045626a9c21f17c6cf0e44e125127240.ppt
- Количество слайдов: 60
 PHARMACOTHERAPY for SMOKING CESSATION
PHARMACOTHERAPY for SMOKING CESSATION
 “CIGARETTE SMOKING… is the chief, single, avoidable cause of death in our society and the most important public health issue of our time. ” C. Everett Koop, M. D. , former U. S. Surgeon General
“CIGARETTE SMOKING… is the chief, single, avoidable cause of death in our society and the most important public health issue of our time. ” C. Everett Koop, M. D. , former U. S. Surgeon General
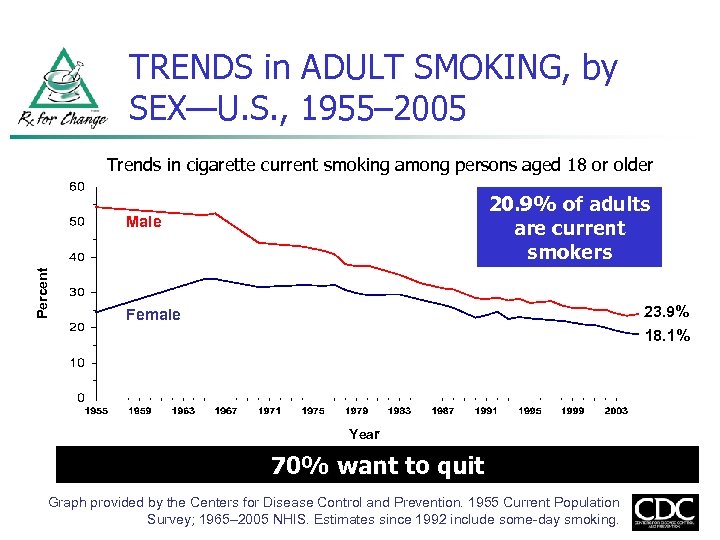 TRENDS in ADULT SMOKING, by SEX—U. S. , 1955– 2005 Trends in cigarette current smoking among persons aged 18 or older 20. 9% of adults are current smokers Percent Male 23. 9% Female 18. 1% Year 70% want to quit Graph provided by the Centers for Disease Control and Prevention. 1955 Current Population Survey; 1965– 2005 NHIS. Estimates since 1992 include some-day smoking.
TRENDS in ADULT SMOKING, by SEX—U. S. , 1955– 2005 Trends in cigarette current smoking among persons aged 18 or older 20. 9% of adults are current smokers Percent Male 23. 9% Female 18. 1% Year 70% want to quit Graph provided by the Centers for Disease Control and Prevention. 1955 Current Population Survey; 1965– 2005 NHIS. Estimates since 1992 include some-day smoking.
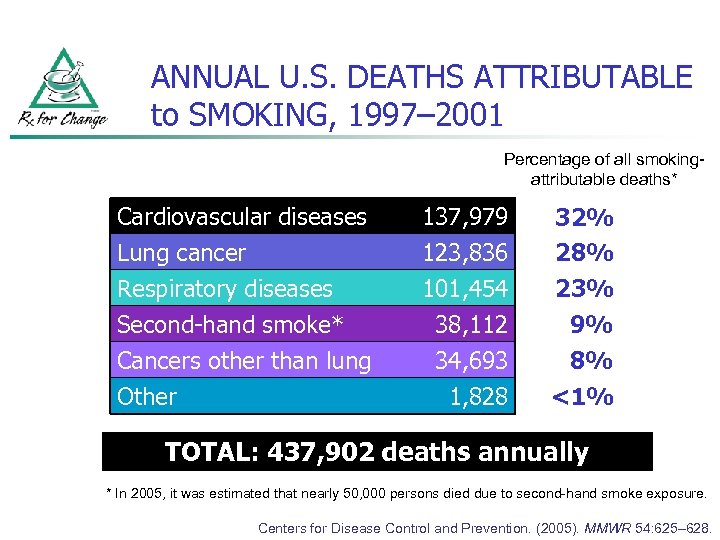 ANNUAL U. S. DEATHS ATTRIBUTABLE to SMOKING, 1997– 2001 Percentage of all smokingattributable deaths* Cardiovascular diseases Lung cancer Respiratory diseases Second-hand smoke* 137, 979 123, 836 101, 454 38, 112 Cancers other than lung Other 34, 693 1, 828 32% 28% 23% 9% 8% <1% TOTAL: 437, 902 deaths annually * In 2005, it was estimated that nearly 50, 000 persons died due to second-hand smoke exposure. Centers for Disease Control and Prevention. (2005). MMWR 54: 625– 628.
ANNUAL U. S. DEATHS ATTRIBUTABLE to SMOKING, 1997– 2001 Percentage of all smokingattributable deaths* Cardiovascular diseases Lung cancer Respiratory diseases Second-hand smoke* 137, 979 123, 836 101, 454 38, 112 Cancers other than lung Other 34, 693 1, 828 32% 28% 23% 9% 8% <1% TOTAL: 437, 902 deaths annually * In 2005, it was estimated that nearly 50, 000 persons died due to second-hand smoke exposure. Centers for Disease Control and Prevention. (2005). MMWR 54: 625– 628.
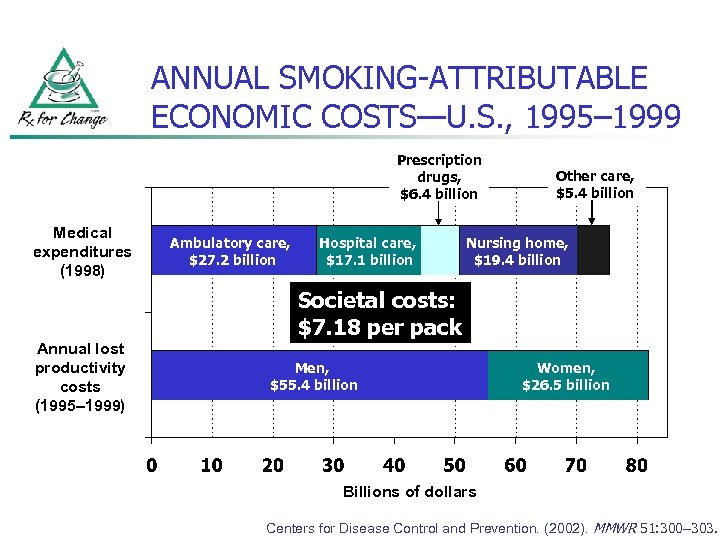 ANNUAL SMOKING-ATTRIBUTABLE ECONOMIC COSTS—U. S. , 1995– 1999 Prescription drugs, $6. 4 billion Medical expenditures (1998) Ambulatory care, $27. 2 billion Hospital care, $17. 1 billion Other care, $5. 4 billion Nursing home, $19. 4 billion Societal costs: $7. 18 per pack Annual lost productivity costs (1995– 1999) Men, $55. 4 billion Women, $26. 5 billion Billions of dollars Centers for Disease Control and Prevention. (2002). MMWR 51: 300– 303.
ANNUAL SMOKING-ATTRIBUTABLE ECONOMIC COSTS—U. S. , 1995– 1999 Prescription drugs, $6. 4 billion Medical expenditures (1998) Ambulatory care, $27. 2 billion Hospital care, $17. 1 billion Other care, $5. 4 billion Nursing home, $19. 4 billion Societal costs: $7. 18 per pack Annual lost productivity costs (1995– 1999) Men, $55. 4 billion Women, $26. 5 billion Billions of dollars Centers for Disease Control and Prevention. (2002). MMWR 51: 300– 303.
 2004 REPORT of the SURGEON GENERAL: HEALTH CONSEQUENCES OF SMOKING FOUR MAJOR CONCLUSIONS: n n Smoking harms nearly every organ of the body, causing many diseases and reducing the health of smokers in general. Quitting smoking has immediate as well as long-term benefits, reducing risks for diseases caused by smoking and improving health in general. Smoking cigarettes with lower machine-measured yields of tar and nicotine provides no clear benefit to health. The list of diseases caused by smoking has been expanded. U. S. Department of Health and Human Services. (2004). The Health Consequences of Smoking: A Report of the Surgeon General.
2004 REPORT of the SURGEON GENERAL: HEALTH CONSEQUENCES OF SMOKING FOUR MAJOR CONCLUSIONS: n n Smoking harms nearly every organ of the body, causing many diseases and reducing the health of smokers in general. Quitting smoking has immediate as well as long-term benefits, reducing risks for diseases caused by smoking and improving health in general. Smoking cigarettes with lower machine-measured yields of tar and nicotine provides no clear benefit to health. The list of diseases caused by smoking has been expanded. U. S. Department of Health and Human Services. (2004). The Health Consequences of Smoking: A Report of the Surgeon General.
 2006 REPORT of the SURGEON GENERAL: INVOLUNTARY EXPOSURE to TOBACCO SMOKE n n Second-hand smoke causes premature death and disease in nonsmokers (children and adults) Children: n n n There is no safe level of second-hand smoke. Increased risk for sudden infant death syndrome (SIDS), acute respiratory infections, ear problems, and more severe asthma Respiratory symptoms and slowed lung growth if parents smoke Adults: n Immediate adverse effects on cardiovascular system n Increased risk for coronary heart disease and lung cancer n Millions of Americans are exposed to smoke in their homes/workplaces n Indoor spaces: eliminating smoking fully protects nonsmokers n Separating smoking areas, cleaning the air, and ventilation are ineffective USDHHS. (2006). The Health Consequences of Involuntary Exposure to Tobacco Smoke: Report of the Surgeon General.
2006 REPORT of the SURGEON GENERAL: INVOLUNTARY EXPOSURE to TOBACCO SMOKE n n Second-hand smoke causes premature death and disease in nonsmokers (children and adults) Children: n n n There is no safe level of second-hand smoke. Increased risk for sudden infant death syndrome (SIDS), acute respiratory infections, ear problems, and more severe asthma Respiratory symptoms and slowed lung growth if parents smoke Adults: n Immediate adverse effects on cardiovascular system n Increased risk for coronary heart disease and lung cancer n Millions of Americans are exposed to smoke in their homes/workplaces n Indoor spaces: eliminating smoking fully protects nonsmokers n Separating smoking areas, cleaning the air, and ventilation are ineffective USDHHS. (2006). The Health Consequences of Involuntary Exposure to Tobacco Smoke: Report of the Surgeon General.
 QUITTING: HEALTH BENEFITS Time Since Quit Date Circulation improves, walking becomes easier Lung function increases up to 30% Excess risk of CHD decreases to half that of a continuing smoker Lung cancer death rate drops to half that of a continuing smoker Risk of cancer of mouth, throat, esophagus, bladder, kidney, pancreas decrease Lung cilia regain normal function 2 weeks to 3 months 1 to 9 months Ability to clear lungs of mucus increases Coughing, fatigue, shortness of breath decrease 1 year 5 years Risk of stroke is reduced to that of people who have never smoked after 15 years Risk of CHD is similar to that of people who have never smoked 10 years
QUITTING: HEALTH BENEFITS Time Since Quit Date Circulation improves, walking becomes easier Lung function increases up to 30% Excess risk of CHD decreases to half that of a continuing smoker Lung cancer death rate drops to half that of a continuing smoker Risk of cancer of mouth, throat, esophagus, bladder, kidney, pancreas decrease Lung cilia regain normal function 2 weeks to 3 months 1 to 9 months Ability to clear lungs of mucus increases Coughing, fatigue, shortness of breath decrease 1 year 5 years Risk of stroke is reduced to that of people who have never smoked after 15 years Risk of CHD is similar to that of people who have never smoked 10 years
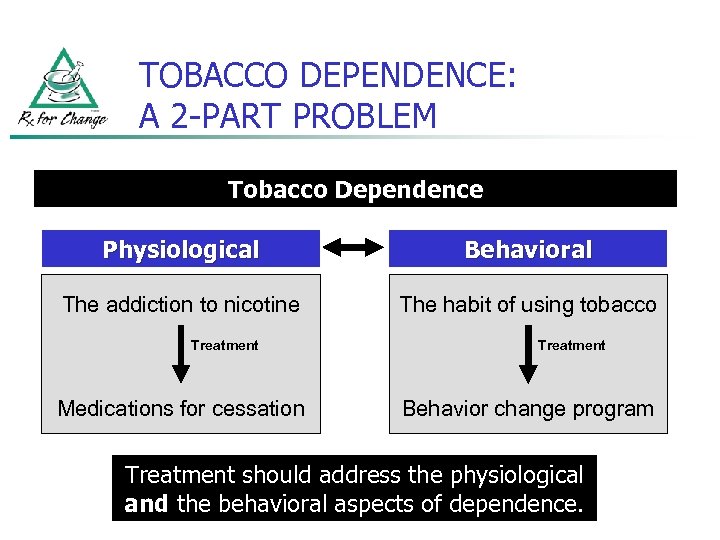 TOBACCO DEPENDENCE: A 2 -PART PROBLEM Tobacco Dependence Physiological Behavioral The addiction to nicotine The habit of using tobacco Treatment Medications for cessation Treatment Behavior change program Treatment should address the physiological and the behavioral aspects of dependence.
TOBACCO DEPENDENCE: A 2 -PART PROBLEM Tobacco Dependence Physiological Behavioral The addiction to nicotine The habit of using tobacco Treatment Medications for cessation Treatment Behavior change program Treatment should address the physiological and the behavioral aspects of dependence.
 CLINICAL PRACTICE GUIDELINE for TREATING TOBACCO USE and DEPENDENCE n n Released June 2000 Sponsored by the Agency for Healthcare Research and Quality of the U. S. Public Heath Service with n n n Centers for Disease Control and Prevention National Cancer Institute National Institute for Drug Addiction National Heart, Lung, & Blood Institute Robert Wood Johnson Foundation www. surgeongeneral. gov/tobacco/
CLINICAL PRACTICE GUIDELINE for TREATING TOBACCO USE and DEPENDENCE n n Released June 2000 Sponsored by the Agency for Healthcare Research and Quality of the U. S. Public Heath Service with n n n Centers for Disease Control and Prevention National Cancer Institute National Institute for Drug Addiction National Heart, Lung, & Blood Institute Robert Wood Johnson Foundation www. surgeongeneral. gov/tobacco/
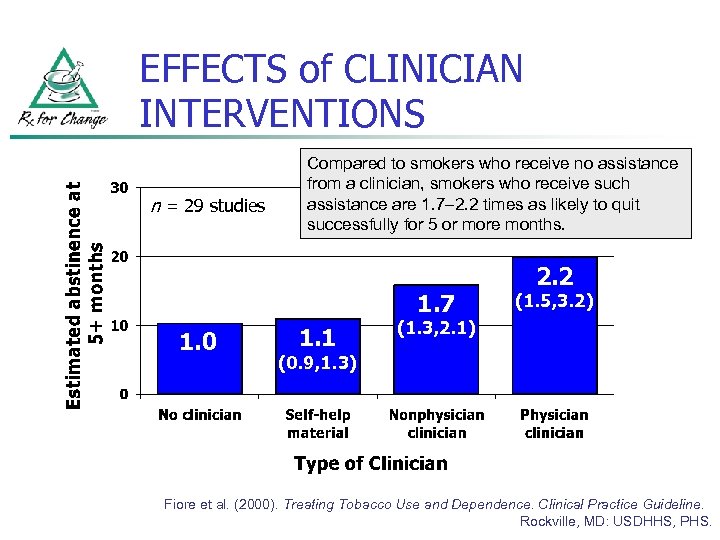 EFFECTS of CLINICIAN INTERVENTIONS n = 29 studies Compared to smokers who receive no assistance from a clinician, smokers who receive such assistance are 1. 7– 2. 2 times as likely to quit successfully for 5 or more months. 1. 7 1. 0 1. 1 2. 2 (1. 5, 3. 2) (1. 3, 2. 1) (0. 9, 1. 3) Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
EFFECTS of CLINICIAN INTERVENTIONS n = 29 studies Compared to smokers who receive no assistance from a clinician, smokers who receive such assistance are 1. 7– 2. 2 times as likely to quit successfully for 5 or more months. 1. 7 1. 0 1. 1 2. 2 (1. 5, 3. 2) (1. 3, 2. 1) (0. 9, 1. 3) Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
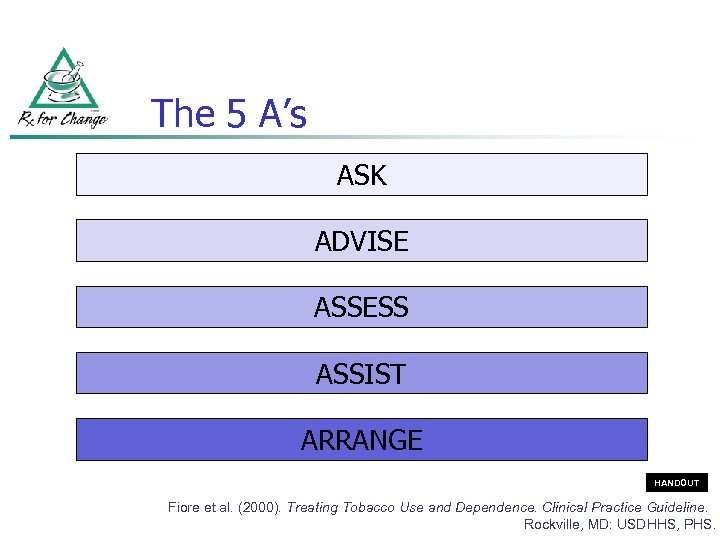 The 5 A’s ASK ADVISE ASSESS ASSIST ARRANGE HANDOUT Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
The 5 A’s ASK ADVISE ASSESS ASSIST ARRANGE HANDOUT Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
 The 5 A’s n (cont’d) ASK Ask about tobacco use n “Do you ever smoke or use any type of tobacco? ” n n n “I take time to ask all of my patients about tobacco use—because it’s important. ” “Medication X often is used for conditions linked with or caused by smoking. Do you, or does someone in your household smoke? ” “Condition X often is caused or worsened by smoking. Do you, or does someone in your household smoke? ”
The 5 A’s n (cont’d) ASK Ask about tobacco use n “Do you ever smoke or use any type of tobacco? ” n n n “I take time to ask all of my patients about tobacco use—because it’s important. ” “Medication X often is used for conditions linked with or caused by smoking. Do you, or does someone in your household smoke? ” “Condition X often is caused or worsened by smoking. Do you, or does someone in your household smoke? ”
 The 5 A’s n (cont’d) ADVISE tobacco users to quit (clear, strong, personalized, sensitive) n n “It’s important that you quit as soon as possible, and I can help you. ” “I realize that quitting is difficult. It is the most important thing you can do to protect your health now and in the future. I have training to help my patients quit, and when you are ready, I will work with you to design a specialized treatment plan. ”
The 5 A’s n (cont’d) ADVISE tobacco users to quit (clear, strong, personalized, sensitive) n n “It’s important that you quit as soon as possible, and I can help you. ” “I realize that quitting is difficult. It is the most important thing you can do to protect your health now and in the future. I have training to help my patients quit, and when you are ready, I will work with you to design a specialized treatment plan. ”
 The 5 A’s (cont’d) n ASSESS Assess readiness to make a quit attempt n Assist ASSIST with the quit attempt n Not ready to quit: provide motivation (the 5 R’s) n Ready to quit: design a treatment plan n Recently quit: relapse prevention
The 5 A’s (cont’d) n ASSESS Assess readiness to make a quit attempt n Assist ASSIST with the quit attempt n Not ready to quit: provide motivation (the 5 R’s) n Ready to quit: design a treatment plan n Recently quit: relapse prevention
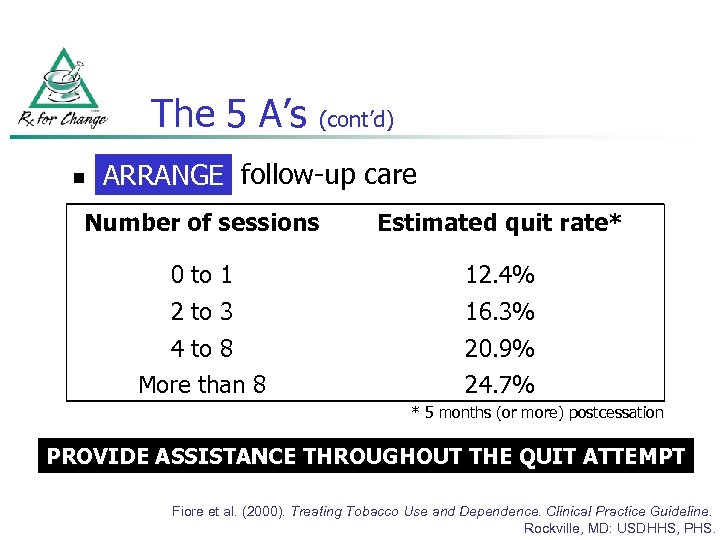 The 5 A’s n (cont’d) Arrange ARRANGE follow-up care Number of sessions Estimated quit rate* 0 to 1 12. 4% 2 to 3 16. 3% 4 to 8 More than 8 20. 9% 24. 7% * 5 months (or more) postcessation PROVIDE ASSISTANCE THROUGHOUT THE QUIT ATTEMPT Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
The 5 A’s n (cont’d) Arrange ARRANGE follow-up care Number of sessions Estimated quit rate* 0 to 1 12. 4% 2 to 3 16. 3% 4 to 8 More than 8 20. 9% 24. 7% * 5 months (or more) postcessation PROVIDE ASSISTANCE THROUGHOUT THE QUIT ATTEMPT Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
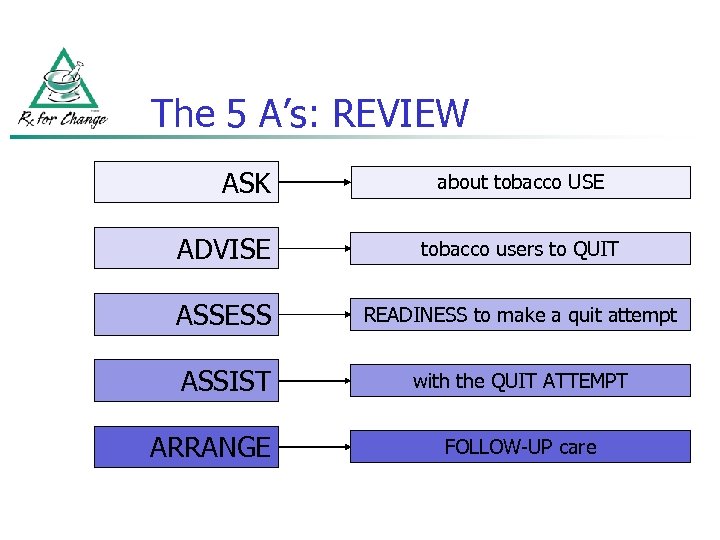 The 5 A’s: REVIEW ASK about tobacco USE ADVISE tobacco users to QUIT ASSESS READINESS to make a quit attempt ASSIST with the QUIT ATTEMPT ARRANGE FOLLOW-UP care
The 5 A’s: REVIEW ASK about tobacco USE ADVISE tobacco users to QUIT ASSESS READINESS to make a quit attempt ASSIST with the QUIT ATTEMPT ARRANGE FOLLOW-UP care
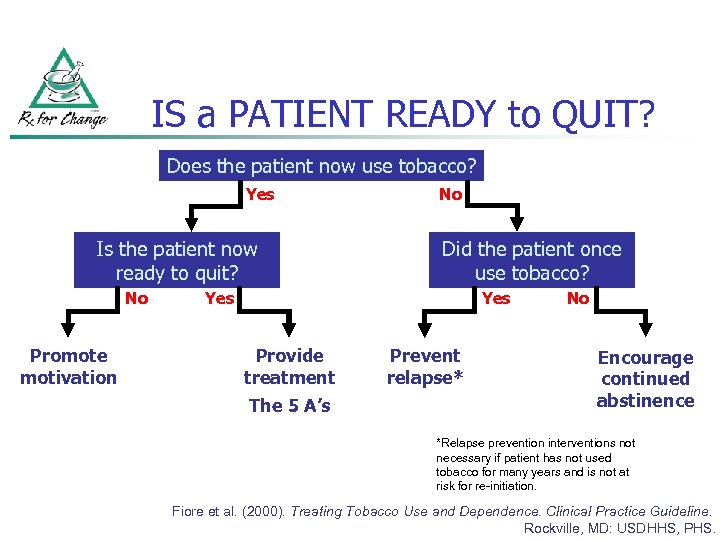 IS a PATIENT READY to QUIT? Does the patient now use tobacco? Yes Is the patient now ready to quit? No Promote motivation No Did the patient once use tobacco? Yes Provide treatment The 5 A’s Prevent relapse* No Encourage continued abstinence *Relapse prevention interventions not necessary if patient has not used tobacco for many years and is not at risk for re-initiation. Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
IS a PATIENT READY to QUIT? Does the patient now use tobacco? Yes Is the patient now ready to quit? No Promote motivation No Did the patient once use tobacco? Yes Provide treatment The 5 A’s Prevent relapse* No Encourage continued abstinence *Relapse prevention interventions not necessary if patient has not used tobacco for many years and is not at risk for re-initiation. Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
 PHARMACOTHERAPY “All patients attempting to quit should be encouraged to use effective pharmacotherapies for smoking cessation except in the presence of special circumstances. ” Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
PHARMACOTHERAPY “All patients attempting to quit should be encouraged to use effective pharmacotherapies for smoking cessation except in the presence of special circumstances. ” Fiore et al. (2000). Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: USDHHS, PHS.
 PHARMACOLOGIC METHODS: FIRST-LINE THERAPIES Three general classes of FDA-approved drugs for smoking cessation: § Nicotine replacement therapy (NRT) § § Psychotropics § § Nicotine gum, patch, lozenge, nasal spray, inhaler Sustained-release bupropion Partial nicotinic receptor agonist § Varenicline Currently, no medications have an FDA indication for use in spit tobacco cessation.
PHARMACOLOGIC METHODS: FIRST-LINE THERAPIES Three general classes of FDA-approved drugs for smoking cessation: § Nicotine replacement therapy (NRT) § § Psychotropics § § Nicotine gum, patch, lozenge, nasal spray, inhaler Sustained-release bupropion Partial nicotinic receptor agonist § Varenicline Currently, no medications have an FDA indication for use in spit tobacco cessation.
 NRT: RATIONALE for USE n n Reduces physical withdrawal from nicotine Allows patient to focus on behavioral and psychological aspects of tobacco cessation NRT APPROXIMATELY DOUBLES QUIT RATES.
NRT: RATIONALE for USE n n Reduces physical withdrawal from nicotine Allows patient to focus on behavioral and psychological aspects of tobacco cessation NRT APPROXIMATELY DOUBLES QUIT RATES.
 NICOTINE PHARMACODYNAMICS: WITHDRAWAL EFFECTS n Depression n Insomnia n Irritability/frustration/anger n Anxiety n Difficulty concentrating n Restlessness n Increased appetite/weight gain n Decreased heart rate n Cravings* * Not considered a withdrawal symptom by DSM-IV criteria. Most symptoms peak 24– 48 hr after quitting and subside within 2 – 4 weeks. HANDOUT American Psychiatric Association. (1994). DSM-IV. Hughes et al. (1991). Arch Gen Psychiatry 48: 52– 59. Hughes & Hatsukami. (1998). Tob Control 7: 92– 93.
NICOTINE PHARMACODYNAMICS: WITHDRAWAL EFFECTS n Depression n Insomnia n Irritability/frustration/anger n Anxiety n Difficulty concentrating n Restlessness n Increased appetite/weight gain n Decreased heart rate n Cravings* * Not considered a withdrawal symptom by DSM-IV criteria. Most symptoms peak 24– 48 hr after quitting and subside within 2 – 4 weeks. HANDOUT American Psychiatric Association. (1994). DSM-IV. Hughes et al. (1991). Arch Gen Psychiatry 48: 52– 59. Hughes & Hatsukami. (1998). Tob Control 7: 92– 93.
 NRT: PRODUCTS Polacrilex gum n n Nicorette (OTC) Generic nicotine gum (OTC) Lozenge n n Nasal spray n Inhaler Commit (OTC) Generic nicotine lozenge (OTC) Transdermal patch n n Nicotrol NS (Rx) Nicoderm CQ (OTC) Generic nicotine patches (OTC, Rx) n Nicotrol (Rx)
NRT: PRODUCTS Polacrilex gum n n Nicorette (OTC) Generic nicotine gum (OTC) Lozenge n n Nasal spray n Inhaler Commit (OTC) Generic nicotine lozenge (OTC) Transdermal patch n n Nicotrol NS (Rx) Nicoderm CQ (OTC) Generic nicotine patches (OTC, Rx) n Nicotrol (Rx)
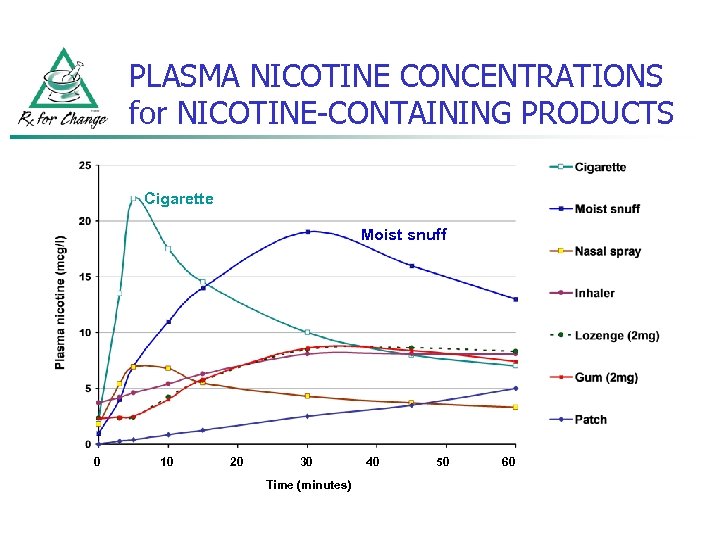 PLASMA NICOTINE CONCENTRATIONS for NICOTINE-CONTAINING PRODUCTS Cigarette Moist snuff 0 10 20 30 Time (minutes) 40 50 60
PLASMA NICOTINE CONCENTRATIONS for NICOTINE-CONTAINING PRODUCTS Cigarette Moist snuff 0 10 20 30 Time (minutes) 40 50 60
 NRT: PRECAUTIONS n Patients with underlying cardiovascular disease n Recent myocardial infarction (within past 2 weeks) n Serious arrhythmias n Serious or worsening angina NRT products may be appropriate for these patients if they are under medical supervision.
NRT: PRECAUTIONS n Patients with underlying cardiovascular disease n Recent myocardial infarction (within past 2 weeks) n Serious arrhythmias n Serious or worsening angina NRT products may be appropriate for these patients if they are under medical supervision.
 NRT: PRECAUTIONS n (cont’d) Patients with other underlying conditions n Active temporomandibular joint disease (gum only) n Pregnancy n Lactation NRT products may be appropriate for these patients if they are under medical supervision.
NRT: PRECAUTIONS n (cont’d) Patients with other underlying conditions n Active temporomandibular joint disease (gum only) n Pregnancy n Lactation NRT products may be appropriate for these patients if they are under medical supervision.
 NRT: OTHER CONSIDERATIONS n n NRT is not FDA-approved for use in children or adolescents Nonprescription sales (patch, gum, lozenge) are restricted to adults ≥ 18 years of age n n NRT use in minors requires a prescription Patients should stop using all forms of tobacco upon initiation of the NRT regimen
NRT: OTHER CONSIDERATIONS n n NRT is not FDA-approved for use in children or adolescents Nonprescription sales (patch, gum, lozenge) are restricted to adults ≥ 18 years of age n n NRT use in minors requires a prescription Patients should stop using all forms of tobacco upon initiation of the NRT regimen
 NICOTINE GUM Nicorette (Glaxo. Smith. Kline); generics n Resin complex n n n Nicotine Polacrilin Sugar-free chewing gum base Contains buffering agents to enhance buccal absorption of nicotine Available: 2 mg, 4 mg; regular, Fresh. Mint, Fruit Chill, mint, & orange flavor
NICOTINE GUM Nicorette (Glaxo. Smith. Kline); generics n Resin complex n n n Nicotine Polacrilin Sugar-free chewing gum base Contains buffering agents to enhance buccal absorption of nicotine Available: 2 mg, 4 mg; regular, Fresh. Mint, Fruit Chill, mint, & orange flavor
 NICOTINE GUM: SUMMARY ADVANTAGES n n n Gum use may satisfy oral cravings. Gum use may delay weight gain. Patients can titrate therapy to manage withdrawal symptoms. DISADVANTAGES n n n Gum chewing may not be socially acceptable. Gum is difficult to use with dentures. Patients must use proper chewing technique to minimize adverse effects.
NICOTINE GUM: SUMMARY ADVANTAGES n n n Gum use may satisfy oral cravings. Gum use may delay weight gain. Patients can titrate therapy to manage withdrawal symptoms. DISADVANTAGES n n n Gum chewing may not be socially acceptable. Gum is difficult to use with dentures. Patients must use proper chewing technique to minimize adverse effects.
 NICOTINE LOZENGE Commit (Glaxo. Smith. Kline); generics n Nicotine polacrilex formulation n n Delivers ~25% more nicotine than equivalent gum dose Sugar-free, mint or cherry flavor (boxed or POP-PAC) Contains buffering agents to enhance buccal absorption of nicotine Available: 2 mg, 4 mg
NICOTINE LOZENGE Commit (Glaxo. Smith. Kline); generics n Nicotine polacrilex formulation n n Delivers ~25% more nicotine than equivalent gum dose Sugar-free, mint or cherry flavor (boxed or POP-PAC) Contains buffering agents to enhance buccal absorption of nicotine Available: 2 mg, 4 mg
 NICOTINE LOZENGE: SUMMARY ADVANTAGES n n n Lozenge use may satisfy oral cravings. The lozenge is easy to use and conceal. Patients can titrate therapy to manage withdrawal symptoms. DISADVANTAGES n Gastrointestinal side effects (nausea, hiccups, and heartburn) may be bothersome.
NICOTINE LOZENGE: SUMMARY ADVANTAGES n n n Lozenge use may satisfy oral cravings. The lozenge is easy to use and conceal. Patients can titrate therapy to manage withdrawal symptoms. DISADVANTAGES n Gastrointestinal side effects (nausea, hiccups, and heartburn) may be bothersome.
 TRANSDERMAL NICOTINE PATCH Nicoderm CQ (Glaxo. Smith. Kline); generic n n n Nicotine is well absorbed across the skin Delivery to systemic circulation avoids hepatic firstpass metabolism Plasma nicotine levels are lower and fluctuate less than with smoking
TRANSDERMAL NICOTINE PATCH Nicoderm CQ (Glaxo. Smith. Kline); generic n n n Nicotine is well absorbed across the skin Delivery to systemic circulation avoids hepatic firstpass metabolism Plasma nicotine levels are lower and fluctuate less than with smoking
 TRANSDERMAL NICOTINE PATCH: SUMMARY ADVANTAGES n n n The patch provides consistent nicotine levels. The patch is easy to use and conceal. Fewer compliance issues are associated with patch use. DISADVANTAGES n Patients cannot titrate the dose. n Allergic reactions to the adhesive may occur. n Patients with dermatologic conditions should not use the patch.
TRANSDERMAL NICOTINE PATCH: SUMMARY ADVANTAGES n n n The patch provides consistent nicotine levels. The patch is easy to use and conceal. Fewer compliance issues are associated with patch use. DISADVANTAGES n Patients cannot titrate the dose. n Allergic reactions to the adhesive may occur. n Patients with dermatologic conditions should not use the patch.
 NICOTINE NASAL SPRAY Nicotrol NS (Pfizer) Aqueous solution of nicotine in a 10 -ml spray bottle n Each metered dose actuation delivers n 50 µl spray n 0. 5 mg nicotine n ~100 doses/bottle n Rapid absorption across nasal mucosa n
NICOTINE NASAL SPRAY Nicotrol NS (Pfizer) Aqueous solution of nicotine in a 10 -ml spray bottle n Each metered dose actuation delivers n 50 µl spray n 0. 5 mg nicotine n ~100 doses/bottle n Rapid absorption across nasal mucosa n
 NICOTINE NASAL SPRAY: SUMMARY ADVANTAGES n Patients can easily titrate therapy to rapidly manage withdrawal symptoms. DISADVANTAGES n n n Nasal/throat irritation may be bothersome. Nasal spray has higher dependence potential. Patients with chronic nasal disorders or severe reactive airway disease should not use the spray.
NICOTINE NASAL SPRAY: SUMMARY ADVANTAGES n Patients can easily titrate therapy to rapidly manage withdrawal symptoms. DISADVANTAGES n n n Nasal/throat irritation may be bothersome. Nasal spray has higher dependence potential. Patients with chronic nasal disorders or severe reactive airway disease should not use the spray.
 NICOTINE INHALER Nicotrol Inhaler (Pfizer) n Nicotine inhalation system consists of n n Mouthpiece Cartridge with porous plug containing 10 mg nicotine Delivers 4 mg nicotine vapor, absorbed across buccal mucosa May satisfy hand-to-mouth ritual of smoking
NICOTINE INHALER Nicotrol Inhaler (Pfizer) n Nicotine inhalation system consists of n n Mouthpiece Cartridge with porous plug containing 10 mg nicotine Delivers 4 mg nicotine vapor, absorbed across buccal mucosa May satisfy hand-to-mouth ritual of smoking
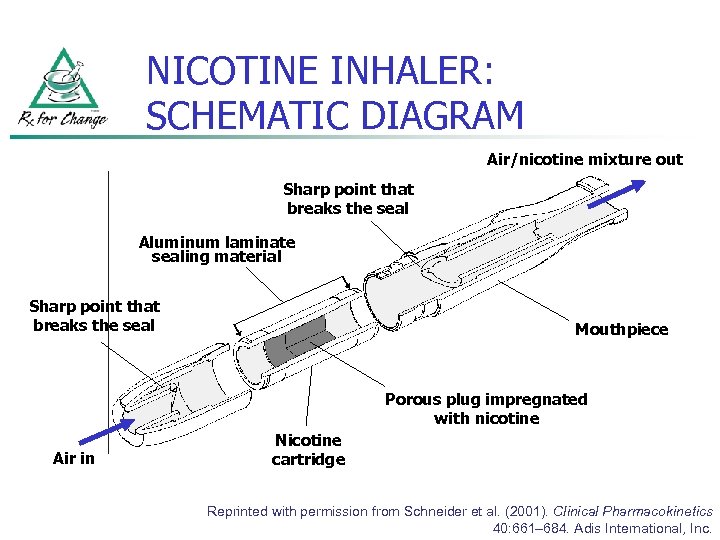 NICOTINE INHALER: SCHEMATIC DIAGRAM Air/nicotine mixture out Sharp point that breaks the seal Aluminum laminate sealing material Sharp point that breaks the seal Mouthpiece Porous plug impregnated with nicotine Air in Nicotine cartridge Reprinted with permission from Schneider et al. (2001). Clinical Pharmacokinetics 40: 661– 684. Adis International, Inc.
NICOTINE INHALER: SCHEMATIC DIAGRAM Air/nicotine mixture out Sharp point that breaks the seal Aluminum laminate sealing material Sharp point that breaks the seal Mouthpiece Porous plug impregnated with nicotine Air in Nicotine cartridge Reprinted with permission from Schneider et al. (2001). Clinical Pharmacokinetics 40: 661– 684. Adis International, Inc.
 NICOTINE INHALER: SUMMARY ADVANTAGES n n Patients can easily titrate therapy to manage withdrawal symptoms. The inhaler mimics the hand-to-mouth ritual of smoking. DISADVANTAGES n n n The initial throat or mouth irritation can be bothersome. Cartridges should not be stored in very warm conditions or used in very cold conditions. Patients with underlying bronchospastic disease must use the inhaler with caution.
NICOTINE INHALER: SUMMARY ADVANTAGES n n Patients can easily titrate therapy to manage withdrawal symptoms. The inhaler mimics the hand-to-mouth ritual of smoking. DISADVANTAGES n n n The initial throat or mouth irritation can be bothersome. Cartridges should not be stored in very warm conditions or used in very cold conditions. Patients with underlying bronchospastic disease must use the inhaler with caution.
 BUPROPION SR Zyban (Glaxo. Smith. Kline); generic n n n Nonnicotine cessation aid Sustained-release antidepressant Oral formulation
BUPROPION SR Zyban (Glaxo. Smith. Kline); generic n n n Nonnicotine cessation aid Sustained-release antidepressant Oral formulation
 BUPROPION: MECHANISM of ACTION n Atypical antidepressant thought to affect levels of various brain neurotransmitters n n n Dopamine Norepinephrine Clinical effects n craving for cigarettes n symptoms of nicotine withdrawal
BUPROPION: MECHANISM of ACTION n Atypical antidepressant thought to affect levels of various brain neurotransmitters n n n Dopamine Norepinephrine Clinical effects n craving for cigarettes n symptoms of nicotine withdrawal
 BUPROPION: CONTRAINDICATIONS n Patients with a seizure disorder n Patients taking n n Wellbutrin, Wellbutrin SR, Wellbutrin XL MAO inhibitors in preceding 14 days Patients with a current or prior diagnosis of anorexia or bulimia nervosa Patients undergoing abrupt discontinuation of alcohol or sedatives (including benzodiazepines)
BUPROPION: CONTRAINDICATIONS n Patients with a seizure disorder n Patients taking n n Wellbutrin, Wellbutrin SR, Wellbutrin XL MAO inhibitors in preceding 14 days Patients with a current or prior diagnosis of anorexia or bulimia nervosa Patients undergoing abrupt discontinuation of alcohol or sedatives (including benzodiazepines)
 BUPROPION: WARNINGS and PRECAUTIONS Bupropion should be used with extreme caution in the following populations: n Patients with a history of seizure n Patients with a history of cranial trauma n n Patients taking medications that lower the seizure threshold (antipsychotics, antidepressants, theophylline, systemic steroids) Patients with severe hepatic cirrhosis
BUPROPION: WARNINGS and PRECAUTIONS Bupropion should be used with extreme caution in the following populations: n Patients with a history of seizure n Patients with a history of cranial trauma n n Patients taking medications that lower the seizure threshold (antipsychotics, antidepressants, theophylline, systemic steroids) Patients with severe hepatic cirrhosis
 BUPROPION: USE in PREGNANCY n n n Category C drug Use only if clearly indicated Attempt nondrug treatment first
BUPROPION: USE in PREGNANCY n n n Category C drug Use only if clearly indicated Attempt nondrug treatment first
 BUPROPION SR: DOSING Patients should begin therapy 1 to 2 weeks PRIOR to their quit date to ensure that therapeutic plasma levels of the drug are achieved. Initial treatment n 150 mg po q AM x 3 days Then… n n 150 mg po bid Duration, 7– 12 weeks
BUPROPION SR: DOSING Patients should begin therapy 1 to 2 weeks PRIOR to their quit date to ensure that therapeutic plasma levels of the drug are achieved. Initial treatment n 150 mg po q AM x 3 days Then… n n 150 mg po bid Duration, 7– 12 weeks
 BUPROPION: ADVERSE EFFECTS Common side effects include the following: n Insomnia (avoid bedtime dosing) n Dry mouth Less common but reported effects: n Tremor n Skin rash
BUPROPION: ADVERSE EFFECTS Common side effects include the following: n Insomnia (avoid bedtime dosing) n Dry mouth Less common but reported effects: n Tremor n Skin rash
 BUPROPION: ADDITIONAL PATIENT EDUCATION n n Dose tapering not necessary when discontinuing treatment If no significant progress toward abstinence by seventh week, therapy is unlikely to be effective n n Discontinue treatment Reevaluate and restart at later date
BUPROPION: ADDITIONAL PATIENT EDUCATION n n Dose tapering not necessary when discontinuing treatment If no significant progress toward abstinence by seventh week, therapy is unlikely to be effective n n Discontinue treatment Reevaluate and restart at later date
 BUPROPION SR: SUMMARY ADVANTAGES n n Bupropion is an oral formulation with twicea-day dosing. Bupropion might be beneficial for patients with depression. DISADVANTAGES n n The seizure risk is increased. Several contraindications and precautions preclude use.
BUPROPION SR: SUMMARY ADVANTAGES n n Bupropion is an oral formulation with twicea-day dosing. Bupropion might be beneficial for patients with depression. DISADVANTAGES n n The seizure risk is increased. Several contraindications and precautions preclude use.
 VARENICLINE Chantix (Pfizer) n n n Nonnicotine cessation aid Partial nicotinic receptor agonist Oral formulation
VARENICLINE Chantix (Pfizer) n n n Nonnicotine cessation aid Partial nicotinic receptor agonist Oral formulation
 VARENICLINE: MECHANISM of ACTION n Binds with high affinity and selectivity at 4 2 neuronal nicotinic acetylcholine receptors n n n Stimulates low-level agonist activity Competitively inhibits binding of nicotine Clinical effects n n symptoms of nicotine withdrawal Blocks dopaminergic stimulation responsible for reinforcement & reward associated with smoking
VARENICLINE: MECHANISM of ACTION n Binds with high affinity and selectivity at 4 2 neuronal nicotinic acetylcholine receptors n n n Stimulates low-level agonist activity Competitively inhibits binding of nicotine Clinical effects n n symptoms of nicotine withdrawal Blocks dopaminergic stimulation responsible for reinforcement & reward associated with smoking
 VARENICLINE : USE in PREGNANCY and LACTATION n n Category C drug Use only if potential benefit justifies potential risk Attempt nondrug treatment first Unknown if drug excreted in human breast milk
VARENICLINE : USE in PREGNANCY and LACTATION n n Category C drug Use only if potential benefit justifies potential risk Attempt nondrug treatment first Unknown if drug excreted in human breast milk
 VARENICLINE: DOSING Patients should begin therapy 1 week PRIOR to their quit date. The dose is gradually increased to minimize treatment-related nausea and insomnia. Treatment Day Initial dose titration Dose Day 1 to day 3 0. 5 mg qd Day 4 to day 7 0. 5 mg bid Day 8 to end of treatment* 1 mg bid * Up to 12 weeks
VARENICLINE: DOSING Patients should begin therapy 1 week PRIOR to their quit date. The dose is gradually increased to minimize treatment-related nausea and insomnia. Treatment Day Initial dose titration Dose Day 1 to day 3 0. 5 mg qd Day 4 to day 7 0. 5 mg bid Day 8 to end of treatment* 1 mg bid * Up to 12 weeks
 VARENICLINE: ADVERSE EFFECTS Common side effects (≥ 5% and twice the rate observed in placebo-treated patients) include: n Nausea n Sleep disturbances (insomnia, abnormal dreams) n Constipation n Flatulence n Vomiting
VARENICLINE: ADVERSE EFFECTS Common side effects (≥ 5% and twice the rate observed in placebo-treated patients) include: n Nausea n Sleep disturbances (insomnia, abnormal dreams) n Constipation n Flatulence n Vomiting
 VARENICLINE: ADDITIONAL PATIENT EDUCATION n n Doses should be taken after eating, with a full glass of water Nausea and insomnia are side effects that are usually temporary. n n If symptoms persist, notify your health care provider Dose tapering not necessary when discontinuing treatment
VARENICLINE: ADDITIONAL PATIENT EDUCATION n n Doses should be taken after eating, with a full glass of water Nausea and insomnia are side effects that are usually temporary. n n If symptoms persist, notify your health care provider Dose tapering not necessary when discontinuing treatment
 VARENICLINE: SUMMARY ADVANTAGES n n Varenicline is an oral formulation with twice-aday dosing. Varenicline offers a new mechanism of action for persons who previously failed using other medications. DISADVANTAGES n n May induce nausea in up to one third of patients. Post-marketing surveillance data not yet available.
VARENICLINE: SUMMARY ADVANTAGES n n Varenicline is an oral formulation with twice-aday dosing. Varenicline offers a new mechanism of action for persons who previously failed using other medications. DISADVANTAGES n n May induce nausea in up to one third of patients. Post-marketing surveillance data not yet available.
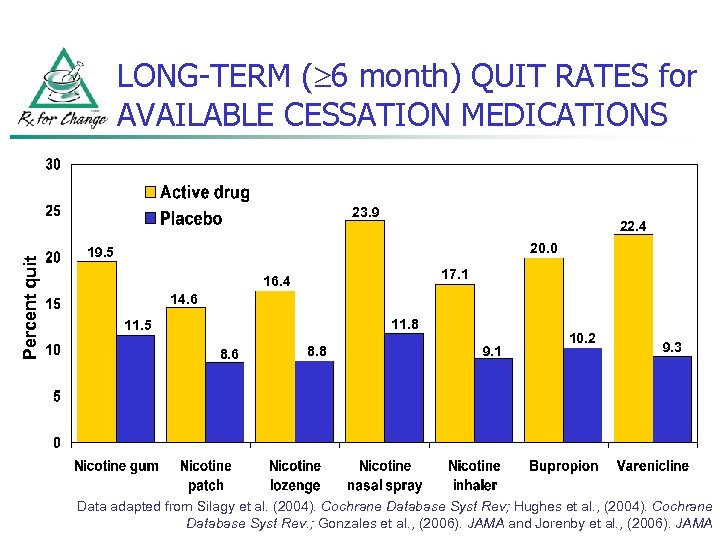 LONG-TERM ( 6 month) QUIT RATES for AVAILABLE CESSATION MEDICATIONS Percent quit 23. 9 22. 4 20. 0 19. 5 17. 1 16. 4 14. 6 11. 8 11. 5 8. 6 8. 8 9. 1 10. 2 9. 3 Data adapted from Silagy et al. (2004). Cochrane Database Syst Rev; Hughes et al. , (2004). Cochrane Database Syst Rev. ; Gonzales et al. , (2006). JAMA and Jorenby et al. , (2006). JAMA
LONG-TERM ( 6 month) QUIT RATES for AVAILABLE CESSATION MEDICATIONS Percent quit 23. 9 22. 4 20. 0 19. 5 17. 1 16. 4 14. 6 11. 8 11. 5 8. 6 8. 8 9. 1 10. 2 9. 3 Data adapted from Silagy et al. (2004). Cochrane Database Syst Rev; Hughes et al. , (2004). Cochrane Database Syst Rev. ; Gonzales et al. , (2006). JAMA and Jorenby et al. , (2006). JAMA
 COMBINATION PHARMACOTHERAPY Reserve for patients unable to quit using monotherapy. n Combination NRT Long-acting formulation (patch) n Produces relatively constant levels of nicotine PLUS Short-acting formulation (gum, lozenge, inhaler, nasal spray) n n n Allows for acute dose titration as needed for withdrawal symptoms Bupropion SR + NRT The safety and efficacy of combination of varenicline with NRT or bupropion has not been established.
COMBINATION PHARMACOTHERAPY Reserve for patients unable to quit using monotherapy. n Combination NRT Long-acting formulation (patch) n Produces relatively constant levels of nicotine PLUS Short-acting formulation (gum, lozenge, inhaler, nasal spray) n n n Allows for acute dose titration as needed for withdrawal symptoms Bupropion SR + NRT The safety and efficacy of combination of varenicline with NRT or bupropion has not been established.
 COMPLIANCE IS KEY to QUITTING n Promote compliance with prescribed regimens. n Use according to dosing schedule, NOT as needed. n Consider telling the patient: n “When you use a cessation product it is important to read all the directions thoroughly before using the product. The products work best in alleviating withdrawal symptoms when used correctly, and according to the recommended dosing schedule. ”
COMPLIANCE IS KEY to QUITTING n Promote compliance with prescribed regimens. n Use according to dosing schedule, NOT as needed. n Consider telling the patient: n “When you use a cessation product it is important to read all the directions thoroughly before using the product. The products work best in alleviating withdrawal symptoms when used correctly, and according to the recommended dosing schedule. ”
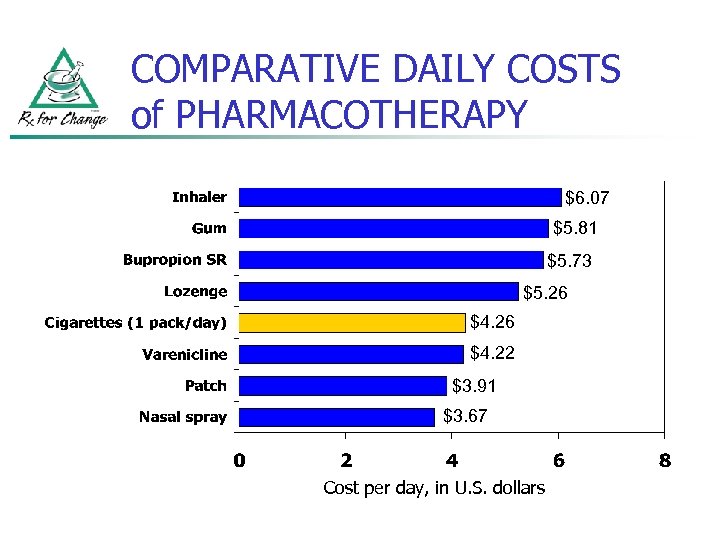 COMPARATIVE DAILY COSTS of PHARMACOTHERAPY $6. 07 $5. 81 $5. 73 $5. 26 $4. 22 $3. 91 $3. 67 Cost per day, in U. S. dollars
COMPARATIVE DAILY COSTS of PHARMACOTHERAPY $6. 07 $5. 81 $5. 73 $5. 26 $4. 22 $3. 91 $3. 67 Cost per day, in U. S. dollars
 The RESPONSIBILITY of HEALTH PROFESSIONALS It is inconsistent to provide health care and —at the same time— remain silent (or inactive) about a major health risk. TOBACCO CESSATION is an important component of THERAPY.
The RESPONSIBILITY of HEALTH PROFESSIONALS It is inconsistent to provide health care and —at the same time— remain silent (or inactive) about a major health risk. TOBACCO CESSATION is an important component of THERAPY.
 BRIEF COUNSELING: ASK, ADVISE, REFER n Brief interventions have been shown to be effective n In the absence of time or expertise: n Ask, advise, and refer to other resources, such as local programs or the toll-free quitline 1 -800 -QUIT-NOW This brief intervention can be achieved in 30 seconds.
BRIEF COUNSELING: ASK, ADVISE, REFER n Brief interventions have been shown to be effective n In the absence of time or expertise: n Ask, advise, and refer to other resources, such as local programs or the toll-free quitline 1 -800 -QUIT-NOW This brief intervention can be achieved in 30 seconds.


