52c2e9cf9b3b9e5a4bc5cde2cdfcdad1.ppt
- Количество слайдов: 28
 PERC: Envisioning the Next Generation of EPath NAACCR Annual Meeting June 10, 2008, Denver Jennifer Seiffert, MLIS, CTR, Northrop Grumman Contractor to NPCR, CDC jenesei@comcast. net Joseph Rogers, NPCR, CDC jdr 0@cdc. gov
PERC: Envisioning the Next Generation of EPath NAACCR Annual Meeting June 10, 2008, Denver Jennifer Seiffert, MLIS, CTR, Northrop Grumman Contractor to NPCR, CDC jenesei@comcast. net Joseph Rogers, NPCR, CDC jdr 0@cdc. gov
 Outline • • • PERC History PERC Members PERC Goals Project Vision Cancer Care Ontario Activities
Outline • • • PERC History PERC Members PERC Goals Project Vision Cancer Care Ontario Activities
 Note • Presentation will emphasize Collaborative Staging (CS) because that is my role on PERC
Note • Presentation will emphasize Collaborative Staging (CS) because that is my role on PERC
 History (1) • PERT (Pathology Electronic Reporting Task Force) established in 2007 • PERT grew out of several prior activities: – SNOMED Surgical Pathology Working Group • Reviewed and updated the SNOMED Anatomic Pathology subset (took over a year) – College of American Pathologists (CAP) Cancer Committee
History (1) • PERT (Pathology Electronic Reporting Task Force) established in 2007 • PERT grew out of several prior activities: – SNOMED Surgical Pathology Working Group • Reviewed and updated the SNOMED Anatomic Pathology subset (took over a year) – College of American Pathologists (CAP) Cancer Committee
 History (2) • In 2008, PERT was upgraded to PERC – A full Committee of CAP – Reports to SNOMED STS [SNOMED Terminology Solutions] Oversight Committee
History (2) • In 2008, PERT was upgraded to PERC – A full Committee of CAP – Reports to SNOMED STS [SNOMED Terminology Solutions] Oversight Committee
 History (3) • PERC has established two subcommittees: – Data Modeling (back end) group for modeling how CAP checklist data can best be collected electronically, interoperably, and mapped to other standards, including CS • Includes CS and NAACCR representatives – User Interface (UI) (front end) group for modeling interface for pathologists entering checklist data electronically
History (3) • PERC has established two subcommittees: – Data Modeling (back end) group for modeling how CAP checklist data can best be collected electronically, interoperably, and mapped to other standards, including CS • Includes CS and NAACCR representatives – User Interface (UI) (front end) group for modeling interface for pathologists entering checklist data electronically
 History (4) • Priority being given to UI work, getting standardized electronic checklists to pathologists and LIS vendors ASAP • Decision has been made, for current work, to – Include mapping to CS for items with one-to-one non-algorithmic maps – Postpone mapping to CS items requiring reference to multiple data items or algorithms to 2 nd phase of project after easy mappings done
History (4) • Priority being given to UI work, getting standardized electronic checklists to pathologists and LIS vendors ASAP • Decision has been made, for current work, to – Include mapping to CS for items with one-to-one non-algorithmic maps – Postpone mapping to CS items requiring reference to multiple data items or algorithms to 2 nd phase of project after easy mappings done
 PERC Members May 2008 • • John Madden, MD, Ph. D Co-Chair Duke University Monica de Baca, MD, Co-Chair Physicians Laboratory George Birdsong, MD Emory University Kenneth Gerlach, MPH, CTR CDC/NPCR Lori Havener, CTR NAACCR Mary Kennedy, MPH CAP Robert Knapp, MD Pathology Laboratory, P. C. • • • Gemma Lee Cancer Care Ontario Andrea Mac. Lean Cancer Care Ontario Richard Moldwin, MD, Ph. D CAP Douglas Murphy CAP Wendy Scharber, RHIT, CTR Registry Widgets Jennifer Seiffert, MLIS, CTR Collaborative Staging Mike Smith, MD CAP James Sorace, MD, MS ASPE, HHS Henry Travers, MD President, WASPa. LM
PERC Members May 2008 • • John Madden, MD, Ph. D Co-Chair Duke University Monica de Baca, MD, Co-Chair Physicians Laboratory George Birdsong, MD Emory University Kenneth Gerlach, MPH, CTR CDC/NPCR Lori Havener, CTR NAACCR Mary Kennedy, MPH CAP Robert Knapp, MD Pathology Laboratory, P. C. • • • Gemma Lee Cancer Care Ontario Andrea Mac. Lean Cancer Care Ontario Richard Moldwin, MD, Ph. D CAP Douglas Murphy CAP Wendy Scharber, RHIT, CTR Registry Widgets Jennifer Seiffert, MLIS, CTR Collaborative Staging Mike Smith, MD CAP James Sorace, MD, MS ASPE, HHS Henry Travers, MD President, WASPa. LM
 PERC Goals (1) • To advance the computerized representation of the CAP checklists – Create FRAMEWORK for • electronic forms (standardized input) • data repositories (retrievable output) – Ensure electronic versions accurately represent the CAP Cancer Committee’s intended meaning
PERC Goals (1) • To advance the computerized representation of the CAP checklists – Create FRAMEWORK for • electronic forms (standardized input) • data repositories (retrievable output) – Ensure electronic versions accurately represent the CAP Cancer Committee’s intended meaning
 PERC Goals (2) • To create an electronic representation of the CAP Cancer Protocol checklists which – Is aligned with (upcoming) AJCC and Collaborative Staging Systems. – Allows for historical representation – Is amply designed for addition of medical specialties, coding, and/or staging systems
PERC Goals (2) • To create an electronic representation of the CAP Cancer Protocol checklists which – Is aligned with (upcoming) AJCC and Collaborative Staging Systems. – Allows for historical representation – Is amply designed for addition of medical specialties, coding, and/or staging systems
 Project Vision (1) • CAP provides software tools for computerized implementations of cancer checklists • Path labs and vendors implement standardized checklist templates for synoptic cancer reports as part of full path reports
Project Vision (1) • CAP provides software tools for computerized implementations of cancer checklists • Path labs and vendors implement standardized checklist templates for synoptic cancer reports as part of full path reports
 Project Vision (2) • Central cancer registries receive standardized synoptic e. Path and can automatically populate standard NAACCR items, including CS • Registries consolidate e. Path with other reports (clinical, demographic)
Project Vision (2) • Central cancer registries receive standardized synoptic e. Path and can automatically populate standard NAACCR items, including CS • Registries consolidate e. Path with other reports (clinical, demographic)
 Aspects of Framework (1) Template Editor • Software application and database for creating and distributing computerized checklists • Written specifically for PERC work • Enables automated data capture • Provides mapping to other standards incl. SNOMED, NAACCR, CS • Facilitates reuse of data for CS, research, ca. Big, tissue banking, etc.
Aspects of Framework (1) Template Editor • Software application and database for creating and distributing computerized checklists • Written specifically for PERC work • Enables automated data capture • Provides mapping to other standards incl. SNOMED, NAACCR, CS • Facilitates reuse of data for CS, research, ca. Big, tissue banking, etc.
 Aspects of Framework (2) Consistency across checklists to improve accuracy. Examples: • vascular invasion and lymphatic invasion standardized to “lymph/vascular invasion” • mitotic activity vs. mitotic count • tumor configuration vs. tumor features • Standardizing “flavors of null” (indeterminate, no data, not reported, etc. )
Aspects of Framework (2) Consistency across checklists to improve accuracy. Examples: • vascular invasion and lymphatic invasion standardized to “lymph/vascular invasion” • mitotic activity vs. mitotic count • tumor configuration vs. tumor features • Standardizing “flavors of null” (indeterminate, no data, not reported, etc. )
 Aspects of Framework (3) Use appropriate dialog controls to improve accuracy Group of Radio Buttons Check Boxes Choose one: o Squamous cell carcinoma o Adenocarcinoma o Large cell carcinoma o Small cell carcinoma Choose as many as apply: q Embryonal carcinoma q Choriocarcinoma q Yolk sac tumor q Teratoma
Aspects of Framework (3) Use appropriate dialog controls to improve accuracy Group of Radio Buttons Check Boxes Choose one: o Squamous cell carcinoma o Adenocarcinoma o Large cell carcinoma o Small cell carcinoma Choose as many as apply: q Embryonal carcinoma q Choriocarcinoma q Yolk sac tumor q Teratoma
 Aspects of Framework (4) Increased Atomicity Current Checklist Format Possible modification ___ Tumor 2 cm or less without extra-parenchymal extension (T 1). Tumor size: ___ Tumor 2 cm but not more than 4 cm without extracapsular extension (T 2). Extraparenchymal extension Positive Negative Indeterminate ___ Tumor more than 4 cm and/or tumor having extraparenchymal extension (T 3). _______ cm CS algorithm could derive p. T.
Aspects of Framework (4) Increased Atomicity Current Checklist Format Possible modification ___ Tumor 2 cm or less without extra-parenchymal extension (T 1). Tumor size: ___ Tumor 2 cm but not more than 4 cm without extracapsular extension (T 2). Extraparenchymal extension Positive Negative Indeterminate ___ Tumor more than 4 cm and/or tumor having extraparenchymal extension (T 3). _______ cm CS algorithm could derive p. T.
 Aspects of Framework (5) Context Sensitivity ___ Cannot be assessed ___ Benign glands at surgical margin _X_ Margins uninvolved by invasive CA ___ Margin(s) involved by invasive CA ___ Cannot be assessed ___ Benign glands at surgical margin ___ Margins uninvolved by invasive CA _X_ Margin(s) involved by invasive CA ___ Unifocal ___ Multifocal ___ Unifocal _X__ Multifocal ___ Apical ___ Bladder neck ___ Anterior ___ Lateral ___ Postero-lateral ___ Posterior ___ Other(s) (specify): ___ Apical _X_ Bladder neck ___ Anterior _X_ Lateral ___ Postero-lateral ___ Posterior ___ Other(s) (specify):
Aspects of Framework (5) Context Sensitivity ___ Cannot be assessed ___ Benign glands at surgical margin _X_ Margins uninvolved by invasive CA ___ Margin(s) involved by invasive CA ___ Cannot be assessed ___ Benign glands at surgical margin ___ Margins uninvolved by invasive CA _X_ Margin(s) involved by invasive CA ___ Unifocal ___ Multifocal ___ Unifocal _X__ Multifocal ___ Apical ___ Bladder neck ___ Anterior ___ Lateral ___ Postero-lateral ___ Posterior ___ Other(s) (specify): ___ Apical _X_ Bladder neck ___ Anterior _X_ Lateral ___ Postero-lateral ___ Posterior ___ Other(s) (specify):
 NPCR’s Goal • Increase quality of cancer surveillance data and efficiency of collection by increasing use of CAP checklists and synoptic reporting – How? Make them easier to use by pathologists, lab system vendors, registries
NPCR’s Goal • Increase quality of cancer surveillance data and efficiency of collection by increasing use of CAP checklists and synoptic reporting – How? Make them easier to use by pathologists, lab system vendors, registries
 Project Vision Review(1) • CAP provides software tools for computerized implementations of cancer checklists • Path labs and vendors implement standardized checklist templates for synoptic cancer reports as part of full path reports
Project Vision Review(1) • CAP provides software tools for computerized implementations of cancer checklists • Path labs and vendors implement standardized checklist templates for synoptic cancer reports as part of full path reports
 Project Vision Review (2) • Central cancer registries receive standardized synoptic e. Path and can automatically populate standard NAACCR items, including CS • Registries consolidate e. Path with other reports (clinical, demographic)
Project Vision Review (2) • Central cancer registries receive standardized synoptic e. Path and can automatically populate standard NAACCR items, including CS • Registries consolidate e. Path with other reports (clinical, demographic)
 PERC Activities for 2008 • Weekly teleconferences of each subgroup • Goal to have model electronic data entry forms for selected checklists published in 2008, using TNM 6 th ed. , for pathologists and LIS vendors to begin implementing • Will probably recommend simple interim database approach for LIS vendors— limited mapping
PERC Activities for 2008 • Weekly teleconferences of each subgroup • Goal to have model electronic data entry forms for selected checklists published in 2008, using TNM 6 th ed. , for pathologists and LIS vendors to begin implementing • Will probably recommend simple interim database approach for LIS vendors— limited mapping
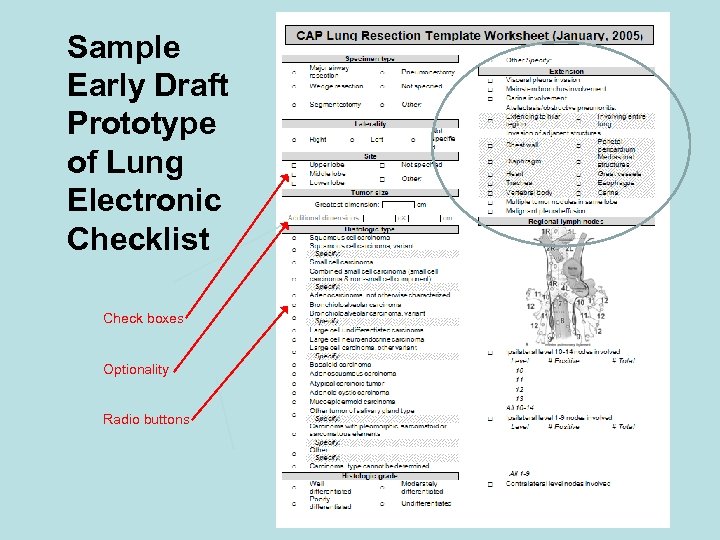 Sample Early Draft Prototype of Lung Electronic Checklist Check boxes Optionality Radio buttons
Sample Early Draft Prototype of Lung Electronic Checklist Check boxes Optionality Radio buttons
 Lung Draft Prototype Detail • Pathologist checks all that apply • Choices are nested, grouped • Mapping to CS Extension available behind the scenes • Database can be built behind the scenes • Algorithm needed for combinations checked. 45 20 70 40 55 60 56 60 70 71 70 70 75 65 45
Lung Draft Prototype Detail • Pathologist checks all that apply • Choices are nested, grouped • Mapping to CS Extension available behind the scenes • Database can be built behind the scenes • Algorithm needed for combinations checked. 45 20 70 40 55 60 56 60 70 71 70 70 75 65 45
 Cancer Care Ontario Activities • Real-world test of some of the PERC concepts – 90% of path reports in province of Ontario are received electronically – Modified CAP/CS aligned checklists implemented in participating hospitals – Central registry will capture CS items from synoptic reports mapped to CS and derive stage – Feasibility and accuracy will be assessed – Also testing a computerized path requisition form for surgeons to complete
Cancer Care Ontario Activities • Real-world test of some of the PERC concepts – 90% of path reports in province of Ontario are received electronically – Modified CAP/CS aligned checklists implemented in participating hospitals – Central registry will capture CS items from synoptic reports mapped to CS and derive stage – Feasibility and accuracy will be assessed – Also testing a computerized path requisition form for surgeons to complete
 Cancer Care Ontario Activities • Collaborative Staging Colorectal Pilot Project. – Modified front end of checklist to align w/CS. – Pathologists complete checklist, data are extracted from hospital information system, CCO coders supplement staging data w/radiology, surgery and clinical info • Other Sites to come: breast, lung, prostate, endometrium • e. Surgery and e. Radiology feasibility assessment underway
Cancer Care Ontario Activities • Collaborative Staging Colorectal Pilot Project. – Modified front end of checklist to align w/CS. – Pathologists complete checklist, data are extracted from hospital information system, CCO coders supplement staging data w/radiology, surgery and clinical info • Other Sites to come: breast, lung, prostate, endometrium • e. Surgery and e. Radiology feasibility assessment underway
 Issues from Cancer Care Ontario (1) • Multiple path reports on same patient/tumor – Need guidelines for pathologists—registry would prefer separate synoptic report for each reportable tumor – Need guidelines for registries, consolidating multiple reports from multiple specimens – Need to determine if reports pertain to stage AT DIAGNOSIS
Issues from Cancer Care Ontario (1) • Multiple path reports on same patient/tumor – Need guidelines for pathologists—registry would prefer separate synoptic report for each reportable tumor – Need guidelines for registries, consolidating multiple reports from multiple specimens – Need to determine if reports pertain to stage AT DIAGNOSIS
 Issues from Cancer Care Ontario (2) • Determining which lab results to capture from multiple tests for SSFs/tumor markers • Can algorithm be written to select the appropriate test results from lab information system? – Usually need highest value prior to treatment – Rules are site- and test-specific
Issues from Cancer Care Ontario (2) • Determining which lab results to capture from multiple tests for SSFs/tumor markers • Can algorithm be written to select the appropriate test results from lab information system? – Usually need highest value prior to treatment – Rules are site- and test-specific
 Information about CDC’s Cancer Prevention & Control Programs www. cdc. gov/cancer The findings and conclusions in this presentation are those of the presenter, and do not necessarily represent the views of the Centers for Disease Control and Prevention.
Information about CDC’s Cancer Prevention & Control Programs www. cdc. gov/cancer The findings and conclusions in this presentation are those of the presenter, and do not necessarily represent the views of the Centers for Disease Control and Prevention.


