9aaeacf555112400c7f46c53488f8d5e.ppt
- Количество слайдов: 26
 PHYSICS AND TECHNIQUE OF LOWTEMPERATURE PLASMA, PLASMACHEMISTRY AND PLASMA TECHNOLOGY FOR MATERIAL PROCESSING Introduction Lecturer : Prof. Alexander Pushkarev, Doctor of Sciences, Department of Electrophysics and High Voltage Technique Tomsk Polytechnic University 1
PHYSICS AND TECHNIQUE OF LOWTEMPERATURE PLASMA, PLASMACHEMISTRY AND PLASMA TECHNOLOGY FOR MATERIAL PROCESSING Introduction Lecturer : Prof. Alexander Pushkarev, Doctor of Sciences, Department of Electrophysics and High Voltage Technique Tomsk Polytechnic University 1
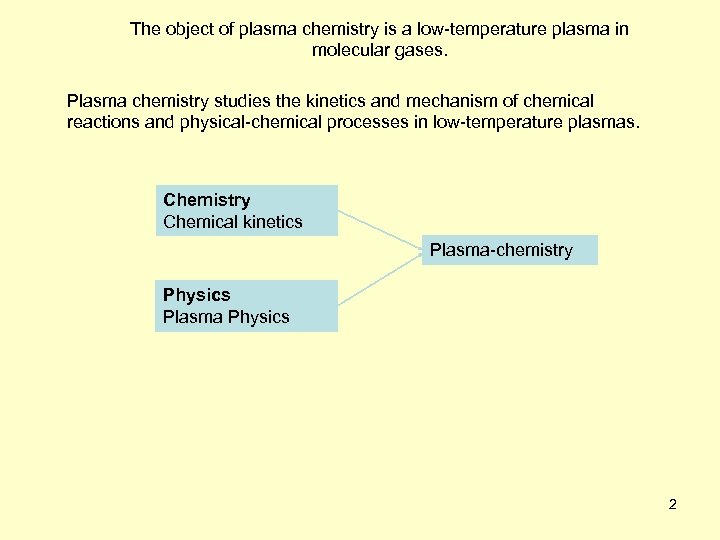 The object of plasma chemistry is a low-temperature plasma in molecular gases. Plasma chemistry studies the kinetics and mechanism of chemical reactions and physical-chemical processes in low-temperature plasmas. Chemistry Chemical kinetics Plasma-chemistry Physics Plasma Physics 2
The object of plasma chemistry is a low-temperature plasma in molecular gases. Plasma chemistry studies the kinetics and mechanism of chemical reactions and physical-chemical processes in low-temperature plasmas. Chemistry Chemical kinetics Plasma-chemistry Physics Plasma Physics 2
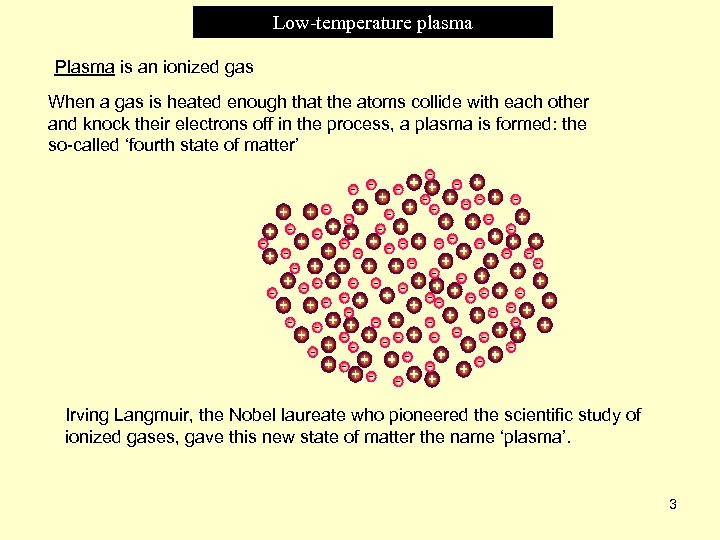 Low-temperature plasma Plasma is an ionized gas When a gas is heated enough that the atoms collide with each other and knock their electrons off in the process, a plasma is formed: the so-called ‘fourth state of matter’ Irving Langmuir, the Nobel laureate who pioneered the scientific study of ionized gases, gave this new state of matter the name ‘plasma’. 3
Low-temperature plasma Plasma is an ionized gas When a gas is heated enough that the atoms collide with each other and knock their electrons off in the process, a plasma is formed: the so-called ‘fourth state of matter’ Irving Langmuir, the Nobel laureate who pioneered the scientific study of ionized gases, gave this new state of matter the name ‘plasma’. 3
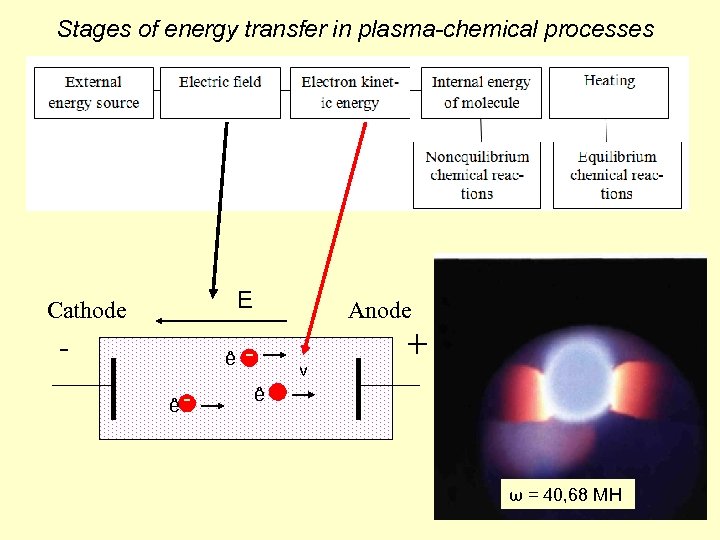 Stages of energy transfer in plasma-chemical processes E Cathode - Anode ê ê v + ê ω = 40, 68 МH 4
Stages of energy transfer in plasma-chemical processes E Cathode - Anode ê ê v + ê ω = 40, 68 МH 4
 Lectures Schedule (25 lectures, 54 hours) LECTURE № 1. INTRODUCTION Determination of plasma chemistry. The object and the main features of plasma chemistry. LECTURE № 2. BASIC CONCEPTS OF CHEMICAL KINETICS The subject of chemical kinetics. The rate of chemical reactions. The law of mass action. The rate constant of chemical reaction. Arennius equation. The order of reaction. Maxwell distribution. The internal energy of the molecule. . LECTURE № 3. INTRODUCTION TO PLASMA PHYSICS The definition of plasma, quasineutrality of the plasma, The Debye shielding. The Debye radius. The potential of a particle in a plasma. Plasma oscillations. The classical and the degenerate plasma. Ideal and non-ideal plasma. The degree of ionization. The Saha equation. 5
Lectures Schedule (25 lectures, 54 hours) LECTURE № 1. INTRODUCTION Determination of plasma chemistry. The object and the main features of plasma chemistry. LECTURE № 2. BASIC CONCEPTS OF CHEMICAL KINETICS The subject of chemical kinetics. The rate of chemical reactions. The law of mass action. The rate constant of chemical reaction. Arennius equation. The order of reaction. Maxwell distribution. The internal energy of the molecule. . LECTURE № 3. INTRODUCTION TO PLASMA PHYSICS The definition of plasma, quasineutrality of the plasma, The Debye shielding. The Debye radius. The potential of a particle in a plasma. Plasma oscillations. The classical and the degenerate plasma. Ideal and non-ideal plasma. The degree of ionization. The Saha equation. 5
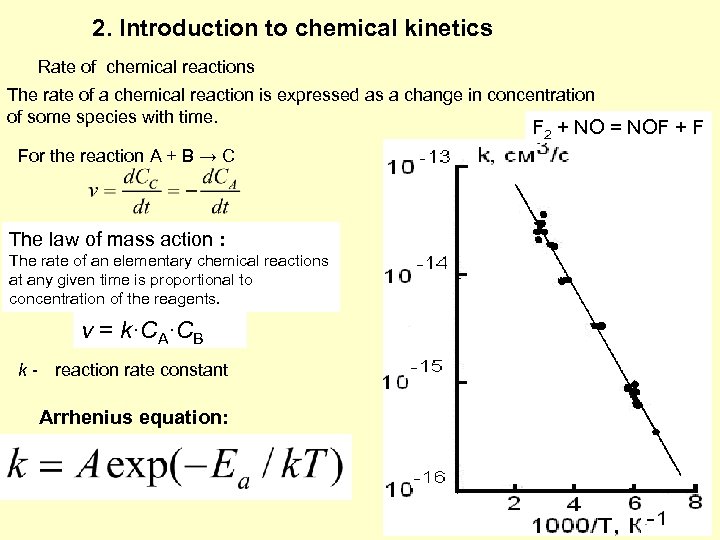 2. Introduction to chemical kinetics Rate of chemical reactions The rate of a chemical reaction is expressed as a change in concentration of some species with time. F 2 + NO = NOF + F For the reaction A + B → C The law of mass action : The rate of an elementary chemical reactions at any given time is proportional to concentration of the reagents. v = k·CA·CB k - reaction rate constant Arrhenius equation: 6
2. Introduction to chemical kinetics Rate of chemical reactions The rate of a chemical reaction is expressed as a change in concentration of some species with time. F 2 + NO = NOF + F For the reaction A + B → C The law of mass action : The rate of an elementary chemical reactions at any given time is proportional to concentration of the reagents. v = k·CA·CB k - reaction rate constant Arrhenius equation: 6
 LECTURE № 4. ELEMENTARY PROCESSES IN PLASMA The rate of elementary processes. The collision cross-section. Elastic collisions of electrons with atoms. . Inelastic elementary processes of the first kind. Excitation though collisions between electrons and neutral particles. Selective vibrational excitation. Direct and stepwise ionization in the plasma. The interaction of charged particles with solid surfaces. LECTURE № 5. ELEMENTARY PROCESSES OF THE SECOND KIND Recombination. Impact recombination. The dissociative recombination. Elementary processes in a discharge plasma, Coronal balance, Elwert formula. Relaxation channels of the excited particles in plasma. Luminescence spectra of the excited molecules. The spectroscopy. Dissociative relaxation of excited molecules. Dissociation of vibrationally excited molecules. Treanor mechanism.
LECTURE № 4. ELEMENTARY PROCESSES IN PLASMA The rate of elementary processes. The collision cross-section. Elastic collisions of electrons with atoms. . Inelastic elementary processes of the first kind. Excitation though collisions between electrons and neutral particles. Selective vibrational excitation. Direct and stepwise ionization in the plasma. The interaction of charged particles with solid surfaces. LECTURE № 5. ELEMENTARY PROCESSES OF THE SECOND KIND Recombination. Impact recombination. The dissociative recombination. Elementary processes in a discharge plasma, Coronal balance, Elwert formula. Relaxation channels of the excited particles in plasma. Luminescence spectra of the excited molecules. The spectroscopy. Dissociative relaxation of excited molecules. Dissociation of vibrationally excited molecules. Treanor mechanism.
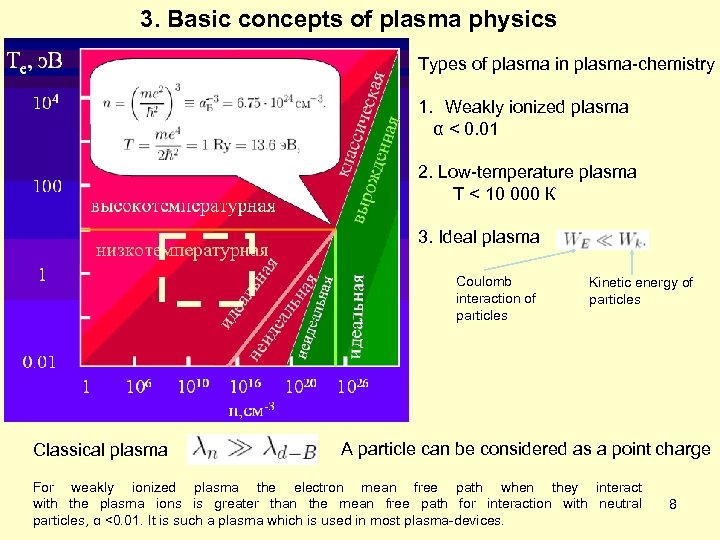 3. Basic concepts of plasma physics Types of plasma in plasma-chemistry 1. Weakly ionized plasma α < 0. 01 2. Low-temperature plasma Т < 10 000 К 3. Ideal plasma Coulomb interaction of particles Classical plasma Kinetic energy of particles A particle can be considered as a point charge For weakly ionized plasma the electron mean free path when they interact with the plasma ions is greater than the mean free path for interaction with neutral particles, α <0. 01. It is such a plasma which is used in most plasma-devices. 8
3. Basic concepts of plasma physics Types of plasma in plasma-chemistry 1. Weakly ionized plasma α < 0. 01 2. Low-temperature plasma Т < 10 000 К 3. Ideal plasma Coulomb interaction of particles Classical plasma Kinetic energy of particles A particle can be considered as a point charge For weakly ionized plasma the electron mean free path when they interact with the plasma ions is greater than the mean free path for interaction with neutral particles, α <0. 01. It is such a plasma which is used in most plasma-devices. 8
 LECTURE № 6. INTRODUCTION TO THE GAS DISCHARGE PHYSICS The mechanisms of electric discharge. Tawsend discharge. Paschen curve. Cathode processes. LECTURE № 7. TYPES OF GAS DISCHARGES. APPLICATIONS Glow discharge: combustion conditions, design and application. The arc discharge. Sliding discharge. Corona discharge. RF capacitive discharge. RF inductive discharge. Microwave discharge.
LECTURE № 6. INTRODUCTION TO THE GAS DISCHARGE PHYSICS The mechanisms of electric discharge. Tawsend discharge. Paschen curve. Cathode processes. LECTURE № 7. TYPES OF GAS DISCHARGES. APPLICATIONS Glow discharge: combustion conditions, design and application. The arc discharge. Sliding discharge. Corona discharge. RF capacitive discharge. RF inductive discharge. Microwave discharge.
 LECTURE № 8. THE SPECIFIC CHARACTERISTICS OF PLASMACHEMICAL REACTIONS Quasi-equilibrium plasma-chemical processes. Nonequilibrium plasma-chemical processes. Basic concepts in non-equilibrium chemical kinetics. Principles of the organization of plasma chemical processes. LECTURE № 9. TYPES OF REACTIONS IN PLASMA CHEMISTRY Monomolecular reaction. Bimolecular and termolecular reactions. Impact-radiative recombination. Recombination of heavy particles into the molecule on the surface of solids (heterogeneous recombination).
LECTURE № 8. THE SPECIFIC CHARACTERISTICS OF PLASMACHEMICAL REACTIONS Quasi-equilibrium plasma-chemical processes. Nonequilibrium plasma-chemical processes. Basic concepts in non-equilibrium chemical kinetics. Principles of the organization of plasma chemical processes. LECTURE № 9. TYPES OF REACTIONS IN PLASMA CHEMISTRY Monomolecular reaction. Bimolecular and termolecular reactions. Impact-radiative recombination. Recombination of heavy particles into the molecule on the surface of solids (heterogeneous recombination).
 TYPES OF REACTIONS IN PLASMA CHEMISTRY The most common reactions are • Dissociative ionization of molecules • Dissociation though the electronically excited state • Dissociative electron attachment to molecules • Stepwise dissociation by electron impact • Dissociative recombination of molecular ions though collisions of electrons with heavy particles 11
TYPES OF REACTIONS IN PLASMA CHEMISTRY The most common reactions are • Dissociative ionization of molecules • Dissociation though the electronically excited state • Dissociative electron attachment to molecules • Stepwise dissociation by electron impact • Dissociative recombination of molecular ions though collisions of electrons with heavy particles 11
 LECTURE № 10. The TEU-500 Pulsed Electron Accelerator
LECTURE № 10. The TEU-500 Pulsed Electron Accelerator
 Experimental installation Reactor volume– 6 l. Gas pressure– up to 2 atm. Temperature –up to 150 °С Electron energy: up to 500 k. Ev Extracted current: 5 -7 к. А Pulse duration: 60 ns Pulse energy: up to 200 J 13
Experimental installation Reactor volume– 6 l. Gas pressure– up to 2 atm. Temperature –up to 150 °С Electron energy: up to 500 k. Ev Extracted current: 5 -7 к. А Pulse duration: 60 ns Pulse energy: up to 200 J 13
 Lecture № 11. Diagnostic equipment of the TEU-500 electron Accelerator 1. Rogowski loop 2. Faraday cup , R = 0, 0485 Оhm 3. Capacitive voltage divider 4. Differential voltage divider 5. Liquid-voltage divider 6. Total absorption calorimeter 14
Lecture № 11. Diagnostic equipment of the TEU-500 electron Accelerator 1. Rogowski loop 2. Faraday cup , R = 0, 0485 Оhm 3. Capacitive voltage divider 4. Differential voltage divider 5. Liquid-voltage divider 6. Total absorption calorimeter 14
 Lecture № 12. Short-Pulsed Ion Accelerator and its application Introduction 1. Principles of high-power pulsed ion beam (HPIB) formation 2. Review of high-power pulsed ion accelerators for material science application (Russia, USA, Japan, China) 3. Fields of accelerator application 4. Scientific technical base of HVRI at TPU, Tomsk 5. Idea of SPIA creation (100 ke. V, 1 k. A, 10 ns), accelerator draft 6. Perspectives of SPIA application for material analyses 7. SPIA application in the field of material science
Lecture № 12. Short-Pulsed Ion Accelerator and its application Introduction 1. Principles of high-power pulsed ion beam (HPIB) formation 2. Review of high-power pulsed ion accelerators for material science application (Russia, USA, Japan, China) 3. Fields of accelerator application 4. Scientific technical base of HVRI at TPU, Tomsk 5. Idea of SPIA creation (100 ke. V, 1 k. A, 10 ns), accelerator draft 6. Perspectives of SPIA application for material analyses 7. SPIA application in the field of material science
 LECTURE № 13. NON-EQUILIBRIUM EXCITATION OF MOLECULES Chemical excitation. Formation of a non-equilibrium energy distribution of molecules in the gasdynamic gas expansion. Vibrational excitation of molecules by electron impact. Features of the plasma-chemical processes initiated by a pulsed electron beam. LECTURE № 14. METHODS OF MATHEMATICAL MODELING OF PLASMA CHEMICAL PROCESSES LECTURE № 15. CHAIN GAS-PHASE PROCESSES The induction period. Classification of chain processes. Unbranched chain reactions. Chain reaction with quadratic chain branching. Chain reactions with degenerate branching. Chain reactions with energy branching. Chain reactions involving an electron.
LECTURE № 13. NON-EQUILIBRIUM EXCITATION OF MOLECULES Chemical excitation. Formation of a non-equilibrium energy distribution of molecules in the gasdynamic gas expansion. Vibrational excitation of molecules by electron impact. Features of the plasma-chemical processes initiated by a pulsed electron beam. LECTURE № 14. METHODS OF MATHEMATICAL MODELING OF PLASMA CHEMICAL PROCESSES LECTURE № 15. CHAIN GAS-PHASE PROCESSES The induction period. Classification of chain processes. Unbranched chain reactions. Chain reaction with quadratic chain branching. Chain reactions with degenerate branching. Chain reactions with energy branching. Chain reactions involving an electron.
 CHAIN GAS-PHASE PROCESSES Chain processes are the chemical and nuclear transformation, where a large number of elementary reactions (chain) occur due to an active particle (free radical, an atom with an unpaired electron, an excited particle - an ion, a molecule - in chemical reactions, a neutron - in nuclear transformations). The active particle is regenerated as a result of the elementary process. 17
CHAIN GAS-PHASE PROCESSES Chain processes are the chemical and nuclear transformation, where a large number of elementary reactions (chain) occur due to an active particle (free radical, an atom with an unpaired electron, an excited particle - an ion, a molecule - in chemical reactions, a neutron - in nuclear transformations). The active particle is regenerated as a result of the elementary process. 17
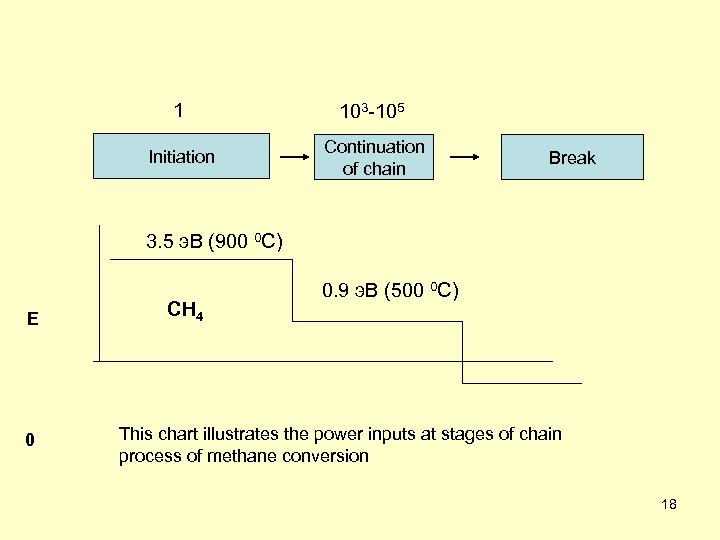 1 103 -105 Initiation Continuation of chain Break 3. 5 э. В (900 0 С) Е 0 CH 4 0. 9 э. В (500 0 С) This chart illustrates the power inputs at stages of chain process of methane conversion 18
1 103 -105 Initiation Continuation of chain Break 3. 5 э. В (900 0 С) Е 0 CH 4 0. 9 э. В (500 0 С) This chart illustrates the power inputs at stages of chain process of methane conversion 18
 UNDER EXTERNAL ACTION Chain oxidation of hydrogen. The induction period of ignition under external influence in the oxygen-hydrog The oscillatory nature of the ignition of mixture 2 H 2 + O 2 when initiated by a p The stoichiometric oxygen-hydrogen mixture. Condensation of water vapor. LECTURE № 17. CONVERSION OF METHANE IN LOW-TEMPERATURE P Plasma pyrolysis of methane. Pyrolysis of methane into the high molecular weight hydrocarbons. The use of catalysts in the plasma pyrolysis of methane. The partial oxidation of methane in the low-temperature plasma. Carbon dioxide conversion of methane plasma. Steam conversion of methane. Combined methods of plasma-chemical conversion of methane. LECTURE № 18. CHAIN PLASMA-CHEMICAL CONVERSION OF METHANE Radiation-thermal cracking of methane. Oxidation of methane under equilibrium conditions at low pressure. Chain oxidation of methane under the external action.
UNDER EXTERNAL ACTION Chain oxidation of hydrogen. The induction period of ignition under external influence in the oxygen-hydrog The oscillatory nature of the ignition of mixture 2 H 2 + O 2 when initiated by a p The stoichiometric oxygen-hydrogen mixture. Condensation of water vapor. LECTURE № 17. CONVERSION OF METHANE IN LOW-TEMPERATURE P Plasma pyrolysis of methane. Pyrolysis of methane into the high molecular weight hydrocarbons. The use of catalysts in the plasma pyrolysis of methane. The partial oxidation of methane in the low-temperature plasma. Carbon dioxide conversion of methane plasma. Steam conversion of methane. Combined methods of plasma-chemical conversion of methane. LECTURE № 18. CHAIN PLASMA-CHEMICAL CONVERSION OF METHANE Radiation-thermal cracking of methane. Oxidation of methane under equilibrium conditions at low pressure. Chain oxidation of methane under the external action.
 LECTURE № 19. PLASMA-CHEMICAL SYNTHESIS OF NANOSIZED PARTICLES Plasma-chemical synthesis of nanosized metal oxides. Synthesis of the nanodispersed titanium dioxide by a pulsed electron beam. Non-equilibrium plasma-chemical synthesis of nanosized composite oxides. LECTURE № 20. PLASMA-CHEMICAL METHODS FOR THE PREPARATION OF CARBON NANOSTRUCTURES Alotropn Allotropic forms of carbon. Fullerenes. Non-equilibrium mechanism of synthesis of carbon nanostructures. Formation of the fullerenes from the soot. LECTURE № 21. APPLICATION OF LOW-TEMPERATURE PLASMA IN CHEMICAL MANUFACTURE Synthesis of nitrogen oxides. Plasma-chemical processing of polymers. Types of discharges used for plasma processing of polymers. Mechanisms of plasma interaction with polymers.
LECTURE № 19. PLASMA-CHEMICAL SYNTHESIS OF NANOSIZED PARTICLES Plasma-chemical synthesis of nanosized metal oxides. Synthesis of the nanodispersed titanium dioxide by a pulsed electron beam. Non-equilibrium plasma-chemical synthesis of nanosized composite oxides. LECTURE № 20. PLASMA-CHEMICAL METHODS FOR THE PREPARATION OF CARBON NANOSTRUCTURES Alotropn Allotropic forms of carbon. Fullerenes. Non-equilibrium mechanism of synthesis of carbon nanostructures. Formation of the fullerenes from the soot. LECTURE № 21. APPLICATION OF LOW-TEMPERATURE PLASMA IN CHEMICAL MANUFACTURE Synthesis of nitrogen oxides. Plasma-chemical processing of polymers. Types of discharges used for plasma processing of polymers. Mechanisms of plasma interaction with polymers.
 LECTURE № 21. PLASMA-CHEMICAL PROCESSING OF MEDICAL POLYMERS Plasma-chemical cleaning and sterilization of the surface. Crosslinking of surface modification and physico-mechanical characteristics in the plasma of inert gases. Functionalization and regulation of hydrophilic-hydrophobic surface properties of polymers in the gas-discharge plasma. LECTURE № 22. PLASMA-CHEMICAL METHODS OF WASTE TREATMENT Plasma methods of solid waste processing. The oxidation of sulfur dioxide SO 2 in the flue gases. Destruction of nitrogen oxides in gaseous wastes. Decomposition of organic compounds.
LECTURE № 21. PLASMA-CHEMICAL PROCESSING OF MEDICAL POLYMERS Plasma-chemical cleaning and sterilization of the surface. Crosslinking of surface modification and physico-mechanical characteristics in the plasma of inert gases. Functionalization and regulation of hydrophilic-hydrophobic surface properties of polymers in the gas-discharge plasma. LECTURE № 22. PLASMA-CHEMICAL METHODS OF WASTE TREATMENT Plasma methods of solid waste processing. The oxidation of sulfur dioxide SO 2 in the flue gases. Destruction of nitrogen oxides in gaseous wastes. Decomposition of organic compounds.
 International Symposium on Theoretical and Applied Plasmachemistry Russian Conference ФНТП-2007» 1 – 5 June 2007
International Symposium on Theoretical and Applied Plasmachemistry Russian Conference ФНТП-2007» 1 – 5 June 2007
 International Congress on Radiation Physics and Chemistry of Inorganic Materials, High Current Electronics, and Modification of Materials with Particle Beams and Plasma Flows Tomsk, Russia
International Congress on Radiation Physics and Chemistry of Inorganic Materials, High Current Electronics, and Modification of Materials with Particle Beams and Plasma Flows Tomsk, Russia
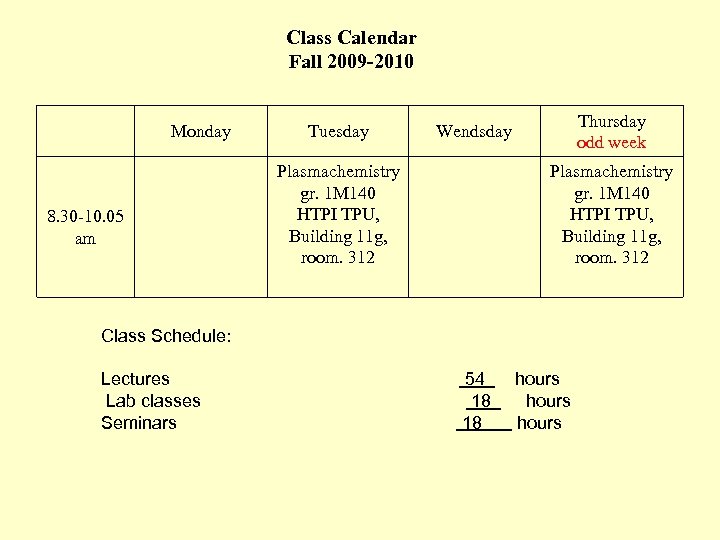 Class Calendar Fall 2009 -2010 Monday 8. 30 -10. 05 am Tuesday Plasmachemistry gr. 1 М 140 HTPI TPU, Building 11 g, room. 312 Thursday odd week Wendsday Plasmachemistry gr. 1 М 140 HTPI TPU, Building 11 g, room. 312 Class Schedule: Lectures 54 hours Lab classes 18 hours Seminars 18 hours
Class Calendar Fall 2009 -2010 Monday 8. 30 -10. 05 am Tuesday Plasmachemistry gr. 1 М 140 HTPI TPU, Building 11 g, room. 312 Thursday odd week Wendsday Plasmachemistry gr. 1 М 140 HTPI TPU, Building 11 g, room. 312 Class Schedule: Lectures 54 hours Lab classes 18 hours Seminars 18 hours
 Seminar/Laboratory Schedule (18 hours) 1. The TEU-500 Pulsed electron accelerator. Its operating principle and application in plasma chemistry 4 hours 2. Seminar on gas discharges: combustion conditions, the mechanism and application 4 hours 3. The installation of combined plasma-chemical processing of metal products 4 hours 4. The TEMP-4 M Pulsed ion accelerator. Its operating principle and application in plasma chemistry 2 hours 5. The study of Plasma-chemical conversion of methane on the laboratory instalation 4 hours
Seminar/Laboratory Schedule (18 hours) 1. The TEU-500 Pulsed electron accelerator. Its operating principle and application in plasma chemistry 4 hours 2. Seminar on gas discharges: combustion conditions, the mechanism and application 4 hours 3. The installation of combined plasma-chemical processing of metal products 4 hours 4. The TEMP-4 M Pulsed ion accelerator. Its operating principle and application in plasma chemistry 2 hours 5. The study of Plasma-chemical conversion of methane on the laboratory instalation 4 hours
 Recommended textbooks 1. Introduction to Plasma Physics: R. J. Goldston, and P. H. Rutherford (The University of Texas at Austin). – available online 2. Lectures on Plasma Physics: Professor of physics Richard Fitzpatrick (Cambridge University Press, Cambridge UK, 1994). - available online 3. RAIZER, Yu. P. : ‘Gas discharge physics’ (Springer, 1997) – available online 4. G. V. Cooray – The Lightning Flash / Publisher: The Institution of Engineering and Technology | 2003 -12 -01 | ISBN: 0852967802 | 574 p. – available online 5. House, J. E. Principles of chemical kinetics / James E. House. – 2 nd ed. p. 963. (2007) - available online 6. A. Frinman. Plasma Chemistry. New York: Cambridge University Press, 2008 - available online 7. Pushkarev A. , Isakova J. , Kholodnaya G. , Sazonov R. Sound Waves Generated Due to the Absorption of a Pulsed Electron Beam //Advances in Sound localization, chapter 12, pp. 199 -223 - ISBN 978 -953 -307 -581 -5 - Vienna: INTECH, 2011. - available online
Recommended textbooks 1. Introduction to Plasma Physics: R. J. Goldston, and P. H. Rutherford (The University of Texas at Austin). – available online 2. Lectures on Plasma Physics: Professor of physics Richard Fitzpatrick (Cambridge University Press, Cambridge UK, 1994). - available online 3. RAIZER, Yu. P. : ‘Gas discharge physics’ (Springer, 1997) – available online 4. G. V. Cooray – The Lightning Flash / Publisher: The Institution of Engineering and Technology | 2003 -12 -01 | ISBN: 0852967802 | 574 p. – available online 5. House, J. E. Principles of chemical kinetics / James E. House. – 2 nd ed. p. 963. (2007) - available online 6. A. Frinman. Plasma Chemistry. New York: Cambridge University Press, 2008 - available online 7. Pushkarev A. , Isakova J. , Kholodnaya G. , Sazonov R. Sound Waves Generated Due to the Absorption of a Pulsed Electron Beam //Advances in Sound localization, chapter 12, pp. 199 -223 - ISBN 978 -953 -307 -581 -5 - Vienna: INTECH, 2011. - available online


