2dc2897dd41fa1c0943779c98fc4da0c.ppt
- Количество слайдов: 21
 Partial Oxidation of Propylene to Acrolein Final Design Presentation April 23, 2008 Kerri M. May Megerle L. Scherholz Christopher M. Watts
Partial Oxidation of Propylene to Acrolein Final Design Presentation April 23, 2008 Kerri M. May Megerle L. Scherholz Christopher M. Watts
 Overview • Introduction • Process Background • Design Process ▫ ▫ Determination of Volume Pressure Drop Multiple Reactions Heat Effects • Optimization • Final Design • Conclusion
Overview • Introduction • Process Background • Design Process ▫ ▫ Determination of Volume Pressure Drop Multiple Reactions Heat Effects • Optimization • Final Design • Conclusion
 Introduction • Design of fixed-bed reactor • Production of acrolein by partial oxidation ▫ CH 2 = CH - CH 3 + O 2 → CH 2 = CH – CHO + H 2 O • 13, 500 Mtons/year with a 2 week downtime ▫ Corresponds to 0. 007941 kmol/s • Original design: ideal/isobaric/isothermal • Final design: pressure drop, multiple reactions and heat effects • Optimized using selectivity and gain
Introduction • Design of fixed-bed reactor • Production of acrolein by partial oxidation ▫ CH 2 = CH - CH 3 + O 2 → CH 2 = CH – CHO + H 2 O • 13, 500 Mtons/year with a 2 week downtime ▫ Corresponds to 0. 007941 kmol/s • Original design: ideal/isobaric/isothermal • Final design: pressure drop, multiple reactions and heat effects • Optimized using selectivity and gain
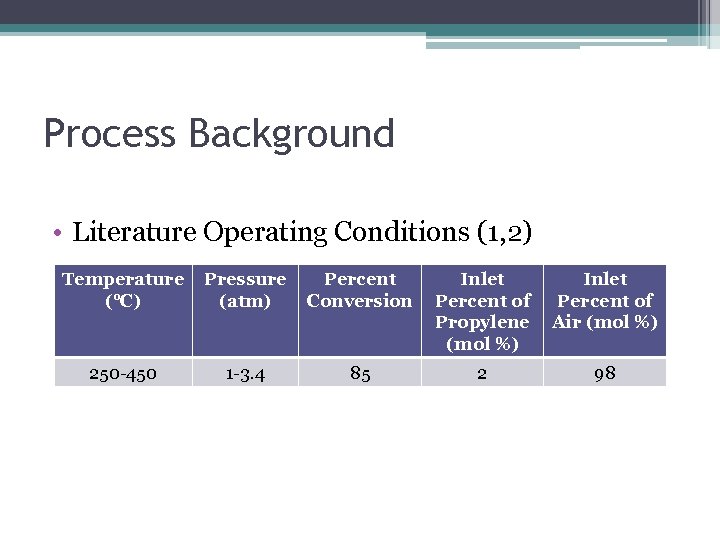 Process Background • Literature Operating Conditions (1, 2) Temperature (°C) Pressure (atm) Percent Conversion Inlet Percent of Propylene (mol %) Inlet Percent of Air (mol %) 250 -450 1 -3. 4 85 2 98
Process Background • Literature Operating Conditions (1, 2) Temperature (°C) Pressure (atm) Percent Conversion Inlet Percent of Propylene (mol %) Inlet Percent of Air (mol %) 250 -450 1 -3. 4 85 2 98
 Process Background Continued • Assumptions Parameter Value Particle Size 5 mm (3) Bulk Density 1415 kg-cat/m 3 -rxtr (4) Packed Bed Void Fraction 0. 38 (4) Tube Diameter 1 in. (0. 0254 m) Viscosity of Air at 390°C 3. 15 x 10 -5 kg/m-s (5) Coolant Temperature 673 K (390°C) Overall Heat Transfer Coefficient 227 J/W-m 2 -K (3) • Given for final design • Deviations for other models discussed
Process Background Continued • Assumptions Parameter Value Particle Size 5 mm (3) Bulk Density 1415 kg-cat/m 3 -rxtr (4) Packed Bed Void Fraction 0. 38 (4) Tube Diameter 1 in. (0. 0254 m) Viscosity of Air at 390°C 3. 15 x 10 -5 kg/m-s (5) Coolant Temperature 673 K (390°C) Overall Heat Transfer Coefficient 227 J/W-m 2 -K (3) • Given for final design • Deviations for other models discussed
 Process Background Continued • Stoichiometric Flow Rates Inlet Compositions Outlet Compositions Mole (kmol/s) Propylene 0. 0093420221 0. 0014013 Oxygen 0. 0888951791 0. 0809545 Inert Nitrogen 0. 0382188797 0. 3821888 Acrolein 0 0. 0079407 Water 0 0. 0079407 Total 0. 4804259982 0. 480426
Process Background Continued • Stoichiometric Flow Rates Inlet Compositions Outlet Compositions Mole (kmol/s) Propylene 0. 0093420221 0. 0014013 Oxygen 0. 0888951791 0. 0809545 Inert Nitrogen 0. 0382188797 0. 3821888 Acrolein 0 0. 0079407 Water 0 0. 0079407 Total 0. 4804259982 0. 480426
 Process Background Continued • Catalyst chosen based on kinetics ▫ Bismuth molybdate (6) • Co-current Heat Exchanger Fluid ▫ Exothermic reaction ▫ Molten Salt used as coolant fluid ▫ Sodium tetrasulfide (7) Melting temperature (294°C)
Process Background Continued • Catalyst chosen based on kinetics ▫ Bismuth molybdate (6) • Co-current Heat Exchanger Fluid ▫ Exothermic reaction ▫ Molten Salt used as coolant fluid ▫ Sodium tetrasulfide (7) Melting temperature (294°C)
 Process Background Continued ▫ Selectivity of Acrolein ▫ Selectivity of Other Profitable Products ▫ Gain
Process Background Continued ▫ Selectivity of Acrolein ▫ Selectivity of Other Profitable Products ▫ Gain
 Process Background Continued • Reaction Kinetics of Byproducts (6, 8) ▫ Reaction Pathway ▫ Assumptions: Steady State Single-site oxygen adsorption Rate of oxidation of acrolein to carbon oxides is negligible compared to other rates
Process Background Continued • Reaction Kinetics of Byproducts (6, 8) ▫ Reaction Pathway ▫ Assumptions: Steady State Single-site oxygen adsorption Rate of oxidation of acrolein to carbon oxides is negligible compared to other rates
 Process Background Continued • Reaction rates for the formation of acrolein and byproducts (6, 8) Where: r 2 = rate of formation of acrolein, kmol/kgcat-s r 3 co 2 = rate of formation of carbon dioxide, kmol/kgcat-s r 3 co = rate of formation of carbon monoxide, kmol/kgcat-s r 4 = rate of formation of acetaldehyde, kmol/kgcat-s s ka = rate constant for oxygen adsorption, (kmol-m 3)1/2/kgcat-s k 12 = rate constant for propylene reaction to acrolein, m 3/kgcat-s k 13 co 2 = rate constant for propylene reaction to carbon dioxides, m 3/kgcat-s k 13 co = rate constant for propylene reaction to carbon monoxide, m 3/kgcat-s k 14 = rate constant for propylene reaction acetaldehyde, m 3/kgcat-s Co = concentration of oxygen, kmol/m 3 Cp = concentration of propylene, kmol/m 3 n 12 = number of moles of oxygen which react with one mole of propylene to produce acrolein, kmol/kmol n 13 co 2 = number of moles oxygen which react with one mole of propylene to product carbon dioxide, kmol/kmol n 13 co = number of moles of oxygen which react with one mole of propylene to produce carbon monoxide, kmol/kmol n 14 = number of moles of oxygen which react with one mole of propylene to produce acetaldehyde, kmol/kmol
Process Background Continued • Reaction rates for the formation of acrolein and byproducts (6, 8) Where: r 2 = rate of formation of acrolein, kmol/kgcat-s r 3 co 2 = rate of formation of carbon dioxide, kmol/kgcat-s r 3 co = rate of formation of carbon monoxide, kmol/kgcat-s r 4 = rate of formation of acetaldehyde, kmol/kgcat-s s ka = rate constant for oxygen adsorption, (kmol-m 3)1/2/kgcat-s k 12 = rate constant for propylene reaction to acrolein, m 3/kgcat-s k 13 co 2 = rate constant for propylene reaction to carbon dioxides, m 3/kgcat-s k 13 co = rate constant for propylene reaction to carbon monoxide, m 3/kgcat-s k 14 = rate constant for propylene reaction acetaldehyde, m 3/kgcat-s Co = concentration of oxygen, kmol/m 3 Cp = concentration of propylene, kmol/m 3 n 12 = number of moles of oxygen which react with one mole of propylene to produce acrolein, kmol/kmol n 13 co 2 = number of moles oxygen which react with one mole of propylene to product carbon dioxide, kmol/kmol n 13 co = number of moles of oxygen which react with one mole of propylene to produce carbon monoxide, kmol/kmol n 14 = number of moles of oxygen which react with one mole of propylene to produce acetaldehyde, kmol/kmol
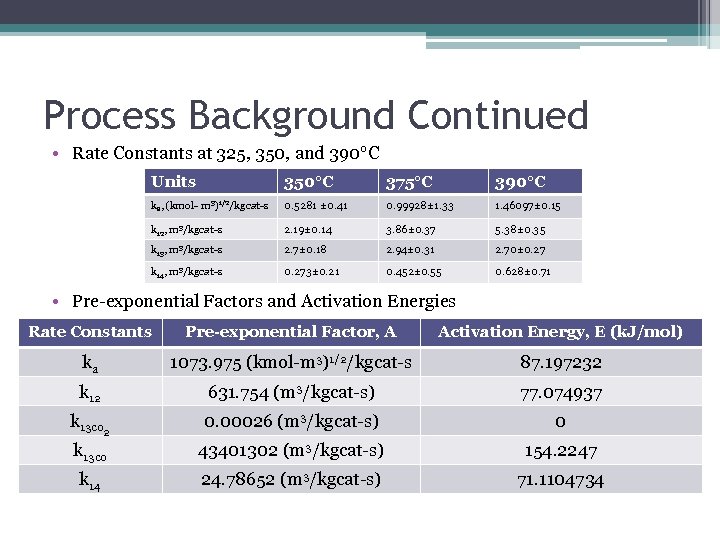 Process Background Continued • Rate Constants at 325, 350, and 390°C Units 350°C 375°C 390°C ka, (kmol- m 3)1/2/kgcat-s 0. 5281 ± 0. 41 0. 99928± 1. 33 1. 46097± 0. 15 k 12, m 3/kgcat-s 2. 19± 0. 14 3. 86± 0. 37 5. 38± 0. 35 k 13, m 3/kgcat-s 2. 7± 0. 18 2. 94± 0. 31 2. 70± 0. 27 k 14, m 3/kgcat-s 0. 273± 0. 21 0. 452± 0. 55 0. 628± 0. 71 • Pre-exponential Factors and Activation Energies Rate Constants Pre-exponential Factor, A Activation Energy, E (k. J/mol) ka 1073. 975 (kmol-m 3)1/2/kgcat-s 87. 197232 k 12 631. 754 (m 3/kgcat-s) 77. 074937 k 13 co 2 0. 00026 (m 3/kgcat-s) 0 k 13 co 43401302 (m 3/kgcat-s) 154. 2247 k 14 24. 78652 (m 3/kgcat-s) 71. 1104734
Process Background Continued • Rate Constants at 325, 350, and 390°C Units 350°C 375°C 390°C ka, (kmol- m 3)1/2/kgcat-s 0. 5281 ± 0. 41 0. 99928± 1. 33 1. 46097± 0. 15 k 12, m 3/kgcat-s 2. 19± 0. 14 3. 86± 0. 37 5. 38± 0. 35 k 13, m 3/kgcat-s 2. 7± 0. 18 2. 94± 0. 31 2. 70± 0. 27 k 14, m 3/kgcat-s 0. 273± 0. 21 0. 452± 0. 55 0. 628± 0. 71 • Pre-exponential Factors and Activation Energies Rate Constants Pre-exponential Factor, A Activation Energy, E (k. J/mol) ka 1073. 975 (kmol-m 3)1/2/kgcat-s 87. 197232 k 12 631. 754 (m 3/kgcat-s) 77. 074937 k 13 co 2 0. 00026 (m 3/kgcat-s) 0 k 13 co 43401302 (m 3/kgcat-s) 154. 2247 k 14 24. 78652 (m 3/kgcat-s) 71. 1104734
 Design Process Reactor 1 Reactor 2 Reactor 3 Reactor 4 Volume Pressure Drop Mult. Reactions Heat Effects Volume (m 3) 21696. 1 4174. 6 22. 51 19. 19 Num. Tubes (1” Dia. ) N/A 683600 17920 16880 Reactor Dia. (m) 13. 6946 21 3. 4 3. 3 Reactor Len. (m) 147. 298 12. 05 2. 4792 2. 24 Cat. Weight (kg-cat) 3. 07 x 107 5. 91 x 106 31850 27150 Particle Size (mm) N/A 3 5 5 Nitrogen Feed (kmol/s) 0. 382188797 0. 3821888 0. 4491963 0. 439638 Oxygen Feed (kmol/s) 0. 088895179 0. 08889518 0. 0979275 0. 095847 Propylene Feed (kmol/s) 0. 009342022 0. 000934202 0. 0117625 0. 011512 Inlet Temp. (°C) 350 390 Inlet Pressure (atm) 1 3 3 3 Pressure Drop (%) N/A 0. 37 7. 97 7. 82 Acrolein Prod. (kmol/s) 0. 007953 0. 0079428 0. 0079426 0. 0079369 Propylene Conversion (%) 85. 13 85. 02 84. 99 85. 01
Design Process Reactor 1 Reactor 2 Reactor 3 Reactor 4 Volume Pressure Drop Mult. Reactions Heat Effects Volume (m 3) 21696. 1 4174. 6 22. 51 19. 19 Num. Tubes (1” Dia. ) N/A 683600 17920 16880 Reactor Dia. (m) 13. 6946 21 3. 4 3. 3 Reactor Len. (m) 147. 298 12. 05 2. 4792 2. 24 Cat. Weight (kg-cat) 3. 07 x 107 5. 91 x 106 31850 27150 Particle Size (mm) N/A 3 5 5 Nitrogen Feed (kmol/s) 0. 382188797 0. 3821888 0. 4491963 0. 439638 Oxygen Feed (kmol/s) 0. 088895179 0. 08889518 0. 0979275 0. 095847 Propylene Feed (kmol/s) 0. 009342022 0. 000934202 0. 0117625 0. 011512 Inlet Temp. (°C) 350 390 Inlet Pressure (atm) 1 3 3 3 Pressure Drop (%) N/A 0. 37 7. 97 7. 82 Acrolein Prod. (kmol/s) 0. 007953 0. 0079428 0. 0079426 0. 0079369 Propylene Conversion (%) 85. 13 85. 02 84. 99 85. 01
 Optimization • Acrolein Selectivity § Greater at increased temperatures § Improved when coolant and inlet temperatures are equal § Higher pressure, higher selectivity § Other Usable Product Selectivity § Decreased at increased temperatures § Favored at lower pressures § Greater when coolant temperature less than the inlet temperature
Optimization • Acrolein Selectivity § Greater at increased temperatures § Improved when coolant and inlet temperatures are equal § Higher pressure, higher selectivity § Other Usable Product Selectivity § Decreased at increased temperatures § Favored at lower pressures § Greater when coolant temperature less than the inlet temperature
 Optimization Continued • Gain ▫ Greater at increased inlet temperature ▫ Independent of coolant and inlet temperature relationship • Optimization Conclusion: ▫ Focus on selectivity opposed to gain
Optimization Continued • Gain ▫ Greater at increased inlet temperature ▫ Independent of coolant and inlet temperature relationship • Optimization Conclusion: ▫ Focus on selectivity opposed to gain
 Final Design • Operating Conditions ▫ Temperature- 390°C ▫ Pressure- 3 atm • Reactor Configurations ▫ ▫ Volume- 19. 08 m 3 Diameter- 3. 4 m Length- 2. 01 m Number of Tubes- 17920 (1” Dia. )
Final Design • Operating Conditions ▫ Temperature- 390°C ▫ Pressure- 3 atm • Reactor Configurations ▫ ▫ Volume- 19. 08 m 3 Diameter- 3. 4 m Length- 2. 01 m Number of Tubes- 17920 (1” Dia. )
 Final Design Continued Inlet Flows Polymath Aspen Plus ® (kmol/s) Outlet (kmol/s) Nitrogen 0. 439638 Oxygen 0. 095847 0. 0832387 0. 0821155 Propylene 0. 011512 0. 0017208 0. 00170713 Acrolein 0 0. 0079412 0. 00795529 Acetyldehyde 0 0. 0009053 0. 000906563 Carbon Monoxide 0 0. 0005578 0. 000561055 Carbon Dioxide 0 0. 0031814 0. 00317457 Water 0 0. 0116804 0. 0116909 Total 0. 546997 0. 5488637 0. 547749008 Pressure (Pa) 303975 284200 284080 Temperature (K) 663 665. 5059 665. 644
Final Design Continued Inlet Flows Polymath Aspen Plus ® (kmol/s) Outlet (kmol/s) Nitrogen 0. 439638 Oxygen 0. 095847 0. 0832387 0. 0821155 Propylene 0. 011512 0. 0017208 0. 00170713 Acrolein 0 0. 0079412 0. 00795529 Acetyldehyde 0 0. 0009053 0. 000906563 Carbon Monoxide 0 0. 0005578 0. 000561055 Carbon Dioxide 0 0. 0031814 0. 00317457 Water 0 0. 0116804 0. 0116909 Total 0. 546997 0. 5488637 0. 547749008 Pressure (Pa) 303975 284200 284080 Temperature (K) 663 665. 5059 665. 644
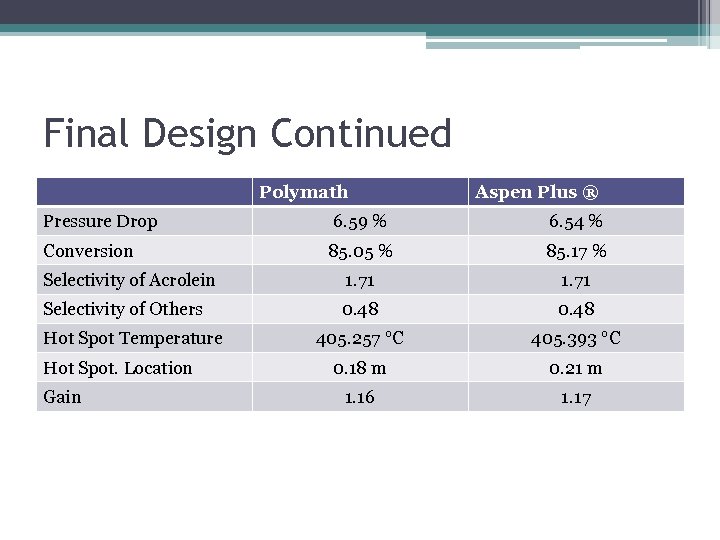 Final Design Continued Polymath Pressure Drop Aspen Plus ® 6. 59 % 6. 54 % 85. 05 % 85. 17 % Selectivity of Acrolein 1. 71 Selectivity of Others 0. 48 405. 257 °C 405. 393 °C 0. 18 m 0. 21 m 1. 16 1. 17 Conversion Hot Spot Temperature Hot Spot. Location Gain
Final Design Continued Polymath Pressure Drop Aspen Plus ® 6. 59 % 6. 54 % 85. 05 % 85. 17 % Selectivity of Acrolein 1. 71 Selectivity of Others 0. 48 405. 257 °C 405. 393 °C 0. 18 m 0. 21 m 1. 16 1. 17 Conversion Hot Spot Temperature Hot Spot. Location Gain
 Final Design Continued • Temperature Profile
Final Design Continued • Temperature Profile
 Conclusions • Reactor volume decreased with complexity increase • Selectivity crucial to optimization • Final model discussed would operate viably • Changed reactor dimensions to optimize final design
Conclusions • Reactor volume decreased with complexity increase • Selectivity crucial to optimization • Final model discussed would operate viably • Changed reactor dimensions to optimize final design
 Questions?
Questions?
 Works Cited 1. 2. 3. 4. 5. 6. 7. 8. Maganlal, Rashmikant, et al. Vapor phase oxidation of propylene to acrolein. 6437193 United States, August 20, 2002. Chemical Database Property Constants. DIPPR Database [Online]. Available from Rowan Hall 3 rd Floor Computer Lab. (Accessed on 1/24/2008). La. Marca, Concetta, Ph. D. Chemical Reaction Engineering Design Project. February 2008. Chemical Engineering Department, Rowan University, Glassboro. Transient Kinetics from the TAP Reactor System: Application to the Oxidation of Propylene to Acrolein. Creten, Glenn, Lafyatis, David S. , and Froment, Gilbert F. Belgium: Journal of Catalysis, 1994, Vol. 154. Chemical Database Property Constants. DIPPR Database [Online]. Available from Rowan Hall 3 rd Floor Computer Lab. (Accessed on 1/24/2008). The reaction network for the oxidation of propylene over a bismuth molybdate catalyst. Tan, H. S. , Downie, J. and Bacon, D. W. Kingston : The Canadian Journal of Chemical Engineering, 1989, Vol. 67 Physical Properties Data Compilations Relevant to Energy Storage. II. Molten Salts: Data on Single and Multi-Component Salt Systems. G. J. Janz, C. B. Allen, N. P. Bansal, R. M. Murphy, and R. P. T. Tomkins Molten Salts Data Center, Rensselaer Polytechnic Institute, NSRDS-NBS 61 -II, April 1979 The kinetics of the oxidation of propylene over a bismuth molybdate catalyst. Tan, H. S. , Downie, J. and Bacon, D. W. Kingston : The Canadian Journal of Chemical Engineering, 1988, Vol. 66
Works Cited 1. 2. 3. 4. 5. 6. 7. 8. Maganlal, Rashmikant, et al. Vapor phase oxidation of propylene to acrolein. 6437193 United States, August 20, 2002. Chemical Database Property Constants. DIPPR Database [Online]. Available from Rowan Hall 3 rd Floor Computer Lab. (Accessed on 1/24/2008). La. Marca, Concetta, Ph. D. Chemical Reaction Engineering Design Project. February 2008. Chemical Engineering Department, Rowan University, Glassboro. Transient Kinetics from the TAP Reactor System: Application to the Oxidation of Propylene to Acrolein. Creten, Glenn, Lafyatis, David S. , and Froment, Gilbert F. Belgium: Journal of Catalysis, 1994, Vol. 154. Chemical Database Property Constants. DIPPR Database [Online]. Available from Rowan Hall 3 rd Floor Computer Lab. (Accessed on 1/24/2008). The reaction network for the oxidation of propylene over a bismuth molybdate catalyst. Tan, H. S. , Downie, J. and Bacon, D. W. Kingston : The Canadian Journal of Chemical Engineering, 1989, Vol. 67 Physical Properties Data Compilations Relevant to Energy Storage. II. Molten Salts: Data on Single and Multi-Component Salt Systems. G. J. Janz, C. B. Allen, N. P. Bansal, R. M. Murphy, and R. P. T. Tomkins Molten Salts Data Center, Rensselaer Polytechnic Institute, NSRDS-NBS 61 -II, April 1979 The kinetics of the oxidation of propylene over a bismuth molybdate catalyst. Tan, H. S. , Downie, J. and Bacon, D. W. Kingston : The Canadian Journal of Chemical Engineering, 1988, Vol. 66


