8abb0f792fa5da6d9d5e7ac477526c00.ppt
- Количество слайдов: 26
 Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 Slide 1 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 1
Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 Slide 1 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 1
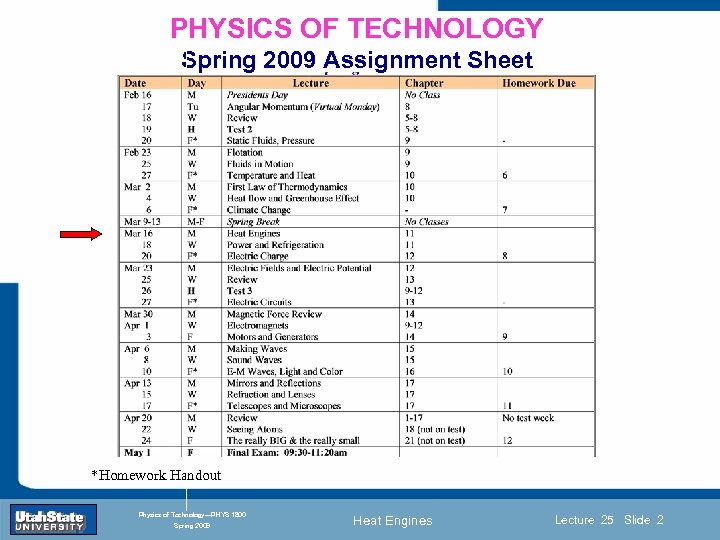 PHYSICS OF TECHNOLOGY Spring 2009 Assignment Sheet Introduction Section 0 Lecture 1 Slide 2 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 *Homework Handout Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 2
PHYSICS OF TECHNOLOGY Spring 2009 Assignment Sheet Introduction Section 0 Lecture 1 Slide 2 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 *Homework Handout Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 2
 Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 Slide 3 Review of Thermodynamics INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 3
Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 Slide 3 Review of Thermodynamics INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 3
 Describing Motion and Interactions Position—where you are in space (L or meter) Velocity—how fast position is changing with time (LT-1 or m/s) Acceleration—how fast velocity is changing with time (LT-2 or m/s 2) Force— what is required to change to motion of a body (MLT-2 or kg-m/s 2 or N) Inertia (mass)— a measure of the force needed to change the motion of a body (M) Energy—the potential for an object to do work. (ML 2 T-2 or kg m 2/s 2 or N-m or J) Work is equal to the force applied times the distance moved. W = F d Kinetic Energy is the energy associated with an object’s motion. KE=½ mv 2 Potential Energy is the energy associated with an objects position. Gravitational potential energy PEgravity=mgh Spring potential energy PEapring= -kx Momentum— the potential of an object to induce motion in another object (MLT-1 or kg-m/s) Introduction Section 0 Lecture 1 Slide 4 Angular Momentum and Rotational Energy— the equivalent constants of motion for rotation (MT-1 or kg/s) and (MLT-2 or kg m/s 2 or N) INTRODUCTION TO Modern Physics PHYX 2710 Pressure— force divided by 2004 area over which the force is applied (ML-1 T-1 or kg/m-s or N/m 2 or Pa) the Fall Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 4
Describing Motion and Interactions Position—where you are in space (L or meter) Velocity—how fast position is changing with time (LT-1 or m/s) Acceleration—how fast velocity is changing with time (LT-2 or m/s 2) Force— what is required to change to motion of a body (MLT-2 or kg-m/s 2 or N) Inertia (mass)— a measure of the force needed to change the motion of a body (M) Energy—the potential for an object to do work. (ML 2 T-2 or kg m 2/s 2 or N-m or J) Work is equal to the force applied times the distance moved. W = F d Kinetic Energy is the energy associated with an object’s motion. KE=½ mv 2 Potential Energy is the energy associated with an objects position. Gravitational potential energy PEgravity=mgh Spring potential energy PEapring= -kx Momentum— the potential of an object to induce motion in another object (MLT-1 or kg-m/s) Introduction Section 0 Lecture 1 Slide 4 Angular Momentum and Rotational Energy— the equivalent constants of motion for rotation (MT-1 or kg/s) and (MLT-2 or kg m/s 2 or N) INTRODUCTION TO Modern Physics PHYX 2710 Pressure— force divided by 2004 area over which the force is applied (ML-1 T-1 or kg/m-s or N/m 2 or Pa) the Fall Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 4
 Dennison’s Laws Thermal Poker (or How to Get a Hot Hand in Physics) • 0 th Law: Full House beats Two Pairs • 1 st Law: We’re playing the same game (but with a wild card) • 2 nd Law: You can’t win in Vegas. • 3 rd Law: In fact, you always loose. • 0 th Law: Defines Temperature • 1 st Law: Conservation of Energy (with heat) • 2 nd Introduction Section 0 Lecture 1 Slide 5 Law: You can’t recover all heat losses (or defining entropy) INTRODUCTION TO Modern Physics PHYX 2710 • 3 rd Fall 2004 Law: You can never get to absolute 0. Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 5
Dennison’s Laws Thermal Poker (or How to Get a Hot Hand in Physics) • 0 th Law: Full House beats Two Pairs • 1 st Law: We’re playing the same game (but with a wild card) • 2 nd Law: You can’t win in Vegas. • 3 rd Law: In fact, you always loose. • 0 th Law: Defines Temperature • 1 st Law: Conservation of Energy (with heat) • 2 nd Introduction Section 0 Lecture 1 Slide 5 Law: You can’t recover all heat losses (or defining entropy) INTRODUCTION TO Modern Physics PHYX 2710 • 3 rd Fall 2004 Law: You can never get to absolute 0. Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 5
 Heat • What is heat? • What is the relationship between quantity of heat and temperature? • What happens to a body (solid, liquid, gas) when thermal energy is added or removed? Thermal Energy Solid: Atoms vibrating in all directions about their fixed equilibrium (lattice) positions. Atoms constantly colliding with each other. Liquid: Atoms still oscillating and colliding with each other but they are free to move so that the long range order (shape) of body is lost. Introduction Section 0 Lecture 1 Slide 6 Gas: No equilibrium position, no oscillations, atoms are free and move in perpetual high-speed “zig-zag” dance punctuated by collisions. INTRODUCTION TO Modern Physics PHYX 2710 Spring 2009 liquid gas Fall 2004 Physics of Technology—PHYS 1800 solid Heat Engines Lecture 25 Slide 6
Heat • What is heat? • What is the relationship between quantity of heat and temperature? • What happens to a body (solid, liquid, gas) when thermal energy is added or removed? Thermal Energy Solid: Atoms vibrating in all directions about their fixed equilibrium (lattice) positions. Atoms constantly colliding with each other. Liquid: Atoms still oscillating and colliding with each other but they are free to move so that the long range order (shape) of body is lost. Introduction Section 0 Lecture 1 Slide 6 Gas: No equilibrium position, no oscillations, atoms are free and move in perpetual high-speed “zig-zag” dance punctuated by collisions. INTRODUCTION TO Modern Physics PHYX 2710 Spring 2009 liquid gas Fall 2004 Physics of Technology—PHYS 1800 solid Heat Engines Lecture 25 Slide 6
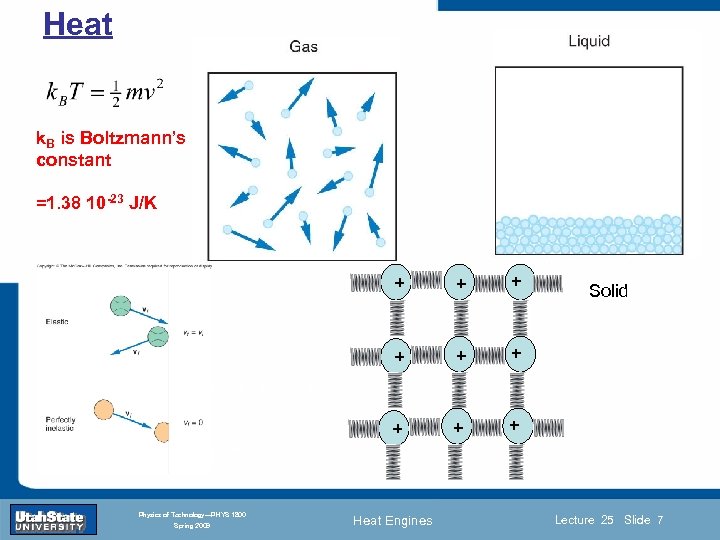 Heat k. B is Boltzmann’s constant + + Lecture 1 + + Section 0 + Introduction + + =1. 38 10 -23 J/K Solid Slide 7 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 7
Heat k. B is Boltzmann’s constant + + Lecture 1 + + Section 0 + Introduction + + =1. 38 10 -23 J/K Solid Slide 7 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 7
 Temperature and Heat • When two objects at different temperatures are placed in contact, heat will flow from the object with the higher temperature to the object with the lower temperature. • Heat added increases temperature, and heat removed decreases temperature. • Heat and temperature are not the same. • Temperature is a quantity that tells us which direction the heat will flow. Introduction Section 0 Heat is a form of energy. Lecture (Here comes conservation of energy!!!) 1 Slide 8 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 8
Temperature and Heat • When two objects at different temperatures are placed in contact, heat will flow from the object with the higher temperature to the object with the lower temperature. • Heat added increases temperature, and heat removed decreases temperature. • Heat and temperature are not the same. • Temperature is a quantity that tells us which direction the heat will flow. Introduction Section 0 Heat is a form of energy. Lecture (Here comes conservation of energy!!!) 1 Slide 8 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 8
 Joule’s Experiment and the First Law of Thermodynamics • Joule’s experiments led to Kelvin’s statement of the first law of thermodynamics. – Both work and heat represent transfers of energy into or out of a system. – If energy is added to a system either as work or heat, the internal energy of the system increases accordingly. • The increase in the internal energy of a system is equal to the amount of heat added to a system minus the amount of work done by the system. U = Q - W Introduction Section 0 Lecture 1 Slide 9 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 9
Joule’s Experiment and the First Law of Thermodynamics • Joule’s experiments led to Kelvin’s statement of the first law of thermodynamics. – Both work and heat represent transfers of energy into or out of a system. – If energy is added to a system either as work or heat, the internal energy of the system increases accordingly. • The increase in the internal energy of a system is equal to the amount of heat added to a system minus the amount of work done by the system. U = Q - W Introduction Section 0 Lecture 1 Slide 9 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 9
 Gas Behavior and The First Law Consider a gas in a cylinder with a movable piston. If the piston is pushed inward by an external force, work is done on the gas, adding energy to the system. • The force exerted on the piston by the gas equals the pressure of the gas times the area of the piston: F = PA • The work done equals the force exerted by the piston times the distance the piston Introduction Section 0 Lecture 1 moves: Slide 10 W = Fd = (PA)d = P V INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 10
Gas Behavior and The First Law Consider a gas in a cylinder with a movable piston. If the piston is pushed inward by an external force, work is done on the gas, adding energy to the system. • The force exerted on the piston by the gas equals the pressure of the gas times the area of the piston: F = PA • The work done equals the force exerted by the piston times the distance the piston Introduction Section 0 Lecture 1 moves: Slide 10 W = Fd = (PA)d = P V INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 10
 Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 INTRODUCTION TO Modern Physics PHYX 2710 Slide 11 Heat Engines Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 11
Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 INTRODUCTION TO Modern Physics PHYX 2710 Slide 11 Heat Engines Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 11
 Heat Engines What is a heat engine? It is a device that uses input heat to generate useful work. From the 1 st Law (Conservation of Energy) In cyclic engines we return to the original state every cycle so Introduction Section 0 Lecture 1 Slide 12 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 12
Heat Engines What is a heat engine? It is a device that uses input heat to generate useful work. From the 1 st Law (Conservation of Energy) In cyclic engines we return to the original state every cycle so Introduction Section 0 Lecture 1 Slide 12 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 12
 Heat Engines What is a heat engine? All heat engines share these main features of operation: – Thermal energy (heat) is introduced into the engine. – Some of this energy is converted to mechanical work. – Some heat (waste heat) is released into the environment at a. Lecture 1 Slide 13 Introduction Section 0 temperature lower than the input temperature. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 13
Heat Engines What is a heat engine? All heat engines share these main features of operation: – Thermal energy (heat) is introduced into the engine. – Some of this energy is converted to mechanical work. – Some heat (waste heat) is released into the environment at a. Lecture 1 Slide 13 Introduction Section 0 temperature lower than the input temperature. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 13
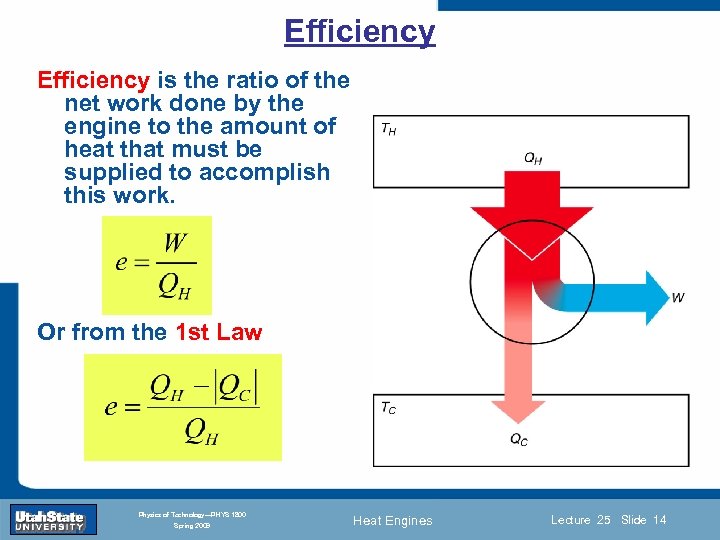 Efficiency is the ratio of the net work done by the engine to the amount of heat that must be supplied to accomplish this work. Or from the 1 st Law Introduction Section 0 Lecture 1 Slide 14 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 14
Efficiency is the ratio of the net work done by the engine to the amount of heat that must be supplied to accomplish this work. Or from the 1 st Law Introduction Section 0 Lecture 1 Slide 14 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 14
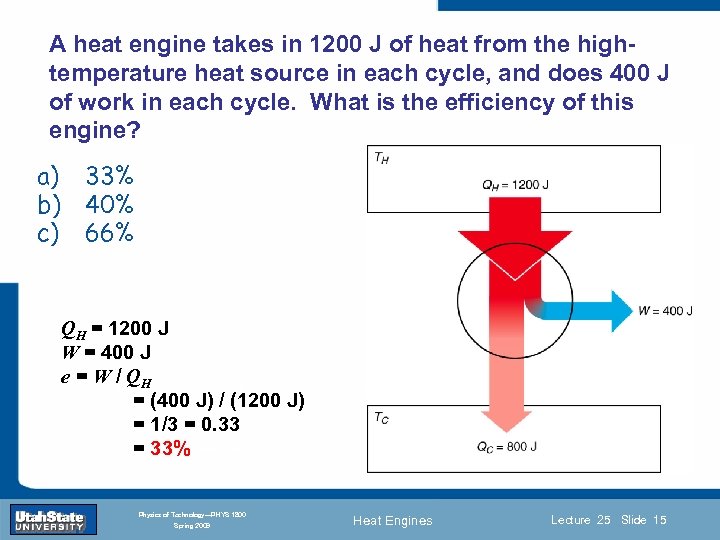 A heat engine takes in 1200 J of heat from the hightemperature heat source in each cycle, and does 400 J of work in each cycle. What is the efficiency of this engine? a) b) c) 33% 40% 66% QH = 1200 J W = 400 J e = W /Introduction Section 0 Lecture 1 QH = (400 J) / (1200 J) = 1/3 = 0. 33 = 33% Slide 15 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 15
A heat engine takes in 1200 J of heat from the hightemperature heat source in each cycle, and does 400 J of work in each cycle. What is the efficiency of this engine? a) b) c) 33% 40% 66% QH = 1200 J W = 400 J e = W /Introduction Section 0 Lecture 1 QH = (400 J) / (1200 J) = 1/3 = 0. 33 = 33% Slide 15 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 15
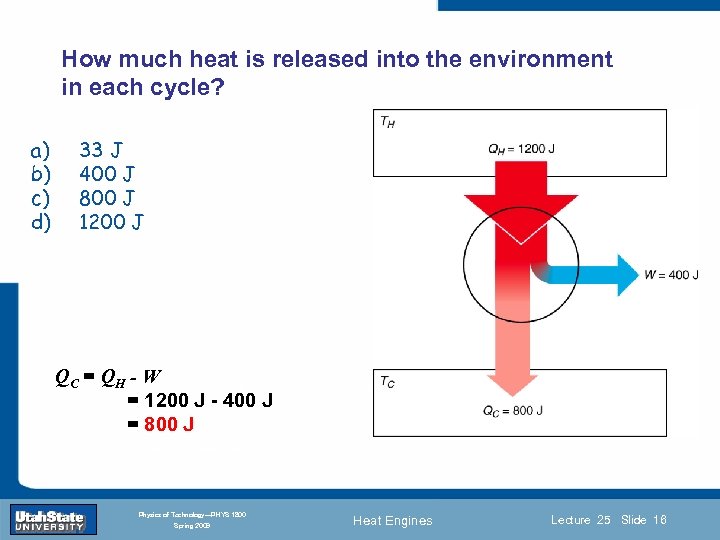 How much heat is released into the environment in each cycle? a) b) c) d) 33 J 400 J 800 J 1200 J QC = QH - W Introduction Section 0 Lecture = 1200 J - 400 J = 800 J 1 Slide 16 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 16
How much heat is released into the environment in each cycle? a) b) c) d) 33 J 400 J 800 J 1200 J QC = QH - W Introduction Section 0 Lecture = 1200 J - 400 J = 800 J 1 Slide 16 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 16
 Carnot Engine and Carnot Cycle • Carnot considered the ideal (most efficient possible) engine for a give TH and TC. • Carnot engine has negligible work lost to friction, turbulence, heat loss, etc. • Carnot also reasoned that the processes should occur without undue turbulence. – The engine is completely reversible: it can be turned around and run the other way at any point in the cycle, because it is always near equilibrium. – This is Carnot’s ideal engine. • The cycle devised by Carnot that an ideal engine would have to follow is called a Carnot cycle. • An. Introduction Section 0 real) engine following this cycle is (ideal, not Lecture 1 Slide 17 called a Carnot engine. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 17
Carnot Engine and Carnot Cycle • Carnot considered the ideal (most efficient possible) engine for a give TH and TC. • Carnot engine has negligible work lost to friction, turbulence, heat loss, etc. • Carnot also reasoned that the processes should occur without undue turbulence. – The engine is completely reversible: it can be turned around and run the other way at any point in the cycle, because it is always near equilibrium. – This is Carnot’s ideal engine. • The cycle devised by Carnot that an ideal engine would have to follow is called a Carnot cycle. • An. Introduction Section 0 real) engine following this cycle is (ideal, not Lecture 1 Slide 17 called a Carnot engine. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 17
 Carnot Efficiency • The efficiency of Carnot’s ideal engine (one using an ideal gas with PV=Nk. BT) is called the Carnot efficiency and is given by: • This is the maximum efficiency possible for any engine taking in heat from a reservoir at absolute temperature TH and releasing heat to a reservoir at temperature TC. Introduction Section 0 Lecture Slide 18 • This provides a useful 1 limiting case. • Even Carnot’s ideal engine is less than 100% efficient. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 18
Carnot Efficiency • The efficiency of Carnot’s ideal engine (one using an ideal gas with PV=Nk. BT) is called the Carnot efficiency and is given by: • This is the maximum efficiency possible for any engine taking in heat from a reservoir at absolute temperature TH and releasing heat to a reservoir at temperature TC. Introduction Section 0 Lecture Slide 18 • This provides a useful 1 limiting case. • Even Carnot’s ideal engine is less than 100% efficient. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 18
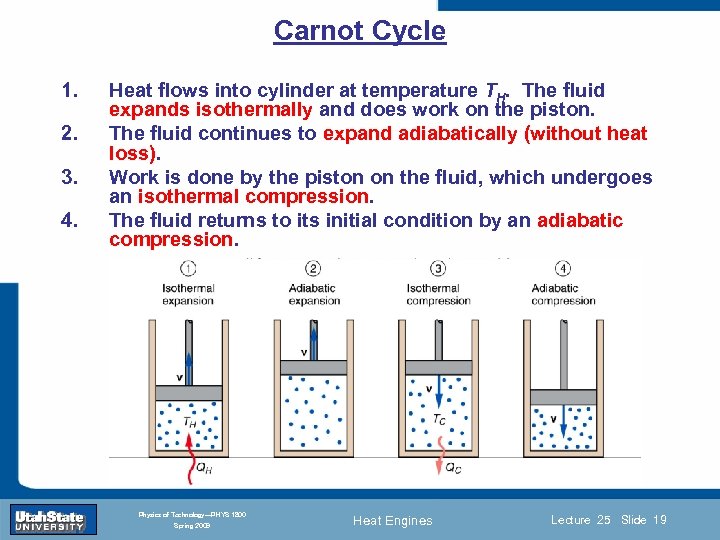 Carnot Cycle 1. 2. 3. 4. Heat flows into cylinder at temperature TH. The fluid expands isothermally and does work on the piston. The fluid continues to expand adiabatically (without heat loss). Work is done by the piston on the fluid, which undergoes an isothermal compression. The fluid returns to its initial condition by an adiabatic compression. Introduction Section 0 Lecture 1 Slide 19 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 19
Carnot Cycle 1. 2. 3. 4. Heat flows into cylinder at temperature TH. The fluid expands isothermally and does work on the piston. The fluid continues to expand adiabatically (without heat loss). Work is done by the piston on the fluid, which undergoes an isothermal compression. The fluid returns to its initial condition by an adiabatic compression. Introduction Section 0 Lecture 1 Slide 19 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 19
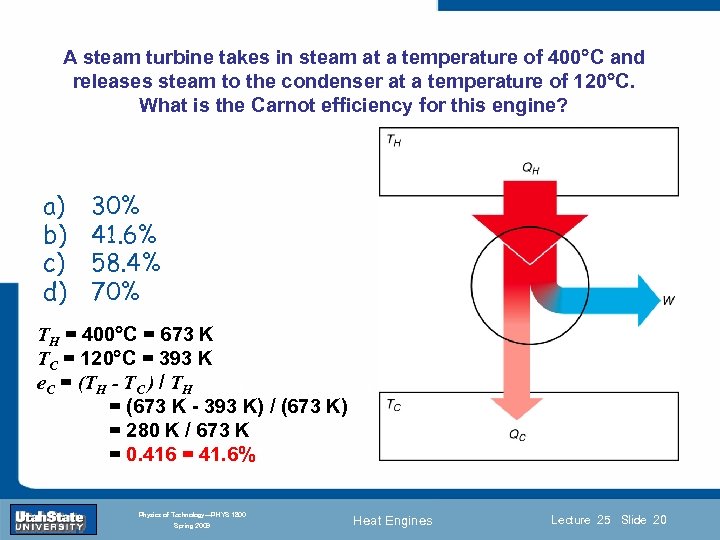 A steam turbine takes in steam at a temperature of 400 C and releases steam to the condenser at a temperature of 120 C. What is the Carnot efficiency for this engine? a) b) c) d) 30% 41. 6% 58. 4% 70% TH = 400 C = 673 K TC = 120 C = 393 K e. C = (TH -Introduction H Section 0 Lecture 1 Slide TC ) / T = (673 K - 393 K) / (673 K) = 280 K / 673 K = 0. 416 = 41. 6% 20 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 20
A steam turbine takes in steam at a temperature of 400 C and releases steam to the condenser at a temperature of 120 C. What is the Carnot efficiency for this engine? a) b) c) d) 30% 41. 6% 58. 4% 70% TH = 400 C = 673 K TC = 120 C = 393 K e. C = (TH -Introduction H Section 0 Lecture 1 Slide TC ) / T = (673 K - 393 K) / (673 K) = 280 K / 673 K = 0. 416 = 41. 6% 20 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 20
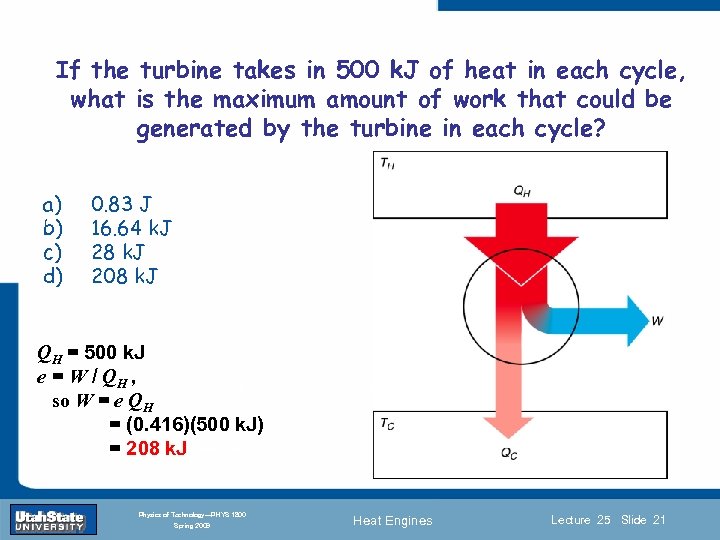 If the turbine takes in 500 k. J of heat in each cycle, what is the maximum amount of work that could be generated by the turbine in each cycle? a) b) c) d) 0. 83 J 16. 64 k. J 28 k. J 208 k. J QH = 500 k. J e = W / QH , Introduction Section 0 Lecture so W = e QH = (0. 416)(500 k. J) = 208 k. J 1 Slide 21 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 21
If the turbine takes in 500 k. J of heat in each cycle, what is the maximum amount of work that could be generated by the turbine in each cycle? a) b) c) d) 0. 83 J 16. 64 k. J 28 k. J 208 k. J QH = 500 k. J e = W / QH , Introduction Section 0 Lecture so W = e QH = (0. 416)(500 k. J) = 208 k. J 1 Slide 21 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 21
 Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 Slide 22 Second Law of Thermodynamics INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 22
Physics of Technology PHYS 1800 Lecture 25 Heat Engines and the 2 nd Law of Thermodynamics Introduction Section 0 Lecture 1 Slide 22 Second Law of Thermodynamics INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 22
 Second Law of Thermodynamics Heat (random motion) is a special form of energy that cannot be fully (with complete efficiency) transformed to other forms of energy. This leads to various forms of the Second Law of Thermodynamics. • You can’t recover all heat losses. • You can’t win in Vegas. • No engine, working in a continuous cycle, can take heat from a reservoir at a single temperature and convert that heat completely to work. • Therefore, no engine can have a greater efficiency than a Carnot Introduction Section between Slide 23 engine operating 0 Lecture 1 the same two temperatures. • Define entropy (something that measures randomness or disorder in an object) to take account of this. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 23
Second Law of Thermodynamics Heat (random motion) is a special form of energy that cannot be fully (with complete efficiency) transformed to other forms of energy. This leads to various forms of the Second Law of Thermodynamics. • You can’t recover all heat losses. • You can’t win in Vegas. • No engine, working in a continuous cycle, can take heat from a reservoir at a single temperature and convert that heat completely to work. • Therefore, no engine can have a greater efficiency than a Carnot Introduction Section between Slide 23 engine operating 0 Lecture 1 the same two temperatures. • Define entropy (something that measures randomness or disorder in an object) to take account of this. INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 23
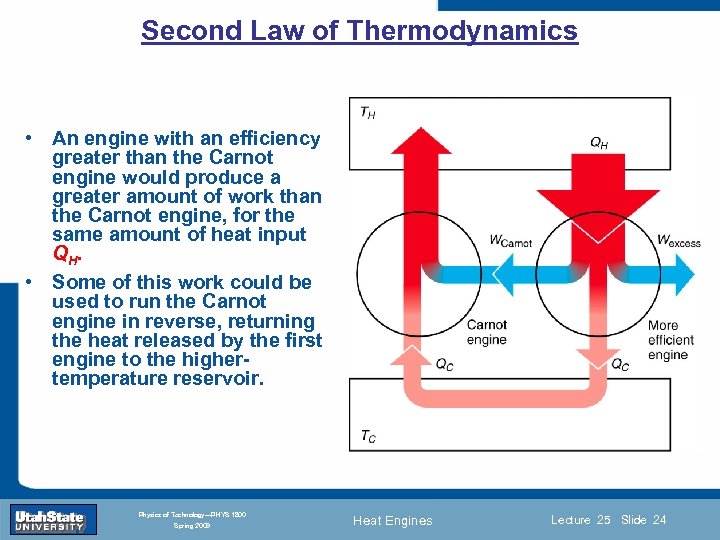 Second Law of Thermodynamics • An engine with an efficiency greater than the Carnot engine would produce a greater amount of work than the Carnot engine, for the same amount of heat input QH. • Some of this work could be used to run the Carnot engine in reverse, returning the heat released by the first engine to the highertemperature reservoir. Introduction Section 0 Lecture 1 Slide 24 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 24
Second Law of Thermodynamics • An engine with an efficiency greater than the Carnot engine would produce a greater amount of work than the Carnot engine, for the same amount of heat input QH. • Some of this work could be used to run the Carnot engine in reverse, returning the heat released by the first engine to the highertemperature reservoir. Introduction Section 0 Lecture 1 Slide 24 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 24
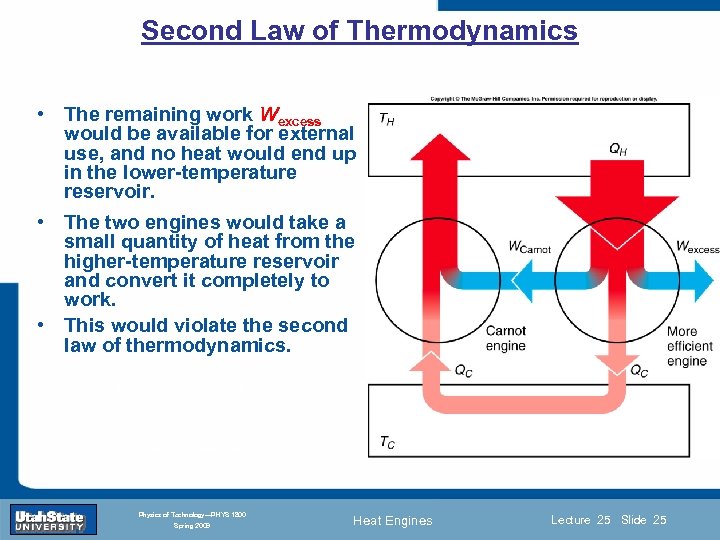 Second Law of Thermodynamics • The remaining work Wexcess would be available for external use, and no heat would end up in the lower-temperature reservoir. • The two engines would take a small quantity of heat from the higher-temperature reservoir and convert it completely to work. • This would violate the second law of thermodynamics. Introduction Section 0 Lecture 1 Slide 25 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 25
Second Law of Thermodynamics • The remaining work Wexcess would be available for external use, and no heat would end up in the lower-temperature reservoir. • The two engines would take a small quantity of heat from the higher-temperature reservoir and convert it completely to work. • This would violate the second law of thermodynamics. Introduction Section 0 Lecture 1 Slide 25 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 25
 Physics of Technology Next Lab/Demo: Fluid Dynamics Temperature Thursday 1: 30 -2: 45 ESLC 46 Ch 9 and 10 Next Class: Wednesday 10: 30 -11: 20 BUS 318 room Slide 26 Review Ch 10 Introduction Section 0 Lecture 1 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 26
Physics of Technology Next Lab/Demo: Fluid Dynamics Temperature Thursday 1: 30 -2: 45 ESLC 46 Ch 9 and 10 Next Class: Wednesday 10: 30 -11: 20 BUS 318 room Slide 26 Review Ch 10 Introduction Section 0 Lecture 1 INTRODUCTION TO Modern Physics PHYX 2710 Fall 2004 Physics of Technology—PHYS 1800 Spring 2009 Heat Engines Lecture 25 Slide 26


