777161929f5b611d02fe7d888056651c.ppt
- Количество слайдов: 48

Pharmacovigilance and the WHO Collaborating Centre for International Drug Monitoring in Uppsala Technical Briefing Seminar in Essential Medicines Policies, Geneva, October 2007 Ronald Meyboom, MD, Ph. D The Uppsala Monitoring Centre www. who-umc. org

16% of hospital admissions are drugrelated (medical ward). Nelson KM, Talbert RL. Pharmacotherapy 1996; 16: 701 -7. Adverse drug reactions are the 5 th leading cause of death in a hospital. Lazarou J. Pomeranz BH, Corey PN. JAMA 1998; 279: 1200 -5. Avoidable in ca. 50 %

Definition of Pharmacovigilance: (WHO, 2002, ISBN 9241590157) • The science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other drug-related problems • Treatment evaluation science

Why pharmacovigilance? • Clinical trials focus on demonstrating efficacy and tolerability (selected ‘healthy’ patients, limited in number and duration) • Incomplete knowledge, e. g. effectiveness, rare but serious adverse reactions, interactions, ‘real-live’ patients, subpopulations • Do not produce all the information needed for the balance of benefit and harm

How pharmacovigilance? • • • Spontaneous Reporting Intensive monitoring (hospital) Prescription Event Monitoring Case Control Surveillance Comprehensive population databases, data-mining • Patient series • Observational studies

Formal studies • Defined aim (identified problem) • Hypothesis testing (problem solving) • Established methods (clinical trial, case control, cohort) • Comparison • Limited in time, drugs, population, place Vigilance • Open question: looking for the unexpected • Hypothesis generation (‘problem raising’) • Exploratory, controversial (SR, PEM, CCS) • No comparison • Continuous, all drugs, total population



Emphasis on • • Early warning Generation of knowledge Dissemination of information Rational and safe use of medicines – Benefit and harm together

Three categories of adverse drug reactions Need different methods for detection An ABC of drug-related problems. Drug Saf 2000; 22: 415 -23 Type A (pharmacological) Type B (hypersensitivity) Type C (more frequent in exposed than in unexposed) Clinical trial Spontaneous reporting Pharmacoepidemiology challenge

Spontaneous Reporting • A country-wide system for the reporting of suspected adverse reactions to drugs • A case report is a notification from a health care professional, describing the history of a patient with a disorder that is suspected to be drug-induced • Limitations because of privacy protection and medical secrecy

Spontaneous Reporting • When different doctors independently report the same unknown and unexpected adverse experience with a drug, this may be a valid early signal • Quantitative: more frequently reported than expected from the background

Advantages of Spontaneous Reporting • • • Effective ‘All’ patients; ‘all’ drugs; many ADRs Continuous Rapid Cheap Not much health care infrastructure needed

Limitations of Spontaneous Reporting • Suspicions, incomplete, uncertain • Underreporting is vast but unknown and variable • Exposure data available? • No frequency measurement • Comparison of drugs difficult • Insensitive to type C adverse effects • Further study for signal testing and explanation

Data assessment in pharmacovigilance 1. Individual case report assessment • • Interest, relevance (new, serious? ) Medical, pharmacological; coding Follow-up Causality assessment 2. Aggregated study and interpretation • • Signal detection Risk factors, interactions Serial (clinicopathological) study Frequency estimation

General design of systems for causality assessment Drug Safety 1997; 17: 374 -389 • Basic questions – Sub-questions • Scores • Overall score • Causality category, e. g. possible, probable, etc

None of the available systems has been validated (i. e. shown to consistently and reproducibly gives a reasonable approximation of the truth) • Validation = ‘proving that a procedure actually leads to the expected results’ • No gold standard • Causality category definitions

• What causality assessment can do assessment cannot do – Decrease disagreement between assessors – Classify relationship likelihood (semiquantitative) – Mark individual case reports – Education / improvement of scientific assessment – Give accurate quantitative measurement of relationship likelihood – Distinguish valid from invalid cases – Prove the connection between drug and event – Quantify the contribution of a drug to the development of an adverse event – Change uncertainty into certainty

Underreporting • • Vast (> 90%) Unknown Variable Biased Difficult to adjust for No frequency calculation Delays signal detection

A signal is a set of data constituting a hypothesis that is relevant to the rational and safe use of a medicine Hypothesis, data, arguments • • • Pharmacological Clinical/pathological Epidemiological Quantitative / qualitative Dynamic; develops over time Drug Safety 1997; 17: 355 -65.
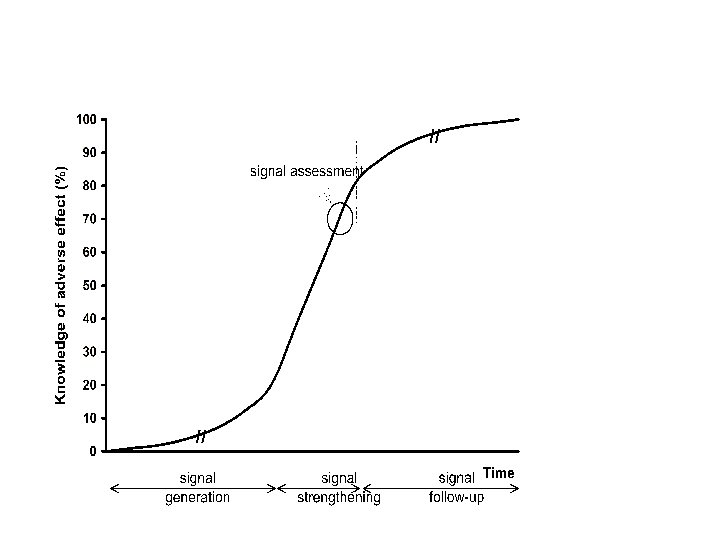

The balance of evidence in a signal • Quantitative strength of the association – number of case reports – statistical disproportionality – drug exposure • Consistency of the data (pattern) • Exposure-response relationship – site, timing, dose, reversibility • Biological plausibility of hypothesis – pharmacological, pathological • Experimental findings – e. g. dechallenge, rechallenge, blood levels, metabolites, drugdependent antibodies • Analogies • Nature and quality of the data – objectivity, documentation, causality assessment

• Signal detection is searching for the unknown. The same data can lead to different conclusions. Since the truth is unknown it is uncertain who is right, but nobody is wrong! • Dilemma: a signal should be early and credible at the same time • Signals may consist of only a few cases and may not be statistically prominent • A signal is a snapshot and changes over time • Signal testing and explanation require further study • Many signals remain unconfirmed – scientific limitations – no funding

WHO Collaborating Centre for International Drug Monitoring The Uppsala Monitoring Centre Stora Torget 3, 75320 Uppsala, Sweden www. who-umc. org

The Uppsala Monitoring Centre • 1968 - WHO Collaborating Centre for International Drug Monitoring, Geneva • 1978 - Moved to Uppsala after agreement between Sweden and WHO • Non-profit foundation with international administrative board • WHO Headquarters responsible for policy • Self-financing • Global pharmacovigilance

The Uppsala Monitoring Centre • • • Director: Prof Ralph Edwards Deputy director: Dr Marie Lindquist International affairs: Sten Olsson Pharmacists (Bio)medics IT specialists Financing (‘Products and Services’) Administrative Together 45
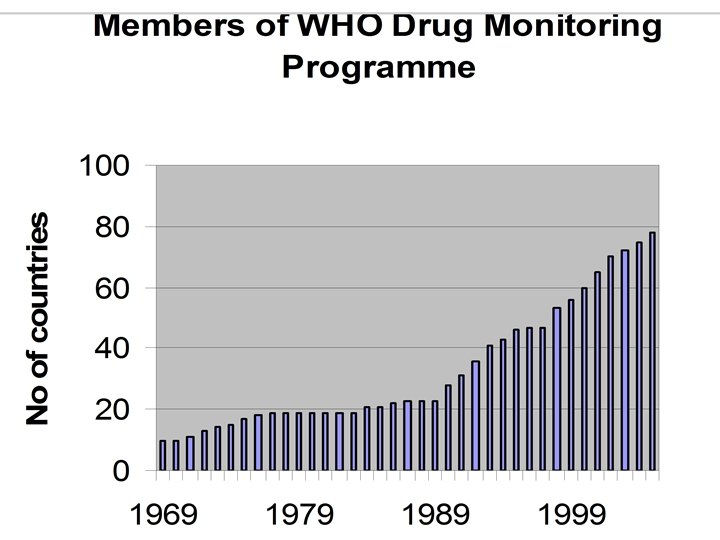

WHO International Pharmacovigilance Programme, March 2006

Aims and activities • Collaboration with National Centres • World-wide collection, analysis and distribution of data – Signal detection and analysis – Pooling of data, comparing experiences • • Communication, exchange of information Technical support Development of methods and tools Improvement of pharmacovigilance around the world
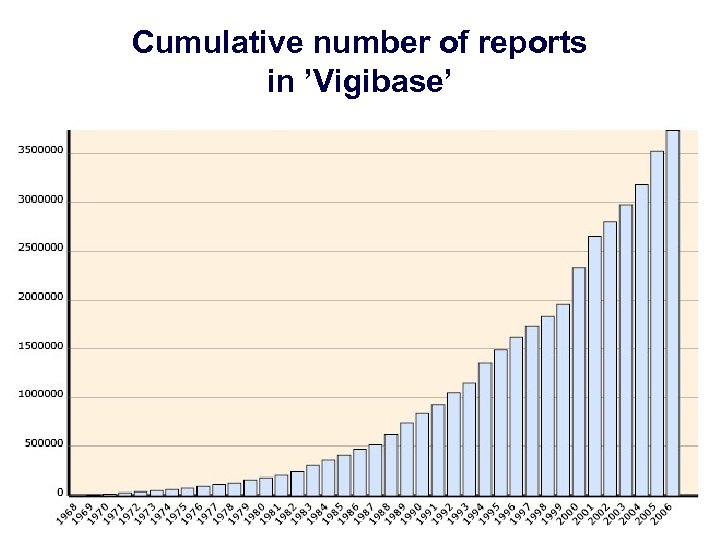
Cumulative number of reports in ’Vigibase’
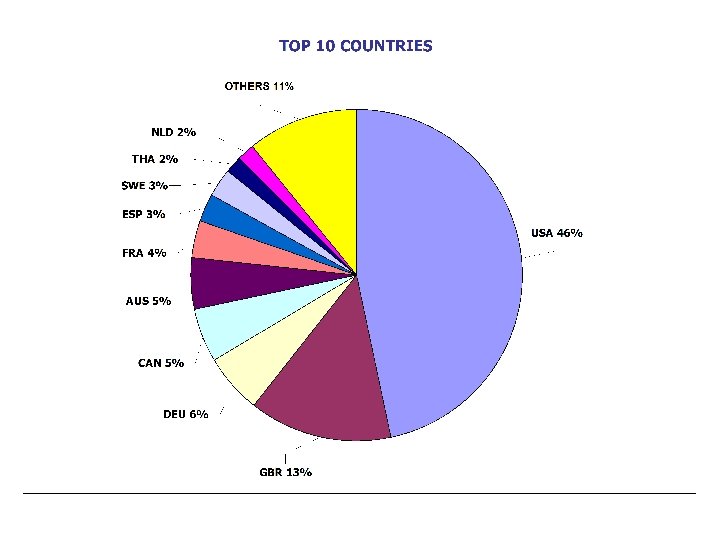

World-wide accumulation and assessment of data • 80 participating National Pharmacovigilance Centres around the world • 3. 5 million case reports • Early warning - acceleration of signal detection • Early signal strengthening by comparing countries

Automated quantitative signal detection • Extremely large numbers of drug adverse reaction combinations • Selects automatically high-interest combinations, using quantitative disproportionality • Manageable subsets of data • No human time needed • No investigators bias • Objective, transparent, reproducible • Flexible / adjustable • Explorative

Signal detection at the Uppsala Monitoring Centre Eur J Clin Pharmacol 1998; 54: 315 -321 A combination of 1. Automated quantitative data mining, using Bayesian statistics and a neural network architecture (Information Component – ‘IC value’) 2. ‘Triage’ 3. Human assessment – National Centres – Review Panel – UMC staff

Triage filter, combining quantitative and qualitative criteria; automatic selection of associations that • IC 025 > 0; two or more countries • Quarterly IC increase of 2 or more • New and serious (WHOART Critical Terms) • Target reaction terms (e. g. SJS), two or more reports, irrespective of IC value ü Literature check
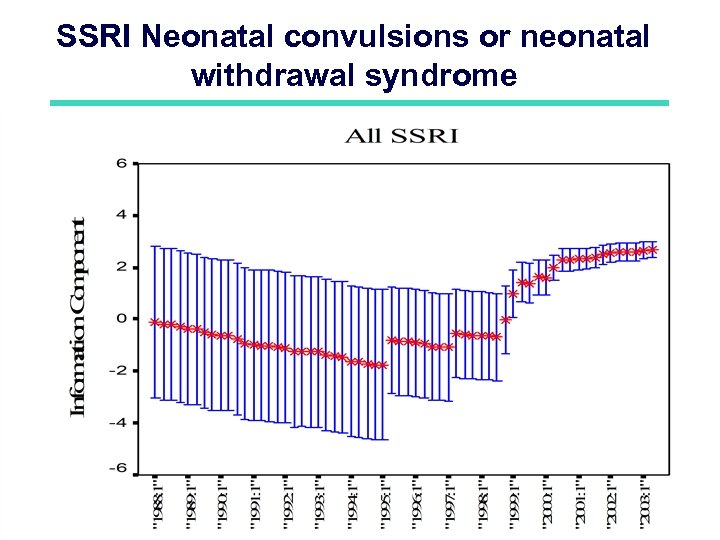
SSRI Neonatal convulsions or neonatal withdrawal syndrome

Example of results in one Quarter (2004) Total number of combinations: No. of associations IC 025 > 0: Triage selection: No. of signals for SIGNAL: 60000 2300 560 28

Signal Review Panel • 40 Experts around the world • Evaluate signals, together with UMC staff and National Centres • Select associations for follow-up • Write signals in the SIGNAL document • Preference for System Organ Class or drug group (ATC)

The SIGNAL document • Sent to all National Centres (national distribution) • Individualized section available to industry • All recipients encouraged to comment on topics presented

Presentations at ISOP annual meeting 2006 • HMG-Co. A inhibitors and pulmonary fibrosis • β-2 -Adrenoceptor agonists and nocturnal enuresis • Systemic effects of intranasal corticosteroids (neuropsychiatric reactions, spontaneous abortion) • Taxoids (paclitaxel and docetaxel) and myocardial infarction • Hypersensitivity reactions to Umckaloabo (Pelargonium sidoides and P. reniforme) • Potentiation of warfarin by glucosamine

Examples of articles • Clark DW, Strandell J. Myopathy including polymyositis: a likely class adverse effect of proton pump inhibitors? Eur J Clin Pharmacol 2006; 62: 473 -9. • Sanz E, et al. Selective serotonin reuptake inhibitors in pregnant women and neonatal withdrawal syndrome. Lancet 2005; 365: 482 -7. • Coulter D, et al. Antipsychotic drugs and heart muscle disorders in international pharmacovigilance: data mining study. BMJ 2001; 322: 1207 -9.

Support to National Centres • • Methodology Terminologies, guidelines Software (VIGIFLOW) Harmonisation, standardisation VIGIMED email discussion group Annual meetings Training Books and brochures

Terminologies, guidelines Links with WHO Geneva, CIOMS, ICH • WHOART • Drug Dictionary • Guidelines for setting up and running of a Pharmacovigilance Centre www. who- umc. org/Dyn. Page. aspx? id=13136&mn=1512#8 • Herbal ATC • Accepted scientific names of therapeutic plants. 2005, ISBN 91 974750 3 3. • WHO guidelines on safety monitoring of herbal medicines

Harmonisation, standardisation • Definitions (Biriell C, Edwards IR. Drug Safety 1994; 23: 95 -9) • WHO causality categories • Reporting adverse drug reactions. Definitions of terms and criteria for their use (CIOMS Council for International Organizations of Medical Sciences. WHO, 1999, Geneva. ISBN 92 9036 071 2. )

Herbal and traditional medicines • UMC Herbals database (Dr Mohamed Farah) • Herbal reviewers panel • Collaboration with – Uppsala University, Sweden – WHO Collaborating Centre, Cape Town, South Africa – Royal Botanical Garden, Kew, UK – University of Exeter, UK – Harvard, US

New development areas • Integrate Chinese ADR database • Patient safety focus including medication errors – World Alliance for Patient Safety • Improved reporting and analysis of vaccine reactions (AEFI) – Flu pandemic planning • Safety surveillance for other Public Health Programmes • Involvement in active surveillance – Cohort Event Monitoring • Data mining analysis of longitudinal patient records

Thank you for your attention info@who-umc. org www. who-umc. org
777161929f5b611d02fe7d888056651c.ppt