cae48f3b8221f0069409aeabf4d231b1.ppt
- Количество слайдов: 32
 Paediatric HIV Research: Status, priorities and ethical considerations Dr Nigel Rollins Child and Adolescent Health and Development, World Health Organization, Geneva International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Paediatric HIV Research: Status, priorities and ethical considerations Dr Nigel Rollins Child and Adolescent Health and Development, World Health Organization, Geneva International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Acknowledgements Siobhan Crowley, Department of HIV Ying Lo Ru, Department of HIV Alasdair Reid, UNAIDS International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Acknowledgements Siobhan Crowley, Department of HIV Ying Lo Ru, Department of HIV Alasdair Reid, UNAIDS International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 2007 global HIV and AIDS estimates Children (<15 years) • Children living with HIV 2. 0 million [1. 9 – 2. 3 million] • New HIV infections in 2007 370 000 [330 000 – 410 000] • Deaths due to AIDS in 2007 270 000 [250 000 – 290 000] International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
2007 global HIV and AIDS estimates Children (<15 years) • Children living with HIV 2. 0 million [1. 9 – 2. 3 million] • New HIV infections in 2007 370 000 [330 000 – 410 000] • Deaths due to AIDS in 2007 270 000 [250 000 – 290 000] International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
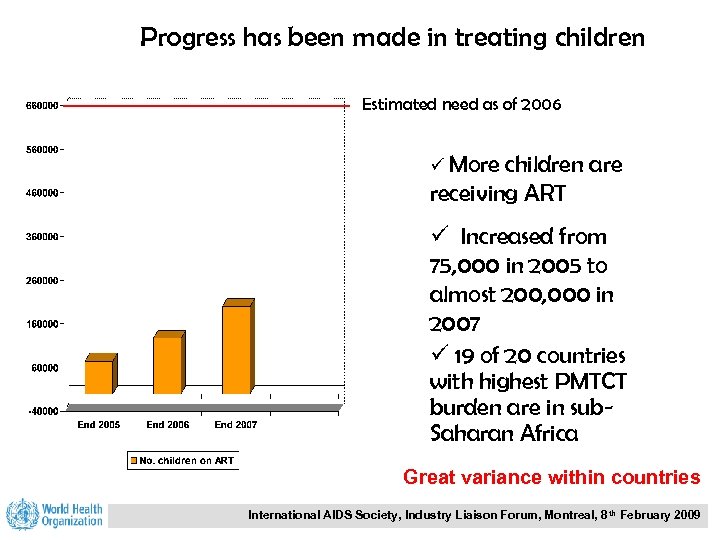 Progress has been made in treating children Estimated need as of 2006 ü More children are receiving ART ü Increased from 75, 000 in 2005 to almost 200, 000 in 2007 ü 19 of 20 countries with highest PMTCT burden are in sub. Saharan Africa Great variance within countries International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Progress has been made in treating children Estimated need as of 2006 ü More children are receiving ART ü Increased from 75, 000 in 2005 to almost 200, 000 in 2007 ü 19 of 20 countries with highest PMTCT burden are in sub. Saharan Africa Great variance within countries International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 ART outcomes - good news across the globe ü National programmes reporting good outcomes ü 1 year survival estimated as 93 -95% ü 2 year survival 91% International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
ART outcomes - good news across the globe ü National programmes reporting good outcomes ü 1 year survival estimated as 93 -95% ü 2 year survival 91% International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
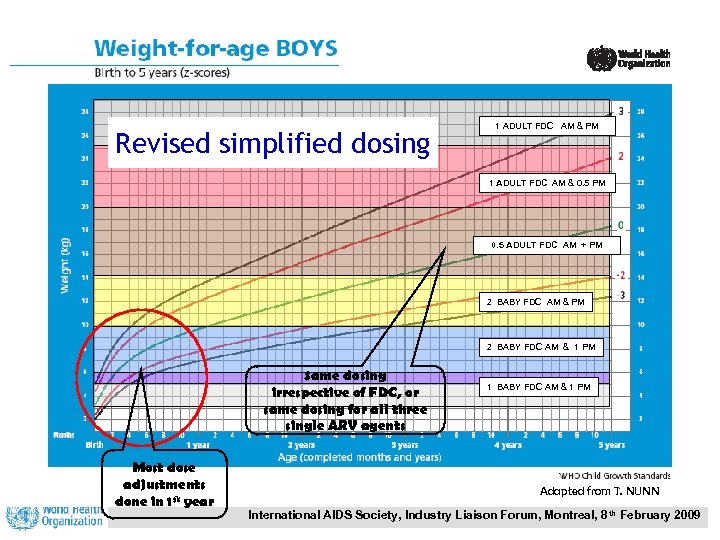 Revised simplified dosing 1 ADULT FDC AM & PM 1 ADULT FDC AM & 0. 5 PM 0. 5 ADULT FDC AM + PM 2 BABY FDC AM & 1 PM Same dosing irrespective of FDC, or same dosing for all three single ARV agents Most dose adjustments done in 1 st year 1 BABY FDC AM & 1 PM Adapted from T. NUNN International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Revised simplified dosing 1 ADULT FDC AM & PM 1 ADULT FDC AM & 0. 5 PM 0. 5 ADULT FDC AM + PM 2 BABY FDC AM & 1 PM Same dosing irrespective of FDC, or same dosing for all three single ARV agents Most dose adjustments done in 1 st year 1 BABY FDC AM & 1 PM Adapted from T. NUNN International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Children everywhere are Starting Treatment Late Baseline Median Age Meta-analysis 1, 195 children from 8 African clinical trials 53% >5 years of age, 70% severe immune deficiency, 12% aged < 12 months (KIDS-ART-LINC) Arrive 2008 Janssens/Cambodia 2007 N=212 George/Haiti Baseline Median CD 4 6. 0 yrs 6% 6. 3 yrs 12% 4. 4 yrs 6% 5. 7 yrs 8% 7. 7 yrs 5% 9. 2 yrs 8. 6% 6. 5 yr 8% 2007 N=100 Wamawala/Kenya 2007 N=67 Reddi/S Africa 2007 N=151 Puthanakit/Thailand 2007 N=107 Kamya/Uganda 2007 N=250 Rouet/Cote d’Ivoire 2006 N=78 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Children everywhere are Starting Treatment Late Baseline Median Age Meta-analysis 1, 195 children from 8 African clinical trials 53% >5 years of age, 70% severe immune deficiency, 12% aged < 12 months (KIDS-ART-LINC) Arrive 2008 Janssens/Cambodia 2007 N=212 George/Haiti Baseline Median CD 4 6. 0 yrs 6% 6. 3 yrs 12% 4. 4 yrs 6% 5. 7 yrs 8% 7. 7 yrs 5% 9. 2 yrs 8. 6% 6. 5 yr 8% 2007 N=100 Wamawala/Kenya 2007 N=67 Reddi/S Africa 2007 N=151 Puthanakit/Thailand 2007 N=107 Kamya/Uganda 2007 N=250 Rouet/Cote d’Ivoire 2006 N=78 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
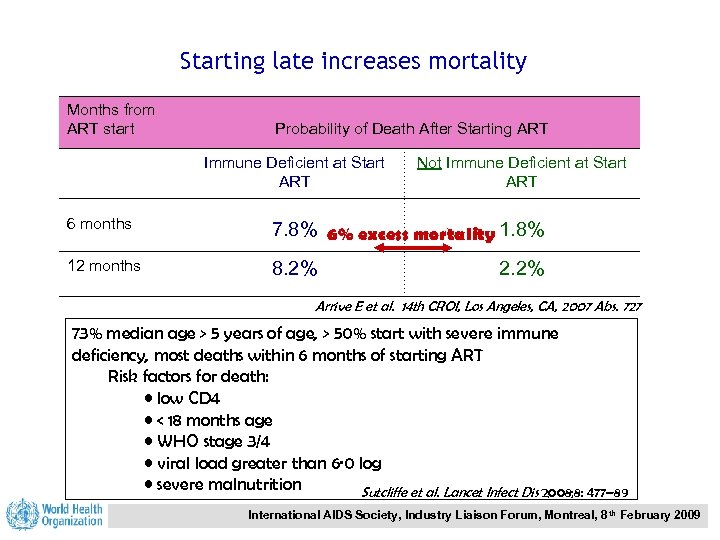 Starting late increases mortality Months from ART start Probability of Death After Starting ART Immune Deficient at Start ART Not Immune Deficient at Start ART 6 months 7. 8% 6% excess mortality 1. 8% 12 months 8. 2% 2. 2% Arrive E et al. 14 th CROI, Los Angeles, CA, 2007 Abs. 727 73% median age > 5 years of age, > 50% start with severe immune deficiency, most deaths within 6 months of starting ART Risk factors for death: • low CD 4 • < 18 months age • WHO stage 3/4 • viral load greater than 6· 0 log • severe malnutrition Sutcliffe et al. Lancet Infect Dis 2008; 8: 477– 89 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Starting late increases mortality Months from ART start Probability of Death After Starting ART Immune Deficient at Start ART Not Immune Deficient at Start ART 6 months 7. 8% 6% excess mortality 1. 8% 12 months 8. 2% 2. 2% Arrive E et al. 14 th CROI, Los Angeles, CA, 2007 Abs. 727 73% median age > 5 years of age, > 50% start with severe immune deficiency, most deaths within 6 months of starting ART Risk factors for death: • low CD 4 • < 18 months age • WHO stage 3/4 • viral load greater than 6· 0 log • severe malnutrition Sutcliffe et al. Lancet Infect Dis 2008; 8: 477– 89 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Research priorities • Early identification of infected children – Diagnostic / screening algorithms e. g. in IMCI – HIV testing algorithms – Universal screening at immunisation clinics • Monitoring of disease progression / ART response in settings with minimal / intermittent lab support – Target weight gain – Clinic-based technology • Feasibility of nurse-led ART initiation / management – Type and methods of support International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Research priorities • Early identification of infected children – Diagnostic / screening algorithms e. g. in IMCI – HIV testing algorithms – Universal screening at immunisation clinics • Monitoring of disease progression / ART response in settings with minimal / intermittent lab support – Target weight gain – Clinic-based technology • Feasibility of nurse-led ART initiation / management – Type and methods of support International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Tuberculosis • 1/3 of the world’s population is infected with TB • Only 5 -10% actually develop TB disease during their lifetime – Risk of progression highest in young children and soon after infection – Increased risk with HIV, malnutrition, immunocompromise • One fifth of all TB cases occur in children and young people • Approximately 11% of all TB cases occur in children <15 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Tuberculosis • 1/3 of the world’s population is infected with TB • Only 5 -10% actually develop TB disease during their lifetime – Risk of progression highest in young children and soon after infection – Increased risk with HIV, malnutrition, immunocompromise • One fifth of all TB cases occur in children and young people • Approximately 11% of all TB cases occur in children <15 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 TB/HIV • Children living with HIV are up to 20 times more likely to develop TB than HIV negative children – Increased risk of exposure – Increased risk of progression from infection to disease • TB is commonest cause of illness & death in people living with HIV in Africa • Data on the situation in children are scant • The burden of HIV in confirmed and clinical cases of TB in children ranges from 11 -70% • Main challenge is making a definitive diagnosis of TB in children International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
TB/HIV • Children living with HIV are up to 20 times more likely to develop TB than HIV negative children – Increased risk of exposure – Increased risk of progression from infection to disease • TB is commonest cause of illness & death in people living with HIV in Africa • Data on the situation in children are scant • The burden of HIV in confirmed and clinical cases of TB in children ranges from 11 -70% • Main challenge is making a definitive diagnosis of TB in children International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Research priorities • Acute need for more research on TB in children, esp those living with HIV – True burden of disease – Better diagnostics that don’t rely on sputum – Clarity on optimal regimens for treatment of TB and MDR TB in combination with ARVs – TB vaccine • safety of BCG in children on ARVs • New more effective TB vaccine International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Research priorities • Acute need for more research on TB in children, esp those living with HIV – True burden of disease – Better diagnostics that don’t rely on sputum – Clarity on optimal regimens for treatment of TB and MDR TB in combination with ARVs – TB vaccine • safety of BCG in children on ARVs • New more effective TB vaccine International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Preventing HIV infection in infants and children Western & Eastern Europe Central Europe & Central Asia <200 North America <500 [<200] [<100] 3200 [2400 – 4300] East Asia Middle East & North Africa Caribbean 1800 [1500 – 2100] Latin America 4600 [4200 – 8300] 5700 [3800 – 8000] Sub-Saharan Africa 330 000 [300 000 – 360 000] 2000 [1200 – 3100] South & South-East Asia 21 000 [14 000 – 29 000] Oceania <1000 Total: 370 000 (330 000 – 410 000) International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Preventing HIV infection in infants and children Western & Eastern Europe Central Europe & Central Asia <200 North America <500 [<200] [<100] 3200 [2400 – 4300] East Asia Middle East & North Africa Caribbean 1800 [1500 – 2100] Latin America 4600 [4200 – 8300] 5700 [3800 – 8000] Sub-Saharan Africa 330 000 [300 000 – 360 000] 2000 [1200 – 3100] South & South-East Asia 21 000 [14 000 – 29 000] Oceania <1000 Total: 370 000 (330 000 – 410 000) International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
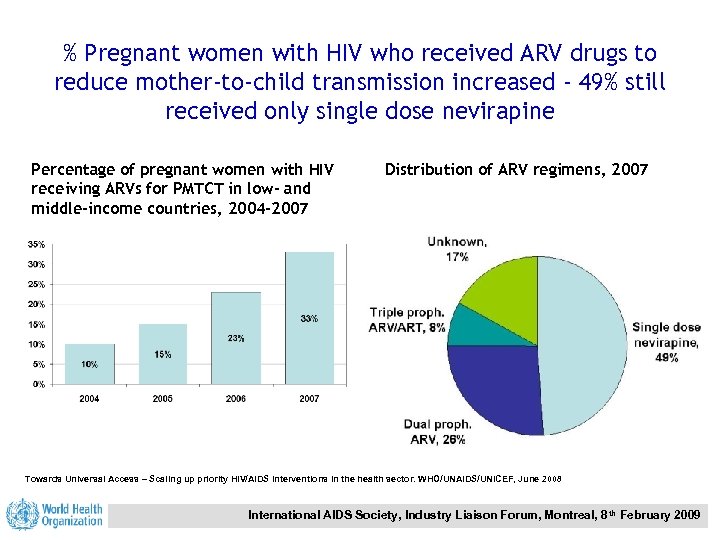 % Pregnant women with HIV who received ARV drugs to reduce mother-to-child transmission increased - 49% still received only single dose nevirapine Percentage of pregnant women with HIV receiving ARVs for PMTCT in low- and middle-income countries, 2004 -2007 Distribution of ARV regimens, 2007 Towards Universal Access – Scaling up priority HIV/AIDS interventions in the health sector. WHO/UNAIDS/UNICEF, June 2008 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
% Pregnant women with HIV who received ARV drugs to reduce mother-to-child transmission increased - 49% still received only single dose nevirapine Percentage of pregnant women with HIV receiving ARVs for PMTCT in low- and middle-income countries, 2004 -2007 Distribution of ARV regimens, 2007 Towards Universal Access – Scaling up priority HIV/AIDS interventions in the health sector. WHO/UNAIDS/UNICEF, June 2008 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
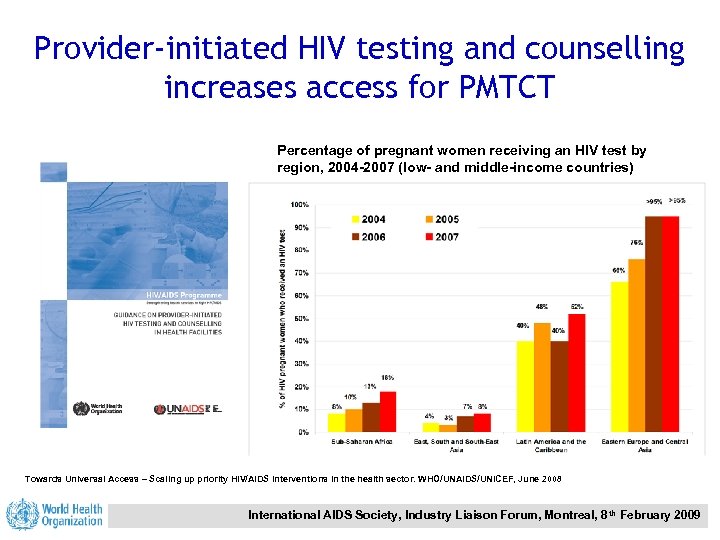 Provider-initiated HIV testing and counselling increases access for PMTCT Percentage of pregnant women receiving an HIV test by region, 2004 -2007 (low- and middle-income countries) Towards Universal Access – Scaling up priority HIV/AIDS interventions in the health sector. WHO/UNAIDS/UNICEF, June 2008 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Provider-initiated HIV testing and counselling increases access for PMTCT Percentage of pregnant women receiving an HIV test by region, 2004 -2007 (low- and middle-income countries) Towards Universal Access – Scaling up priority HIV/AIDS interventions in the health sector. WHO/UNAIDS/UNICEF, June 2008 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
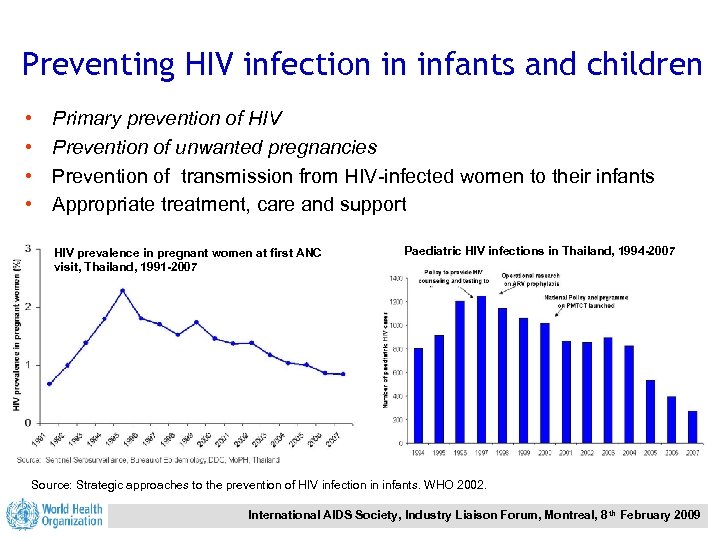 Preventing HIV infection in infants and children • • Primary prevention of HIV Prevention of unwanted pregnancies Prevention of transmission from HIV-infected women to their infants Appropriate treatment, care and support HIV prevalence in pregnant women at first ANC visit, Thailand, 1991 -2007 Paediatric HIV infections in Thailand, 1994 -2007 Source: Strategic approaches to the prevention of HIV infection in infants. WHO 2002. International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Preventing HIV infection in infants and children • • Primary prevention of HIV Prevention of unwanted pregnancies Prevention of transmission from HIV-infected women to their infants Appropriate treatment, care and support HIV prevalence in pregnant women at first ANC visit, Thailand, 1991 -2007 Paediatric HIV infections in Thailand, 1994 -2007 Source: Strategic approaches to the prevention of HIV infection in infants. WHO 2002. International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Recommended first-line ART regimens for eligible pregnant women Mother Antepartum AZT + 3 TC + NVP twice daily Intrapartum AZT + 3 TC + NVP twice daily Postpartum AZT + 3 TC + NVP twice daily Infant * AZT x 7 days* If the mother receives < 4 wks of ART during pregnancy, give 4 wks of infant AZT Source: Antiretroviral drugs for treating pregnant women and preventing HIV infection in infants. WHO 2006 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Recommended first-line ART regimens for eligible pregnant women Mother Antepartum AZT + 3 TC + NVP twice daily Intrapartum AZT + 3 TC + NVP twice daily Postpartum AZT + 3 TC + NVP twice daily Infant * AZT x 7 days* If the mother receives < 4 wks of ART during pregnancy, give 4 wks of infant AZT Source: Antiretroviral drugs for treating pregnant women and preventing HIV infection in infants. WHO 2006 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Recommended ARV-prophylaxis for pregnant women not eligible for ART Mother Antepartum AZT Intrapartum Sd-NVP + AZT/3 TC Postpartum AZT/3 TC for 7 days Infant * Sd-NVP + AZT for 7 days* If the mother receives < 4 wks of ART during pregnancy, give 4 wks of infant AZT Source: Antiretroviral drugs for treating pregnant women and preventing HIV infection in infants. WHO 2006 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Recommended ARV-prophylaxis for pregnant women not eligible for ART Mother Antepartum AZT Intrapartum Sd-NVP + AZT/3 TC Postpartum AZT/3 TC for 7 days Infant * Sd-NVP + AZT for 7 days* If the mother receives < 4 wks of ART during pregnancy, give 4 wks of infant AZT Source: Antiretroviral drugs for treating pregnant women and preventing HIV infection in infants. WHO 2006 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
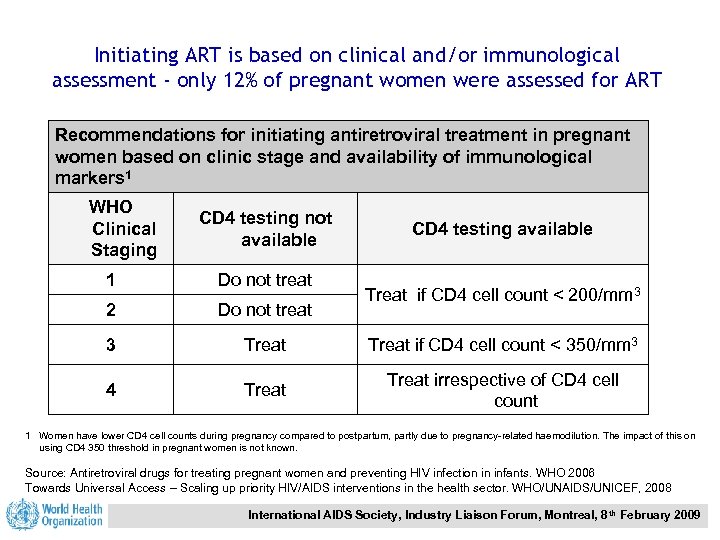 Initiating ART is based on clinical and/or immunological assessment - only 12% of pregnant women were assessed for ART Recommendations for initiating antiretroviral treatment in pregnant women based on clinic stage and availability of immunological markers 1 WHO Clinical Staging CD 4 testing not available CD 4 testing available 1 Do not treat 2 Do not treat 3 Treat if CD 4 cell count < 350/mm 3 4 Treat irrespective of CD 4 cell count Treat if CD 4 cell count < 200/mm 3 1 Women have lower CD 4 cell counts during pregnancy compared to postpartum, partly due to pregnancy-related haemodilution. The impact of this on using CD 4 350 threshold in pregnant women is not known. Source: Antiretroviral drugs for treating pregnant women and preventing HIV infection in infants. WHO 2006 Towards Universal Access – Scaling up priority HIV/AIDS interventions in the health sector. WHO/UNAIDS/UNICEF, 2008 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Initiating ART is based on clinical and/or immunological assessment - only 12% of pregnant women were assessed for ART Recommendations for initiating antiretroviral treatment in pregnant women based on clinic stage and availability of immunological markers 1 WHO Clinical Staging CD 4 testing not available CD 4 testing available 1 Do not treat 2 Do not treat 3 Treat if CD 4 cell count < 350/mm 3 4 Treat irrespective of CD 4 cell count Treat if CD 4 cell count < 200/mm 3 1 Women have lower CD 4 cell counts during pregnancy compared to postpartum, partly due to pregnancy-related haemodilution. The impact of this on using CD 4 350 threshold in pregnant women is not known. Source: Antiretroviral drugs for treating pregnant women and preventing HIV infection in infants. WHO 2006 Towards Universal Access – Scaling up priority HIV/AIDS interventions in the health sector. WHO/UNAIDS/UNICEF, 2008 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 HIV and infant feeding technical consultation Geneva, October 25 -27, 2006 CONSENSUS STATEMENT • The most appropriate infant feeding option for an HIV-infected mother should continue to depend on her individual circumstances………… • Exclusive breastfeeding is recommended for HIV-infected women for the first 6 months of life unless replacement feeding is acceptable, feasible, affordable, sustainable and safe (AFASS) for them and their infants before that time. • When replacement feeding is acceptable, feasible, affordable, sustainable and safe, avoidance of all breastfeeding by HIVinfected women is recommended. International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
HIV and infant feeding technical consultation Geneva, October 25 -27, 2006 CONSENSUS STATEMENT • The most appropriate infant feeding option for an HIV-infected mother should continue to depend on her individual circumstances………… • Exclusive breastfeeding is recommended for HIV-infected women for the first 6 months of life unless replacement feeding is acceptable, feasible, affordable, sustainable and safe (AFASS) for them and their infants before that time. • When replacement feeding is acceptable, feasible, affordable, sustainable and safe, avoidance of all breastfeeding by HIVinfected women is recommended. International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
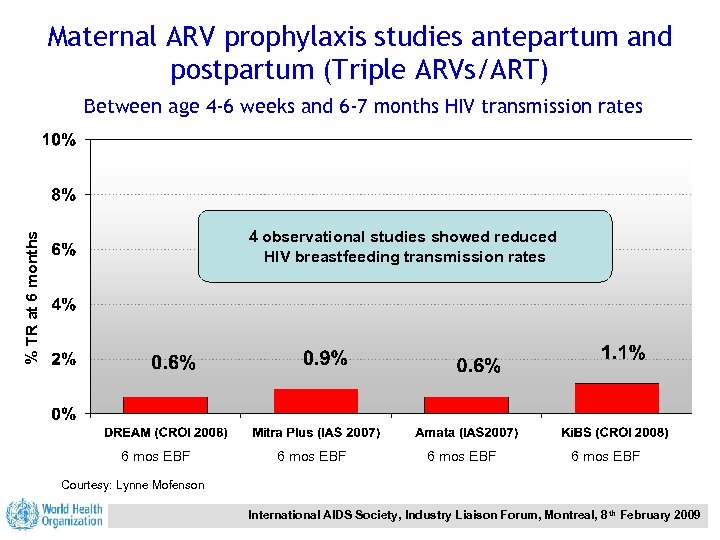 Maternal ARV prophylaxis studies antepartum and postpartum (Triple ARVs/ART) Between age 4 -6 weeks and 6 -7 months HIV transmission rates % TR at 6 months 4 observational studies showed reduced HIV breastfeeding transmission rates 6 mos EBF Courtesy: Lynne Mofenson International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Maternal ARV prophylaxis studies antepartum and postpartum (Triple ARVs/ART) Between age 4 -6 weeks and 6 -7 months HIV transmission rates % TR at 6 months 4 observational studies showed reduced HIV breastfeeding transmission rates 6 mos EBF Courtesy: Lynne Mofenson International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
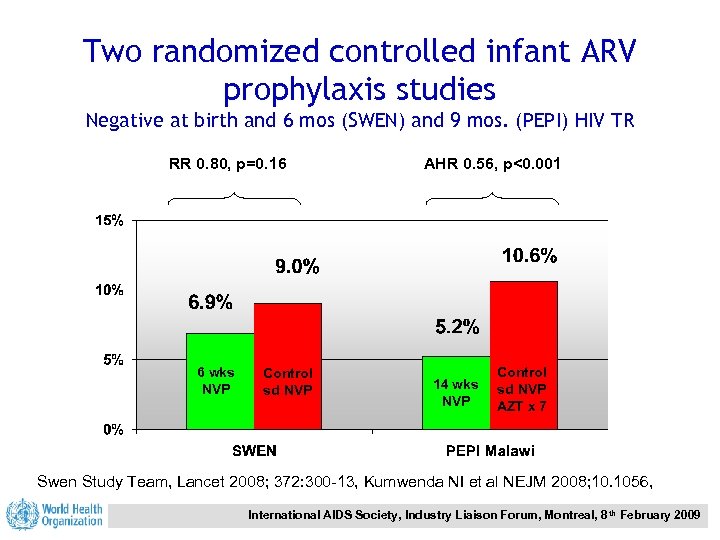 Two randomized controlled infant ARV prophylaxis studies Negative at birth and 6 mos (SWEN) and 9 mos. (PEPI) HIV TR RR 0. 80, p=0. 16 6 wks NVP Control sd NVP AHR 0. 56, p<0. 001 14 wks NVP Control sd NVP AZT x 7 Swen Study Team, Lancet 2008; 372: 300 -13, Kumwenda NI et al NEJM 2008; 10. 1056, International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Two randomized controlled infant ARV prophylaxis studies Negative at birth and 6 mos (SWEN) and 9 mos. (PEPI) HIV TR RR 0. 80, p=0. 16 6 wks NVP Control sd NVP AHR 0. 56, p<0. 001 14 wks NVP Control sd NVP AZT x 7 Swen Study Team, Lancet 2008; 372: 300 -13, Kumwenda NI et al NEJM 2008; 10. 1056, International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Research priorities Maternal triple ARV regimens for prophylaxis • • • Impact of stopping triple ARVs on women's health Optimal regimens differentiated by maternal CD 4 Safety for the mother and the infant Transmitted HIV drug resistance to the infant Optimum duration of ARV prophylaxis (? until EBF discontinued or lifelong) • Feasibility and cost International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Research priorities Maternal triple ARV regimens for prophylaxis • • • Impact of stopping triple ARVs on women's health Optimal regimens differentiated by maternal CD 4 Safety for the mother and the infant Transmitted HIV drug resistance to the infant Optimum duration of ARV prophylaxis (? until EBF discontinued or lifelong) • Feasibility and cost International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Research priorities • Extended infant ARV prophylaxis – Optimal duration and safety of prolonged nevirapine, or other ARVs e. g. 3 TC, administration to HIV negative infants – Resistance and impact on prophylaxis and future treatment options in infants who become infected despite prophylaxis – Feasibility – under what circumstances would infant prophylaxis be preferred to maternal prophylaxis • Infant feeding – Under what circumstances to recommend a woman to stop breastfeeding at about 6 months? – Can an infant be adequately fed from 6 -12 months without any milk and still achieve normal growth and development? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Research priorities • Extended infant ARV prophylaxis – Optimal duration and safety of prolonged nevirapine, or other ARVs e. g. 3 TC, administration to HIV negative infants – Resistance and impact on prophylaxis and future treatment options in infants who become infected despite prophylaxis – Feasibility – under what circumstances would infant prophylaxis be preferred to maternal prophylaxis • Infant feeding – Under what circumstances to recommend a woman to stop breastfeeding at about 6 months? – Can an infant be adequately fed from 6 -12 months without any milk and still achieve normal growth and development? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 ‘PMTCT’ • As a term it has – Constrained our understanding of what is needed – Focussed the outcome of value to be what happens to ‘the child’ – Partitioned service delivery • Greater understanding of the relationship between treatment and prevention – Requires a different way of thinking, and organisation of services – To achieve HIV-free survival of the child, it is essential to protect maternal health and survival • ‘Integrated Care of Women and Children in the context of HIV’ International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
‘PMTCT’ • As a term it has – Constrained our understanding of what is needed – Focussed the outcome of value to be what happens to ‘the child’ – Partitioned service delivery • Greater understanding of the relationship between treatment and prevention – Requires a different way of thinking, and organisation of services – To achieve HIV-free survival of the child, it is essential to protect maternal health and survival • ‘Integrated Care of Women and Children in the context of HIV’ International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
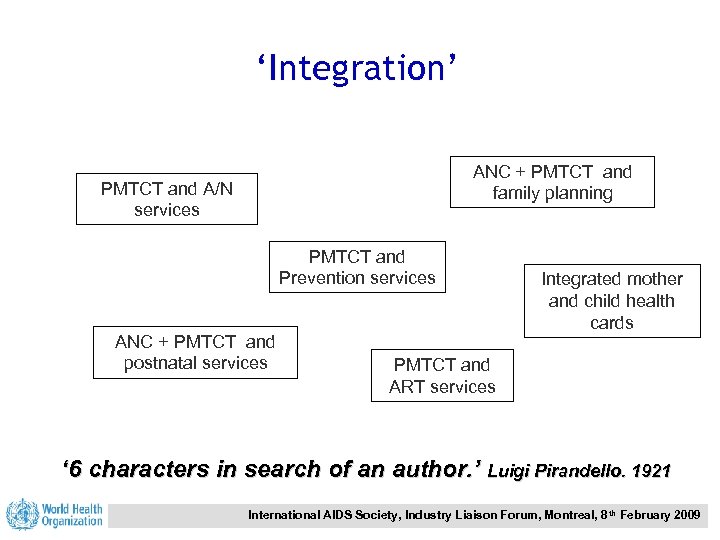 ‘Integration’ ANC + PMTCT and family planning PMTCT and A/N services PMTCT and Prevention services ANC + PMTCT and postnatal services Integrated mother and child health cards PMTCT and ART services ‘ 6 characters in search of an author. ’ Luigi Pirandello. 1921 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
‘Integration’ ANC + PMTCT and family planning PMTCT and A/N services PMTCT and Prevention services ANC + PMTCT and postnatal services Integrated mother and child health cards PMTCT and ART services ‘ 6 characters in search of an author. ’ Luigi Pirandello. 1921 International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
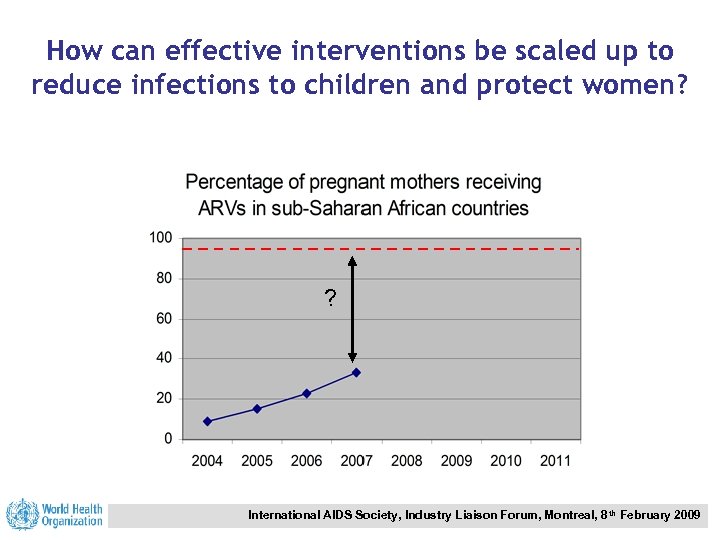 How can effective interventions be scaled up to reduce infections to children and protect women? ? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
How can effective interventions be scaled up to reduce infections to children and protect women? ? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 From proof of principle to implementation • Cannot simply extrapolate practices implemented at one pilot site (even 2 -3 sites) and expect the same outcomes – Promotion of exclusive breastfeeding International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
From proof of principle to implementation • Cannot simply extrapolate practices implemented at one pilot site (even 2 -3 sites) and expect the same outcomes – Promotion of exclusive breastfeeding International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
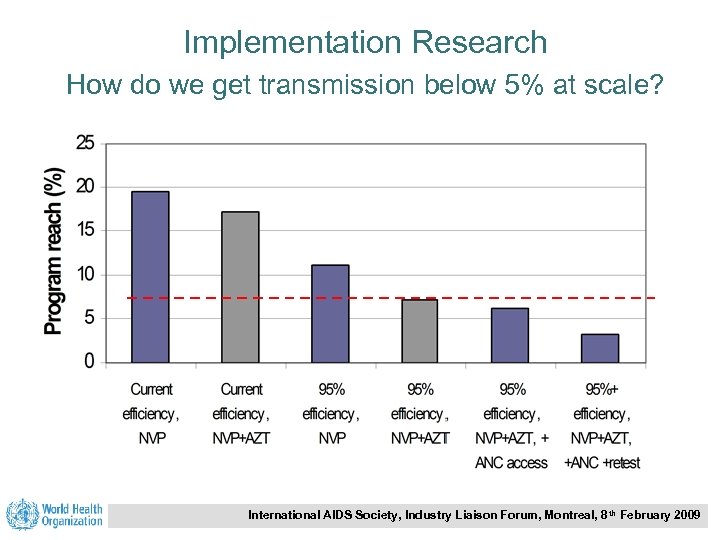 Implementation Research How do we get transmission below 5% at scale? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Implementation Research How do we get transmission below 5% at scale? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Why do academics and funders not, more commonly, engage these questions? • Implementation research not seen as a serious science • Requires a different way of looking at problems - unfamiliar territory • Out there – not in here • Not in control (of the system) • Needs to work with non-researchers and Do. H – multidisciplinary team • Different skills and perspectives International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Why do academics and funders not, more commonly, engage these questions? • Implementation research not seen as a serious science • Requires a different way of looking at problems - unfamiliar territory • Out there – not in here • Not in control (of the system) • Needs to work with non-researchers and Do. H – multidisciplinary team • Different skills and perspectives International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Other ethical challenges in paediatric HIV research • How to balance the child’s interests (survival) vs. the mother’s interests (confidentiality)? • Developing models of care that might place child at risk because of dependency e. g. hospital or home for MDR / XDR TB Rx. • How to assess understanding in the assent process? • If effective vaccines are identified, how can these be given to infants or children in order to prevent infections in adolescence? • How to respect the sexual and reproductive rights of HIVinfected adolescents in order to achieve risk-reduction interventions for other? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Other ethical challenges in paediatric HIV research • How to balance the child’s interests (survival) vs. the mother’s interests (confidentiality)? • Developing models of care that might place child at risk because of dependency e. g. hospital or home for MDR / XDR TB Rx. • How to assess understanding in the assent process? • If effective vaccines are identified, how can these be given to infants or children in order to prevent infections in adolescence? • How to respect the sexual and reproductive rights of HIVinfected adolescents in order to achieve risk-reduction interventions for other? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
 Other ethical challenges in paediatric HIV research • What responsibility do research centres have to help local services improve their quality of care vs. setting up parallel structures? • Is it reasonable to research models of care that cannot be scaled-up to address the greater need? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009
Other ethical challenges in paediatric HIV research • What responsibility do research centres have to help local services improve their quality of care vs. setting up parallel structures? • Is it reasonable to research models of care that cannot be scaled-up to address the greater need? International AIDS Society, Industry Liaison Forum, Montreal, 8 th February 2009


