cb4bdd808791e864396122dd200f7bd4.ppt
- Количество слайдов: 50

PARGLUVA® (Muraglitazar) Bristol-Myers Squibb and Merck & Co. , Inc. New Drug Application 21 -865 Clinical Safety Julie Golden, M. D. Medical Officer Division of Metabolic and Endocrine Drug Products September 9, 2005 Silver Spring, MD Center for Drug Evaluation and Research

Outline • • • Background Subject Disposition Safety Issues Concerns/Points to Consider Questions for Discussion Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 2

Regulatory Issues • First PPAR-γ (troglitazone) removed from the market in 2000 due to cases of idiosyncratic liver failure • PPARs and carcinogenicity: all clinical studies > 6 mo currently on clinical hold unless preclinical carcinogenicity studies submitted to the Agency Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 3

Background • Safety profile of muraglitazar is generally consistent with PPAR-γ and PPAR-α pharmacology • Potential concerns with PPAR compounds – PPAR-γ: edema, weight gain, congestive heart failure, anemia, neutropenia – PPAR-α: myopathy, cholelithiasis Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 4

Clinical Safety Database • 22 Clinical Pharmacology Studies • 6 Phase 2 b and 3 Studies – 5 Type 2 Diabetes Studies • 2 Monotherapy • 3 Combination Therapy – 1 Mixed Dyslipidemia Study Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 5

Clinical Safety Database • 3226 subjects received at least one dose of muraglitazar in the Phase 2/3 studies – Type 2 diabetes: 2969 – Monotherapy: 1560 – Combination therapy: 1409 • ~ 2000 subjects received muraglitazar for at least 24 weeks • ~ 700 subjects received muraglitazar for at least 104 weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 6

Type 2 Diabetes Studies, Treatment Groups Study CV 168006 Mur 0. 5 mg Mur 1. 5 mg Studies CV 168018, CV 168021, CV 168022 Mur 2. 5 mg Mur 5 mg Placebo Mur 5 mg Mur 10 mg Study CV 168025 Mur 20 mg Pio 15 mg Mur 5 mg Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 Pio 30 mg 7

Pooling • Muraglitazar ≤ 5 mg: – CV 168006: Mur 0. 5 mg, 1. 5 mg, 5 mg (titration possible) – Phase 3 Studies: Mur 2. 5 and 5 mg • Pioglitazone ≤ 45 mg: – CV 168006: Pio 15 mg (titration to 45 mg possible) – CV 168025: Pio 30 mg Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 8

Pooling • Monotherapy: CV 168006 and CV 168018 • Combination therapy – CV 168021: + glyburide – CV 168022 and CV 168025: + metformin Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 9
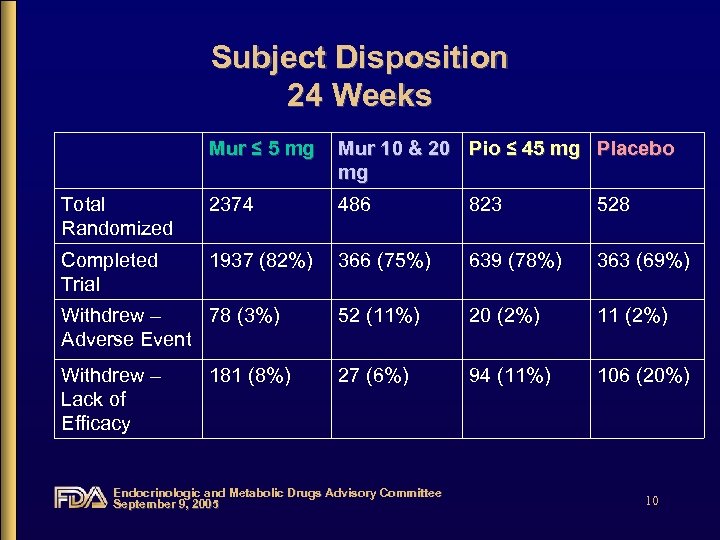
Subject Disposition 24 Weeks Mur ≤ 5 mg Mur 10 & 20 Pio ≤ 45 mg Placebo mg Total Randomized 2374 486 823 528 Completed Trial 1937 (82%) 366 (75%) 639 (78%) 363 (69%) Withdrew – 78 (3%) Adverse Event 52 (11%) 20 (2%) 11 (2%) Withdrew – Lack of Efficacy 27 (6%) 94 (11%) 106 (20%) 181 (8%) Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 10
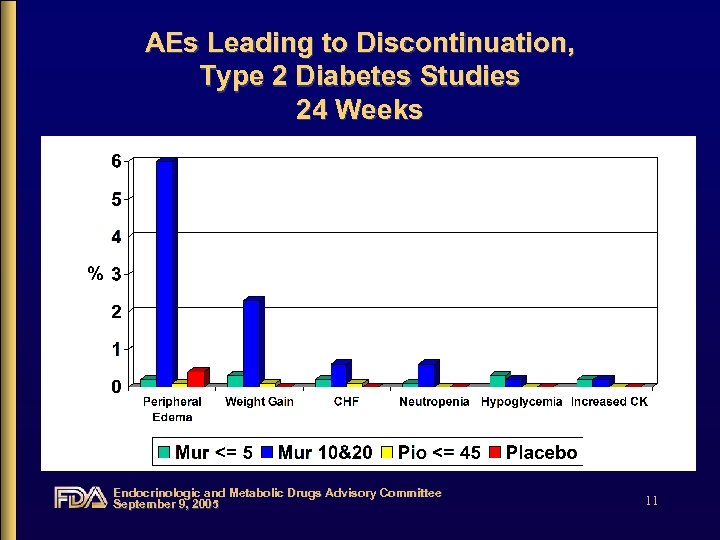
AEs Leading to Discontinuation, Type 2 Diabetes Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 11

Safety Issues • Deaths • Edema Events • Heart Failure Events • Weight Gain • Cardiovascular Events Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 12

Deaths Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 13
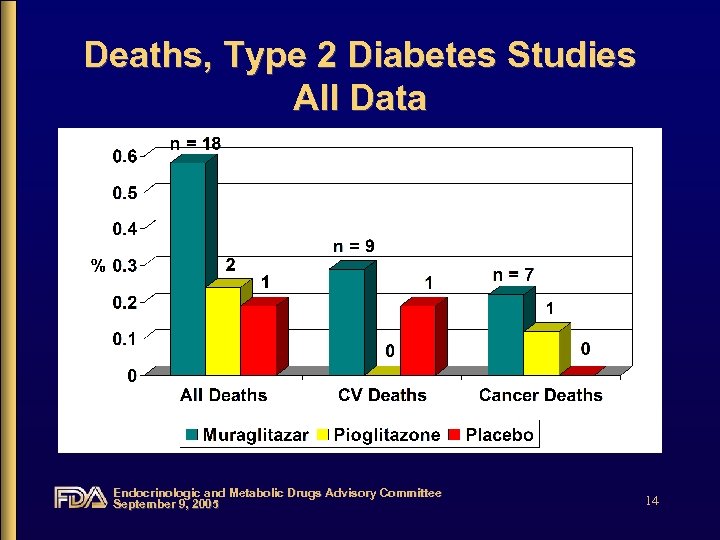
Deaths, Type 2 Diabetes Studies All Data Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 14

Cardiovascular Deaths • • 52 yo male (Mur 2. 5 + Met): myocardial infarction 54 yo male (Mur 5 + Met): myocardial infarction* 67 yo male (Mur 5 + Met): sudden death 53 yo male (Mur 5 + Met): stroke 56 yo female (Mur 5 + Gly): myocardial infarction 66 yo female (Mur 5 + Met): sudden death* 61 yo male (Mur 5 + Met): “found dead” 60 yo female (Mur 5 + Met): stroke • 59 yo male (Mur 20): myocardial infarction* • 62 yo male (Placebo + Gly): pulmonary embolism Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 15

Subject CV 168022 -153 -2 Muraglitazar 5 mg + metformin • 54 year-old white male with 6 -year history of type 2 diabetes, obesity, hypertension, coronary artery disease, and coronary thrombosis • Day 115: CHF symptoms – 1 day hospitalization, single dose of furosemide 40 mg I. V. • Day 117: normal chest x-ray • Day 118: – body weight increase of 10 lbs. from Day 90 – elevated NT-pro. BNP (1236 pg/m. L; screening 548 pg/m. L) – physical exam and outpatient cardiac exam refused • Day 125: found dead in home – death certificate: myocardial infarction and occult coronary artery disease Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 16

Subject CV 168025 -193 -9 Muraglitazar 5 mg + metformin • 66 year-old white female with 4 -year history of type 2 diabetes, CHF, hypertension, peripheral vascular disease, transient ischemic attack, bilateral lower extremity fluid retention, atrial flutter, mitral insufficiency, obesity, and tobacco use • Day 201: dyspnea; reported as CHF • Day 202: sudden death – autopsy not performed; cause of death reported as myocardial infarction Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 17

Subject CV 168006 -11 -8 Muraglitazar 20 mg • 59 year old white male with 2 -year history of type 2 diabetes, previous tobacco use, hypertension, iron overload with fatty liver, and psoriasis • Day 43: bilateral pitting edema in ankles • Day 49: admitted with myocardial infarction and CHF – increasing exertional dyspnea for the previous month – markedly elevated cardiac enzyme levels – clinical deterioration; intubation • Day 50: – cardiac catheterization: 99% left main arterial distal stenosis – echocardiogram: moderately dilated left ventricle with severe global hypokinesis; ejection fraction 15 - 20% • Day 60: life support withdrawn; patient died Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 18

Cancer Deaths • • 69 yo male (Mur 1. 5): lung cancer 69 yo female (Mur 1. 5): acute myeloid leukemia 49 yo male (Mur 2. 5 + Met): hepatocellular carcinoma 56 yo female (Mur 5 + Met): breast cancer 70 yo female (Mur 5): lung cancer 63 yo female (Mur 5 + Met): lung cancer 50 yo female (Mur 5 + Met): pancreatic cancer • 66 yo male (Pio 15): throat cancer Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 19

Deaths Study CV 168025 Muraglitazar 5 mg + Metformin Pioglitazone 30 mg + Metformin N 587 572 Total Deaths 6 (1. 02%) 1 (0. 17%) Cardiovascular Deaths 5 (0. 85%) 0 (0. 0%) 5 cardiovascular deaths from 3 sites (out of 234): Site 193 = Netherlands, 2 deaths, 8 randomized subjects Site 241 = Russia, 2 deaths, 38 randomized subjects Site 314 = Finland, 1 death, 2 randomized subjects Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 20

Edema Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 21

Edema-Related Predefined Preferred Terms • • • Fluid retention/overload Generalized edema Peripheral edema Swelling Hypervolemia Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 22
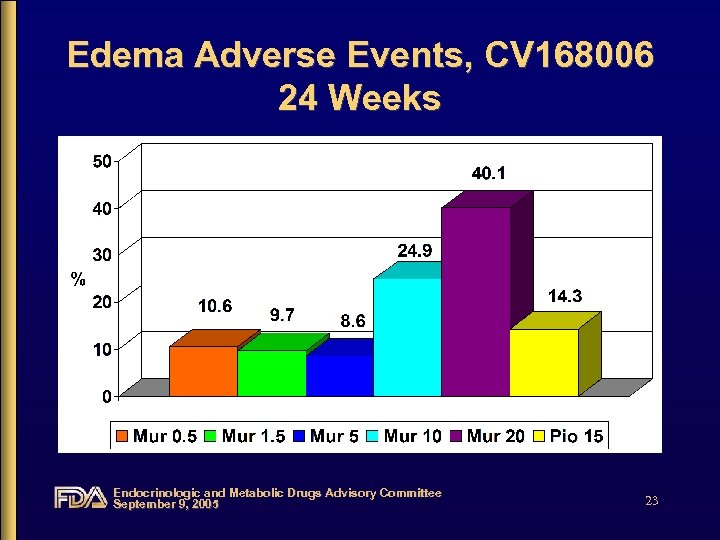
Edema Adverse Events, CV 168006 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 23
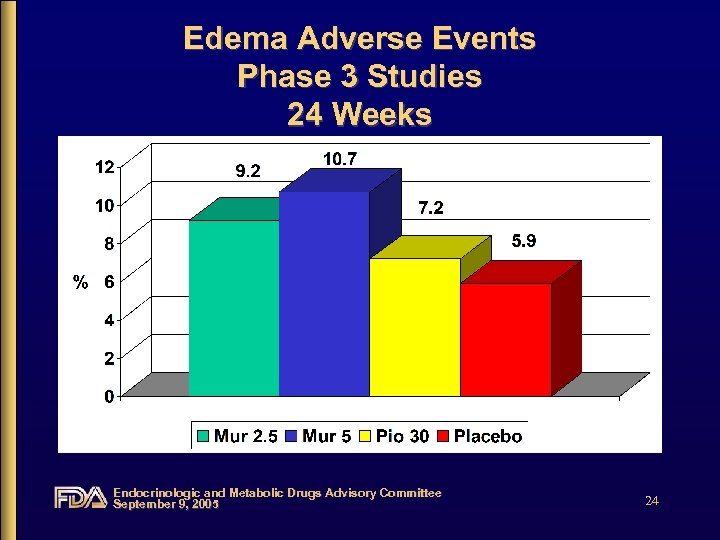
Edema Adverse Events Phase 3 Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 24
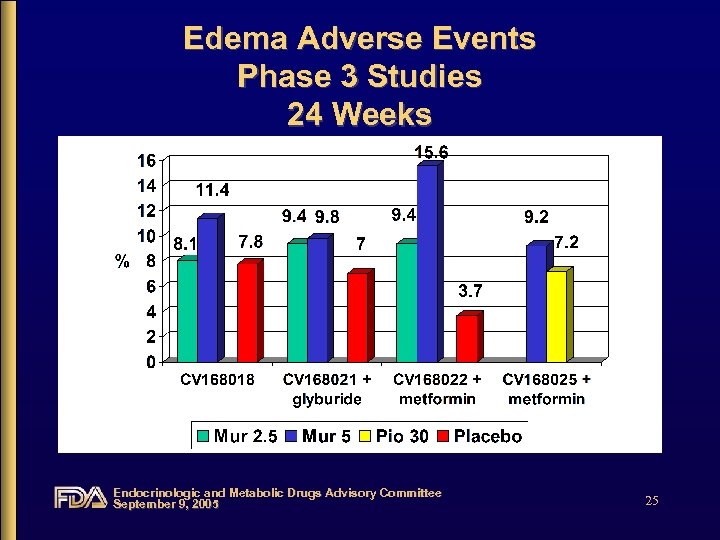
Edema Adverse Events Phase 3 Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 25

Congestive Heart Failure Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 26
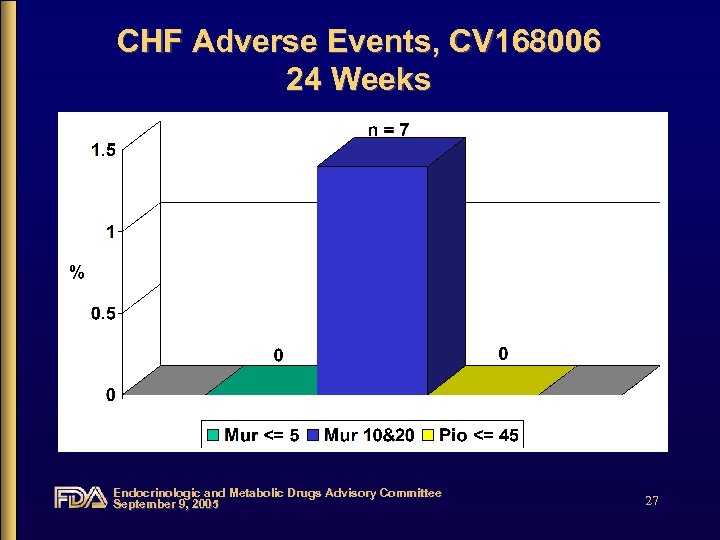
CHF Adverse Events, CV 168006 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 27
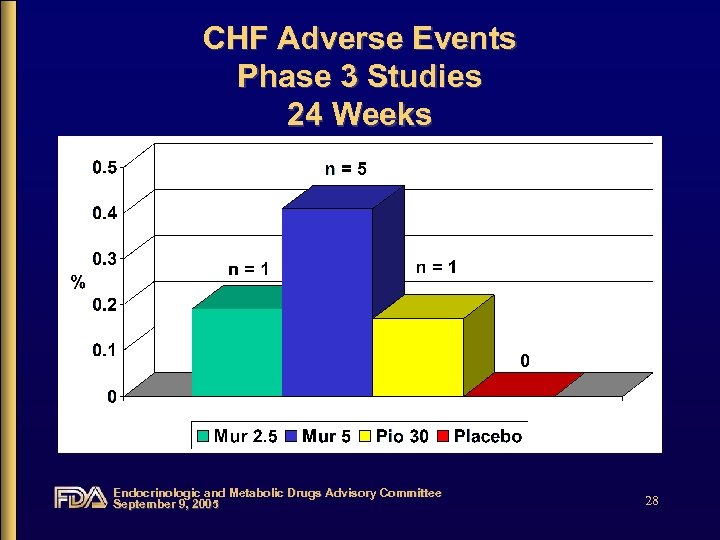
CHF Adverse Events Phase 3 Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 28
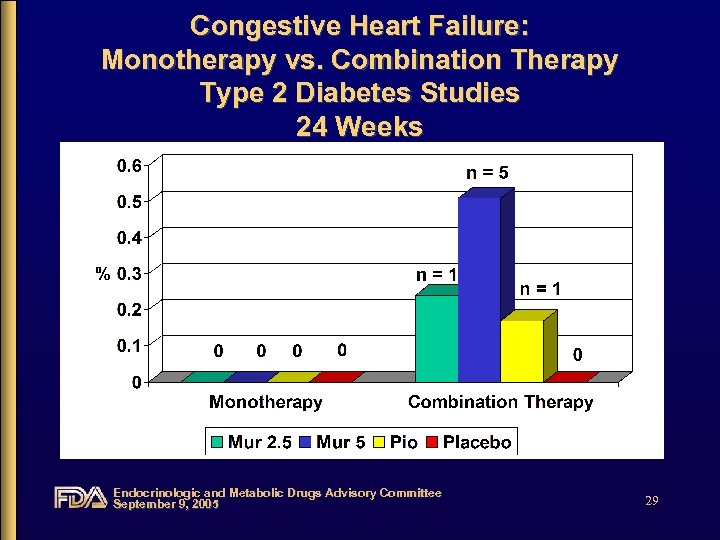
Congestive Heart Failure: Monotherapy vs. Combination Therapy Type 2 Diabetes Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 29

CHF Adjudication Committee: Predefined Preferred Terms • • • Cardiac failure Pulmonary edema Ventricular failure Dyspnea Edema (moderate or greater intensity) Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 30
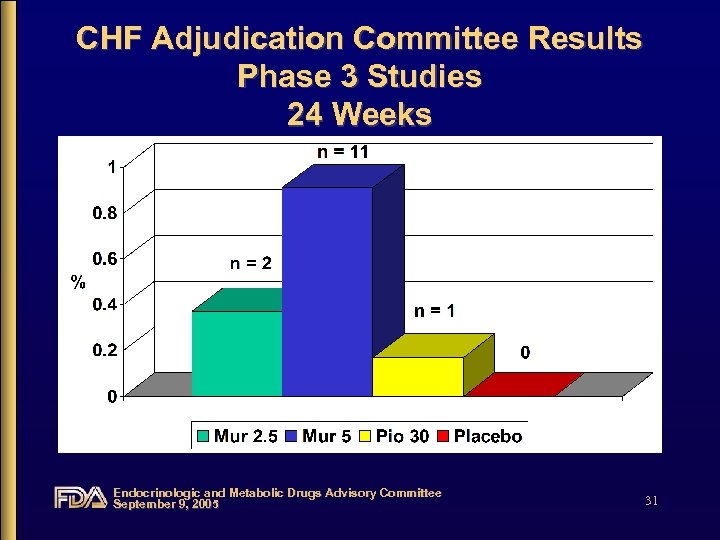
CHF Adjudication Committee Results Phase 3 Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 31

Weight Gain Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 32
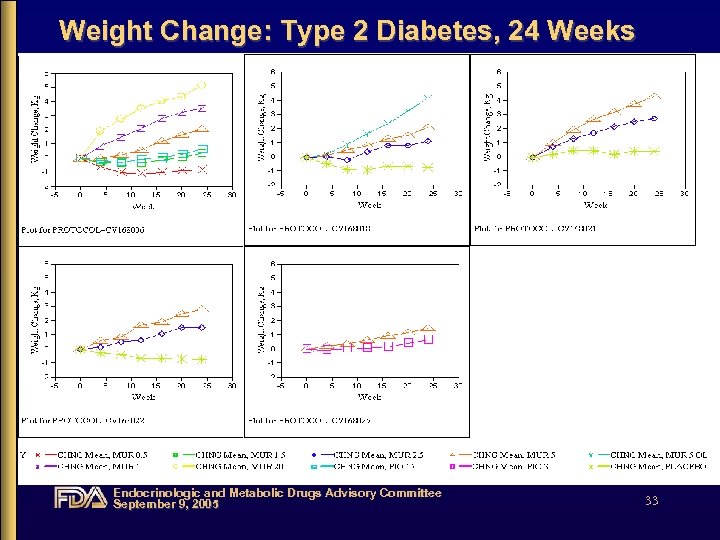
Weight Change: Type 2 Diabetes, 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 33

Cardiovascular Events Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 34

Cardiovascular Adverse Events: Predefined Preferred Terms • • Myocardial infarction Coronary revascularization Coronary artery disease Angina/myocardial ischemia Cardiac death Stroke Transient ischemic attack * NOT congestive heart failure Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 35
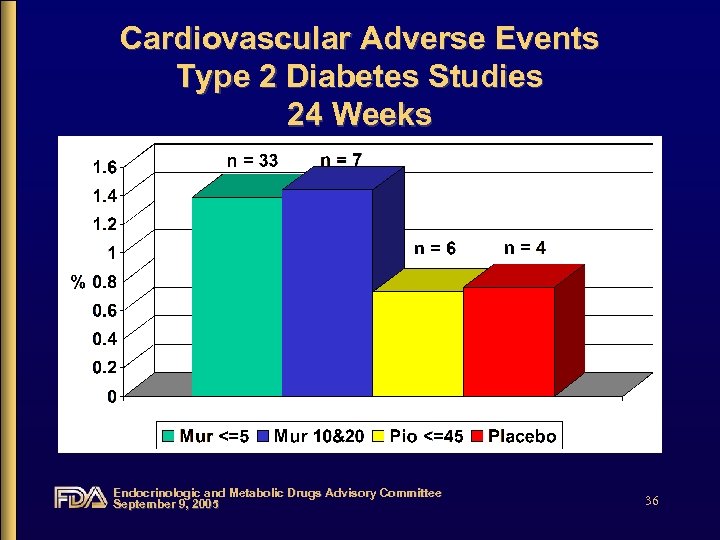
Cardiovascular Adverse Events Type 2 Diabetes Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 36
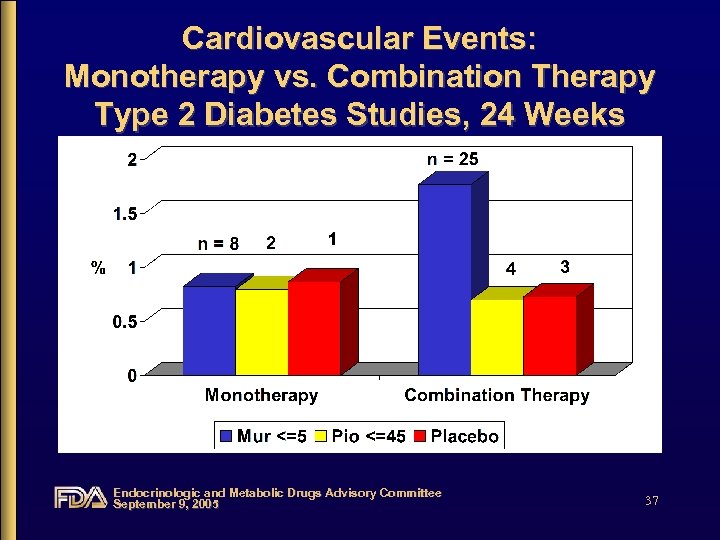
Cardiovascular Events: Monotherapy vs. Combination Therapy Type 2 Diabetes Studies, 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 37
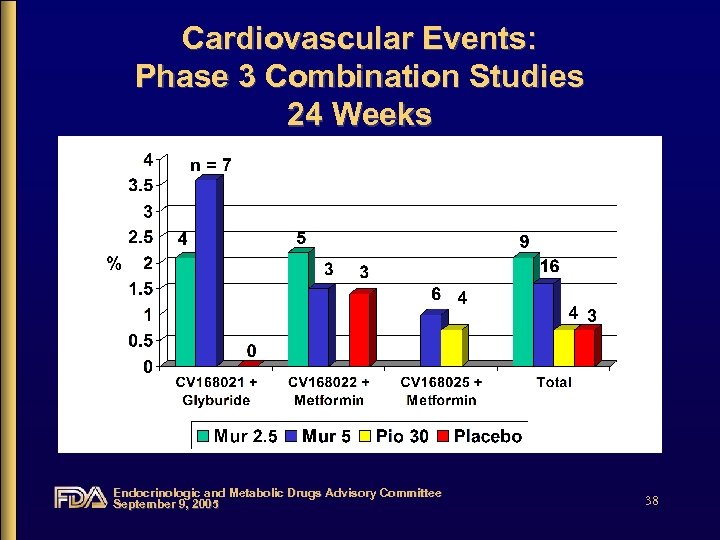
Cardiovascular Events: Phase 3 Combination Studies 24 Weeks Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 38

Cardiovascular Events: Study CV 168021 24 Weeks Muraglitazar 2. 5 mg: Muraglitazar 5 mg: • 64 yo M: Non-Q wave MI, day 85 • 68 yo M: MI, day 1 (prior to first dose) • 52 yo F: TIA, day 31 • 54 yo F: Stroke, d 26; Brain tumor, day 43 • 64 yo M: Unstable angina, day 11 • 62 yo M: Coronary artery disease, day 111 • 59 yo F: Stroke, day 9 • 70 yo M: Angina, days 17, 37, 56, and 66 • 52 yo F: TIA, day 78 • 46 yo M: Myocardial ischemia, day 85 • 60 yo M: Stroke, day 139 Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 39

Inclusion/Exclusion Criteria Baseline Characteristics Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 40

Inclusion/Exclusion Criteria • No history of MI, coronary angioplasty or bypass graft(s), valvular disease or repair, unstable angina pectoris, TIA or cerebrovascular accidents within 6 months prior to entry into the study • Monotherapy: No antihyperglycemic therapy more than 3 consecutive or a total of 7 nonconsecutive days 4 -6 weeks prior to Screening • Combination Therapy: Receiving treatment with sulfonylurea (CV 168021) or metformin (CV 168022, CV 168025) for at least 6 weeks prior to Screening Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 41
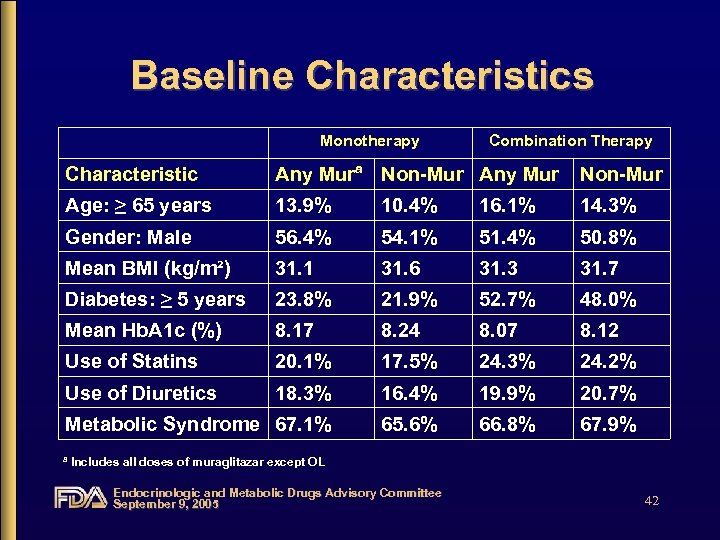
Baseline Characteristics Monotherapy Combination Therapy Characteristic Any Murª Non-Mur Any Mur Non-Mur Age: ≥ 65 years 13. 9% 10. 4% 16. 1% 14. 3% Gender: Male 56. 4% 54. 1% 51. 4% 50. 8% Mean BMI (kg/m²) 31. 1 31. 6 31. 3 31. 7 Diabetes: ≥ 5 years 23. 8% 21. 9% 52. 7% 48. 0% Mean Hb. A 1 c (%) 8. 17 8. 24 8. 07 8. 12 Use of Statins 20. 1% 17. 5% 24. 3% 24. 2% Use of Diuretics 18. 3% 16. 4% 19. 9% 20. 7% Metabolic Syndrome 67. 1% 65. 6% 66. 8% 67. 9% ª Includes all doses of muraglitazar except OL Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 42
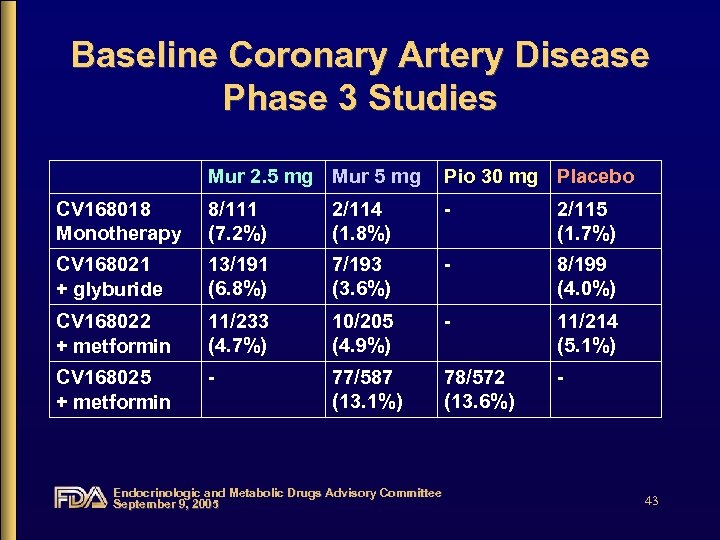
Baseline Coronary Artery Disease Phase 3 Studies Mur 2. 5 mg Mur 5 mg Pio 30 mg Placebo CV 168018 Monotherapy 8/111 (7. 2%) 2/114 (1. 8%) - 2/115 (1. 7%) CV 168021 + glyburide 13/191 (6. 8%) 7/193 (3. 6%) - 8/199 (4. 0%) CV 168022 + metformin 11/233 (4. 7%) 10/205 (4. 9%) - 11/214 (5. 1%) CV 168025 + metformin - 77/587 (13. 1%) 78/572 (13. 6%) - Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 43

Concerns/Points to Consider Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 44

Deaths • Most deaths in the muraglitazartreatment group were due to cardiovascular events and cancer • Congestive heart failure may have contributed to the cardiac event leading to death in 3 subjects on muraglitazar • No clear pattern has emerged Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 45

Deaths • Most cardiovascular deaths were from one study – CV 168025: metformin add-on, active control – 5 cardiovascular deaths from 3 study sites Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 46

Cardiovascular Events • Diverse events with no clear pattern • Increased incidence in combination studies • Imbalance driven by one study – CV 168021: glyburide add-on, placebocontrolled study Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 47

Cardiovascular Events • When combination studies pooled, events were not dose-related • Inconsistent rate of events in placebo groups from combination studies • Low number of events, particularly in comparator groups, make incidence rates unstable Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 48

Questions for Discussion • Is it likely that the excess of cardiovascular deaths and events in the muraglitazar group are related to the dose-related fluid retention? • If not, is there a plausible pharmacological explanation? • Are subjects on combination therapy (i. e. , longer history of diabetes) or with a relevant medical history more vulnerable to the adverse effects of muraglitazar? – Cardiovascular events – Fluid-related events Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 49

Acknowledgements • Medical Review Team – Robert Misbin, M. D. – Eric Colman, M. D. – David Orloff, M. D. • Pharmacology/Toxicology Review Team – John Colerangle, Ph. D. – Jeri El Hage, Ph. D. • Biopharmaceutics Review • Statistical Review Team – Lee Ping Pian, Ph. D. – Jaya Vaidyanathan, Ph. D. – J. Todd Sahlroot, Ph. D. – Hae Young Ahn, Ph. D. • Project Management – Jena Weber Endocrinologic and Metabolic Drugs Advisory Committee September 9, 2005 50
cb4bdd808791e864396122dd200f7bd4.ppt