bf6a393fb980e8173e6ad9524a8ebf34.ppt
- Количество слайдов: 91

Parasitology Course BIOL 2272 By Fred Opperdoes

Definition of parasitism • Parasitology describes the relationships between two organisms, i. e. the host organism and the parasite. When one organism gives shelter and food to another organism two totally different situations may exist: – Without any harm being done, or in the case where some hosts may benefit from the presence of a parasite, in which case we normally speak of: • mutualism: a situation where two organisms live in some contact and benefit from each others presence. They exchange food or provide shelter or protection, but may still be able to live an independent life. • symbiosis: a situation where two organisms live in close contact and benefit from each others presence. They have become dependent upon each others presence and are unable to survive independently. • endosymbiosis: bacteria have invaded the cytoplasm of a eukaryotic host cell. Host cell and bacterium have become dependent upon each other. In the case large amounts of genetic material have been moved from the bacterial genome to the nucleus of the host an irreversible situation is reached which led to the formation of: – chloroplasts, photosynthetic organelles that have originated from photosynthetic cyanobacteria – mitochondria, organelles involved in oxidative phosphorylation that have originated from the alpha subdivision of the proteobacteria.

Definition of parasitism (cont. ) – • With harm being done. • When one organism lives at the expense of its host we can distinct several situations: – – – * * * viral infections bacterial infections parasitic diseases NB: Normally a true parasite does not kill its host

Parasitic associations are very common in Nature • • There is no major taxonomic group that has no parasitism associated with it. Only viruses have no parasites ! Host organism Viruses Prokaryotes Eubacteria and Archaebacteria Eukaryotes Protozoa Fungi Metazoan organisms • Parasite bacteriophages/plasmids viruses and bacteria viruses and plasmids viruses and bacteria, protozoa and fungi and metazoan organisms In most cases, however, the associations presented in the above table are not regarded as true parasitism and are dealt with by such disciplines as virology, microbiology and mycology (the study of fungal infections). Only when metazoan organisms are infected by protozoans, or by helminths, or infested by organisms that live in or upon the host's skin (ectoparasites), this is regarded as a case of parasitism.

What is parasitology (1) • Parasitology is the study of parasitism and due to the nature of parasitism it can not be well defined. • Parasitology can be divided in : • The study of the parasite itself. It is important to know how the parasite functions in order to be able to combat it. This comprises all disciplines of biology and beyond. Host-Parasite relationships – This comprises all disciplines that deal with the host-parasite interface and the way the two organisms interact with each other. The most important disciplines are: – Physiology – Biochemistry – Cell biology – Immunology – Pharmacology •

What is parasitology (2) Immunology of parasitic infections. This deals with the description of the immunological response by the host and the way the parasite tries to evade the host's immune attack. This comprises such disciplines as: Humural and cellular immunology Molecular immunology Chemotherapy of parasitic infections. This deals with the study of the effect of drugs on the parasite and host and the treatment by drugs that would lead to the killing of the parasite and the recovery of the diseased host. The disciplines involved are: Organic chemistry Pharmacology Biochemistry Medicine Epidemiology tries to describe the ways the diseases spread amongst the population. In the case of parasitic infections not only the host, but also the parasite and the vector should be included. The disciplines are: Tropical hygiene Entomology Geography

Different types of parasites • Due to the great variety in parasitic organisms in Nature they can be divided in many different classes: Type of parasite Examples (Viruses) (Bacteria) Phytoparasites Zooparasites Ectoparasites Endoparasites Yeasts (Candida), Fungi Bugs, lice (punaises, poux) bloodvessels: Schistosoma intestin: Taenia tissues: Trichinella cells: Leishmania, Toxoplasma

Different types of parasites (2) • Parasites can be divided into different classes of parasite : – Obligatory parasites. – These parasites can only survive in a host and therefore go directly from one host to another. This may involve complex life cycles. – Examples are: • Trichomonas vaginalis • Taenia • Trichinella

Trichomonas vaginalis (trichomoniasis) • Light microscopic picture of Trichomonas vaginalis in culture • Trichomonas vaginalis is a sexually transmitted disease (STD), although transmission by other routes (such as soiled towels) has been documented. There is no cyst in the life cycle, so transmission is via the trophozoite stage only. Most people infected with trichomoniasis are asymptomatic. It is one of the most frequent sexually transmitted diseases in man. 30 to 50 % of all women in western countries may be infected. In males the infection normally does not lead to any symptoms, while in females this may lead to an increased secretion, characterized by a white discharge from the genital tract and itching. Diagnosis depends on finding trophozoites in secretions of the genital tract from men or women. In cases where the numbers of organisms are very low, the trophozoites can be cultured to increase their numbers.
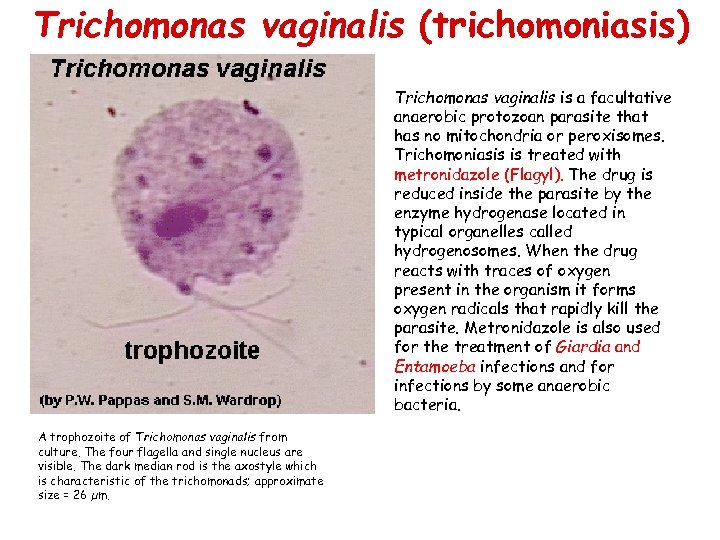
Trichomonas vaginalis (trichomoniasis) • A trophozoite of Trichomonas vaginalis from culture. The four flagella and single nucleus are visible. The dark median rod is the axostyle which is characteristic of the trichomonads; approximate size = 26 µm. Trichomonas vaginalis is a facultative anaerobic protozoan parasite that has no mitochondria or peroxisomes. Trichomoniasis is treated with metronidazole (Flagyl). The drug is reduced inside the parasite by the enzyme hydrogenase located in typical organelles called hydrogenosomes. When the drug reacts with traces of oxygen present in the organism it forms oxygen radicals that rapidly kill the parasite. Metronidazole is also used for the treatment of Giardia and Entamoeba infections and for infections by some anaerobic bacteria.

Different types of parasites (3) • Temporary parasites. – These parasites spend only part of their lives as a parasite and another part as free-living organism. – Examples are: • Fasciola hepatica [Liver fluke (douve)] • Schistosoma • Ascaris • Haemonchus

Different types of parasites (4) • Facultative parasites. – These organisms are normally free living and infect a host only by accident. – Examples: some free-living amoeba such as: • Naegleria • Acanthamoeba Trophozoites of Acanthamoeba castellani • Fungi such as Candida (vaginal candidosis, athlete foot)

Naegleria • Naegleria trophozoites in brain tissue The genus Naegleria includes several species of free-living uninucleate amoebae that live in aquatic environment (ponds, lakes, sewage). It belongs to a family of amoebae (Valkampfiidae) whose members can transform into flagellate stage, which is a transient, non-feeding non-dividing form; the amoebal stage can also transform into a resistant cyst stage. The species Naegleria fowleri is occasionally pathogenic to a variety of mammals including humans. Following direct intranasal contact, usually by swimming, N. fowleri invade the olfactory bulb and progress along the olfactory nerves to the brain, thereby causing an acute and fulminating disease called primary amoebic meningoencephalitis.

Acanthamoeba • Acanthamoeba trophozoites in culture after immunofluorescence staining Acanthamoeba spp. (Rhizopoda; hartmanellidae) is a free-living amoeba which is common in a variety of habitats, including fresh water, soil, and sewage. In humans they can cause a sub acute or chronic infection called granulomatous amoebic encephalitis. Acanthamoeba is frequently found in contact lenses solutions, thereby causing vision-threatening keratitis. Despite morphological similarities with Naegleria and other free-living amoebae, sequence comparisons arising from small and large ribosomal RNA subunits suggest Acantamoeba being as closely related to higher plants and vertebrates as they are to various protist genera.

Athlete foot or Tinea pedis • Athlete foot or tinea pedis is a common condition in occupational groups and communities. In boarding schools and amongst coal miners it may reach epidemic proportions. The commonest organisms are T. interdigitale and T. rubrum and the high rate of infection is associated with its increased opportunity to spread for instance in shower rooms, where the parasite dwells in the wooden 'duck' boards in the showers. Preventive measures consist in the removal of those wooden boards from the showers, regular foot inspection and the use of bathroom footwear. • Treatment consist in the application onto the infected skin of miconazole nitrate ointment (Daktarin, Janssen Pharmaceutica), the use of miconazole-nitrate foot powder in socks and shoes and the oral use of Sporanox (isoconazole nitrate). The mode of action of these imadazoles is based upon their capacity to inhibit the action of fungal cytochrome P 450 that is involved in the biosynthesis of ergosterol, an essential sterol of the fungal plasma membrane. •

Vaginal candidosis • The disease In recent years there has been a dramatic rise in the incidence of vaginal candidosis. Such Candida infections have been associated with a number of factors most notably diabetis mellitus and pregnancy. The contraceptive pill and an increased sexual activity have also been held responsible. Recurrent and even chronic infections in women are common. • Treatment consist in the use of oral fungicides or the application of miconazole nitrate ointment (Daktarin, Janssen Pharmaceutica) into the vagina (for mode of action see athlete foot).

Types of host • Like the parasites, the hosts can be divided into several classes as well. These are: – Definitive host (DH). A definitive host is an organism that hosts the adult (sexual) form of the parasite – Intermediate host (IH). An intermediate host is an organism that hosts the asexual form of the parasite (only when there is an obligatory passage through the host). Intermediate hosts can be divided into two groups: – Passive IH (molluscs in the case of Schistosoma) – Active IH (tsetse fly in the case of trypanosomes) • Also the vectors can be divided into two different types: – Biological vectors • Examples: haematophagous athropodes such as mosquitoes and other biting insects – Mechanical vectors • Examples: flies for transport of amoebal cysts
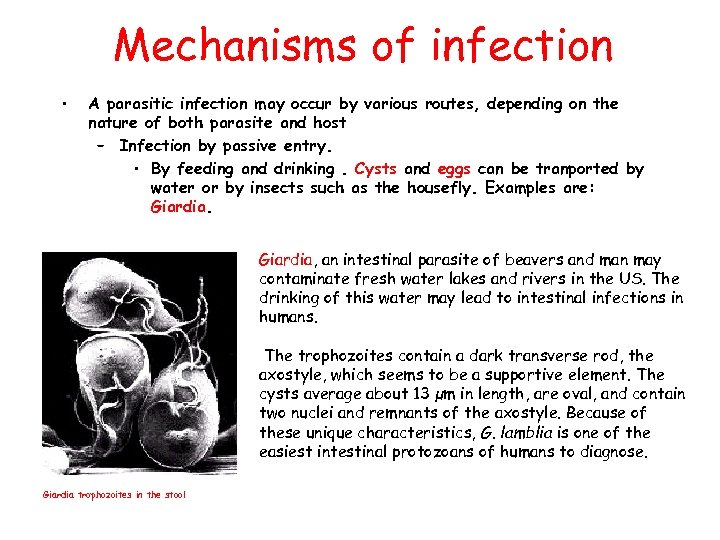
Mechanisms of infection • • A parasitic infection may occur by various routes, depending on the nature of both parasite and host – Infection by passive entry. • By feeding and drinking. Cysts and eggs can be tranported by water or by insects such as the housefly. Examples are: Giardia, an intestinal parasite of beavers and man may contaminate fresh water lakes and rivers in the US. The drinking of this water may lead to intestinal infections in humans. The trophozoites contain a dark transverse rod, the axostyle, which seems to be a supportive element. The cysts average about 13 µm in length, are oval, and contain two nuclei and remnants of the axostyle. Because of these unique characteristics, G. lamblia is one of the easiest intestinal protozoans of humans to diagnose. Giardia trophozoites in the stool

Giardia lamblia • G. lamblia trophozoites adhere closely to the lining of the small intestine, and in heavy infections much of the lining of the small intestine can be covered with trophozoites. The symptoms associated with giardiasis range from none (in light infections) to severe, chronic diarrhea (in heavy infections), but not dysentery. • One person can pass millions of G. lamblia cysts each day, and most infections probably result from water or food contaminated with human sewage. Open sewers in city streets and contamination of drinking water with this sewage undoubtedly results in many infections. However, in some countries the use of human fecal material ("night soil") as a fertilizer is also an important source of infection. Many cases of "traveler's diarrhea" are caused by Giardia. Even in developed countries potable water can be contaminated with small amounts of sewage, especially when septic systems are build too close to wells. G. lamblia is found throughout the world. • The parasite is also transmitted through anal sexual intercourse by homosexuals males.

Mechanisms of infection (2) – Entamoeba, an intestinal parasite of humans is transmitted in the form of cysts by the house fly. – – Entamoeba trophozoites in the stool

Entamoeba histolytica • Entamoeba histolytica is a facultative anaerobic protist that forms trophozoites as well as cysts. • The life cycle of Entamoeba histolytica involves trophozoites that live in the host's large intestine and cysts that are passed in the host's feces. Humans are infected by ingesting cysts, most often via food or water contaminated with human fecal material. The trophozoites can destroy the tissues that line the host's large intestine, so of the amoeba infecting the human gastrointestinal tract, E. histolytica is potentially the most pathogenic. In most infected humans the symptoms of "amoebiasis" are intermittent and mild (various gastrointestinal upsets, including colitis and diarrhea). In more severe cases the gastrointestinal tract hemorrhages, resulting in dysentery. In some cases the trophozoites will enter the circulatory system and infect other organs, most often the liver (hepatic amoebiasis), or they may penetrate the gastrointestnal tract resulting in acute peritonitis; such cases are often fatal. As with most of the amoeba, infections of E. histolytica are often diagnosed by demonstrating cysts or trophozoites in a stool sample.
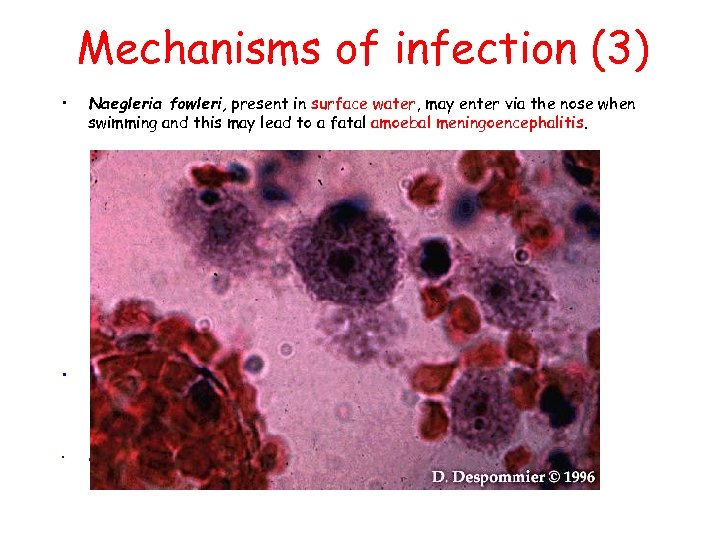
Mechanisms of infection (3) • Naegleria fowleri, present in surface water, may enter via the nose when swimming and this may lead to a fatal amoebal meningoencephalitis. • • Naegleria fowleri in brain tissue
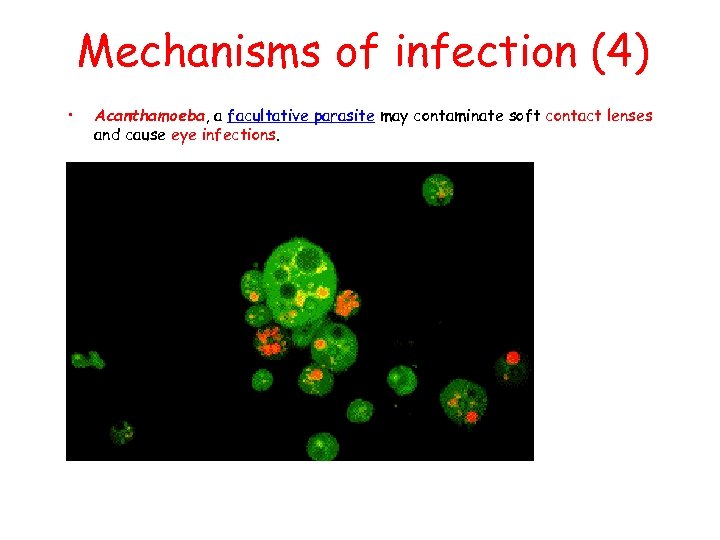
Mechanisms of infection (4) • Acanthamoeba, a facultative parasite may contaminate soft contact lenses and cause eye infections. • Acanthamoeba trophozoites in culture
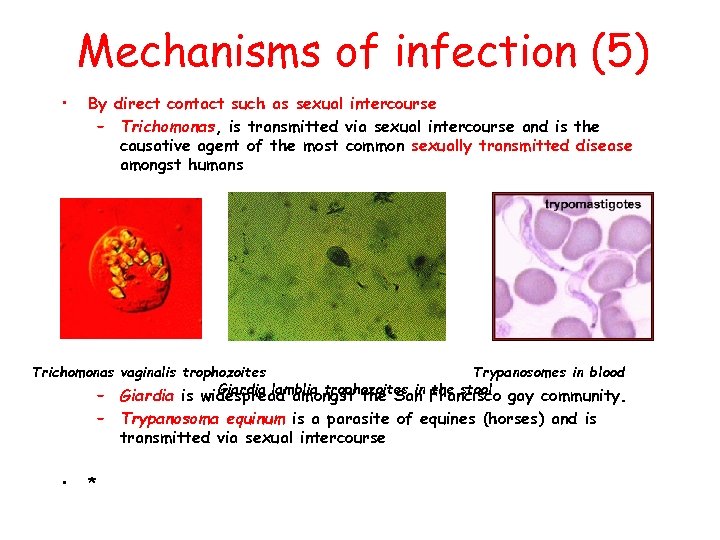
Mechanisms of infection (5) • By direct contact such as sexual intercourse – Trichomonas, is transmitted via sexual intercourse and is the causative agent of the most common sexually transmitted disease amongst humans Trichomonas vaginalis trophozoites Trypanosomes in blood Giardia lamblia trophozoites in the stool – Giardia is widespread amongst the San Francisco gay community. – Trypanosoma equinum is a parasite of equines (horses) and is transmitted via sexual intercourse • *

Mechanisms of infection (5) • Active entry – By biting (haematophagous) insects that serve as the active vectors in the transmission of disease: • mosquitoes transmit malaria and filariasis • black flies transmit onchocerciasis or river blindness • tsetse flies transmit sleeping sickness in Africa Anopheles mosquito • sandflies transmit leishmaniasis Tsetse fly sandfly – By highly specialized developmental forms in the life cycle of the parasite • Schistosoma cercaria actively penetrate human skin

Parasite reservoirs • A parasite reservoir (PR) is the biotope where the parasite lives. In general it is the Definitive Host, where lives the sexual stage of the parasite, producing large amounts of larvae or eggs, that serves as the PR. A PR can be: • Human reservoir – Human is the sole host (schistosomiasis) – Human is an accidental host (leishmaniasis) • Animal reservoir – 1. domestic animals – 2. wild animals

How do parasites survive inside an immunocompetent host ? (1) • • Ectoparasites – Infestation leads to local lesions of minor to moderate importance. This leads only to: allergic reactions (itching) and immunological reactions (without result) – None of these reactions really harm the parasite, but do harm the host Intracellular parasites – These parasites try to escape any immunological reactions mounted by the host by hiding themselves inside the host cells where the immune system cannot reach them. – Examples are: • Toxoplasma in lymphocytes, • Plasmodium in erythrocytes, • Leishmania, in macrophages, • T. cruzi in muscle cells.

How do parasites survive inside an immunocompetent host ? (2) • Extracellular parasites – Some parasites cover their cell surface with host serum proteins to avoid recognition by the immune system of the host – Examples are: • Schistosoma worms that cover themselves with host serum albumin • Rodent trypanosomes that cover themselves with ablastin (Ig. E) • Antigenic variation in the African trypanosomes that live freely in the bloodstream and body fluids of the host is another effective mechanism of evasion. • Cyst formation by Entamoeba and other amoeboid parasites

• • Harmful effects of the parasites on the host (1) Many parasites cause harmful effects to their host, but in most cases these effects are not of such importance that the host is being killed. Such effects comprise: – Wasting (cachexia, spoliatrices) African trypanosomiasis and leishmaniasis may lead to severe loss of weight in both animals and man. – Superinfections In the case of (muco)cutaneous leishmaniasis ulcerations may lead to superinfections with bacteria – Production of toxic compounds It is thought that the African trypanosome, when in the central nervous system, produces aromatic amino-acid analogues that may influence brain function. – Immunodepression Malaria, bilharziosis, etc. , lead to a certain degree of immune suppression which renders the infected host more susceptible to other diseases. – Allergic reactions In the case of onchocerciasis (river blindness) the presence of the filarial worms under the skin may lead to depigmentation due to allergic reactions.

Harmful effects of the parasites on the host (2) – Anaphylactic shock • may be induced by the sudden release of large amounts of parasite internal antigens into the bloodstream. • In malaria this occurs when the merozoites are released in waves from infected erythrocytes. • In African trypanosomiasis or sleeping sickness this occurs when the immune response leads to the massive killing and lysis of the circulating parasites. • Also drug treatment leading to a massive killing of the parasites may result in anaphylactic shock. – Mechanical damage • In the case of malaria the lysis of erythrocytes does lead to haemolysis and anaemia. • In the case of ascaris infection the presence of the worms in the small intestine may lead to intestinal occlusions – Irritative reflexes (intestinal contractions: ascaris) – Irritation of skin and tissues by ecto- and endoparasites

Prophylaxis (1) • Prophylaxis is regarded the prevention of disease by taking special measures. These measures may comprise a great variety of individual as well as collective measures • Prophylactic measures and their examples are: – – – Isolate the parasite reservoir (PR) In the case of a zoonozis such as leishmaniasis it would be sufficient to remove the infected dogs from the houshold. Sterilize PR In the case of schistosomiasis the PR can be sterilized either by treatment of the population by chemotherapy with drugs or by hygienic measures such as the placement of latrines in or just outside the villages. Eliminate intermediate host In the case of schistosomiasis treatment of lakes and rivers with molluscicides would interupt transmission via the snail as intermediate host.

Prophylaxis (2) – – • Eliminate vectors Treatment of surface water with larvicides or the application of insecticides in the house in the case of malaria will reduce or prevent transmission. Isolate vectors The use of bed nets, impregnated or not, with insecticides is an effective protection against mosquito bites. Vaccinate This would be the most efficient protective measure. However, so far there is no vaccine available for any parasitic disease of humans. Carry out chemoprophylaxis The preventive use of chloroquine (Nivaquine) for long periods when travelling in areas where malaria is endemic Depending of the nature of the parasite, its host and its vector different measures or a combination of measures have to be taken.

The Kinetoplastida • • Trypanosomiases and leishmaniases are caused by haemoflagellated protozoan parasites that belong to the family of the Trypanosomatidae, or trypanosomes, are members of the protist order Kinetoplastida, which can be divided in Bodonina and Trypanosomatina. Contrary to the Bodonina, which can be divided in the free-living Bodonidae and the parasitic Cryptobiae, all Trypanosomatidae, without exception, are parasitic. They are flagellated and parasitise all major groups of multicellular organisms such as insects, plants and animals. They can either be extracellular or intracellular and have a great variety of shapes, depending on the stage of the life cycle and their location within the host.

The Kinetoplastida • The following parasite-host associations are known: • Trypanosomatidae – Insect parasites • Crithidia • Herpetomonas – Plant parasites • Phytomonas – Amphibian parasites • Trypanosoma mega – Fish parasites • Trypanosoma – Reptiles • Leptomonas – Animals • Trypanosoma • Leishmania Cryptobiae Fishes Trypanoplasma Bodonidae free-living
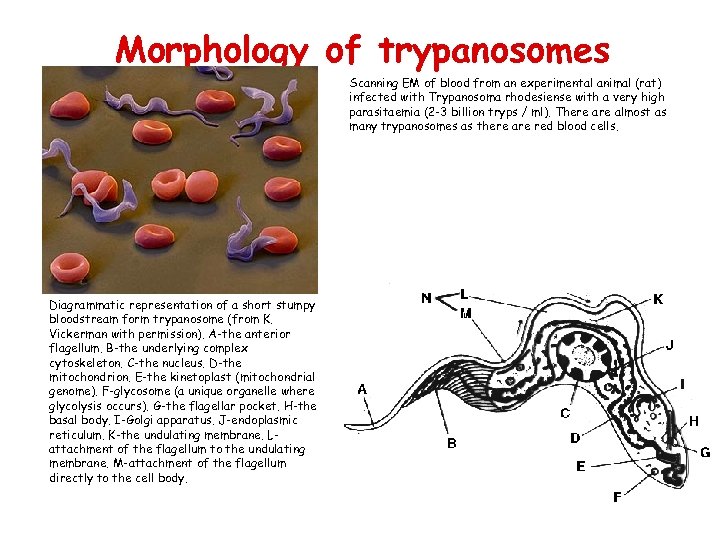
Morphology of trypanosomes Scanning EM of blood from an experimental animal (rat) infected with Trypanosoma rhodesiense with a very high parasitaemia (2 -3 billion tryps / ml). There almost as many trypanosomes as there are red blood cells. Diagrammatic representation of a short stumpy bloodstream form trypanosome (from K. Vickerman with permission). A-the anterior flagellum. B-the underlying complex cytoskeleton. C-the nucleus. D-the mitochondrion. E-the kinetoplast (mitochondrial genome). F-glycosome (a unique organelle where glycolysis occurs). G-the flagellar pocket. H-the basal body. I-Golgi apparatus. J-endoplasmic reticulum. K-the undulating membrane. Lattachment of the flagellum to the undulating membrane. M-attachment of the flagellum directly to the cell body.

• • Trypanosomes infecting man and animals African trypanosomes The genus Trypanosoma includes several species that infect wild and domesticated animals in Africa, particularly hoofed animals, and humans. The African trypanosomes : T. rhodesiense, T. gambiense, T. brucei, T. evansi T. congolense, T. vivax and T. suis, are all transmitted via tsetse flies (Glossina spp. ). The species that cause human African trypanosomiasis ("sleeping sickness") also infect wild animals and can be transmitted from these animals to humans (zoonotic infections). As their name implies, most African trypanosomes are restricted to Africa, although a few species such as T. evansi and T. vivax, have been imported into South America.

Trypanosomes infecting man and animals (2) • After a bloodmeal the bloodform trypanosomes, called trypomastigotes, transform and develop in the insect's midgut, first as procyclic insect stages. After some time they move to the proventriculus, where they transform to epimastigotes and from where they infect the salivary glands. Once in the salivary glands they transform to metacyclic trypomastigotes, which are infectious to the mammalian host. Once in the mammal they develop as long slender trypomastigotes, first in the area of the bite that becomes an ulcering chancre, from where they move towards the lymphe glands and the bloodstream, where they divide every 5 -7 hours. In general this phase of the disease leads to recurrent attacks of fever and to the wasting (cachexia, loss of weight) of man and animal. From there the trypanosomes cross the blood-brain barrier to invade the central nervous system and this leads to the typical symptoms of sleeping sickness, i. e. a disturbed day and night rhythm, change in personal character and coma. When not treated the course of the disease will be a fatal one resulting in death.

American trypanosomes • A few species of Trypanosoma are also found in the New World. T. cruzi, causing Chagas' disease in latin America is only distantly related to the African trypanosomes. It is an intracellular parasite that is transmitted via blood-sucking bugs (Triatoma). Chagas' disease is found throughout much of central and northern South America, Central America, and Mexico. T. cruzi is found in a number of animals other than humans, including dogs, cats and rodents, and the infection can be transmitted from these animals to humans.
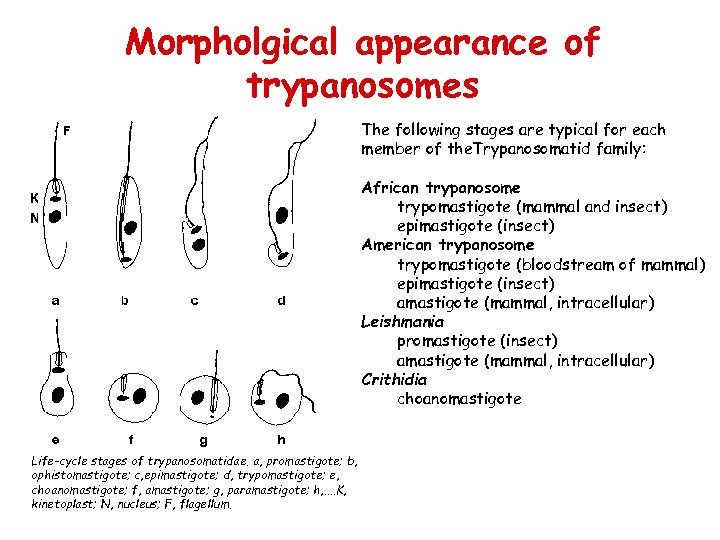
Morpholgical appearance of trypanosomes The following stages are typical for each member of the. Trypanosomatid family: African trypanosome trypomastigote (mammal and insect) epimastigote (insect) American trypanosome trypomastigote (bloodstream of mammal) epimastigote (insect) amastigote (mammal, intracellular) Leishmania promastigote (insect) amastigote (mammal, intracellular) Crithidia choanomastigote Life-cycle stages of trypanosomatidae. a, promastigote; b, ophistomastigote; c, epimastigote; d, trypomastigote; e, choanomastigote; f, amastigote; g, paramastigote; h, . . K, kinetoplast; N, nucleus; F, flagellum.
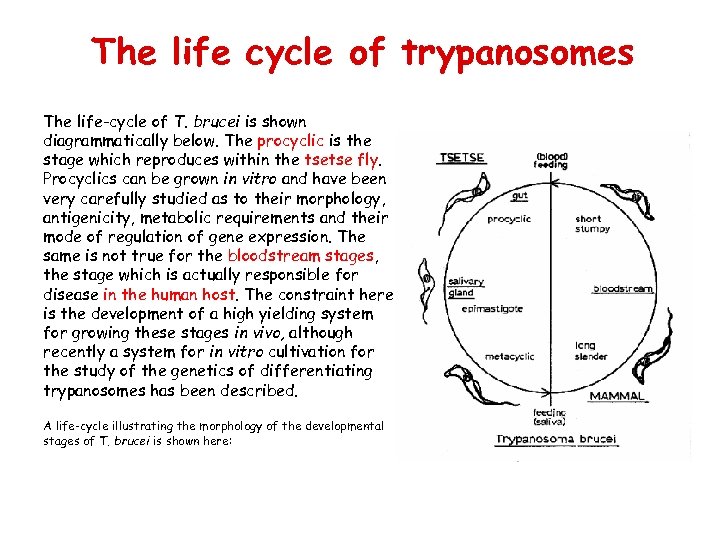
The life cycle of trypanosomes The life-cycle of T. brucei is shown diagrammatically below. The procyclic is the stage which reproduces within the tsetse fly. Procyclics can be grown in vitro and have been very carefully studied as to their morphology, antigenicity, metabolic requirements and their mode of regulation of gene expression. The same is not true for the bloodstream stages, the stage which is actually responsible for disease in the human host. The constraint here is the development of a high yielding system for growing these stages in vivo, although recently a system for in vitro cultivation for the study of the genetics of differentiating trypanosomes has been described. A life-cycle illustrating the morphology of the developmental stages of T. brucei is shown here:

Trypanosomes, their hosts and the diseases they cause

African trypanosomiasis or sleeping sickness • Sleeping sickness is highly focalized. In West Africa, left of the dividing line, tsetse flies transmit Trypanosoma brucei gambiense, responsible for a more slowly developing type of disease, the course of which may take several years. In East Africa the causative agent is Trypanosoma brucei rhodesiense. This organism causes a fulminant type of disease that may lead to the death of the host within several weeks to months. The deteriorating situation in Central Africa has led to major outbreaks of the disease, with recently hundreds of thousands of cases in Congo alone. Tsetse fly taking a blood meal. Geographical distribution of tsetse in Africa

Prevalence of African Human Trypanosomiasis (HAT) or sleeping sickness

• Symptoms and pathology Metacyclic trypanosomes injected into the host by tsetse, develop first in the skin leading to a typical ulceration called the chancre. From there the trypanosomes move to the lymphe nodes after which they invade the bloodstream. Giemsa-stained smear of blood infected with Trypanosoma rhodesiense • Once in the bloodstream the trypanosomes divide as long slender trypomastigotes of one specific variable antigen type (VAT) until the majority of them are killed by the host's antibodies directed against this VAT. However, a small percentage of them expresses a different VAT and this survives and will establish the next wave of infection and so on, each wave of infection causes a new peak of parasitaemia, high fever and malaise. Course of parasitaemia

Symptoms and pathology (2) • After some time the trypanosomes invade the central nervous system, resulting in the typical symptoms of sleeping sickness, such as an altered day and night rhythm, a change of character and eventually coma. When left untreated the outcome of African trypanosomiasis is always fatal.

Diagnosis of HAT (1) • Surveillance Is carried out by mobile teams which visit on a routine basis the rural areas of tropical Africa. With the deteriorating situation in Central Africa, this routine screening of the population has come to an almost complete halt. • Diagnosis By checking for a swelling of the lymphe nodes in Mobileneck. This is the village sign, in the team visiting an African first addition to the typical chancre at the place of the bite of the tsetse fly, and of the presence of trypanosomes in the body. Other tests are the analysis of a thick smear of blood for the presence of alive trypanosomes, or that of a giemsa-stained blood smear. Other less laborious tests are the CATT (card agglutination) test for the presence of circulating antibodies. DEAE cellulose column to separate trypanosomes from blood components. • •

Diagnosis of HAT (1 a) Collection of a drop of finger blood and the subsequent analysis of a wet drop for the presence of trypanosomes

Diagnosis of HAT (2) • HAT is known in two stages – Early stage disease Trypanosomes are present in the circulation only. – Late stage disease Trypanosomes have infected the central nervous system. • Once an infection has been confirmed by the presence of trypanosomes in the circulation, it has to be established whether the patient is still in the early or already in the late phase of the disease. This is decisive for the further treatment of the patient. • The presence of trypanosomes in the central nervous system can only be diagnosed by analyzing the fluid from the spine after a lumbar punction. Presence of either trypanosomes, or an increased white blood cell count, or elevated Ig. M, are all indications for central nervous system involvement.

Control (1) • Insecticides It will be impossible to eradicate tsetse from the African continent. Thus control measures must exist in maintaining the lowest possible level of exposure of the population to the infective bite of a tsetse fly. Control measures consist in low-cost ground spraying techniques from fixed wing aeroplanes with residual insecticides such as endosulfan of riverine areas. However, such measures have little selectivity and are normally not designed for sleeping sickness alone.

Control (2) • Tsetse traps These are cheap, cost-effective and community based. The most successful is the biconical trap made of blue tissue. Its blue colour attracts the flies which then enter the trap via holes in the lower part of the bicone. Once inside, the flies try to escape upwards towards the sun light and are trapped and killed in a trapping device attached to the apex of the upper cone that often contains some diesel oil. A reduction of the tsetse burden by 90 - 95 % can so easily be obtained in and around villages. The drawbacks include the cost of maintaining the traps, for once the bait expires the flies will soon re-enter the area.

Treatment of HAT • Treatment of the early phase of the disease is with either pentamidine in West Africa or suramin in East Africa and with organic arsenicals (Arsobal, Melarsoprol, Mel B) for the late stage of the disease. The latter treatment is not without danger and may lead in 5 -10% of cases to a fatal encephalopathy. In the case of melarsoprolrefractive cases Eflornitine is recommended. Trypanosomiasis victim, dying from an acute encephalopathy after treatment of the late stage of trpanosomiasis with the drug melarsoprol. • There are drugs that are widely used for the treatment of nagana or cattle trypanosomiasis such as ethidium, isomethamidium or berenil. These drugs are effective, both for treatment and for prophylaxis, but are supposed to intercalate into the DNA and therefore suspect to be mutagenic and thus cannot be used for the treatment of humans.

Drugs in use for HAT (1) • Pentamidine (Lomidine) is used only for the early phase of the disease, since it does not cross the blood-brain barrier. • It is used against T. gambiense, but cannot be used against T. rhodesiense, because of resistance in some areas. It should also not be used in areas where pentamidine has been widely employed for chemoprophylaxis, such as in the former Belgian Congo. • As chemoprophylaxis pentamidine as an intramuscular injection of 4 mg/kg has been assumed to protect for several months. This is not recommended anymore since the dose is subcurative and may mask an underlying infection. Moreover, the prophylactic use of pentamidine has provoked resitance in several areas. • The recommended dose for treatment is 4 mg of pentamidine base per kg of body weight given intramuscularly in a total of 7 injections daily or on alternating days.

Drugs in use for HAT (2) • • • Suramin has been developed in 1916 as a spin off of the German dye industry. It is a highly charged compound and so it is believed not to cross the blood-brain barrier. Therefore, monotherapy should only be used in the early stage of the disease. It is the preferred drug for treatment of the infection in East Africa. The drug is administered intravenously at a dosage of 20 mg/kg. It circulates in the blood in tight association with serum albumin and low density lipoproteins and is taken up slowly by both host and parasite, probably by receptor mediated endocytosis. Suramin is deposited in the renal tubes. Therefore it should not be administered to patients with renal disease. Urine should be checked before and during treatment for proteinuria. Suramin is also used for the treatment of onchocerciasis or river blindness in West Africa and therefore it is preferred not to be used for the treatment of sleeping sickness in this region.

Drugs in use for HAT (3) • • Melarsoprol (Arsobal, Mel B) is in use for trypanosomiasis since 1947. It is very effective in the treatment of both the early and the late stage of sleeping sickness, however, due to the risks associated with its use the drug is used only for the treatment of the late stage of the disease, both in West and in East Africa. • Treatment may be associated with an acute reactive encephalopathy in 5 - 10 % of the cases, which may result in paralysis, brain damage or even death.

Drugs in use for HAT (4) • • • Difluoromethyl ornithine (DFMO) or Eflornithine is a newly developed drug that is effective in the treatment of both the early and the late stage of West African sleeping sickness. It is not effective in the treatment of T. rhodesiense infections in East Africa. It is an ornithine analogue that inhibits the enzyme ornithine decarboxylase, the first enzyme in the synthesis of polyamines, essential in cell division and in the protection against oxidant stress in the trypanosome. Due to rapid excretion of the drug, the compound has to be given in very large quantities. A full treatment takes 400 grams of Eflornithine over a total period of two weeks, first as an infusion, followed by oral adminsitration in fruit juice. This drug is also called the "resurrection" drug, since comatous patients treated with Eflornithine may wake up rapidly and resume their activities.

Trypanosomiasis and nutrition (1) • • Nagana is the name the native population gives to cattle trypanosomiasis and it is caused by the trypanosomes: T. brucei, T. congolense and T. vivax which are transmitted by various members of the tsetse fly species Glossina. Imported cattle such as the Zebu are very sensitive to trypanosomiasis and after infection they rapidly lose weight and die within several months. As a consequence it is very difficult, if not impossible, to breed cattle within the tsetse belt Only in the Sahel, where it is too dry for tsetse, cattle breeding occurs at a significant scale. However, in this area the herds ravage the sparse vegetation and so are thought to be directly responsible for the spreading of the vast Sahara desert towards the south. Zebu cattle

Trypanosomiasis and nutrition (2) Differences in nutritional income between Africa and the European Union due to trypanosomiasis

Trypanosomiasis and nutrition (3) Influence of tsetse on cattle breeding in Africa

Rodent trypanosomes • • • Most trypanosomes are capable of infecting rodents such as mice and rats. Rodents serve widely as the experimental hosts and such experimental infections are used as models of the disease. The naturally occurring trypanosomes of rodents are: – – Trypanosoma lewisi (rat) Trypanosoma musculi (mouse) These trypanosomes are transmitted between rodents through the bite of fleas that live in the fur of the host animal and that feed on its blood. These trypanosomes are well adapted to their respective hosts and have a high degree of host specificity. Since these trypanosomes are extracellular parasites they are exposed to the humural immune response of the host at all times.
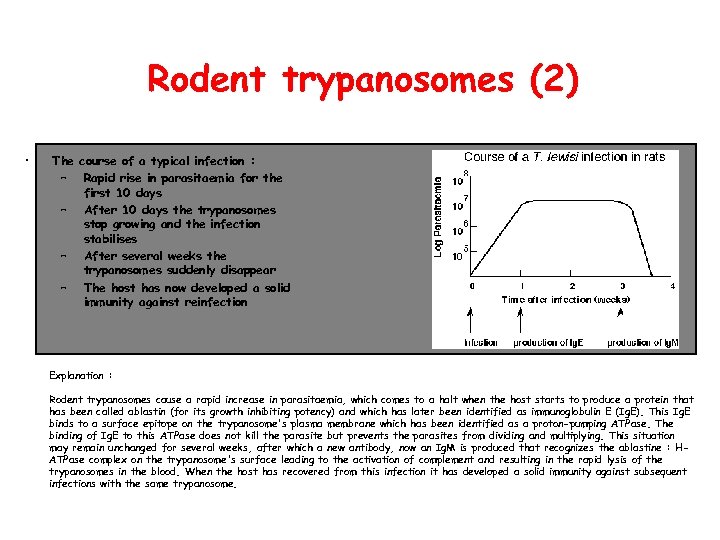
Rodent trypanosomes (2) • The course of a typical infection : – Rapid rise in parasitaemia for the first 10 days – After 10 days the trypanosomes stop growing and the infection stabilises – After several weeks the trypanosomes suddenly disappear – The host has now developed a solid immunity against reinfection Explanation : Rodent trypanosomes cause a rapid increase in parasitaemia, which comes to a halt when the host starts to produce a protein that has been called ablastin (for its growth inhibiting potency) and which has later been identified as immunoglobulin E (Ig. E). This Ig. E binds to a surface epitope on the trypanosome's plasma membrane which has been identified as a proton-pumping ATPase. The binding of Ig. E to this ATPase does not kill the parasite but prevents the parasites from dividing and multiplying. This situation may remain unchanged for several weeks, after which a new antibody, now an Ig. M is produced that recognizes the ablastine : HATPase complex on the trypanosome's surface leading to the activation of complement and resulting in the rapid lysis of the trypanosomes in the blood. When the host has recovered from this infection it has developed a solid immunity against subsequent infections with the same trypanosome.

Antigenic variation in African trypanosomes • • African trypanosomes are extra-cellular parasites. Thus they are exposed to the mammalian humural immune system at all times. To evade the immune response trypanosomes are covered with a surface coat consisting of a monomolecular layer of a glycoprotein called Variable Surface coat Glycoprotein or VSG. This protein is being changed every 5 -7 days, which results in a new VAT (variable antigen type). Immunoglobulins directed against these VSGs recognise the trypanosomes. Binding of antibodies to the tryps will lead to lysis of the trypanosomes by complement and disappearance of the tryps from the blood. However, a minority will have changed its VSG and is not recognised by the circulating antibodies. This population will multiply and expand unlimited, until new antibodies will be produced against this variant, etc. .

Structure of trypanosome VSG Schematic representation of the protein chain of a typical VSG. The N-terminus contains a signal peptide which is cleaved off from the mature protein. The variable region is different for each VSG and shows little or no identity with other VSGs. The hydrophobic tail contains a recognition signal for attachment to the glycolipid anchor (see below). When the anchor is attached the last 20 amino acids are cleaved off. Structure of the glycolipid anchor for VSG. The variable surface glycoprotein (VSG) is attached to the membrane via a glycolipid anchor consisting of ethanolamine, a glycan structure containing several mannose moieties, a glucosamine and a phosphoinositol that is linked to a 1, 2 -dimyristoylglycerol burried in the plasma membrane.

Recycling of the surface coat • The entire VSG surface of a trypanosome is recycled every seven minutes by a process of VSG endocytosis and exocytosis. This rapid rate of recycling is incompatible with the slow speed of diffusion of VSG proteins along the length of the trypanosome (45 minutes). This was reconciled by the discovery of a VSG ‘fast lane’, along the flagellar attachment zone, that rapidly facilitates the movement of VSG along the length of the trypanosome before lateral diffusion. This mechanism for the rapid recycling of the surface coat probably also offers a mechanism for clearing antibodies attached to the trypanosome surface.

Antigenic variation in African trypanosomes (2) • • • It is assumed that antigenic variation may continue for ever and certainly for as long as the host is alive, since there are: – at least 100 different VATs that have been identified in T. equiperdum – at least 1000 different genes that are present in the genome of T. brucei. Moreover, trypanosomes have a very high capacity for homologous recombination. This means that different members of the VSG gene family may recombine to form new variants. This is a process that may go on for ever. When a host is infected with a single cloned trypanosome there is a semiordered sequence of appearance of VAT's and every new VAT can be totally different from the previous one (ie. no immunological cross-reactivity). In general the VSGs that appear in an infection can be divided into early, medium and late VSGs. They have the tendency to appear in this order, but this is not a strict rule.

Molecular mechanisms involved in antigenic variation • Several mechanisms have been proposed to explain the observation of antigenic variation at the molecular level. These are all based on the observation that only VSG genes located at, or near, a chromosome end (called a telomere) are expressed (this does not hold for metacyclic VSG genes). VSG genes make part of a large gene family but only a limited number of VSG genes are present in expression sites on telomeres. Only one expression site can be active at a given time, explaining why only one type of VSG is expressed at a time. The mechanisms of VSG activation proposed are shown in the figure below.

Properties of the kinetoplast • • • Kinetoplast DNA consists of a network, containing a complex of 10 000 concatenated minicircles and some 50 maxicircles The maxicircles are highly conserved and contain the genetic information for components of the mitochondrial respiratory chain, similar to the DNA of other mitochondria. The minicircles are heterogeneous in sequence. The maxicircle genes are often incomplete and may lack a start codon (ATG). Sometimes they even contain less than 50% of the coding information and do not even hybridize with their corresponding mature RNA. Linear map of the 22 -kb maxicircle of T. brucei.
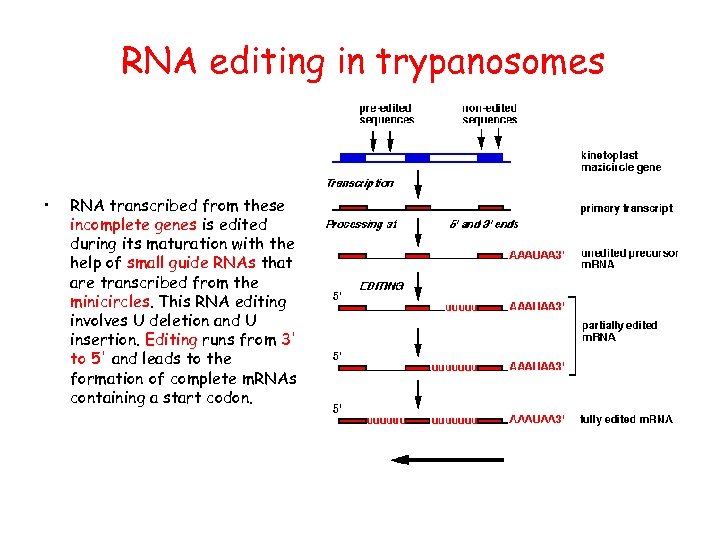
RNA editing in trypanosomes • RNA transcribed from these incomplete genes is edited during its maturation with the help of small guide RNAs that are transcribed from the minicircles. This RNA editing involves U deletion and U insertion. Editing runs from 3' to 5' and leads to the formation of complete m. RNAs containing a start codon.

Pan-editing of mitochondrial RNA in Kinetoplastida. • The sequence shown below represents a part of a mature m. RNA that has been created by a process of pan-editing (pan-editing is editing throughout the entire length of the m. RNA). Upper case letters are those also present in the DNA. Uridines which have been inserted by the editing process are indicated as lower case us in magenta and those that have been removed are indicated with an asterix (*). Note that owing to the editing process a start codon is being created so allowing for the translation of the mature m. RNA. UCCu. Auu. A*Au. UUUUUGu. UA**UAu AGuuuuuu. AA*UGUUGuuu. GGu. A *uuuuuuu. Au. UG*UGuuu. AGuuuu. Guuu. G****GU NB: Also note that the mature m. RNA would not be able to hybridise with the corresponding Gu. Guu. Auu. G**UUUUGAGAuu. G • gene present in the kinetoplast DNA and thus cannot be detected as such.

Dyskinetoplastic trypanosomes • In the absence of any selective pressure on the mitochondrial activity (no developmental passage through an insect vector), dyskinetoplastic strains may develop spontaneously. The intermediate stages in this development are: – 1, mutation followed by deletion of maxicircle genes; – 2, loss of guide RNAs and loss of heterogeneity of the minicircles; – 3, loss of maxicircles; – 4, complete loss of the minicircle complex. • Naturally occurring species with incomplete k. DNA or without any k. DNA at all are: T. evansi, T. equiperdum and T. equinum. They cannot develop in the tsetse fly and are transmitted by horseflies (T. evansi) or by copulation (T. equiperdum, T. equinum).

DNA transcription and mini exons Orientation of genes on chromosomes • In Trypanosomatidae only a few promoters have been identified so far. These are the VSG promoter, the ribosomal RNA promoter, the tubulin promoter and the PARP promoter, for which abundant gene products are required. It is assumed that for most other gene products DNA is continuously transcribed in the form of a single polycistronic messenger. By a mechanism called trans-splicing, all mature messenger RNAs will receive the same 36 nucleotide long leader sequence, the so-called "spliced leader" or "miniexon". Trans-splicing of RNA

Glycosomes of T. brucei bloodforms visualised by serial sectioning Three major types of organelles are shown: the nucleus, the single mitochondrion and the microbodies Glycosome Nucleus Glycolytic enzymes are sequestered in some 65 microbodies, called glycosomes that are distributed throughout the cytoplasm Mitochondrion From: Tetley and Vickerman, 1991
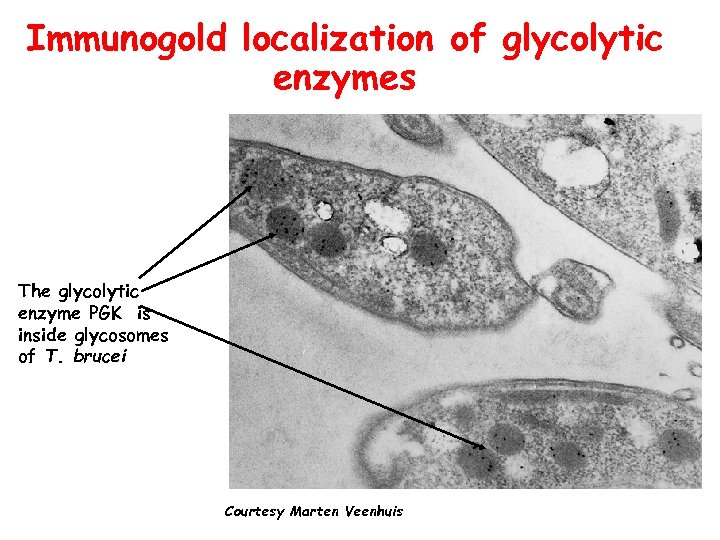
Immunogold localization of glycolytic enzymes The glycolytic enzyme PGK is inside glycosomes of T. brucei Courtesy Marten Veenhuis

Compartmentation of glycolysis in bloodstream form trypanosomes Three compartments 1. Glycosome, where glucose is converted to phosphoglycerate 2. Mitochondrion, where glycosomal NADH is reoxidized via an alternative oxidase 3. Cytosol, where takes place the net synthesis of ATP and production of pyruvate

American Trypanosomiasis or Chagas' Disease • A few species of Trypanosoma are also found in the New World. From the standpoint of human health, the most important is Trypanosoma cruzi which causes American trypanosomiasis or Chagas' disease is found throughout much of central and northern South America, Central America, and Mexico. Distribution of Chagas ’ disease. Among an estimated population of 360 million inhabitants at least 90 million persons (25%) are considered at risk of infection and 16 -18 million people are infected. It is generally expected that about 30% of the infected population will develop clinically overt disease. Thus it is expected that in total some 5 million people have the clinical symptoms of Chagas' disease. T. cruzi is found in a number of animals other than humans, including dogs, cats and rodents, and infections of such reservoirs in the United States have been reported. However, it remains unclear whether the T. cruzi found in these reservoirs can actually be transmitted to humans.

The triatomine bug, vector of Chagas ’ disease

Geographical distribution of the vector

Life cycle of Trypanosoma cruzi

Trypomastigote form of T. cruzi • 1, sub-pellicular microtubules 2, acidocalcisomes 3, mitochondrion 4, kinetoplast 5, axoneme 6, Golgi apparatus 7, nucleus 8, nucleolus 9, flagellum According to W. de Souza

Life-cycle stages of T. cruzi

American Trypanosomiasis or Chagas' Disease (2) • • In humans, T. cruzi is found as both an intracellular form, the amastigote, and as a trypomastigote form in the blood. The vector for Chagas' Disease, a "true bug" (Hemiptera) such a Triatoma, ingests amastigotes or trypomastigotes when it feeds. In the vector the parasite reproduces asexually as epimastigotes, and metacyclic trypomastigotes are found in the vector's hindgut. The vector defecates on the host's skin at the same time as it feeds, and the metacyclic trypomastigotes enter the host's body, most often by being "rubbed in" to the vector's bite or the mucous membranes of the eye, nose, or mouth. Intracellular amastigotes epimastigote Triatoma Trypomastigotes in blood

Entry sites for trypanosomes The local inflammation caused by the entry of T. cruzi is called chagoma. Chagoma of the eye, Romaña ’s sign, is seen in 90 % of patients diagnosed as recently infected.

Symptoms and pathology of Chagas' disease (2) • Trypanosomes are taken up by macrophages and multiply intracellularly as amastigotes in the cytoplasm of the host cells. When these cells are loaded with amastigotes the latter transform to trypomastigotes and the host cells bursts open, releasing the trypomastigotes into the circulating blood, causing high parasiteamia and fever. These are the symptoms of the early stage of Chagas' disease. When antibodies against the trypomastigotes develop, parasiteamia goes down and the trypomastigotes almost completely disappear from the blood to such a level that they cannot be detected other than by xenodiagnosis or by such sophisticated techniques as PCR (polymerase chain reaction). Xenodiagnosis
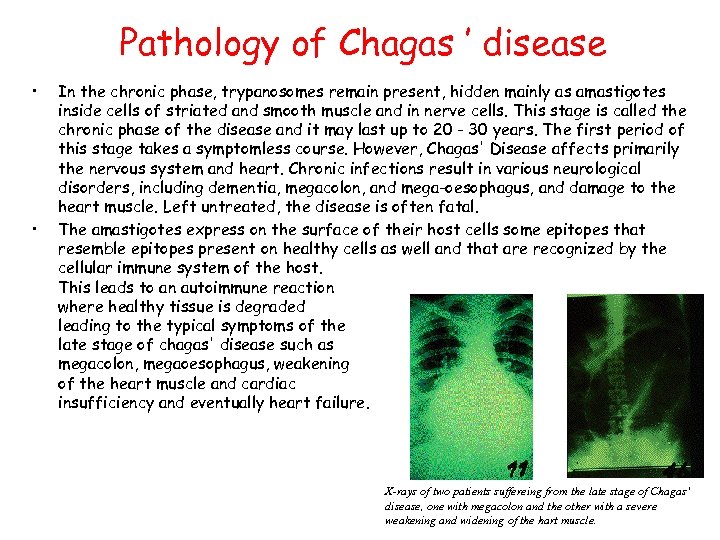
Pathology of Chagas ’ disease • • In the chronic phase, trypanosomes remain present, hidden mainly as amastigotes inside cells of striated and smooth muscle and in nerve cells. This stage is called the chronic phase of the disease and it may last up to 20 - 30 years. The first period of this stage takes a symptomless course. However, Chagas' Disease affects primarily the nervous system and heart. Chronic infections result in various neurological disorders, including dementia, megacolon, and mega-oesophagus, and damage to the heart muscle. Left untreated, the disease is often fatal. The amastigotes express on the surface of their host cells some epitopes that resemble epitopes present on healthy cells as well and that are recognized by the cellular immune system of the host. This leads to an autoimmune reaction where healthy tissue is degraded leading to the typical symptoms of the late stage of chagas' disease such as megacolon, megaoesophagus, weakening of the heart muscle and cardiac insufficiency and eventually heart failure. X-rays of two patients suffereing from the late stage of Chagas' disease, one with megacolon and the other with a severe weakening and widening of the hart muscle.

Aneurysm of the heart

Various degrees of megacolon and megaoesophagus

Chagas’ disease and blood transfusion • A severe problem related to Chagas' disease is the presence of undetectable amounts of trypanosomes in the blood of asymptomatic carriers. If their blood is used for blood transfusion, trypanosomes are transferred from an immune donor to a non-immune individual, which may lead to a fulminating parasitaemia and sudden death of the latter. • For this purpose blood banks in South America have to treat all blood with gentian violet for 24 h at 4° C in order to kill any trypomastigotes. • An inconvenience related to this treatment is that the blood turns into purple and that some recipients do refuse infusion with blood treated this way.

Immunology of Chagas' disease • • In the circulation of Chagas patients large amounts of Ig. M and Ig. G can be detected which: – lyse insect parasites (metacyclic epimastigotes) and thus protect against superinfection, – do not lyse trypomastigotes (blood forms) – have no effect on the intracellular amastigotes The actively dividing parasite itself hides away inside the host cells and cannot be reached by the immune system. A major problem is the development of autoimmune disease: T. cruzi amastigotes and neuronal cells share common antigenic determinants: the recognition of the parasite epitopes leads to the recognition of self which provokes an autoimmune disease that affects nerve cells and muscle cells.

Chagas’ disease and vaccination • Since the antigens responsible for the onset of autoimmune disease have not yet been characterized it is impossible to develop vaccins based on whole organisms, since any such vaccin may provoke the same kind of autoimmune disease without ever having been in contact with the parasite. • Therefore, the only solution would be the development of so-called subunit vaccins (see malaria).

Treatment of Chagas' Disease • • • The treatment of Chagas' disease is indicated for patients with the acute form of the disease or in congenital disease. No information is available about the efficacy of drug treatment in preventing the development of chronic disease. In these cases only symptomatic treatment is advised. The following drugs are available: – Nifurtimox (Lampit, a nitrofuran derivative) is active against intracellular amastigotes as well as against the trypomastigotes. It is given in daily doses of 10 mg per kg of body weight to adults and 15 mg/kg to children for 60 -90 days. Since the drug causes oxidative stress its use should be avoided in cases of glucose-6 -phosphate dehydrogenase deficiency. – Benznidazole (Radanil, a nitroimidazole derivative) is active against intracellular amastigotes as well as against the trypomastigotes. It is given in daily doses of 5 -10 mg/kg for 30 -60 days. Since the drug causes oxidative stress its use should be avoided in cases of glucose-6 -phosphate dehydrogenase deficiency. – Allopurinol, an experimental drug presently in use for the treatment of gout. – Gentian or crystal violet is used for blood transfusion for the inactivation of trypanosomes in the blood. Efficacy of the treatment. A problem associated with both nifurtimox and benznidazole is that their therapeutic window (difference between their ED 50 and LD 50) is rather small. Both drugs are usually effective, but figures from Brazil indicate that treatment may also fail.

Mode of action of anti-Chagas ’ drugs • • Nifurtimox and Benznidazole are thought to increase oxidant stress of the parasites leading to their killing or inhibition of growth. Allopurinol is supposed to function as an alternative substrate for the enzyme hypoxanthine guanine phosphoribosyl transferase (HGPRTase), so allowing the incorporation of allopurinol riboside into RNA, which leads to the inhibition of protein synthesis in the parasite.

Prophylaxis • • It is advised not to spend the night with the rural population inside the primitive housing often infested with the triatomine bug, the vector of Chagas disease. All blood for blood transfusion should be treated overnight with Gentian violet. Houses in rural areas where Chagas disease is endemic should be treated with insecticides or with insecticide-containing paints. Where possible proper housing with stone brick walls should be provided to the population.
bf6a393fb980e8173e6ad9524a8ebf34.ppt