b2fe1064af4414a2e2946a55e85f1d91.ppt
- Количество слайдов: 34
 Pharmacodynamic Paradigms in Early-Phase Cancer Clinical Trials Workshop Phase 0 Trials In Oncologic Drug Development Hilary Calvert Northern Institute for Cancer Research
Pharmacodynamic Paradigms in Early-Phase Cancer Clinical Trials Workshop Phase 0 Trials In Oncologic Drug Development Hilary Calvert Northern Institute for Cancer Research
 Methodology for Phase I and Phase 0 (translational) Trials Develop trial methodology designed for targeted agents in trials with pharmacodynamic endpoints The use of pharmacodynamic or toxic endpoints present similar problems – magnitude, reproducibility, variability Endpoints – To develop methods that utilise continuously variable (scalar) endpoints rather than yes/no (Boolean) endpoints – To extend these techniques to combination Phase I trials
Methodology for Phase I and Phase 0 (translational) Trials Develop trial methodology designed for targeted agents in trials with pharmacodynamic endpoints The use of pharmacodynamic or toxic endpoints present similar problems – magnitude, reproducibility, variability Endpoints – To develop methods that utilise continuously variable (scalar) endpoints rather than yes/no (Boolean) endpoints – To extend these techniques to combination Phase I trials
 Classical Methodology for Phase I and Translational Trials 1. Traditional – – Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages • • 3. 2. – – – Continual Reassessment (O’Quigley) Stochastic model to predict probability of DLT vs dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge – – – Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals – Pharmacokinetically-guided (Collins) – 4. – – Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients • Inter-patient variability in PKs • • • Assumes linearity Metabolites May not be feasible Disadvantages Accelerated Phase I Design (Simon) Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels
Classical Methodology for Phase I and Translational Trials 1. Traditional – – Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages • • 3. 2. – – – Continual Reassessment (O’Quigley) Stochastic model to predict probability of DLT vs dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge – – – Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals – Pharmacokinetically-guided (Collins) – 4. – – Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients • Inter-patient variability in PKs • • • Assumes linearity Metabolites May not be feasible Disadvantages Accelerated Phase I Design (Simon) Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels
 Classical Methodology for Phase I and Translational Trials 1. – – Traditional Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages • • Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals 2. Pharmacokinetically-guided (Collins) – – 3. – – – Continual Reassessment (O’Quigley) Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients Disadvantages • • Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible Stochastic model to predict probability of DLT vs. dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge 4. – – Accelerated Phase I Design (Simon) Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels
Classical Methodology for Phase I and Translational Trials 1. – – Traditional Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages • • Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals 2. Pharmacokinetically-guided (Collins) – – 3. – – – Continual Reassessment (O’Quigley) Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients Disadvantages • • Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible Stochastic model to predict probability of DLT vs. dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge 4. – – Accelerated Phase I Design (Simon) Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels
 CI-941 – DMP-941 Losoxantrone • Similar to mitoxantrone • Animal models – Activity equal to or better than doxorubicin – No or little cardiotoxicity • One of 3 analogues submitted for clinical development by Warner Lambert • Candidate for AUC-based dose escalation – Preclinical pharmacology established “target” AUC and linearity up to mg/m 2 45
CI-941 – DMP-941 Losoxantrone • Similar to mitoxantrone • Animal models – Activity equal to or better than doxorubicin – No or little cardiotoxicity • One of 3 analogues submitted for clinical development by Warner Lambert • Candidate for AUC-based dose escalation – Preclinical pharmacology established “target” AUC and linearity up to mg/m 2 45
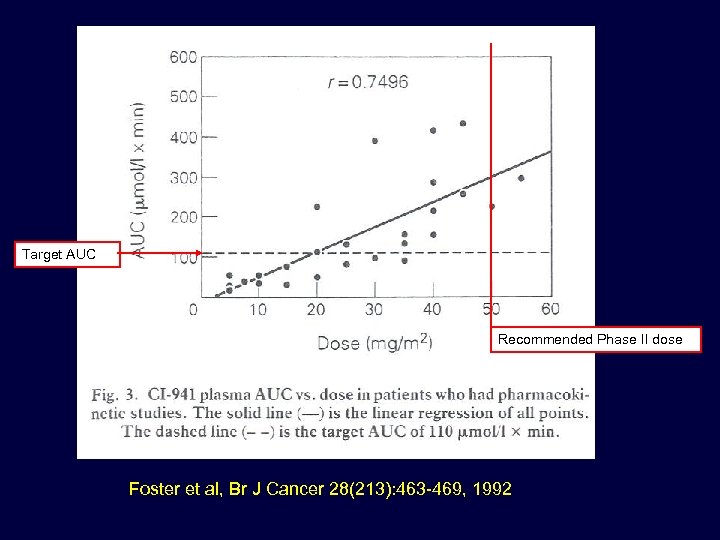 Target AUC Recommended Phase II dose Foster et al, Br J Cancer 28(213): 463 -469, 1992
Target AUC Recommended Phase II dose Foster et al, Br J Cancer 28(213): 463 -469, 1992
 Classical Methodology for Phase I and Translational Trials 1. – – – Traditional Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages – • • 3. Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals 2. Pharmacokinetically-guided (Collins) – – Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients Disadvantages • • Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible Continual Reassessment – – – Stochastic model to predict probability of DLT vs. dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • Methodologically complex • Needs constraints for safety • May take time to converge O'Quigley J et al: Biometrics, 46, 33 -48, 1990 4. – – Accelerated Phase I Design (Simon) Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels
Classical Methodology for Phase I and Translational Trials 1. – – – Traditional Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages – • • 3. Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals 2. Pharmacokinetically-guided (Collins) – – Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients Disadvantages • • Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible Continual Reassessment – – – Stochastic model to predict probability of DLT vs. dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • Methodologically complex • Needs constraints for safety • May take time to converge O'Quigley J et al: Biometrics, 46, 33 -48, 1990 4. – – Accelerated Phase I Design (Simon) Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels
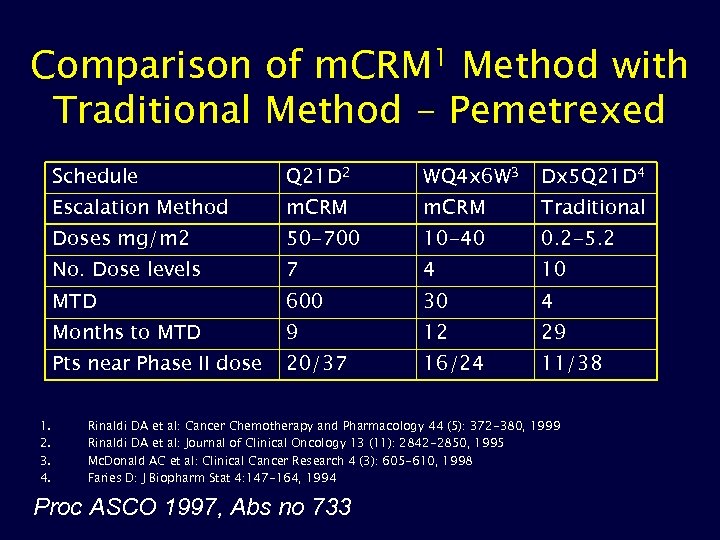 Comparison of m. CRM 1 Method with Traditional Method - Pemetrexed Schedule WQ 4 x 6 W 3 Dx 5 Q 21 D 4 Escalation Method m. CRM Traditional Doses mg/m 2 50 -700 10 -40 0. 2 -5. 2 No. Dose levels 7 4 10 MTD 600 30 4 Months to MTD 9 12 29 Pts near Phase II dose 1. 2. 3. 4. Q 21 D 2 20/37 16/24 11/38 Rinaldi DA et al: Cancer Chemotherapy and Pharmacology 44 (5): 372 -380, 1999 Rinaldi DA et al: Journal of Clinical Oncology 13 (11): 2842 -2850, 1995 Mc. Donald AC et al: Clinical Cancer Research 4 (3): 605 -610, 1998 Faries D: J Biopharm Stat 4: 147 -164, 1994 Proc ASCO 1997, Abs no 733
Comparison of m. CRM 1 Method with Traditional Method - Pemetrexed Schedule WQ 4 x 6 W 3 Dx 5 Q 21 D 4 Escalation Method m. CRM Traditional Doses mg/m 2 50 -700 10 -40 0. 2 -5. 2 No. Dose levels 7 4 10 MTD 600 30 4 Months to MTD 9 12 29 Pts near Phase II dose 1. 2. 3. 4. Q 21 D 2 20/37 16/24 11/38 Rinaldi DA et al: Cancer Chemotherapy and Pharmacology 44 (5): 372 -380, 1999 Rinaldi DA et al: Journal of Clinical Oncology 13 (11): 2842 -2850, 1995 Mc. Donald AC et al: Clinical Cancer Research 4 (3): 605 -610, 1998 Faries D: J Biopharm Stat 4: 147 -164, 1994 Proc ASCO 1997, Abs no 733
 Classical Methodology for Phase I and Translational Trials 1. – – Traditional 2. Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages • • Pharmacokinetically-guided (Collins) – – – Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals – Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients Disadvantages • • Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible 4. Accelerated Phase I Design (Simon) 3. – – – Continual Reassessment (O’Quigley) Stochastic model to predict probability of DLT vs. dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge – – Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels Simon R et al: Journal of the National Cancer Institute 89 (15): 1138 -1147, 1997
Classical Methodology for Phase I and Translational Trials 1. – – Traditional 2. Starting dose Modified Fibonacci escalation Maximum Tolerated Dose (MTD) as an endpoint Disadvantages • • Pharmacokinetically-guided (Collins) – – – Patient inefficient Many patients at ineffective doses Safety risk as MTD is approached No built-in confidence intervals – Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose Escalate in large increments to achieve target AUC in patients Disadvantages • • Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible 4. Accelerated Phase I Design (Simon) 3. – – – Continual Reassessment (O’Quigley) Stochastic model to predict probability of DLT vs. dose Starting dose Dose doubling Add data to model Predict dose with desired probability of DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge – – Starting dose Single patient dose doubling Increase patients per cohort and reduce dose increments when mild (Grade II) toxicity is seen Disadvantages • • Could be hazardous with a steep dose/toxicity relationship Little data at lower dose levels Simon R et al: Journal of the National Cancer Institute 89 (15): 1138 -1147, 1997
 Methodology for Phase I and Translational Trials • • Traditional – – • – – – – • SL Patient inefficient • Many patients at ineffective doses • Safety risk as MTD is approached • No built-in confidence intervals Stochastic model to predict probability of DLT vs dose E S Starting dose O O L Dose doubling ND Add T A to model S data Predict dose with desired probability of FA DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge – – Starting dose Y Modified Fibonacci escalation AD E Maximum Tolerated Dose (MTD) as ST an endpoint. ND A W Disadvantages O Continual Reassessment (O’Quigley) Pharmacokinetically-guided (Collins) • Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose T Escalate in large GE increments to achieve O target AUC in patients DT Disadvantages AR • • H Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible Accelerated Phase I Design (Simon) – Starting dose UL RF – Single patient dose. E doubling HE – Increase patients per cohort and C reduce dose D N increments when mild A (Grade II) toxicity is seen AP E – Disadvantages CH Could be hazardous with a steep • • dose/toxicity relationship Little data at lower dose levels
Methodology for Phase I and Translational Trials • • Traditional – – • – – – – • SL Patient inefficient • Many patients at ineffective doses • Safety risk as MTD is approached • No built-in confidence intervals Stochastic model to predict probability of DLT vs dose E S Starting dose O O L Dose doubling ND Add T A to model S data Predict dose with desired probability of FA DLT Disadvantages • • • Methodologically complex Needs constraints for safety May take time to converge – – Starting dose Y Modified Fibonacci escalation AD E Maximum Tolerated Dose (MTD) as ST an endpoint. ND A W Disadvantages O Continual Reassessment (O’Quigley) Pharmacokinetically-guided (Collins) • Establish Target Area Under the Curve (AUC) from preclinical studies Monitor Pharmacokinetics at starting dose T Escalate in large GE increments to achieve O target AUC in patients DT Disadvantages AR • • H Inter-patient variability in PKs Assumes linearity Metabolites May not be feasible Accelerated Phase I Design (Simon) – Starting dose UL RF – Single patient dose. E doubling HE – Increase patients per cohort and C reduce dose D N increments when mild A (Grade II) toxicity is seen AP E – Disadvantages CH Could be hazardous with a steep • • dose/toxicity relationship Little data at lower dose levels
 Use of Pharmacodynamic Endpoints • Almost always useful as a secondary endpoint – Clinical “proof of principle” of an effect on the target • May be useful as a primary endpoint if – Target is known, is single and is known to mediate therapeutic effect – Level of target suppression needed is known (50%, 99%? ) – Required duration of target effect is known – It is possible to measure all of the above • Methodology required for trials with a Pharmacodynamic endpoint – Requires definition of a dose where an effect of sufficient magnitude is present for sufficiently long in a sufficiently high proportion of the patients – Endpoint is scalar (e. g. , 95%) rather than Boolean (e. g. , DLT present or not) – Interpatient variability and confidence intervals – Prediction of duration of effect • Use of a scalar (continuously variable) methodology will also be of value where toxicity is used as an endpoint
Use of Pharmacodynamic Endpoints • Almost always useful as a secondary endpoint – Clinical “proof of principle” of an effect on the target • May be useful as a primary endpoint if – Target is known, is single and is known to mediate therapeutic effect – Level of target suppression needed is known (50%, 99%? ) – Required duration of target effect is known – It is possible to measure all of the above • Methodology required for trials with a Pharmacodynamic endpoint – Requires definition of a dose where an effect of sufficient magnitude is present for sufficiently long in a sufficiently high proportion of the patients – Endpoint is scalar (e. g. , 95%) rather than Boolean (e. g. , DLT present or not) – Interpatient variability and confidence intervals – Prediction of duration of effect • Use of a scalar (continuously variable) methodology will also be of value where toxicity is used as an endpoint
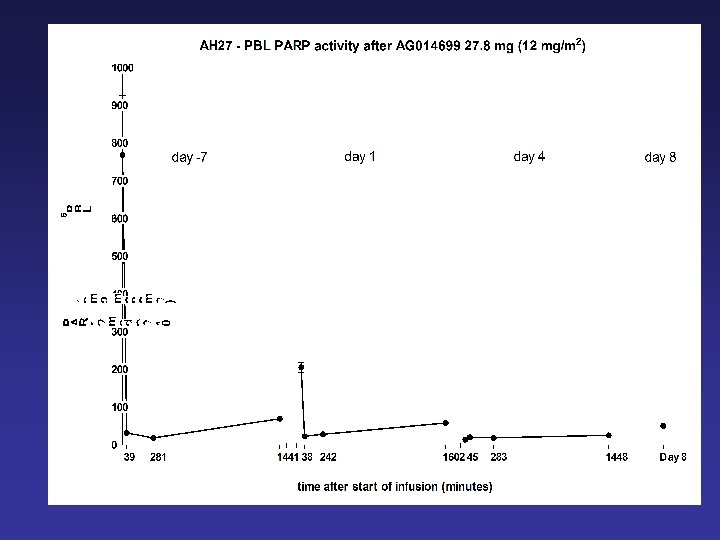
 PARP Inhibitor Phase 1 (0. 5? ) Trial: AG 014699 • Potent inhibitor, IV administration • Not expected to be active as a single agent (BRCA data not known at the time of design) • Expected to potentiate monomethylating agents and Topoisomerase I active compounds • Tumour biopsies required for PD endpoint • Desire for single agent data on PARP inhibitor • Combination study with temozolomide undertaken – PARP inhibitors potentiate temozolomide – Temozolomide active in melanoma – Melanoma patients have multiple lesions, biopsies relatively easy – Single dose of AG 14699 scheduled 1 week before combo
PARP Inhibitor Phase 1 (0. 5? ) Trial: AG 014699 • Potent inhibitor, IV administration • Not expected to be active as a single agent (BRCA data not known at the time of design) • Expected to potentiate monomethylating agents and Topoisomerase I active compounds • Tumour biopsies required for PD endpoint • Desire for single agent data on PARP inhibitor • Combination study with temozolomide undertaken – PARP inhibitors potentiate temozolomide – Temozolomide active in melanoma – Melanoma patients have multiple lesions, biopsies relatively easy – Single dose of AG 14699 scheduled 1 week before combo
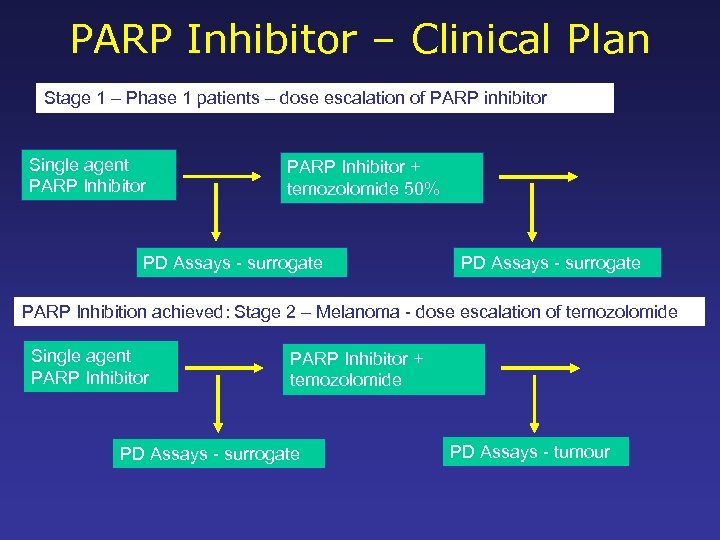 PARP Inhibitor – Clinical Plan Stage 1 – Phase 1 patients – dose escalation of PARP inhibitor Single agent PARP Inhibitor + temozolomide 50% PD Assays - surrogate PARP Inhibition achieved: Stage 2 – Melanoma - dose escalation of temozolomide Single agent PARP Inhibitor + temozolomide PD Assays - surrogate PD Assays - tumour
PARP Inhibitor – Clinical Plan Stage 1 – Phase 1 patients – dose escalation of PARP inhibitor Single agent PARP Inhibitor + temozolomide 50% PD Assays - surrogate PARP Inhibition achieved: Stage 2 – Melanoma - dose escalation of temozolomide Single agent PARP Inhibitor + temozolomide PD Assays - surrogate PD Assays - tumour
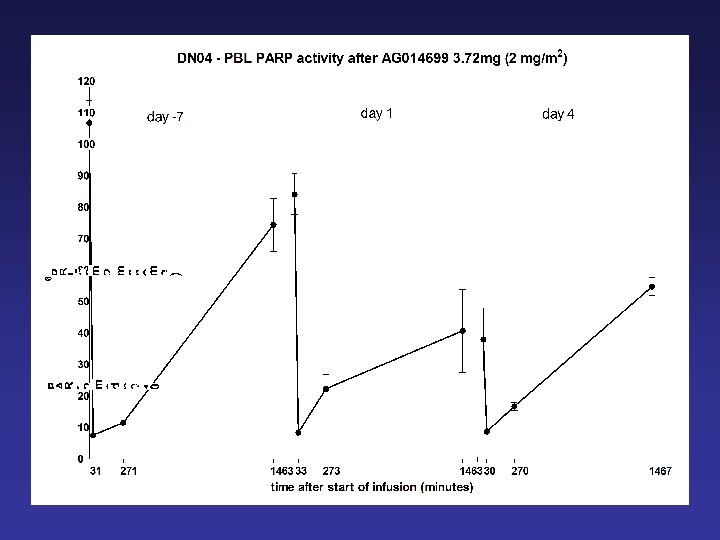
 PD End point • PARP Inhibitory Dose (PID) • Dose of AG-014699 causing ≥ 50% inhibition on PARP-1 ex vivo in peripheral blood lymphocytes 24 hours after 1 st dose, with a plateau in the degree of inhibition between dose levels. • Validated quantified immunoblot using monoclonal antibody against PAR
PD End point • PARP Inhibitory Dose (PID) • Dose of AG-014699 causing ≥ 50% inhibition on PARP-1 ex vivo in peripheral blood lymphocytes 24 hours after 1 st dose, with a plateau in the degree of inhibition between dose levels. • Validated quantified immunoblot using monoclonal antibody against PAR
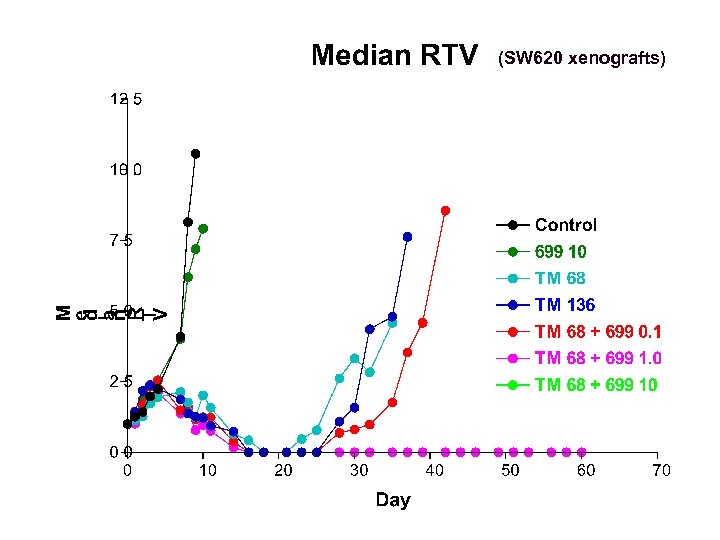 (SW 620 xenografts)
(SW 620 xenografts)
 PARP immunoblot assay with grateful thanks and credit to Alex permeabilised. Bürkle and Ruth Plummer cell suspension expose to NAD+ and oligonucleotide for 6 min PAR formed stop reaction with ice-cold 12. 5µM 699 blot known number of cells on to nylon membrane probe with 1° anti-PAR antibody probe with 2° HRP-conjugated antibody expose to ECL and measure luminescence
PARP immunoblot assay with grateful thanks and credit to Alex permeabilised. Bürkle and Ruth Plummer cell suspension expose to NAD+ and oligonucleotide for 6 min PAR formed stop reaction with ice-cold 12. 5µM 699 blot known number of cells on to nylon membrane probe with 1° anti-PAR antibody probe with 2° HRP-conjugated antibody expose to ECL and measure luminescence
 PARP Assay Validation • Minimise / explain variability – Enzyme stable with freezing? ? – Inhibition stable with freezing – Can inhibition be measured in PBMCs? • Establish procedures for handling samples – Does sampling and transport affect result? – Consistency of assay reagents • Provide standards for acceptability of results – Control samples – Intra- and inter-assay variability • Thanks to Ruth Plummer • Probably 1 -2 person years
PARP Assay Validation • Minimise / explain variability – Enzyme stable with freezing? ? – Inhibition stable with freezing – Can inhibition be measured in PBMCs? • Establish procedures for handling samples – Does sampling and transport affect result? – Consistency of assay reagents • Provide standards for acceptability of results – Control samples – Intra- and inter-assay variability • Thanks to Ruth Plummer • Probably 1 -2 person years
 Schedule: 28 day cycle : Temozolomide : AG 014699 Day: -10 to -4 ↑ 1 4 ↑ ↑ PK PK PK PD PD PD PK PK 8 15 22 28 PK (plasma) and PD (lymphocytes) in Part 1 (any tumour) and 2 (melanoma) First cycle only Comet Biopsy in Part 2 (melanoma patients) only
Schedule: 28 day cycle : Temozolomide : AG 014699 Day: -10 to -4 ↑ 1 4 ↑ ↑ PK PK PK PD PD PD PK PK 8 15 22 28 PK (plasma) and PD (lymphocytes) in Part 1 (any tumour) and 2 (melanoma) First cycle only Comet Biopsy in Part 2 (melanoma patients) only
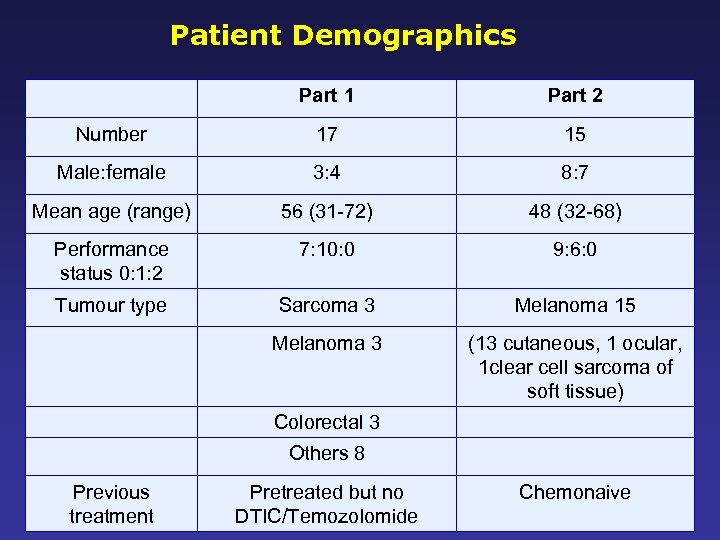 Patient Demographics Part 1 Part 2 Number 17 15 Male: female 3: 4 8: 7 Mean age (range) 56 (31 -72) 48 (32 -68) Performance status 0: 1: 2 7: 10: 0 9: 6: 0 Tumour type Sarcoma 3 Melanoma 15 Melanoma 3 (13 cutaneous, 1 ocular, 1 clear cell sarcoma of soft tissue) Colorectal 3 Others 8 Previous treatment Pretreated but no DTIC/Temozolomide Chemonaive
Patient Demographics Part 1 Part 2 Number 17 15 Male: female 3: 4 8: 7 Mean age (range) 56 (31 -72) 48 (32 -68) Performance status 0: 1: 2 7: 10: 0 9: 6: 0 Tumour type Sarcoma 3 Melanoma 15 Melanoma 3 (13 cutaneous, 1 ocular, 1 clear cell sarcoma of soft tissue) Colorectal 3 Others 8 Previous treatment Pretreated but no DTIC/Temozolomide Chemonaive
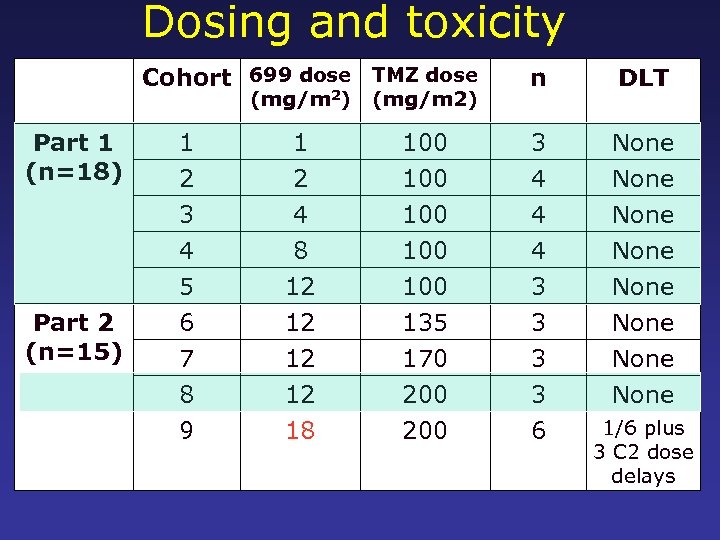 Dosing and toxicity Cohort 699 dose TMZ dose n DLT (mg/m 2) Part 1 (n=18) Part 2 (n=15) (mg/m 2) 1 2 100 3 4 None 3 4 5 6 7 8 9 4 8 12 12 18 100 100 135 170 200 4 4 3 3 6 None None 1/6 plus 3 C 2 dose delays
Dosing and toxicity Cohort 699 dose TMZ dose n DLT (mg/m 2) Part 1 (n=18) Part 2 (n=15) (mg/m 2) 1 2 100 3 4 None 3 4 5 6 7 8 9 4 8 12 12 18 100 100 135 170 200 4 4 3 3 6 None None 1/6 plus 3 C 2 dose delays
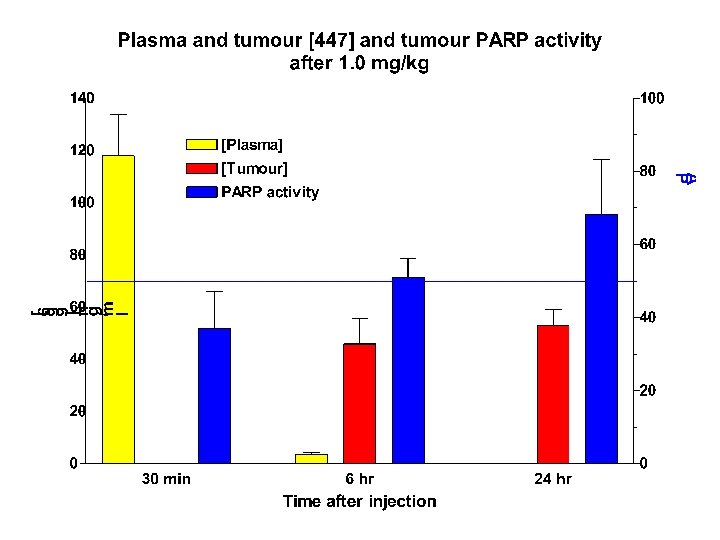
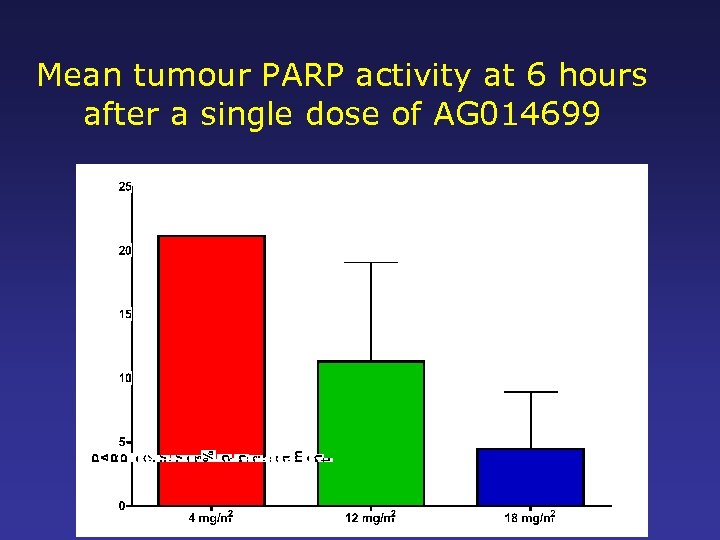 Mean tumour PARP activity at 6 hours after a single dose of AG 014699
Mean tumour PARP activity at 6 hours after a single dose of AG 014699
 AG 14699 Phase 0/1 Trial Interpretation • 12 mg/m 2 AG 14699 causes profound inhibition of PARP in PBMCs and ~90% inhibition in melanoma • 12 mg/m 2 AG 14699 can be given with a “full” dose of temozolomide • Protocol criteria have been met, but: – Might 18 mg/m 2 with a dose-reduction for temozolomide work better? – What would be the variability of the level and duration of tumour inhibition? – Do we need a longer period of inhibition for single agent treatment of BRCA tumours? – What might the effects on PARP homologues be?
AG 14699 Phase 0/1 Trial Interpretation • 12 mg/m 2 AG 14699 causes profound inhibition of PARP in PBMCs and ~90% inhibition in melanoma • 12 mg/m 2 AG 14699 can be given with a “full” dose of temozolomide • Protocol criteria have been met, but: – Might 18 mg/m 2 with a dose-reduction for temozolomide work better? – What would be the variability of the level and duration of tumour inhibition? – Do we need a longer period of inhibition for single agent treatment of BRCA tumours? – What might the effects on PARP homologues be?
 PARP Homologues • PARP-1 most prevalent most of existing data relates to PARP-1 • PARP-2 responsible for residual PARP activity in PARP-1 knockouts PARP-2 knockouts are also viable Double knockouts not viable • PARP-3 Unknown • PARP-4 V-PARP - drug resistance • PARP-5 Tankyrase 1 - involved in telomerase activity • PARP-6 Tankyrase 2 • PARP 7. . upwards ? function
PARP Homologues • PARP-1 most prevalent most of existing data relates to PARP-1 • PARP-2 responsible for residual PARP activity in PARP-1 knockouts PARP-2 knockouts are also viable Double knockouts not viable • PARP-3 Unknown • PARP-4 V-PARP - drug resistance • PARP-5 Tankyrase 1 - involved in telomerase activity • PARP-6 Tankyrase 2 • PARP 7. . upwards ? function
 Two Dimensional CRM Method An ni go ta c xi sm To ity tiv di Ad y rg ne Sy • For any two drugs, there is a range of maximum tolerated dose pairs c • Toxicities may potentiate or antagonise xi To • For every single agent Phase I there will be many combination Phase Is c xi – “Multikinase inhibitors” To • New targeted agents will be used in combination with both traditional cytotoxics and other targeted agents MTD of Drug B Developed in house by James Wright MTD of Drug A
Two Dimensional CRM Method An ni go ta c xi sm To ity tiv di Ad y rg ne Sy • For any two drugs, there is a range of maximum tolerated dose pairs c • Toxicities may potentiate or antagonise xi To • For every single agent Phase I there will be many combination Phase Is c xi – “Multikinase inhibitors” To • New targeted agents will be used in combination with both traditional cytotoxics and other targeted agents MTD of Drug B Developed in house by James Wright MTD of Drug A
 Two Dimensional CRM Method Developed by James Wright, Ph. D Student, 1997 -2000 • • • CRM Methodology requires that the probability of DLT at each level is estimated before the start of the trial (priors) A model relating the probability of DLT to dose is created using the estimated data points As real data accumulate during the course of the trial they are used to modify the model A problem for single agent studies is that the initial estimates may be way out For combination Phase I studies, single agent data are already available, facilitating the estimation of priors Hypothetical example: D 1 0. 00 0. 25 0. 4 0. 6 0. 8 0. 95 0. 00 0. 16 0. 22 0. 30 0. 48 0. 25 0. 16 0. 24 0. 30 0. 40 0. 51 0. 62 0. 60 0. 80 0. 95 0. 30 0. 48 0. 40 0. 51 0. 62 0. 49 0. 60 0. 68 0. 60 0. 77 0. 70 0. 79 0. 84 0. 77 0. 84 0. 88 D 2 0. 00 0. 40 0. 22 0. 30 0. 37 0. 49 0. 60 0. 68 Data derived from single agent Phase I Studies Data estimated from mechanistic knowledge and experience Priors are constructed showing the probability of dose limiting toxicity for each pair of doses
Two Dimensional CRM Method Developed by James Wright, Ph. D Student, 1997 -2000 • • • CRM Methodology requires that the probability of DLT at each level is estimated before the start of the trial (priors) A model relating the probability of DLT to dose is created using the estimated data points As real data accumulate during the course of the trial they are used to modify the model A problem for single agent studies is that the initial estimates may be way out For combination Phase I studies, single agent data are already available, facilitating the estimation of priors Hypothetical example: D 1 0. 00 0. 25 0. 4 0. 6 0. 8 0. 95 0. 00 0. 16 0. 22 0. 30 0. 48 0. 25 0. 16 0. 24 0. 30 0. 40 0. 51 0. 62 0. 60 0. 80 0. 95 0. 30 0. 48 0. 40 0. 51 0. 62 0. 49 0. 60 0. 68 0. 60 0. 77 0. 70 0. 79 0. 84 0. 77 0. 84 0. 88 D 2 0. 00 0. 40 0. 22 0. 30 0. 37 0. 49 0. 60 0. 68 Data derived from single agent Phase I Studies Data estimated from mechanistic knowledge and experience Priors are constructed showing the probability of dose limiting toxicity for each pair of doses
 CRM Method – Illustration with Completed Trial • OSI 211 in combination Phase I with carboplatin OSI 211 – Liposomal Lurtotecan Carboplatin
CRM Method – Illustration with Completed Trial • OSI 211 in combination Phase I with carboplatin OSI 211 – Liposomal Lurtotecan Carboplatin
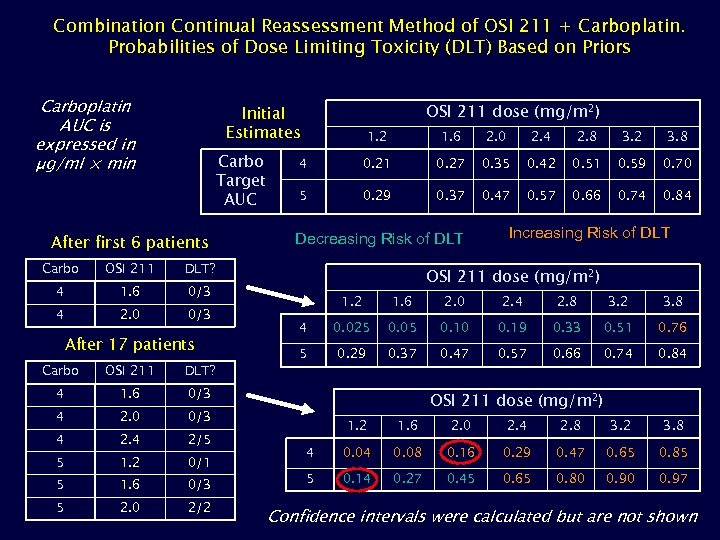 Combination Continual Reassessment Method of OSI 211 + Carboplatin. Probabilities of Dose Limiting Toxicity (DLT) Based on Priors Carboplatin AUC is expressed in µg/ml × min OSI 211 dose (mg/m 2) Initial Estimates Carbo Target AUC After first 6 patients Carbo OSI 211 1. 6 2. 0 0/3 2. 0 2. 4 2. 8 3. 2 3. 8 4 0. 21 0. 27 0. 35 0. 42 0. 51 0. 59 0. 70 5 0. 29 0. 37 0. 47 0. 57 0. 66 0. 74 0. 84 Decreasing Risk of DLT 0/3 4 1. 6 DLT? 4 1. 2 After 17 patients Carbo OSI 211 DLT? 4 1. 6 2. 0 2. 4 2/5 5 1. 2 0/1 5 1. 6 0/3 5 2. 0 2/2 1. 6 2. 0 2. 4 2. 8 3. 2 3. 8 4 0. 025 0. 05 0. 10 0. 19 0. 33 0. 51 0. 76 5 0. 29 0. 37 0. 47 0. 57 0. 66 0. 74 0. 84 0/3 4 OSI 211 dose (mg/m 2) 0/3 4 Increasing Risk of DLT OSI 211 dose (mg/m 2) 1. 2 1. 6 2. 0 2. 4 2. 8 3. 2 3. 8 4 0. 08 0. 16 0. 29 0. 47 0. 65 0. 85 5 0. 14 0. 27 0. 45 0. 65 0. 80 0. 97 Confidence intervals were calculated but are not shown
Combination Continual Reassessment Method of OSI 211 + Carboplatin. Probabilities of Dose Limiting Toxicity (DLT) Based on Priors Carboplatin AUC is expressed in µg/ml × min OSI 211 dose (mg/m 2) Initial Estimates Carbo Target AUC After first 6 patients Carbo OSI 211 1. 6 2. 0 0/3 2. 0 2. 4 2. 8 3. 2 3. 8 4 0. 21 0. 27 0. 35 0. 42 0. 51 0. 59 0. 70 5 0. 29 0. 37 0. 47 0. 57 0. 66 0. 74 0. 84 Decreasing Risk of DLT 0/3 4 1. 6 DLT? 4 1. 2 After 17 patients Carbo OSI 211 DLT? 4 1. 6 2. 0 2. 4 2/5 5 1. 2 0/1 5 1. 6 0/3 5 2. 0 2/2 1. 6 2. 0 2. 4 2. 8 3. 2 3. 8 4 0. 025 0. 05 0. 10 0. 19 0. 33 0. 51 0. 76 5 0. 29 0. 37 0. 47 0. 57 0. 66 0. 74 0. 84 0/3 4 OSI 211 dose (mg/m 2) 0/3 4 Increasing Risk of DLT OSI 211 dose (mg/m 2) 1. 2 1. 6 2. 0 2. 4 2. 8 3. 2 3. 8 4 0. 08 0. 16 0. 29 0. 47 0. 65 0. 85 5 0. 14 0. 27 0. 45 0. 65 0. 80 0. 97 Confidence intervals were calculated but are not shown
 Proposed Enhancements of 2 -Dimensional Phase I Methodology • Use a scalar rather than a Boolean endpoint (e. g. , reduction in neutrophil count rather than MTD) • Modify for use with Pharmacodynamic endpoints
Proposed Enhancements of 2 -Dimensional Phase I Methodology • Use a scalar rather than a Boolean endpoint (e. g. , reduction in neutrophil count rather than MTD) • Modify for use with Pharmacodynamic endpoints
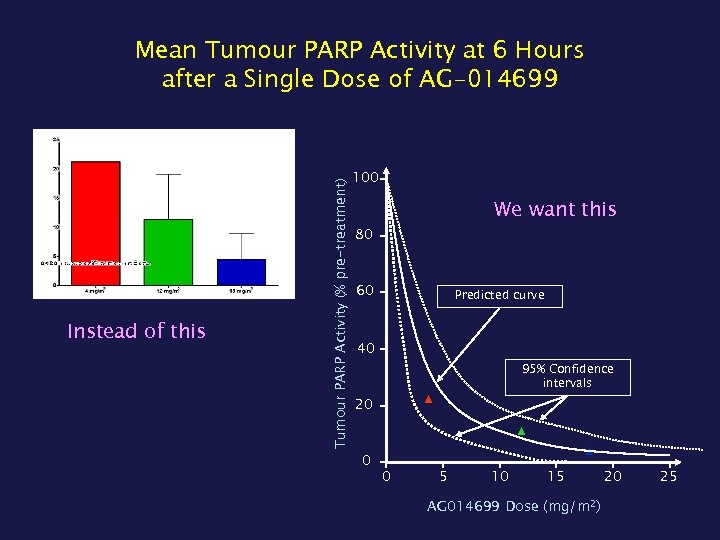 Instead of this Tumour PARP Activity (% pre-treatment) Mean Tumour PARP Activity at 6 Hours after a Single Dose of AG-014699 100 We want this 80 60 Predicted curve 40 95% Confidence intervals ▲ 20 ▲ 0 0 5 10 ▲ 15 AG 014699 Dose (mg/m 2) 20 25
Instead of this Tumour PARP Activity (% pre-treatment) Mean Tumour PARP Activity at 6 Hours after a Single Dose of AG-014699 100 We want this 80 60 Predicted curve 40 95% Confidence intervals ▲ 20 ▲ 0 0 5 10 ▲ 15 AG 014699 Dose (mg/m 2) 20 25
 Methodology for Phase I and Translational Trials - Needs • Trial methodology designed for targeted agents in trials with pharmacodynamic endpoints methods that utilise continuously variable (scalar) endpoints rather than yes/no (Boolean) endpoints • Extension of these techniques to combination Phase I trials – Models to detect trends may be more appropriate than hypothesis-testing • We need to use these methods where available and develop new mathematical models where they are not • Early investment in PD assay development and validation
Methodology for Phase I and Translational Trials - Needs • Trial methodology designed for targeted agents in trials with pharmacodynamic endpoints methods that utilise continuously variable (scalar) endpoints rather than yes/no (Boolean) endpoints • Extension of these techniques to combination Phase I trials – Models to detect trends may be more appropriate than hypothesis-testing • We need to use these methods where available and develop new mathematical models where they are not • Early investment in PD assay development and validation
 Acknowledgements Newcastle Agouron/Pfizer Patients Research nurses Ruth Plummer Nicola Curtin Herbie Newell Roger Griffin Chris Jones Alan Boddy Barbara Durkacz Bernard Golding Plus the other clinical investigators Heidi Steinfeldt Zdenek Hostomsky Raz Dweji Gerrit Los Cancer Research UK
Acknowledgements Newcastle Agouron/Pfizer Patients Research nurses Ruth Plummer Nicola Curtin Herbie Newell Roger Griffin Chris Jones Alan Boddy Barbara Durkacz Bernard Golding Plus the other clinical investigators Heidi Steinfeldt Zdenek Hostomsky Raz Dweji Gerrit Los Cancer Research UK


