4d82168b50cc49ea17cf0b848f84995e.ppt
- Количество слайдов: 14
 PHARMASUM THERAPEUTICS AS 1 WE DEVELOP DRUG TREATMENTS FOR ALZHEIMER’S DISEASE ANDERS FUGELLI, PHD CHIEF EXECUTIVE OFFICER DEMENTIA INVESTMENT FORUM 15 TH APRIL 2015 NOT FOR GENERAL DISTRIBUTION
PHARMASUM THERAPEUTICS AS 1 WE DEVELOP DRUG TREATMENTS FOR ALZHEIMER’S DISEASE ANDERS FUGELLI, PHD CHIEF EXECUTIVE OFFICER DEMENTIA INVESTMENT FORUM 15 TH APRIL 2015 NOT FOR GENERAL DISTRIBUTION
 An attractive investment seeking 5 MNOK (GBP 500 k) • Targeting disease area with high unmet medical need • Team with excellence in drug discovery and commercialisation • Coherent plan • Potential for high-value M&A exit in 5 -7 years • Current investment will deliver value-inflection NOT FOR GENERAL DISTRIBUTION 2
An attractive investment seeking 5 MNOK (GBP 500 k) • Targeting disease area with high unmet medical need • Team with excellence in drug discovery and commercialisation • Coherent plan • Potential for high-value M&A exit in 5 -7 years • Current investment will deliver value-inflection NOT FOR GENERAL DISTRIBUTION 2
 Alzheimer’s disease has a huge unmet medical need § Disease first described in 1906 by the physician Alois Alzheimer; Cells in the brain gradually die and leads to impaired memory, behaviour, sleep and death within 10 years § > 10 % of everyone above 75 yrs suffer from Alzheimer’s § Care and nursing for demented individuals costs > 1% of the global GDP and number is expected to triple within 2050 § There is no cure, only symptomatic treatments Pharmasum is developing drugs targeting the processes that is currently considered to be central to the disease NOT FOR GENERAL DISTRIBUTION 3 Frau Auguste Deter Patient described by Alzheimer
Alzheimer’s disease has a huge unmet medical need § Disease first described in 1906 by the physician Alois Alzheimer; Cells in the brain gradually die and leads to impaired memory, behaviour, sleep and death within 10 years § > 10 % of everyone above 75 yrs suffer from Alzheimer’s § Care and nursing for demented individuals costs > 1% of the global GDP and number is expected to triple within 2050 § There is no cure, only symptomatic treatments Pharmasum is developing drugs targeting the processes that is currently considered to be central to the disease NOT FOR GENERAL DISTRIBUTION 3 Frau Auguste Deter Patient described by Alzheimer
 An international, experienced team Anders Fugelli, Ph. D. , Founder and Chief Executive Officer More than 20 years experience in neuroscience research, pharmaceutical and biotechnology industry – Neurology at GSK, start-up experience Lytix Biopharma Ph. D in Neuropharmacology / biochemistry from Imperial College, London Corporate Finance study module, London Business School John Sigurd Svendsen, Ph. D. , Co-founder and Chief Scientific Officer Professor of organic chemistry in the Faculty of Sciences at the University of Tromsø, Norway; start-up experience Lytix Biopharma Extensive research experience from work in medical biochemistry, synthetic organic chemistry and medicinal chemistry Ph. D from the University of Tromsø - postdoc with Professor K. B. Sharpless (Nobel Laureate, Chemistry, 2000) at MIT. Rich Engh, Ph. D. , Advisor on Structural Biology Professor at the University of Tromsø, Norway Extensive experience protein kinase x-ray crystallography, industry focused R&D (Boehringer Ingelheim, Roche) Ph. D University of Chicago - postdoc with Professor Robert Huber (Nobel Laureate, Chemistry, 1988) at Max-Planck. Pauline Stewart-Long, Ph. D. , Head of Project Management More than 27 years broad experience as Pharmaceuticals R&D Project Manager Previously Head of GSK Global R&D Project Management Henning Mork, Chief Financial Officer More than 25 years financial experience within industry Senior managment experience from listed as well as private, start-up companies Bachelor in Marketing at BI with additional studies in finance and political science NOT FOR GENERAL DISTRIBUTION 4
An international, experienced team Anders Fugelli, Ph. D. , Founder and Chief Executive Officer More than 20 years experience in neuroscience research, pharmaceutical and biotechnology industry – Neurology at GSK, start-up experience Lytix Biopharma Ph. D in Neuropharmacology / biochemistry from Imperial College, London Corporate Finance study module, London Business School John Sigurd Svendsen, Ph. D. , Co-founder and Chief Scientific Officer Professor of organic chemistry in the Faculty of Sciences at the University of Tromsø, Norway; start-up experience Lytix Biopharma Extensive research experience from work in medical biochemistry, synthetic organic chemistry and medicinal chemistry Ph. D from the University of Tromsø - postdoc with Professor K. B. Sharpless (Nobel Laureate, Chemistry, 2000) at MIT. Rich Engh, Ph. D. , Advisor on Structural Biology Professor at the University of Tromsø, Norway Extensive experience protein kinase x-ray crystallography, industry focused R&D (Boehringer Ingelheim, Roche) Ph. D University of Chicago - postdoc with Professor Robert Huber (Nobel Laureate, Chemistry, 1988) at Max-Planck. Pauline Stewart-Long, Ph. D. , Head of Project Management More than 27 years broad experience as Pharmaceuticals R&D Project Manager Previously Head of GSK Global R&D Project Management Henning Mork, Chief Financial Officer More than 25 years financial experience within industry Senior managment experience from listed as well as private, start-up companies Bachelor in Marketing at BI with additional studies in finance and political science NOT FOR GENERAL DISTRIBUTION 4
 A highly qualified incoming Board – R&D and Commercialisation Andrew A Parsons, Ph. D. , Chairman Dr. Parsons has vast experience from preclinial R&D, primarily within the CNS field. He manages Reciprocal Minds, a consultancy company focusing on building strong crosscultural teams. Previously, he was Vice-President and founding member of the GSK system for external innovation - Centre of Excellence for External Drug Discovery (CEEDD). Previously, at GSK Dr. Parsons was head of preclinical R&D and member of the Management Team at Centre of Excellence for Drug Discovery (CEDD) for Neurology and GI. He served as the Head of the global Imigran Scientifc Advisory Board. Øystein Soug, M. Sc. , Non-executive Director Øystein Soug was until recently CFO of the Norwegian biotechnology company Algeta, where he started in 2008. Algeta witnessed extensive growth, with an increase in share price from approx. NOK 8 in Dec. 2008 to around NOK 362 when it was sold to Bayer in March 2014. Mr. Soug has in this period been responsible for building up the Finance, IR, IT and HR functions of the company and gained broad experience through three equity fund raisings and one large out-licensing deal. Prior to Algeta, he worked six years at the Norwegian industrial group Orkla, lastly as CFO of Orkla's Russian arm, and previously within Orkla's Corporate Development division focusing on M&A. Mr. Soug started his career as a banker with Credit Suisse and with the European Bank for Reconstruction and Development. Mr. Soug received an MSc in Economics and Financial Markets from Universität St. Gallen in Switzerland in 1997. Masha Strømme, Ph. D, Non-executive Director Dr. Strømme has a Ph. D in Alzheimer’s disease from University of Oxford, UK and extensive experience in Life Sciences Investment Banking from Morgan Stanley and Altium Capital (Apax) in London. She is member of the Board of Navamedic ASA, Lipi. Dx AS and Mylife products AS. She works as advisor for Artic Securities and Oslo Medtech. John Sigurd Svendsen, Ph. D. , CSO and Non-executive Director See management bio Anders Fugelli, CEO – reporting to Board NOT FOR GENERAL DISTRIBUTION 5
A highly qualified incoming Board – R&D and Commercialisation Andrew A Parsons, Ph. D. , Chairman Dr. Parsons has vast experience from preclinial R&D, primarily within the CNS field. He manages Reciprocal Minds, a consultancy company focusing on building strong crosscultural teams. Previously, he was Vice-President and founding member of the GSK system for external innovation - Centre of Excellence for External Drug Discovery (CEEDD). Previously, at GSK Dr. Parsons was head of preclinical R&D and member of the Management Team at Centre of Excellence for Drug Discovery (CEDD) for Neurology and GI. He served as the Head of the global Imigran Scientifc Advisory Board. Øystein Soug, M. Sc. , Non-executive Director Øystein Soug was until recently CFO of the Norwegian biotechnology company Algeta, where he started in 2008. Algeta witnessed extensive growth, with an increase in share price from approx. NOK 8 in Dec. 2008 to around NOK 362 when it was sold to Bayer in March 2014. Mr. Soug has in this period been responsible for building up the Finance, IR, IT and HR functions of the company and gained broad experience through three equity fund raisings and one large out-licensing deal. Prior to Algeta, he worked six years at the Norwegian industrial group Orkla, lastly as CFO of Orkla's Russian arm, and previously within Orkla's Corporate Development division focusing on M&A. Mr. Soug started his career as a banker with Credit Suisse and with the European Bank for Reconstruction and Development. Mr. Soug received an MSc in Economics and Financial Markets from Universität St. Gallen in Switzerland in 1997. Masha Strømme, Ph. D, Non-executive Director Dr. Strømme has a Ph. D in Alzheimer’s disease from University of Oxford, UK and extensive experience in Life Sciences Investment Banking from Morgan Stanley and Altium Capital (Apax) in London. She is member of the Board of Navamedic ASA, Lipi. Dx AS and Mylife products AS. She works as advisor for Artic Securities and Oslo Medtech. John Sigurd Svendsen, Ph. D. , CSO and Non-executive Director See management bio Anders Fugelli, CEO – reporting to Board NOT FOR GENERAL DISTRIBUTION 5
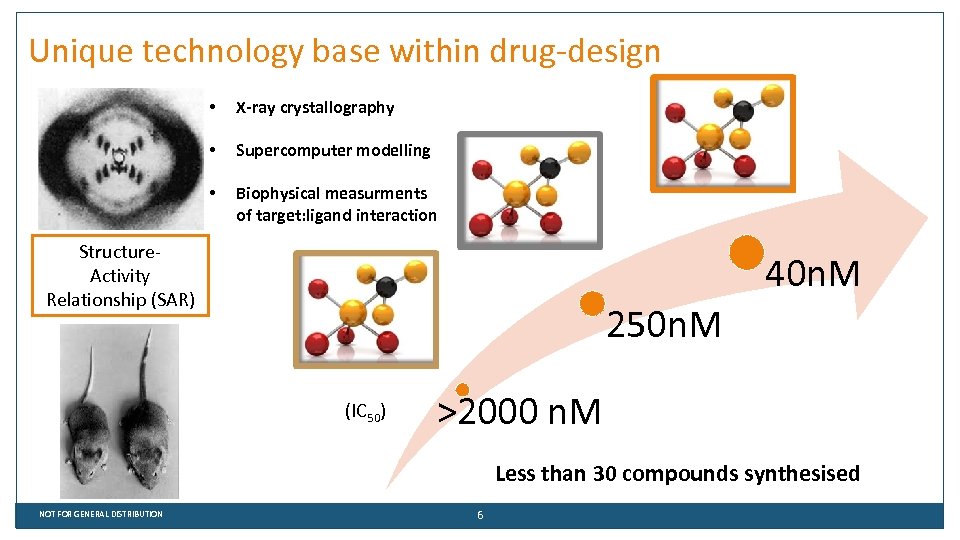 Unique technology base within drug-design • X-ray crystallography • Supercomputer modelling • Biophysical measurments of target: ligand interaction X-ray crystallography Drug design know-how Structure. Activity Relationship (SAR) 40 n. M 250 n. M (IC 50) >2000 n. M Less than 30 compounds synthesised NOT FOR GENERAL DISTRIBUTION 6
Unique technology base within drug-design • X-ray crystallography • Supercomputer modelling • Biophysical measurments of target: ligand interaction X-ray crystallography Drug design know-how Structure. Activity Relationship (SAR) 40 n. M 250 n. M (IC 50) >2000 n. M Less than 30 compounds synthesised NOT FOR GENERAL DISTRIBUTION 6
 Protein kinase enzyme DYRK 1 A is linked to dementia ØClosely ØIs involved in brain development increased in brain of Alzheimer’s patients ØGenetically linked to Alzheimer’s and dementia in other neurological diseases ØDYRK 1 A is involved both with tangles and plaques by promoting their formation This is our drug target NOT FOR GENERAL DISTRIBUTION 7 Normal brain Alzheimer’s
Protein kinase enzyme DYRK 1 A is linked to dementia ØClosely ØIs involved in brain development increased in brain of Alzheimer’s patients ØGenetically linked to Alzheimer’s and dementia in other neurological diseases ØDYRK 1 A is involved both with tangles and plaques by promoting their formation This is our drug target NOT FOR GENERAL DISTRIBUTION 7 Normal brain Alzheimer’s
 Progress last 12 months Ø Total of six proprietary X-ray structures of enzym : ligand (1. 8Å resolution) Ø Documented unique, chemical scaffold (series 2) Ø Key CNS drug-like characteristics established Ø Ø Ø Ø High activity Good selectivity High solubility Stability in liver microsomes Distribution to brain tissue Significant free fraction in brain Effective in cellular models NOT FOR GENERAL DISTRIBUTION 8
Progress last 12 months Ø Total of six proprietary X-ray structures of enzym : ligand (1. 8Å resolution) Ø Documented unique, chemical scaffold (series 2) Ø Key CNS drug-like characteristics established Ø Ø Ø Ø High activity Good selectivity High solubility Stability in liver microsomes Distribution to brain tissue Significant free fraction in brain Effective in cellular models NOT FOR GENERAL DISTRIBUTION 8
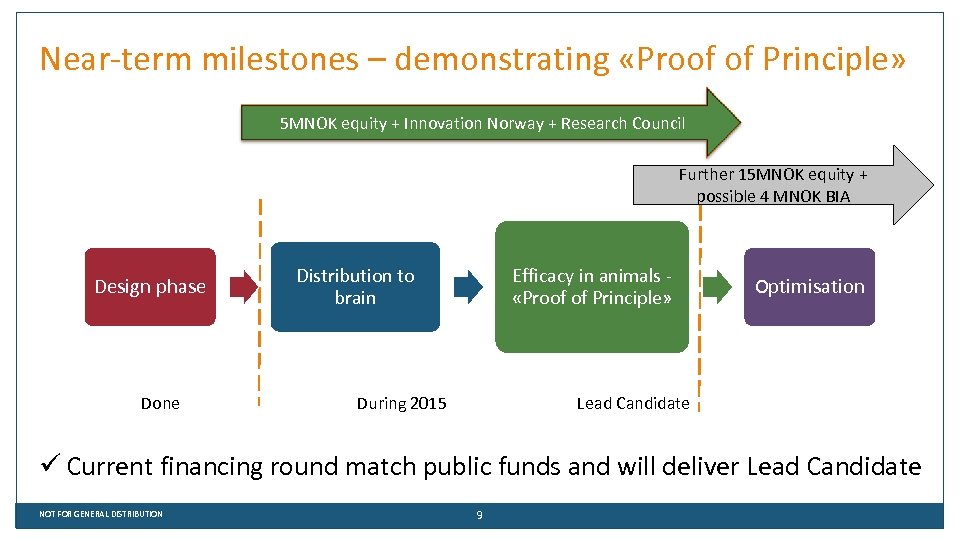 Near-term milestones – demonstrating «Proof of Principle» 5 MNOK equity + Innovation Norway + Research Council Further 15 MNOK equity + possible 4 MNOK BIA Design phase Done Distribution to brain Efficacy in animals «Proof of Principle» During 2015 Optimisation Lead Candidate ü Current financing round match public funds and will deliver Lead Candidate NOT FOR GENERAL DISTRIBUTION 9
Near-term milestones – demonstrating «Proof of Principle» 5 MNOK equity + Innovation Norway + Research Council Further 15 MNOK equity + possible 4 MNOK BIA Design phase Done Distribution to brain Efficacy in animals «Proof of Principle» During 2015 Optimisation Lead Candidate ü Current financing round match public funds and will deliver Lead Candidate NOT FOR GENERAL DISTRIBUTION 9
 Business strategy – build an attractive R&D company «Big Pharma» needs to buy projects and companies to grow Our exit strategy is either: Ø to be acquired, or Ø to pursue development and list the company on the stock exchange For comparison: Ø i. Perian developed an m. Ab drug for Alzheimer against protein Tau Ø Ø Prosject was in preclinical phase, ready for start of phase 1 in 2015 Bristol Myers-Squibb acquired company in March 2014 for total of USD 725 m Ø AC Immune develops m. Abs for Alzheimer Ø Ø m. Ab propject against Tau was in clinical phase 1 In January 2015, Johnson & Johnson bought rights through license worth USD 509 m NOT FOR GENERAL DISTRIBUTION 10
Business strategy – build an attractive R&D company «Big Pharma» needs to buy projects and companies to grow Our exit strategy is either: Ø to be acquired, or Ø to pursue development and list the company on the stock exchange For comparison: Ø i. Perian developed an m. Ab drug for Alzheimer against protein Tau Ø Ø Prosject was in preclinical phase, ready for start of phase 1 in 2015 Bristol Myers-Squibb acquired company in March 2014 for total of USD 725 m Ø AC Immune develops m. Abs for Alzheimer Ø Ø m. Ab propject against Tau was in clinical phase 1 In January 2015, Johnson & Johnson bought rights through license worth USD 509 m NOT FOR GENERAL DISTRIBUTION 10
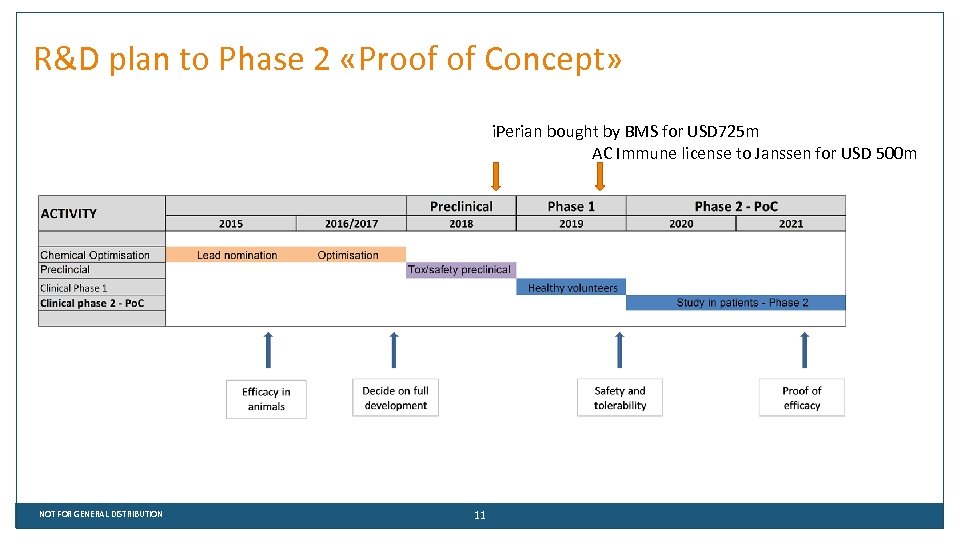 R&D plan to Phase 2 «Proof of Concept» i. Perian bought by BMS for USD 725 m AC Immune license to Janssen for USD 500 m NOT FOR GENERAL DISTRIBUTION 11
R&D plan to Phase 2 «Proof of Concept» i. Perian bought by BMS for USD 725 m AC Immune license to Janssen for USD 500 m NOT FOR GENERAL DISTRIBUTION 11
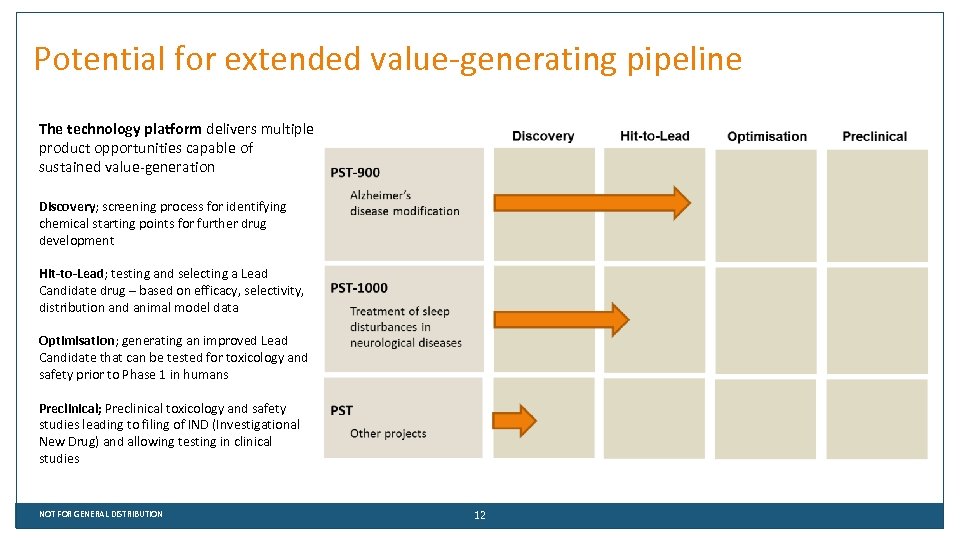 Potential for extended value-generating pipeline The technology platform delivers multiple product opportunities capable of sustained value-generation Discovery; screening process for identifying chemical starting points for further drug development Hit-to-Lead; testing and selecting a Lead Candidate drug – based on efficacy, selectivity, distribution and animal model data Optimisation; generating an improved Lead Candidate that can be tested for toxicology and safety prior to Phase 1 in humans Preclinical; Preclinical toxicology and safety studies leading to filing of IND (Investigational New Drug) and allowing testing in clinical studies NOT FOR GENERAL DISTRIBUTION 12
Potential for extended value-generating pipeline The technology platform delivers multiple product opportunities capable of sustained value-generation Discovery; screening process for identifying chemical starting points for further drug development Hit-to-Lead; testing and selecting a Lead Candidate drug – based on efficacy, selectivity, distribution and animal model data Optimisation; generating an improved Lead Candidate that can be tested for toxicology and safety prior to Phase 1 in humans Preclinical; Preclinical toxicology and safety studies leading to filing of IND (Investigational New Drug) and allowing testing in clinical studies NOT FOR GENERAL DISTRIBUTION 12
 An attractive investment proposition seeking 5 MNOK • Targeting high unmet medical need • Excellence in drug discovery • Coherent plan • Potential for high-value M&A exit in 5 -7 years • Current investment will deliver value-inflection NOT FOR GENERAL DISTRIBUTION 13
An attractive investment proposition seeking 5 MNOK • Targeting high unmet medical need • Excellence in drug discovery • Coherent plan • Potential for high-value M&A exit in 5 -7 years • Current investment will deliver value-inflection NOT FOR GENERAL DISTRIBUTION 13
 Thank you for your attention Anders Fugelli, Founder and CEO Pharmasum Therapeutics Phone: +47 924 81432 Email: afugelli@pharmasum. com NOT FOR GENERAL DISTRIBUTION John Sigurd Svendsen, Founder and CSO Pharmasum Therapeutics Phone: +47 900 95972 Email: john-sigurd. svendsen@uit. no 14
Thank you for your attention Anders Fugelli, Founder and CEO Pharmasum Therapeutics Phone: +47 924 81432 Email: afugelli@pharmasum. com NOT FOR GENERAL DISTRIBUTION John Sigurd Svendsen, Founder and CSO Pharmasum Therapeutics Phone: +47 900 95972 Email: john-sigurd. svendsen@uit. no 14


