5b2a53a6544f04a575e02512af385dd6.ppt
- Количество слайдов: 10

Phy 100: Heat transport Three basic forms of thermal heat transport 1) Conduction (applications); 2) Convection; 3) Radiation.

Heat conduction Consider an old brick house. The brick thickness. Outside winter temperature T=0. Inside temperature is kept at 24 c. 1) Assume the house is heated by Natural gases. Estimate the amount of natural gases used each month in units of GJ ? 2) How much energy can one save by lowering the inside temperatures ?
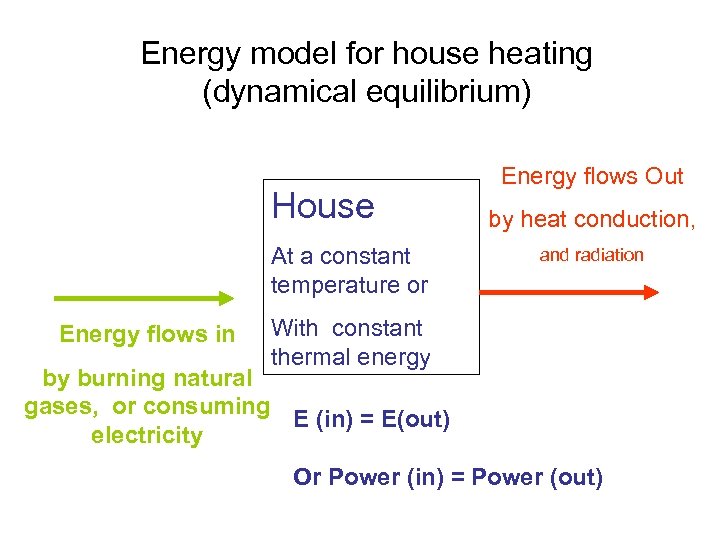
Energy model for house heating (dynamical equilibrium) House At a constant temperature or Energy flows in Energy flows Out by heat conduction, and radiation With constant thermal energy by burning natural gases, or consuming E (in) = E(out) electricity Or Power (in) = Power (out)

Approach 1) Calculate the outgoing energy (conduction loss rate) (you need to know the surface area, thickness and materials etc). 2) To maintain a constant temperature in a house, the conduction loss has to be balanced by the energy supplied by natural gases. 3) This can be converted into the amount of natural gases needed per day or per month.
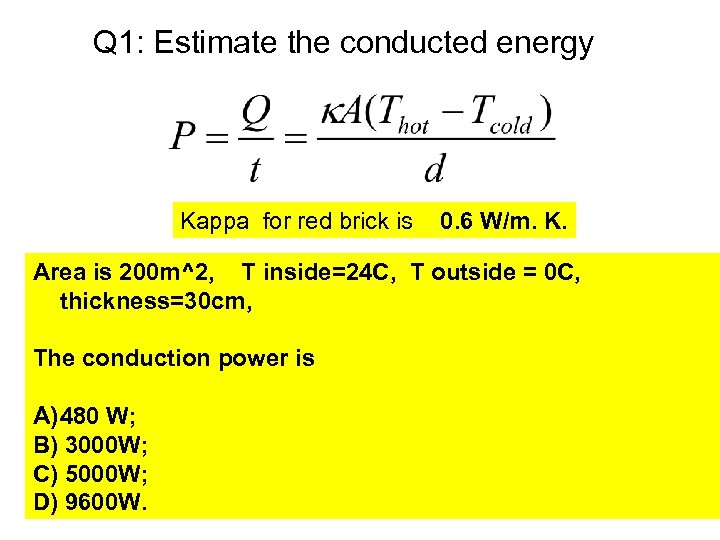
Q 1: Estimate the conducted energy Kappa for red brick is 0. 6 W/m. K. Area is 200 m^2, T inside=24 C, T outside = 0 C, thickness=30 cm, The conduction power is A) 480 W; B) 3000 W; C) 5000 W; D) 9600 W.

Solution A=200 m^2, T inside – T outside = 24 K, d=0. 3 m, Q/t = 9600 W. Q for one day is 829, 440 KJ. Q for one month is 24, 883, 200 KJ = 24. 9 GJ Terasen Gas uses GJ (=billion J) as units. 1 GJ charge is about 10 CAD. Monthly bill for house heating is about 250 CAD.

Q 2: Energy saving In previous calculations, you find that your monthly bill is 250 CAD when you keep the temperature inside at 24 C. If you keep it at 20 C, how much can you save? (Outside temperature is 0 C. ) A) B) C) D) About 40 CAD; About 80 CAD; About 100 CAD; About 120 CAD.

Energy table for natural resources • • • Propane Natural gas Heating oil Coal Wood 50 MJ/kg 47 MJ/kg 43 MJ/kg 29 MJ/kg 16 MJ/kg Also read Table 6. 2 for food. 0. 5 kg/m 3 0. 8 kg/m 3
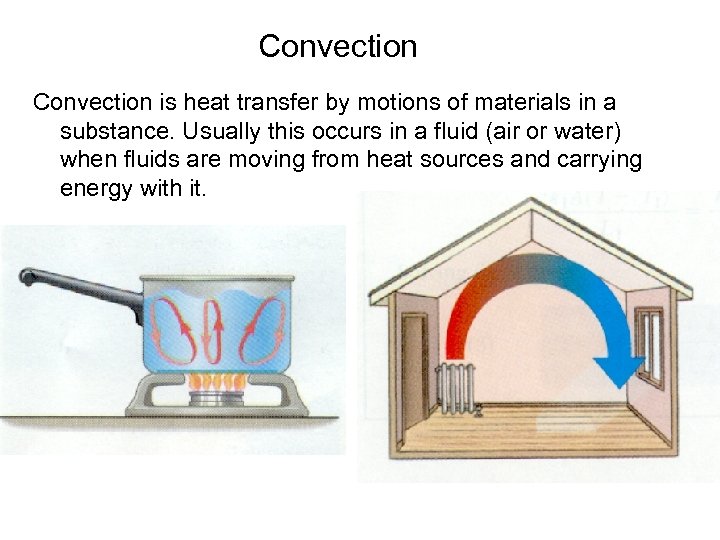
Convection is heat transfer by motions of materials in a substance. Usually this occurs in a fluid (air or water) when fluids are moving from heat sources and carrying energy with it.

Radiation Heat can also be transferred by radiating light (i. e. photons) or electromagnetic waves with different wavelengths. The heat is transferred from the sun to the earth mostly by visible light.
5b2a53a6544f04a575e02512af385dd6.ppt