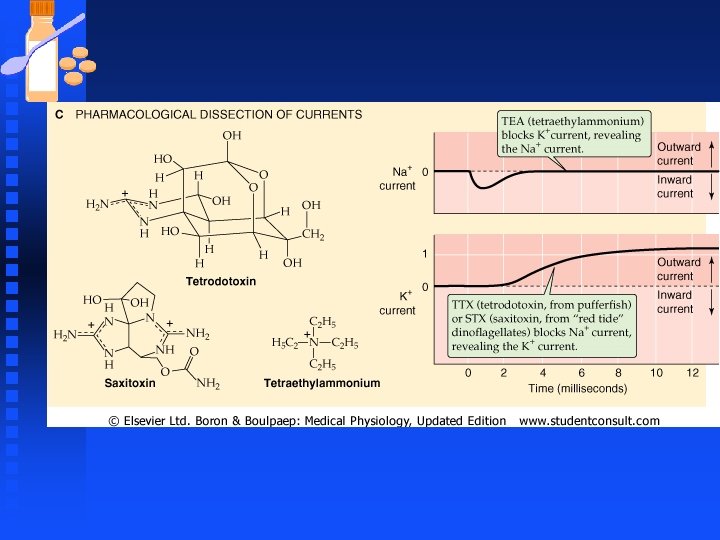

K Channels K channels determine resting potential and regulate the frequency and termination of AP n Types of K channels n u Kv---voltage-gated K channels u Small conductance KCa channels ( Ca 2+-activated K+ channel) including Sk. Ca and Ik. Ca channels u Large conductance KCa channels (Ca 2+-activated K+ channels, including BKCa and Na+-activated K+ channels)

K Channels n Types of K channels continued, u Kir channels (inward rectifier K+ channels) u K 2 P channels (two pore K+ channels) n Kv (Shaker-Related ) K+ channels mediate both the delayed outward rectifier current and the transient A-type current. u A-type currents can determine the interval between successive spikes and the timing of AP
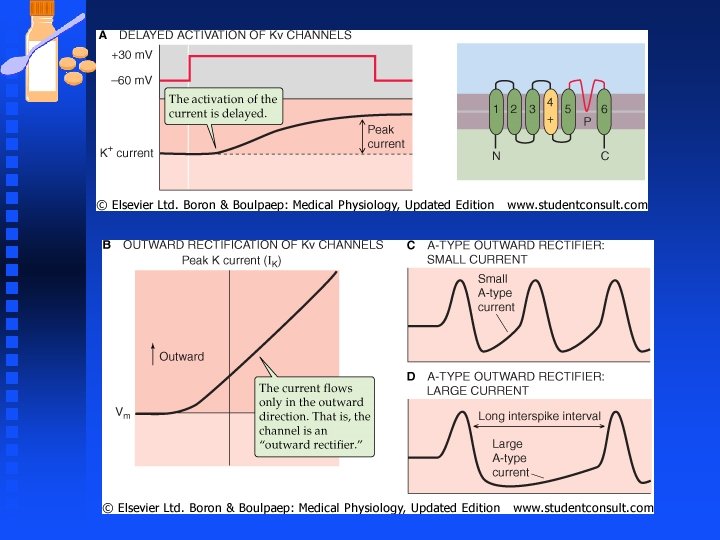

K Channels n Kca channels mediate Ca 2+-activated K+ currents u Opening probability (Po ) increases at positive values of Vm u Po increases as [Ca 2+]i increases u A particular type of KCa channel called maxi-KCa or BK has large conductance (~300 p. S) u KCa channels stabilizes the cell by counteracting repetitive excitation and intracellular Ca 2+ loading
K Channels n KCa channels mediate the afterhyperpolarizing phase of AP in cell bodies of various neurons u Implicated in terminating bursts of AP in bursting neuronal pacemaker cells u [Ca 2+]i increases gradually during repetitive firing and triggers the opening of KCa channels, which results in hyperpolarization and a quiescent interburst period that lasts until Ca pumps lowers [Ca 2+]i u Relaxes smooth muscles

K Channels n Ik. Ca and SKCa are voltage insensitive and are activated by Ca binding protein calmodulin u In some cells these currents regulate AP firing frequency by participating in AP repolarization and afterhyperpolarization.

K Channels n Kir channels (inward rectifier or anomalous rectifier) u Conducts more K+ current in the inward than outward direction u Clamp the Vm close to the equilibrium potential for K+ and prevent excessive loss of intracellular K during repetitive activity and long-duration AP u Lack the S 1 -S 4 voltage-sensing domain so not steeply activated by voltage

K Channels n Kir channels u Conducts K+ current only inwardly in presence of cytosolic magnesium u Inward rectification is due to intracellular block of the channel by Mg 2+ u Mg 2+ binds in a voltage-dependent manner to the inner mouth of the channel. Positive voltage favors binding of Mg 2+ while negative Vm pulls Mg 2+ out of the channel. u Incoming K+ displaces Mg 2+ favoring K+ influx over efflux
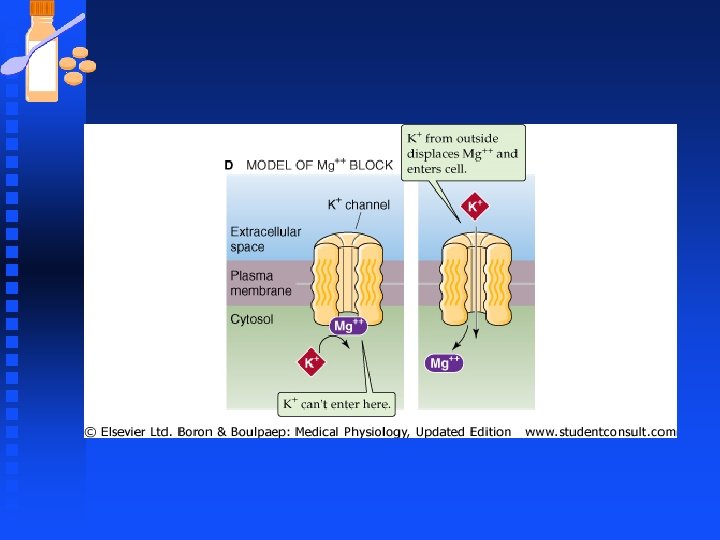

K Channels n G protein-activated inwardly rectifying K+ channels (GIRKs) u Subfamily of Kir u Regulated by βγ subunit of heterotrimeric G proteins e. g stimulation of the vagus nerve slows heartbeat because vagal Ach binds to postsynaptic muscarinic receptors in the heart that are G-protein coupled. Binding releases βγ which diffuses to GIRK, binds and causes channel opening, hyperpolarization , slowing rate at which Vm reaches AP and lower heart rate.

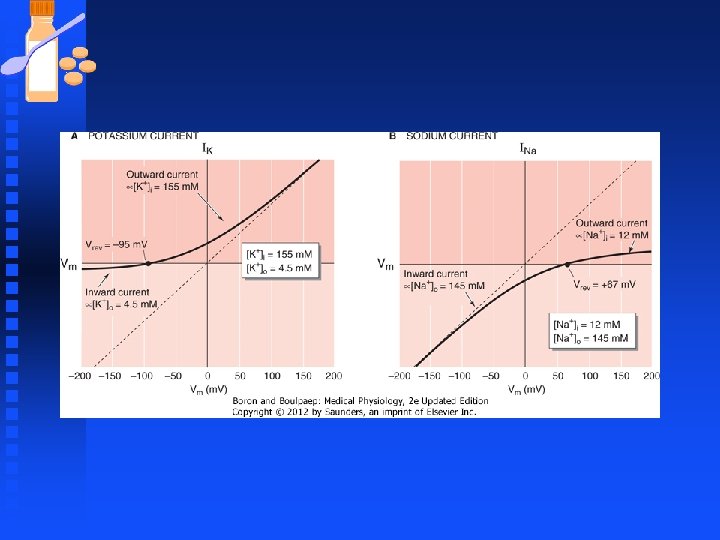
Reference n Cell, Vol. 83, 443 -449, November 3, 1995 u u u n n Identification of Domains Conferring G Protein Regulation on Inward Rectifier Potassium Channels Maya T. Kunkel and Ernest G. Peralta Department of Molecular and Cellular Biology Harvard University Cambridge, Massachusetts 02138 Summary Cardiac m 2 muscarinic acetylcholine receptors reduce heart rate by coupling to heterotrimeric (afly) guanine nucleotide-binding (G) proteins that activate IKach, an inward rectifier K+ channel (IRK). Activation of the GIRK subunit of I x. ACh requires GBv subunits; however, the structural basis of channel regulation is unknown. To determine which sequences confer GBu regulation upon IRKS, we generated chimeric proteins composed of GIRK and RB-IRKS, a related, G protein-insensitive channel. Importantly, a chimeric channel containing the hydrophobic pore region of RB-IRK 2 joined to the amino and carboxyl termini of GIRK exhibited voltageand IRKS help set the resting potential of a cell near the K+ equilibrium potential (EK) by conducting substantial inward K+ currents at membrane potentials negative to EK and small outward currents at voltages positive to EK (Hille, 1992).
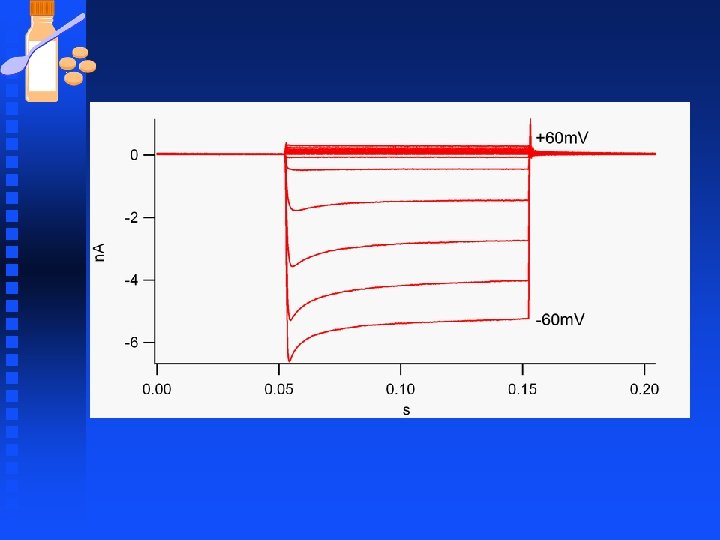

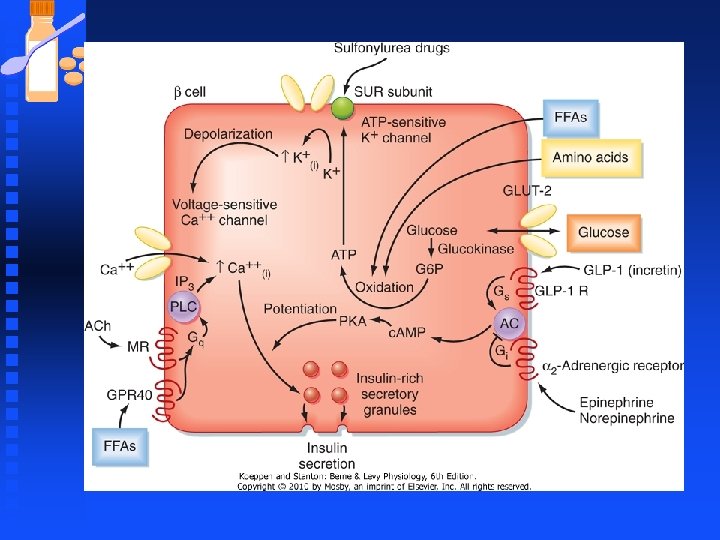
K Channels n G protein-activated inwardly rectifying K+ channels (GIRKs) u Some GIRK channels activated by membrane phospholipid PIP 2 u G-protein coupled receptors that activate phospholipase C release PIP 2, which activates GIRK channels. u Another member of the Kir K+ channels is the KATP channels F Inhibited by intracellular ATP F Activated by ADP

K Channels u Another member of the Kir K+ channels is the KATP channels F Inhibited by intracellular ATP F Activated by ADP u Link between cellular metabolism , membrane excitability and K+ transport. u If ATP levels fall because of O 2 deprivation, KATP channels open and hyperpolarize the cell to suppress firing of action potential, thereby reducing energy expenditure.

K Channels n Two pore or K 2 P channels u Implicated in forming resting membrane potential u Activated by various stimuli e. g membrane stretch, PIP 2, heat, intracellular p. H, general anesthetics.

Synaptic Transmission n Synapse u Specialized gap region between two apposing cells u point of impulse transmission between neurons; impulses are transmitted from pre-synaptic neurons to post-synaptic neurons u Synaptic transmission is the process of cell to cell transfer of electrical signals
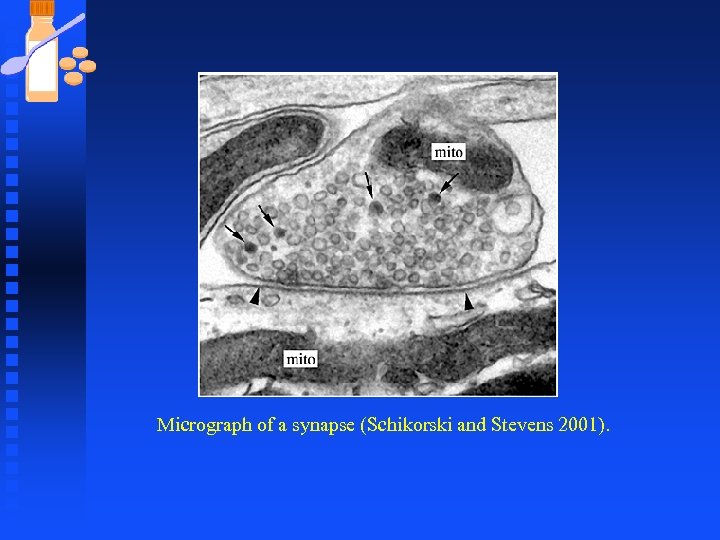
Micrograph of a synapse (Schikorski and Stevens 2001).

Synaptic Transmission Synapses are small (1µm) and presynaptic terminals appear in light microscope as swellings called bouton n Most presynaptic terminals arise from axons and can contact virtually any part of the neuron n Contact site and direction of communication determine the way a synapse is named. n
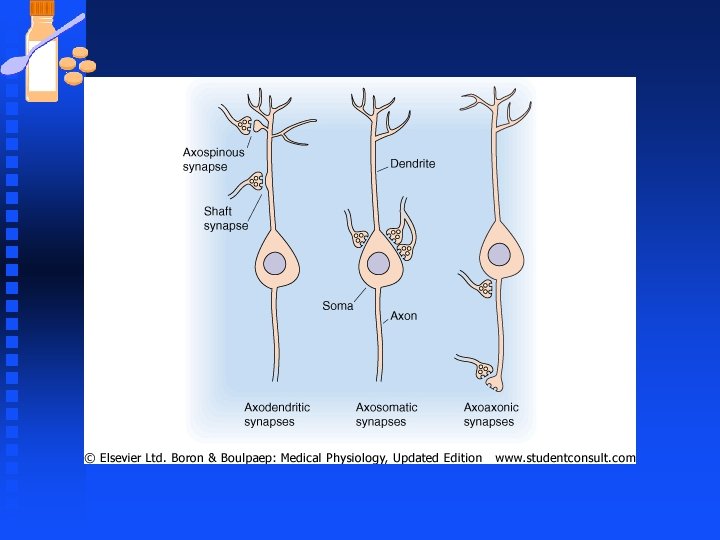

Synaptic Transmission n Synapse u Electrical synapses provide direct electrical continuity between cells by means of gap junctions u Chemical synapses link cells together by a neurotransmitter released from one cell and diffuses to another.
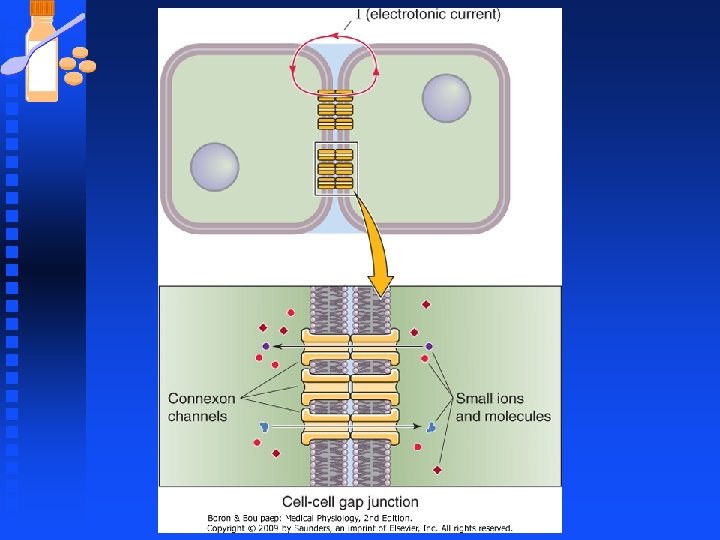

Synaptic Transmission Neurotransmitters can activate metabotropic receptors or inotropic receptors n Glutamate receptors that are ion channels n u Inotropic receptors u Cause rapid opening of ion channels n Glutamate receptors coupled to G proteins u Metabotropic receptors u Produce active α and βγ subunits which initiate cellular responses by interaction with ion channels or second messenger systems

Synaptic Transmission n Excitatory neurotransmitters at neuronal synapses u Glutamate u Aspartate n Inhibitory neurotransmitters u γ-aminobutyric acid (GABA) u Glycine u beta-endorphin, which results in decreased pain perception by the CNS u Other neuronal transmitters include Ach, norepinephrine, serotonin, histamine, peptides

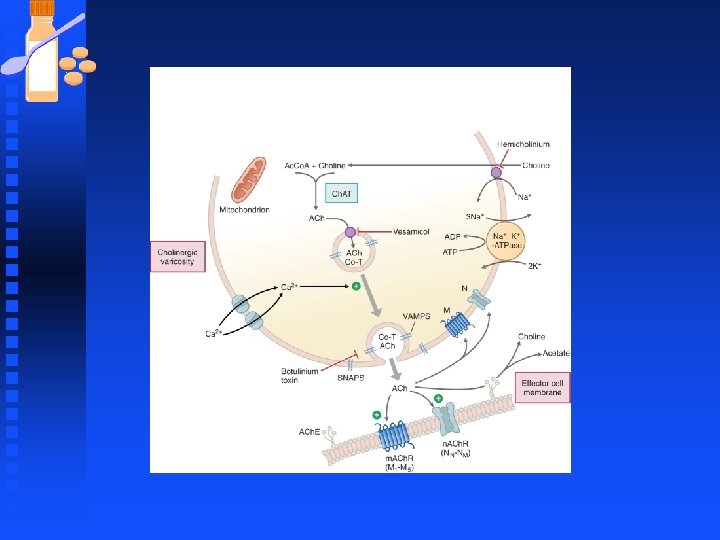
Synaptic Transmission n Steps of synaptic transmission u Packing of NT molecules into vesicles that concentrate at presynaptic terminals u AP depolarizes presynaptic membrane u Depolarization opens voltage-gated Ca 2+ channels and increase [Ca 2+]I u Increased [Ca 2+]I triggers fusion of vesicles with the presynaptic membrane. Synaptotagmin plays a role.

Synaptic Transmission n Steps of synaptic transmission u Transmitter is released into the extracellular space in quantized amounts and diffuse passively across the synaptic cleft u Some transmitter molecules bind to receptors on postsynaptic membrane to cause an event u Transmitter molecules diffuse away and are cleared by diffusion, enzymatic degradation, or active uptake into cells
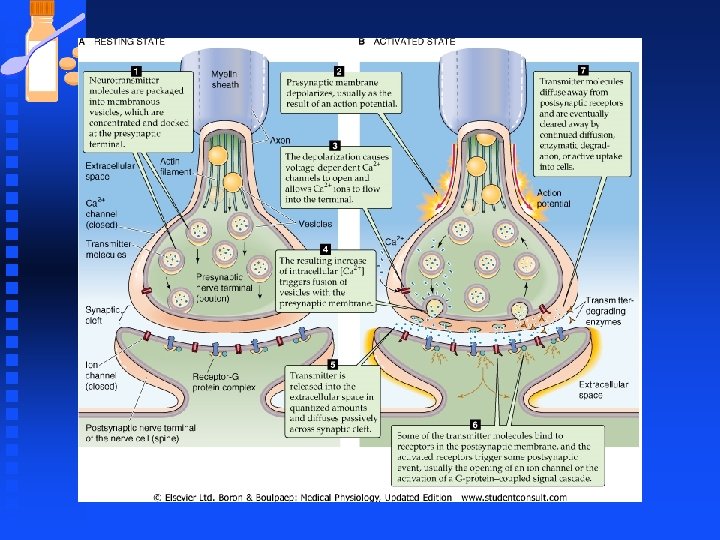
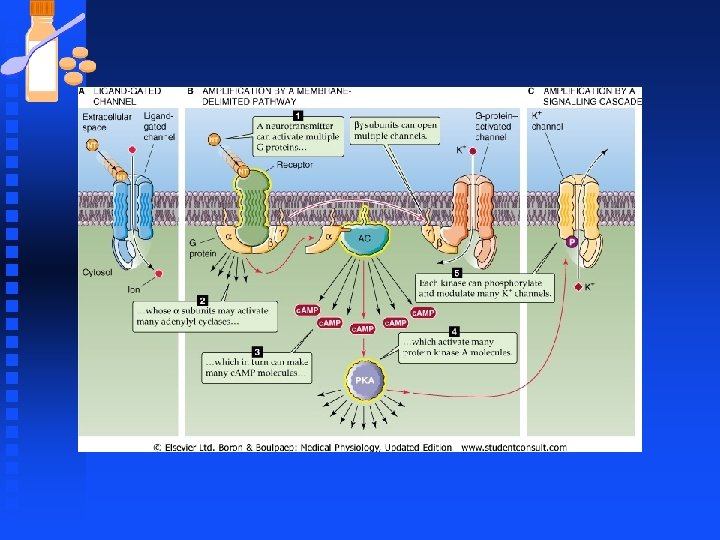

Synaptic Transmission n Neurotransmitters can activate Ionotropic or metabotropic receptors u NT receptors transduce info by F Ligand-gated ion channels F G-protein-coupled receptors u Ach and glutamate are agonists for both types of receptors.

Synaptic Transmission n Glutamate receptors u Glutamate receptors that are ions channels are called ionotropic receptors. u Glutamate receptors that are coupled to G proteins are called metabotropic receptors u These receptors determine the ultimate functional response to transmitter release

Synaptic Transmission n Glutamate receptors u Activation of an ionotropic receptor cause rapid opening of ion channels leading to depolarization or hyperpolarization of postsynaptic membrane u Ionotropic responses mediate fast ionic synaptic responses (milliseconds).

Synaptic Transmission n Glutamate receptors u Activation of metabotropic receptors results in production of active α and βγ subunits, which initiate a wide variety of cellular responses. u Metabotropic receptors mediate slow, biochemically mediated synaptic response (seconds to minutes)
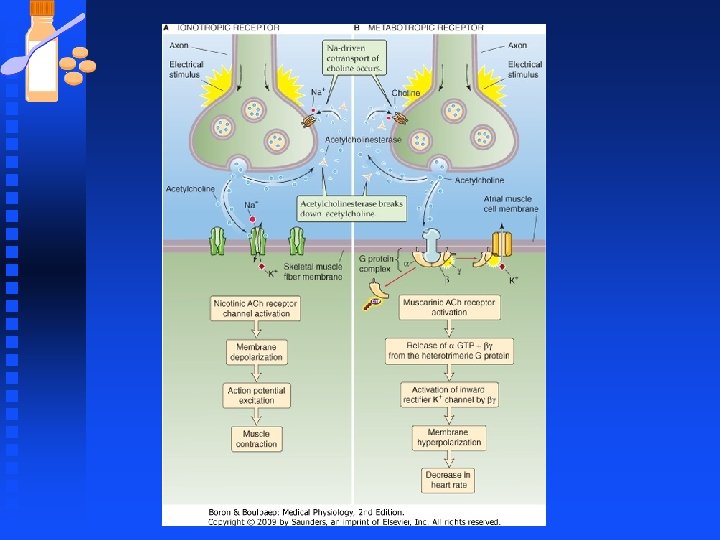

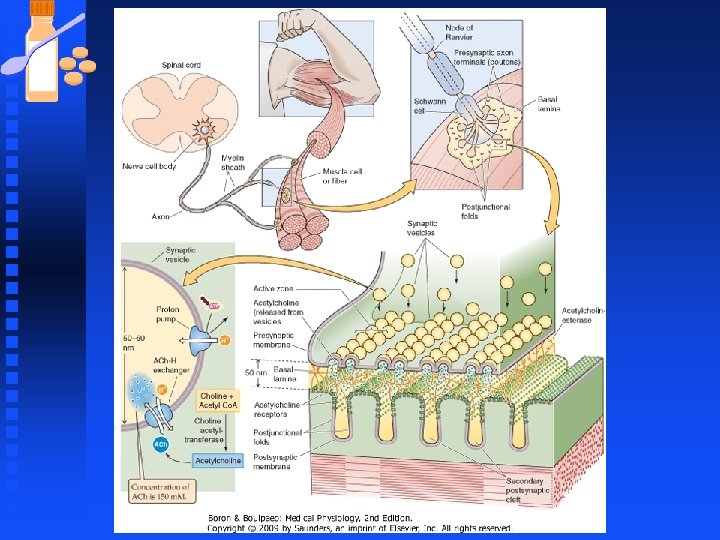
Synaptic Transmission at Neuromuscular Junction n Synapse between motor neurons and skeletal muscle u Motor neurons have cell bodies in the spinal cord. The axons branch extensively near the point of contact with the target muscle u Assembly of muscle fibers innervated by the axon from one motor neuron is called a motor unit.

Synaptic Transmission at Neuromuscular Junction n Synapse between motor neurons and skeletal muscle u Typically an axon contacts the muscle fiber at a single point called neuromuscular junction or the end plate. u An individual end plate consists of a small treelike patch of unmyelinated nerve processes called terminal arborizations u Bulb-shaped endings that finally contact the muscle fibers are called boutons.

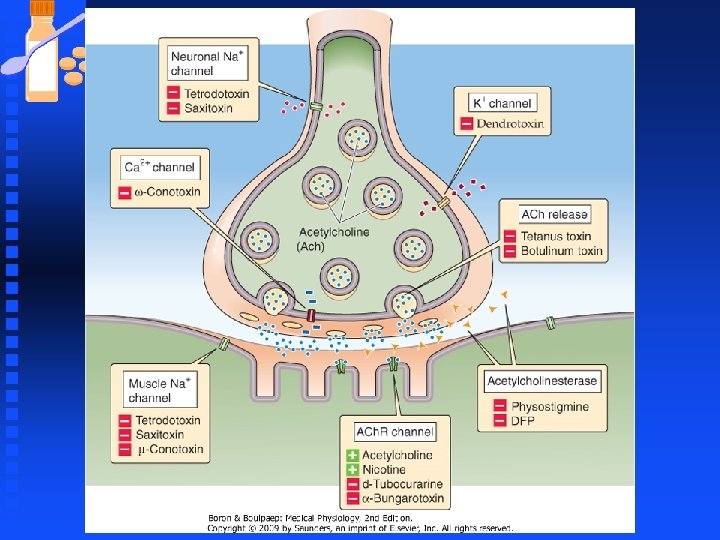
Toxins and Drugs Affecting Synaptic Transmission n Guanidinium neurotoxins prevent depolarization of nerve terminal u Tetrodotoxin and saxitoxin block voltage- dependent sodium channels, thereby preventing the depolarization phase of the action potential n Dendrotoxins facilitate the release of Ach u Block K+ channels thereby inhibiting repolarization of the presynaptic membrane, thereby prolonging the duration of the AP and facilitating transmitter release in response to entry of extra Ca 2+ into the nerve terminal.

Toxins and Drugs Affecting Synaptic Transmission n ω-Conotoxins Blocks Ca 2+-channels u N-type voltage-gated Ca 2+ -channels are blocked by ω-conotoxins thereby preventing transmitter release. n Botulinum and tetanus toxins u Cleave proteins involved in exocytosis, preventing fusion of synaptic vesicles.

Toxins and Drugs Affecting Synaptic Transmission n Botulinum toxin u Produced by Clostridium botulinum u A source of homemade food poisoning u Symptoms include dry mouth, double vision, difficulty in swallowing and speaking. Later GI complications include vomiting, pain and diarrhea. Weakness and paralysis of limbs may follow and ultimately death by paralysis of respiratory muscles

Toxins and Drugs Affecting Synaptic Transmission n Botulinum toxin u Toxin has zinc-dependent endoproteinase activity u Toxin enters nerve terminal and cleave proteins (synaptobrevin, SNAP-25, syntaxin) needed for synaptic vesicle exocytosis u Inhibit neurotransmitter release u Treat disorders of muscle spasms e. g strabismus u Botox used to treat wrinkles in aging

Toxins and Drugs Affecting Synaptic Transmission n Tetanus toxin u Produced by Clostridium tetani u Toxin is called tetanospasmin and is the causative agent of tetanus (“lockjaw”) which is characterized by a general increase in muscle tension and muscle rigidity starting with the muscles of mastication. u Tetanus toxin gains entry by cut or puncture wound and travels along peripheral nerves to the spinal cord, its major site of action.

Toxins and Drugs Affecting Synaptic Transmission n Tetanus toxin u Toxin cleaves synaptobrevin to prevent exocytosis and transmitter release. u Toxin has the greatest effect on interneurons that normally inhibit firing of motor neurons that in turn activate skeletal muscle. u Because the toxin suppresses inhibition of the normal reflex arc, muscle contraction leads to profound spasms, mainly of jaw muscles. u Respiratory arrest, aspiration pneumonia, rib fractures etc. can occur.


Autonomic Nervous System By Lincoln Edwards, D. D. S. ; Ph. D. Ext 42923 email: lpedwards@llu. edu

Autonomic Nervous System n The Autonomic Nervous System Has three divisions u Sympathetic Parasympathetic u Enteric Divisions u F The enteric division of the ANS is a system of afferent neurons, interneurons, and motor neurons that form networks of neurons called plexuses that surround the gastrointestinal tract. It can function as a separate and independent nervous system, but it is normally controlled by the CNS through sympathetic and parasympathetic fibers.

Autonomic Nervous System n Output from the central nervous system (CNS) travels along two anatomically and functionally distinct pathways: Somatic motor neurons, which innervate striated skeletal muscle u Autonomic motor neurons, which innervate smooth muscle, cardiac muscle, secretory epithelia, and glands. All viscera are richly supplied by efferent axons from the ANS that constantly adjust organ function. u
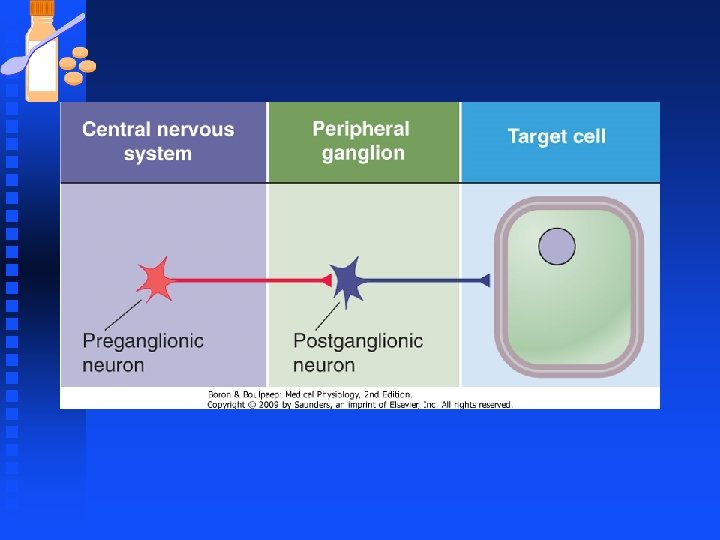

Sympathetic Nervous System Peripheral sympathetic system regulates activity of cardiac and smooth muscle and glandular systems by increasing or decreasing their activity. n Sympathetic stimulants augment ongoing cellular processes. n Transfer of information from sympathetic nerves to effector organs is primarily n mediated by norepinephrine secreted from nerve terminals. n

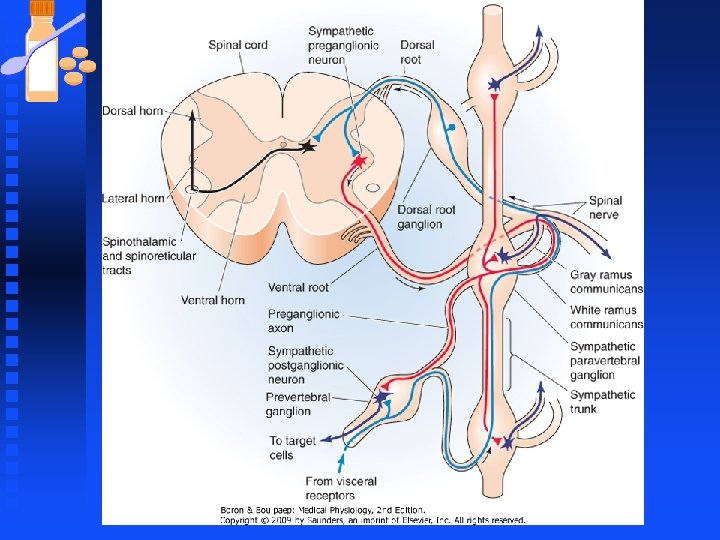
Autonomic Nervous System Autonomic controls visceral functions such as heart rate, blood flow, secretions etc. n The autonomic nervous system is further divided into: n u Sympathetic (thoracolumbar) u Parasympathetic system (craniosacral) n Autonomic fibers are nonmyelinated

Autonomic Nervous System n Parasympathetic fibers u originate from the craniosacral region of the spinal column. n Chemical messengers: u between preganglionic and postganglionic fiber is acetylcholine (ACh). u between the postganglionic fiber and the effector organ (smooth muscle, u cardiac muscle, gland cells) is also ACh. u Parasympathetic activity is referred to as cholinergic.

Autonomic Nervous System n Sympathetic fibers: u originate from nerve fibers in the thoracolumbar region of the spinal column. n Chemical messengers: u between the preganglionic fiber and postganglionic fiber is ACh. u However, the chemical messenger between the postganglionic fiber and the effector organ (smooth muscle, cardiac muscle, gland cells) is u norepinephrine (NE). u Sympathetic activity is referred to as adrenergic.

Autonomic Nervous System Ganglia synapsing in the thoracolumbar region and terminating in the blood vessels are called “sympathetic dilator nerves” n ACh is the transmitter both in the ganglia and the end effector organ (blood vessel). These fibers although “cholinergic” are called “sympathetic” dilator nerves simply because the ganglia originate in the thoracolumbar region. n

Autonomic Nervous System u Activation of these terminal fibers liberates ACh and dilates the blood vessel as opposed to contracting blood vessel. u However, impulses through these nerves are regarded pharmacologically as cholinergic or parasympathetic.
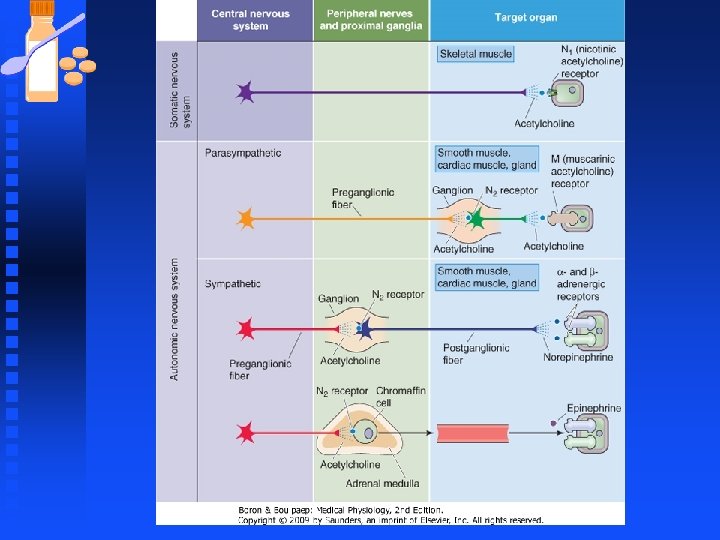

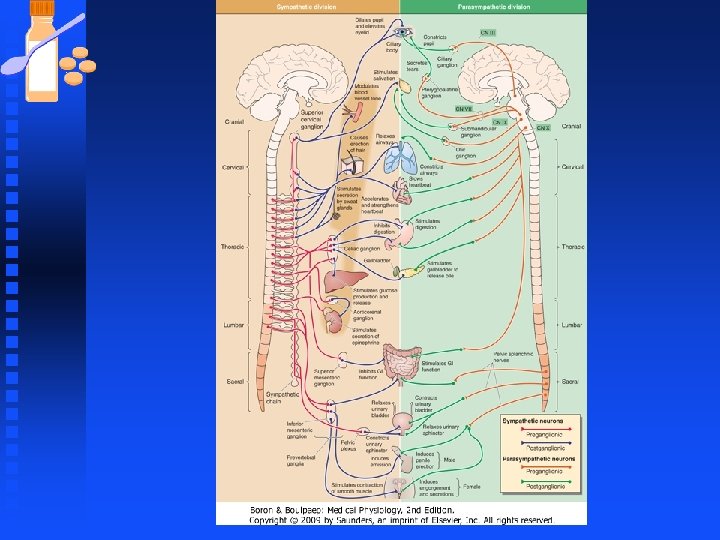
Autonomic Nervous System n Physiological effects of Stimulation of the Autonomic System u The sympathetic and parasympathetic nervous system maintain organ homeostasis and are physiological antagonists in most cases. u In the case of stress (exercise) or emergency (fear, defense) the sympathetic nervous system prepares the individual for the fight or flight response.

Autonomic Nervous System n Parasympathetic system u The parasympathetic system has been called the feed and breed system and is more associated with re-established homeostasis after the exercise or emergency (panic) response.

Autonomic Nervous System n Eye u adrenergic activity acting via α 1 receptors contracts radial muscle in iris = mydriasis (dilation of pupil). Cholinergic activity contracts the sphincter muscle in the iris = miosis (constriction of pupil).

Autonomic Nervous System n Heart u In the heart these two systems have antagonistic actions also u SA node: Adrenergic impulses (sympathetic) activate β 1 receptors increase heart rate. Cholinergic impulses: Vagus nerve releases Ach, which activate M 2 receptors at SA node and decrease heart rate. u Atria: Adrenergic impulses increase contraction (β 1 receptors). Cholinergic u impulses decrease (M 2) contraction

Autonomic Nervous System n Arterioles: u More complex with the sympathetic system, depending on the location of the artery adrenergic impulses can cause contraction or dilation

Autonomic Nervous System n Pulmonary: u Adrenergic impulses (which remember release NE) acting on α 1 -receptors cause contraction in arterioles. u β 2 -receptors: Expressed in the lung vascular bed and activation of these receptors cause dilation. Dilatory effect due to epinephrine (EPI) which can reinforce (by way of α 1 receptors) or antagonize NE by acting on β 2 u receptors. u Cholinergic impulses (M 3 -receptors) cause dilation in the pulmonary vascular bed.

Autonomic Nervous System n . Skin and mucosa: u Express α 1 and α 2 receptors, adrenergic impulses always produce constriction and cholinergic impulses (M 3 receptors) dilate u the blood vessels. u Adrenergic impulses (α 1 -receptors constrict). u Cholinergic impulses F (M 3 -receptors dilate) the blood vessels.

Autonomic Nervous System n Intestine: Two systems clearly antagonize each other: Adrenergic impulses (prejunctional α 2 receptors) decrease motility and tone. u Cholinergic impulses (M 3 receptors) increase motility and tone. u

Autonomic Nervous System n Adrenal medulla: u cholinergic (special case) impulses through the ganglia initiate the secretion of EPI and NE. n Bladder: u Sympathetic relaxes detrusor (β 2 -receptors) and contracts the sphincter (α 1 -receptors). u Parasympathetic innervation does just the opposite contracting the detrusor (M 3 -receptors) relaxing the sphincter (M 3 -receptors).

Autonomic Nervous System Metabolic function of sympathetic system: via the adrenal medulla acting through NE and EPI release. n Liver: n u EPI induce gluconeogenesis (β 2 receptors) and glycogenolysis (β 2 receptors). n Adipocytes: u lipolysis is induced by EPI and NE by their action at β 3 receptors.

Autonomic Nervous System n Kidney: u NE induces renin release by activation of β 1 receptors.

Sympathetic Nervous System The action of norepinephrine is reinforced by secretion of epinephrine, norepinephrine and dopamine secreted from the adrenal medulla n The actions of epinephrine and norepinephrine are modulated by the parasympathetic system and attenuate the effects of the catecholamines. n

Sympathetic Nervous System n Release of norepinephrine from the nerve: u When the nerve varicosity is depolarized calcium enters the varicosity primarily through N-type calcium channels u Calcium influx into the varicosity activates synaptic vesicle proteins which fuse the vesicle membrane with the prejunctional plasma membrane resulting in the release of NE into the synaptic cleft.

Sympathetic Nervous System u Once NE is liberated the signal is transduced by postjunctional receptors located in the effector cell. u NE inhibits own release through prejunctional feedback α 2 -autoreceptors.

Sympathetic Nervous System n Subtypes of alpha α 1 receptors u α 1 A primarily activates calcium channels and mobilizes extracellular calcium. However, activation of α 1 A receptors will produce minor activation of intracellular calcium release mechanisms. u The α 1 B receptor primarily mobilizes intracellular calcium through the second messenger IP 3 and release of calcium from the sarcoplasmic reticulum. However, α 1 B receptor activation can produce a minor activation of calcium channels.

Sympathetic Nervous System The end result of the activation of α 1 receptors is to increase intracellular n calcium in smooth muscle cells and activate the contractile mechanism, n constricting the blood vessel and elevating peripheral vascular resistance. n

Sympathetic Nervous System n Vascular effects u Epinephrine: F increases systolic pressure and has little effect or a decrease in diastolic pressure because of the activation of β 2 receptors in blood vessels in the lung and skeletal muscle. n Norepinephrine u increase systolic and diastolic pressure through the activation of α 1 receptors.

Sympathetic Nervous System n The α 2 -receptor appears to exist as a pre and postjunctional receptor. u Activation of prejunctional α 2 -receptors decreases NE release u α -receptor activation decreases calcium influx via a reduction in opening probability of Ca channels. Reducing Ca influx reduces NE overflow. 2


a3d74a5b0f6ae6861f01b2ff0d443983.ppt